Functions and Mechanisms of Fibroblast Growth Factor (FGF) Signalling in Drosophila melanogaster
Abstract
:1. Drosophila melanogaster as a Versatile Model for FGF Signalling Research
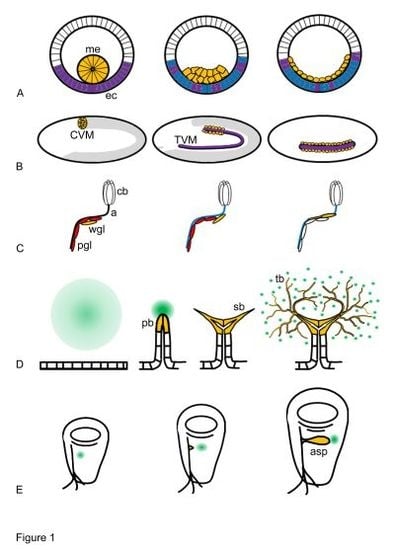
2. Biological Functions of FGF Signalling in Drosophila Development
2.1. Biological Functions of Htl
2.2. Biological Functions of Btl
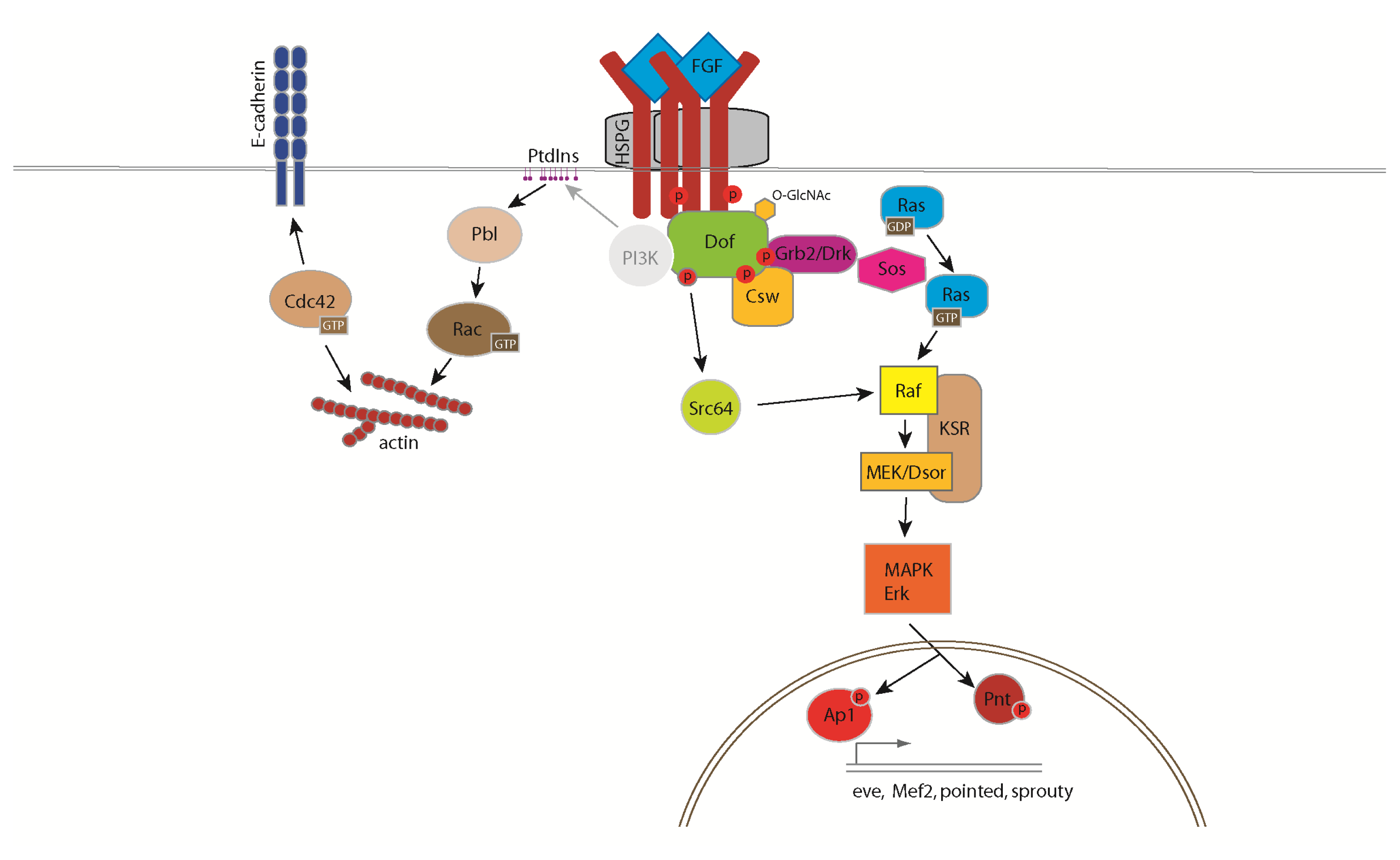
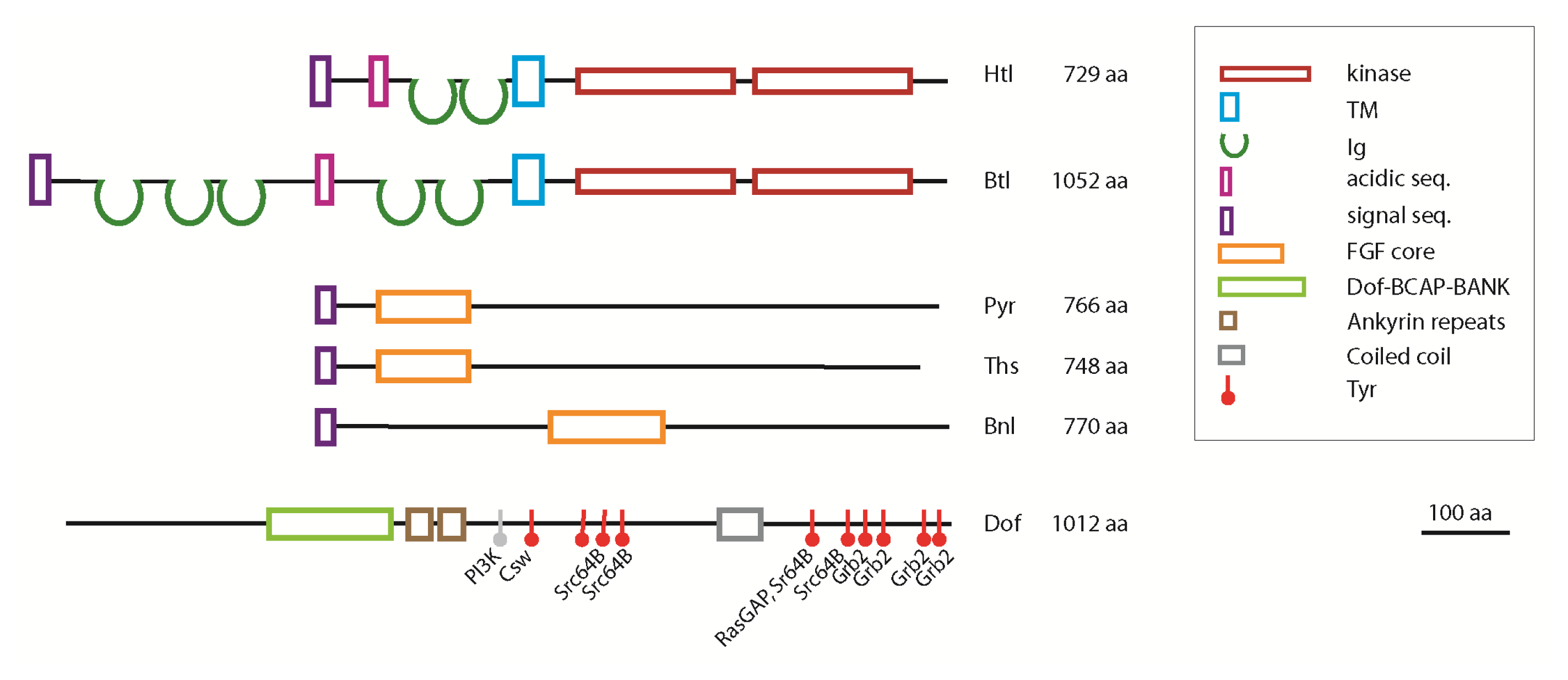
3. The FGF Signalling Pathway in Drosophila
4. Cellular Responses Controlled by FGF Signalling
5. Conclusions
Acknowledgements
References
- Glazer, L.; Shilo, B.Z. The drosophila fgf-r homolog is expressed in the embryonic tracheal system and appears to be required for directed tracheal cell extension. Genes Dev 1991, 5, 697–705. [Google Scholar]
- Itoh, N.; Ornitz, D.M. Fibroblast growth factors: From molecular evolution to roles in development, metabolism and disease. J. Biochem 2010, 149, 121–130. [Google Scholar]
- Zhang, X.; Ibrahimi, O.A.; Olsen, S.K.; Umemori, H.; Mohammadi, M.; Ornitz, D.M. Receptor specificity of the fibroblast growth factor family. The complete mammalian fgf family. J. Biol. Chem 2006, 281, 15694–15700. [Google Scholar]
- Klambt, C.; Glazer, L.; Shilo, B.Z. Breathless, a drosophila fgf receptor homolog, is essential for migration of tracheal and specific midline glial cells. Genes Dev 1992, 6, 1668–1678. [Google Scholar]
- Gryzik, T.; Muller, H.A. Fgf8-like1 and fgf8-like2 encode putative ligands of the fgf receptor htl and are required for mesoderm migration in the Drosophila gastrula. Curr. Biol 2004, 14, 659–667. [Google Scholar]
- Schumacher, S.; Gryzik, T.; Tannebaum, S.; Muller, H.A. The rhogef pebble is required for cell shape changes during cell migration triggered by the Drosophila fgf receptor heartless. Development 2004, 131, 2631–2640. [Google Scholar]
- Stathopoulos, A.; Tam, B.; Ronshaugen, M.; Frasch, M.; Levine, M. Pyramus and thisbe: Fgf genes that pattern the mesoderm of Drosophila embryos. Genes Dev 2004, 18, 687–699. [Google Scholar]
- Beiman, M.; Shilo, B.Z.; Volk, T. Heartless, a Drosophila fgf receptor homolog, is essential for cell migration and establishment of several mesodermal lineages. Genes Dev 1996, 10, 2993–3002. [Google Scholar]
- Reichman-Fried, M.; Shilo, B.Z. Breathless, a Drosophila fgf receptor homolog, is required for the onset of tracheal cell migration and tracheole formation. Mech. Dev 1995, 52, 265–273. [Google Scholar]
- Michelson, A.M.; Gisselbrecht, S.; Buff, E.; Skeath, J.B. Heartbroken is a specific downstream mediator of fgf receptor signalling in Drosophila. Development 1998, 125, 4379–4389. [Google Scholar]
- Lee, T.; Hacohen, N.; Krasnow, M.; Montell, D.J. Regulated breathless receptor tyrosine kinase activity required to pattern cell migration and branching in the Drosophila tracheal system. Genes Dev 1996, 10, 2912–2921. [Google Scholar]
- Metzger, R.J.; Krasnow, M.A. Genetic control of branching morphogenesis. Science 1999, 284, 1635–1639. [Google Scholar]
- Yang, X.; Dormann, D.; Munsterberg, A.E.; Weijer, C.J. Cell movement patterns during gastrulation in the chick are controlled by positive and negative chemotaxis mediated by fgf4 and fgf8. Dev. Cell 2002, 3, 425–437. [Google Scholar]
- Ciruna, B.; Rossant, J. Fgf signaling regulates mesoderm cell fate specification and morphogenetic movement at the primitive streak. Dev. Cell 2001, 1, 37–49. [Google Scholar]
- Klingseisen, A.; Clark, I.B.; Gryzik, T.; Muller, H.A. Differential and overlapping functions of two closely related drosophila fgf8-like growth factors in mesoderm development. Development 2009, 136, 2393–2402. [Google Scholar]
- Clark, I.B.; Muha, V.; Klingseisen, A.; Leptin, M.; Muller, H.A. Fibroblast growth factor signalling controls successive cell behaviours during mesoderm layer formation in Drosophila. Development 2011, 138, 2705–2715. [Google Scholar]
- Kadam, S.; McMahon, A.; Tzou, P.; Stathopoulos, A. Fgf ligands in Drosophila have distinct activities required to support cell migration and differentiation. Development 2009, 136, 739–747. [Google Scholar]
- Gisselbrecht, S.; Skeath, J.B.; Doe, C.Q.; Michelson, A.M. Heartless encodes a fibroblast growth factor receptor (dfr1/dfgf-r2) involved in the directional migration of early mesodermal cells in the Drosophila embryo. Genes Dev 1996, 10, 3003–3017. [Google Scholar]
- Winklbauer, R.; Muller, H.A. Mesoderm layer formation in xenopus and Drosophila gastrulation. Phys. Biol 2011, 8, 045001. [Google Scholar]
- McMahon, A.; Reeves, G.T.; Supatto, W.; Stathopoulos, A. Mesoderm migration in Drosophila is a multi-step process requiring fgf signaling and integrin activity. Development 2010, 137, 2167–2175. [Google Scholar]
- McMahon, A.; Supatto, W.; Fraser, S.E.; Stathopoulos, A. Dynamic analyses of Drosophila gastrulation provide insights into collective cell migration. Science 2008, 322, 1546–1550. [Google Scholar]
- Murray, M.J.; Saint, R. Photoactivatable gfp resolves Drosophila mesoderm migration behaviour. Development 2007, 134, 3975–3983. [Google Scholar]
- Wilson, R.; Vogelsang, E.; Leptin, M. Fgf signalling and the mechanism of mesoderm spreading in Drosophila embryos. Development 2005, 132, 491–501. [Google Scholar]
- Shishido, E.; Higashijima, S.; Emori, Y.; Saigo, K. Two fgf-receptor homologues of Drosophila: One is expressed in mesodermal primordium in early embryos. Development 1993, 117, 751–761. [Google Scholar]
- Shishido, E.; Ono, N.; Kojima, T.; Saigo, K. Requirements of dfr1/heartless, a mesoderm-specific drosophila fgf-receptor, for the formation of heart, visceral and somatic muscles, and ensheathing of longitudinal axon tracts in cns. Development 1997, 124, 2119–2128. [Google Scholar]
- Michelson, A.M.; Gisselbrecht, S.; Zhou, Y.; Baek, K.H.; Buff, E.M. Dual functions of the heartless fibroblast growth factor receptor in development of the Drosophila embryonic mesoderm. Dev. Genet 1998, 22, 212–229. [Google Scholar]
- Dutta, D.; Shaw, S.; Maqbool, T.; Pandya, H.; Vijayraghavan, K. Drosophila heartless acts with heartbroken/dof in muscle founder differentiation. PLoS Biol 2005, 3, e337. [Google Scholar]
- Kadam, S.; Ghosh, S.; Stathopoulos, A. Synchronous and symmetric migration of Drosophila caudal visceral mesoderm cells requires dual input by two fgf ligands. Development 2012, 139, 699–708. [Google Scholar]
- Reim, I.; Hollfelder, D.; Ismat, A.; Frasch, M. The fgf8-related signals pyramus and thisbe promote pathfinding, substrate adhesion, and survival of migrating longitudinal gut muscle founder cells. Dev. Biol 2012, 368, 28–43. [Google Scholar]
- Franzdottir, S.R.; Engelen, D.; Yuva-Aydemir, Y.; Schmidt, I.; Aho, A.; Klambt, C. Switch in fgf signalling initiates glial differentiation in the Drosophila eye. Nature 2009, 460, 758–761. [Google Scholar]
- Avet-Rochex, A.; Kaul, A.K.; Gatt, A.P.; McNeill, H.; Bateman, J.M. Concerted control of gliogenesis by inr/tor and fgf signalling in the Drosophila post-embryonic brain. Development 2012, 139, 2763–2772. [Google Scholar]
- Ghabrial, A.; Luschnig, S.; Metzstein, M.M.; Krasnow, M.A. Branching morphogenesis of the Drosophila tracheal system. Annu. Rev. Cell Dev. Biol 2003, 19, 623–647. [Google Scholar]
- Jarecki, J.; Johnson, E.; Krasnow, M.A. Oxygen regulation of airway branching in Drosophila is mediated by branchless fgf. Cell 1999, 99, 211–220. [Google Scholar]
- Sutherland, D.; Samakovlis, C.; Krasnow, M.A. Branchless encodes a Drosophila fgf homolog that controls tracheal cell migration and the pattern of branching. Cell 1996, 87, 1091–1101. [Google Scholar]
- Cabernard, C.; Affolter, M. Distinct roles for two receptor tyrosine kinases in epithelial branching morphogenesis in Drosophila. Dev. Cell 2005, 9, 831–842. [Google Scholar]
- Chanut-Delalande, H.; Jung, A.C.; Lin, L.; Baer, M.M.; Bilstein, A.; Cabernard, C.; Leptin, M.; Affolter, M. A genetic mosaic analysis with a repressible cell marker screen to identify genes involved in tracheal cell migration during Drosophila air sac morphogenesis. Genetics 2007, 176, 2177–2187. [Google Scholar]
- Chanut-Delalande, H.; Jung, A.C.; Baer, M.M.; Lin, L.; Payre, F.; Affolter, M. The hrs/stam complex acts as a positive and negative regulator of rtk signaling during Drosophila development. PLoS One 2010, 5, e10245. [Google Scholar]
- Ahmad, S.M.; Baker, B.S. Sex-specific deployment of fgf signaling in Drosophila recruits mesodermal cells into the male genital imaginal disc. Cell 2002, 109, 651–661. [Google Scholar]
- Murphy, A.M.; Lee, T.; Andrews, C.M.; Shilo, B.Z.; Montell, D.J. The breathless fgf receptor homolog, a downstream target of Drosophila c/ebp in the developmental control of cell migration. Development 1995, 121, 2255–2263. [Google Scholar]
- Sen, A.; Yokokura, T.; Kankel, M.W.; Dimlich, D.N.; Manent, J.; Sanyal, S.; Artavanis-Tsakonas, S. Modeling spinal muscular atrophy in Drosophila links smn to fgf signaling. J. Cell Biol 2011, 192, 481–495. [Google Scholar]
- Rapraeger, A.C.; Krufka, A.; Olwin, B.B. Requirement of heparan sulfate for bfgf-mediated fibroblast growth and myoblast differentiation. Science 1991, 252, 1705–1708. [Google Scholar]
- Nugent, M.A.; Edelman, E.R. Kinetics of basic fibroblast growth factor binding to its receptor and heparan sulfate proteoglycan: A mechanism for cooperactivity. Biochemistry 1992, 31, 8876–8883. [Google Scholar]
- Perrimon, N.; Lanjuin, A.; Arnold, C.; Noll, E. Zygotic lethal mutations with maternal effect phenotypes in Drosophila melanogaster. II. Loci on the second and third chromosomes identified by p-element-induced mutations. Genetics 1996, 144, 1681–1692. [Google Scholar]
- Hacker, U.; Lin, X.; Perrimon, N. The Drosophila sugarless gene modulates wingless signaling and encodes an enzyme involved in polysaccharide biosynthesis. Development 1997, 124, 3565–3573. [Google Scholar]
- Lin, X.; Buff, E.M.; Perrimon, N.; Michelson, A.M. Heparan sulfate proteoglycans are essential for fgf receptor signaling during Drosophila embryonic development. Development 1999, 126, 3715–3723. [Google Scholar]
- Kamimura, K.; Koyama, T.; Habuchi, H.; Ueda, R.; Masu, M.; Kimata, K.; Nakato, H. Specific and flexible roles of heparan sulfate modifications in Drosophila fgf signaling. J. Cell Biol 2006, 174, 773–778. [Google Scholar]
- Yan, D.; Lin, X. Drosophila glypican dally-like acts in fgf-receiving cells to modulate fgf signaling during tracheal morphogenesis. Dev. Biol 2007, 312, 203–216. [Google Scholar]
- Knox, J.; Moyer, K.; Yacoub, N.; Soldaat, C.; Komosa, M.; Vassilieva, K.; Wilk, R.; Hu, J.; de Vazquez Paz, L.L.; Syed, Q.; et al. Syndecan contributes to heart cell specification and lumen formation during Drosophila cardiogenesis. Dev. Biol. 2011, 356, 279–290. [Google Scholar]
- Vincent, S.; Wilson, R.; Coelho, C.; Affolter, M.; Leptin, M. The Drosophila protein dof is specifically required for fgf signaling. Mol. Cell 1998, 2, 515–525. [Google Scholar]
- Csiszar, A.; Vogelsang, E.; Beug, H.; Leptin, M. A novel conserved phosphotyrosine motif in the drosophila fibroblast growth factor signaling adaptor dof with a redundant role in signal transmission. Mol. Cell Biol 2010, 30, 2017–2027. [Google Scholar]
- Itoh, N.; Ornitz, D.M. Evolution of the fgf and fgfr gene families. Trends Genet 2004, 20, 563–569. [Google Scholar]
- Tulin, S.; Stathopoulos, A. Analysis of thisbe and pyramus functional domains reveals evidence for cleavage of Drosophila fgfs. BMC Dev. Biol 2010, 10, 83. [Google Scholar]
- Klingseisen, A. Function of FGF8-Like1 and -2. Drosophila mesoderm Cell Migration. Ph.D. Dissertation, University of Dundee, Dundee, UK, 2010. [Google Scholar]
- Wang, Q.; Uhlirova, M.; Bohmann, D. Spatial restriction of fgf signaling by a matrix metalloprotease controls branching morphogenesis. Dev. Cell 2010, 18, 157–164. [Google Scholar]
- Battersby, A.; Csiszar, A.; Leptin, M.; Wilson, R. Isolation of proteins that interact with the signal transduction molecule dof and identification of a functional domain conserved between dof and vertebrate bcap. J. Mol. Biol 2003, 329, 479–493. [Google Scholar]
- Petit, V.; Nussbaumer, U.; Dossenbach, C.; Affolter, M. Downstream-of-fgfr is a fibroblast growth factor-specific scaffolding protein and recruits corkscrew upon receptor activation. Mol. Cell Biol 2004, 24, 3769–3781. [Google Scholar]
- Wilson, R.; Battersby, A.; Csiszar, A.; Vogelsang, E.; Leptin, M. A functional domain of dof that is required for fibroblast growth factor signaling. Mol. Cell Biol 2004, 24, 2263–2276. [Google Scholar]
- Imam, F.; Sutherland, D.; Huang, W.; Krasnow, M.A. Stumps, a Drosophila gene required for fibroblast growth factor (fgf)-directed migrations of tracheal and mesodermal cells. Genetics 1999, 152, 307–318. [Google Scholar]
- Turner, N.; Grose, R. Fibroblast growth factor signalling: From development to cancer. Nat. Rev. Cancer 2010, 10, 116–129. [Google Scholar]
- Gabay, L.; Seger, R.; Shilo, B.Z. Map kinase in situ activation atlas during Drosophila embryogenesis. Development 1997, 124, 3535–3541. [Google Scholar]
- Okada, T.; Maeda, A.; Iwamatsu, A.; Gotoh, K.; Kurosaki, T. Bcap: The tyrosine kinase substrate that connects b cell receptor to phosphoinositide 3-kinase activation. Immunity 2000, 13, 817–827. [Google Scholar]
- Yokoyama, K.; Su Ih, I.H.; Tezuka, T.; Yasuda, T.; Mikoshiba, K.; Tarakhovsky, A.; Yamamoto, T. Bank regulates bcr-induced calcium mobilization by promoting tyrosine phosphorylation of ip(3) receptor. EMBO J 2002, 21, 83–92. [Google Scholar]
- Wassarman, D.A.; Therrien, M.; Rubin, G.M. The ras signaling pathway in Drosophila. Curr. Opin. Genet. Dev 1995, 5, 44–50. [Google Scholar]
- Xia, F.; Li, J.; Hickey, G.W.; Tsurumi, A.; Larson, K.; Guo, D.; Yan, S.J.; Silver-Morse, L.; Li, W.X. Raf activation is regulated by tyrosine 510 phosphorylation in Drosophila. PLoS Biol 2008, 6, e128. [Google Scholar]
- Hacohen, N.; Kramer, S.; Sutherland, D.; Hiromi, Y.; Krasnow, M.A. Sprouty encodes a novel antagonist of fgf signaling that patterns apical branching of the Drosophila airways. Cell 1998, 92, 253–263. [Google Scholar]
- Mariappa, D.; Sauert, K.; Marino, K.; Turnock, D.; Webster, R.; van Aalten, D.M.; Ferguson, M.A.; Muller, H.A. Protein O-glcnacylation is required for fibroblast growth factor signaling in. Drosophila. Sci. Signal 2011, 4, ra89. [Google Scholar]
- Ridley, A.J. Rho gtpases and actin dynamics in membrane protrusions and vesicle trafficking. Trends Cell Biol 2006, 16, 522–529. [Google Scholar]
- Prokopenko, S.N.; Brumby, A.; O’Keefe, L.; Prior, L.; He, Y.; Saint, R.; Bellen, H.J. A putative exchange factor for rho1 gtpase is required for initiation of cytokinesis in Drosophila. Genes. Dev 1999, 13, 2301–2314. [Google Scholar]
- van Impel, A.; Schumacher, S.; Draga, M.; Herz, H.M.; Grosshans, J.; Muller, H.A. Regulation of the rac gtpase pathway by the multifunctional rho gef pebble is essential for mesoderm migration in the Drosophila gastrula. Development 2009, 136, 813–822. [Google Scholar]
- Liu, X.F.; Ishida, H.; Raziuddin, R.; Miki, T. Nucleotide exchange factor ect2 interacts with the polarity protein complex par6/par3/protein kinase czeta (pkczeta) and regulates pkczeta activity. Mol. Cell Biol 2004, 24, 6665–6675. [Google Scholar]
- Solski, P.A.; Wilder, R.S.; Rossman, K.L.; Sondek, J.; Cox, A.D.; Campbell, S.L.; Der, C.J. Requirement for C-terminal sequences in regulation of ect2 guanine nucleotide exchange specificity and transformation. J. Biol. Chem 2004, 279, 25226–25233. [Google Scholar]
- Sato, M.; Kornberg, T.B. Fgf is an essential mitogen and chemoattractant for the air sacs of the Drosophila tracheal system. Dev. Cell 2002, 3, 195–207. [Google Scholar]
- Carmena, A.; Gisselbrecht, S.; Harrison, J.; Jimenez, F.; Michelson, A.M. Combinatorial signaling codes for the progressive determination of cell fates in the Drosophila embryonic mesoderm. Genes. Dev 1998, 12, 3910–3922. [Google Scholar]
- Guillemin, K.; Groppe, J.; Ducker, K.; Treisman, R.; Hafen, E.; Affolter, M.; Krasnow, M.A. The pruned gene encodes the Drosophila serum response factor and regulates cytoplasmic outgrowth during terminal branching of the tracheal system. Development 1996, 122, 1353–1362. [Google Scholar]
- Smallhorn, M.; Murray, M.J.; Saint, R. The epithelial-mesenchymal transition of the Drosophila mesoderm requires the rho gtp exchange factor pebble. Development 2004, 131, 2641–2651. [Google Scholar]
- Murray, M.J.; Ng, M.M.; Fraval, H.; Tan, J.; Liu, W.; Smallhorn, M.; Brill, J.A.; Field, S.J.; Saint, R. Regulation of Drosophila mesoderm migration by phosphoinositides and the ph domain of the rho gtp exchange factor pebble. Dev. Biol 2012, 372, 17–27. [Google Scholar]
- Ribeiro, C.; Ebner, A.; Affolter, M. In vivo imaging reveals different cellular functions for fgf and dpp signaling in tracheal branching morphogenesis. Dev. Cell 2002, 2, 677–683. [Google Scholar]
- Lim, J.; Thiery, J.P. Epithelial-mesenchymal transitions: Insights from development. Development 2012, 139, 3471–3486. [Google Scholar]
- Urbano, J.M.; Dominguez-Gimenez, P.; Estrada, B.; Martin-Bermudo, M.D. Ps integrins and laminins: Key regulators of cell migration during Drosophila embryogenesis. PLoS One 2011, 6, e23893. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Muha, V.; Müller, H.-A.J. Functions and Mechanisms of Fibroblast Growth Factor (FGF) Signalling in Drosophila melanogaster. Int. J. Mol. Sci. 2013, 14, 5920-5937. https://doi.org/10.3390/ijms14035920
Muha V, Müller H-AJ. Functions and Mechanisms of Fibroblast Growth Factor (FGF) Signalling in Drosophila melanogaster. International Journal of Molecular Sciences. 2013; 14(3):5920-5937. https://doi.org/10.3390/ijms14035920
Chicago/Turabian StyleMuha, Villö, and Hans-Arno J. Müller. 2013. "Functions and Mechanisms of Fibroblast Growth Factor (FGF) Signalling in Drosophila melanogaster" International Journal of Molecular Sciences 14, no. 3: 5920-5937. https://doi.org/10.3390/ijms14035920