Optimized Production of Biodiesel from Waste Cooking Oil by Lipase Immobilized on Magnetic Nanoparticles
Abstract
:1. Introduction
2. Results and Discussion
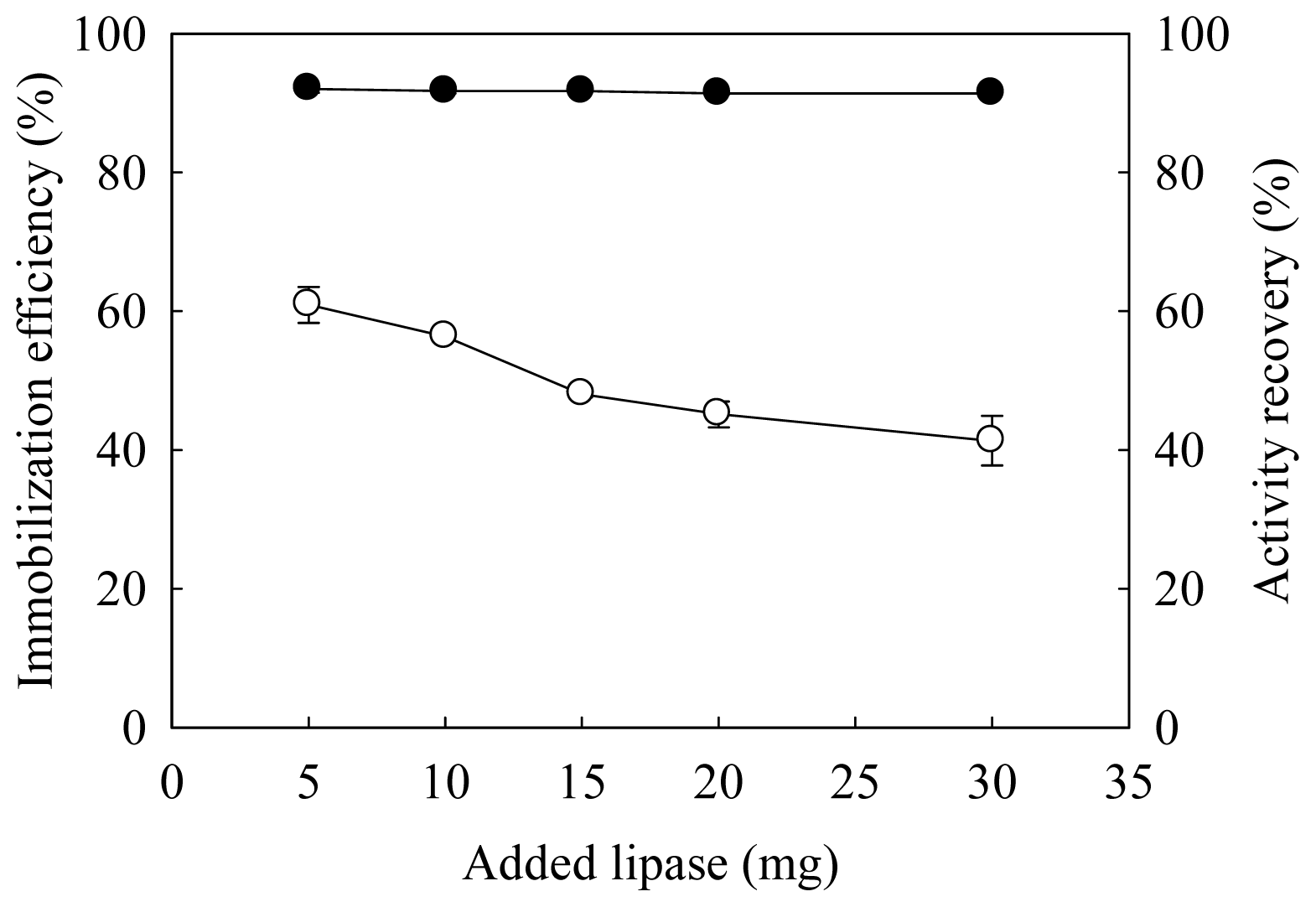
2.1. Effects of Amount of Added Lipase on Immobilization Efficiency and Activity Recovery
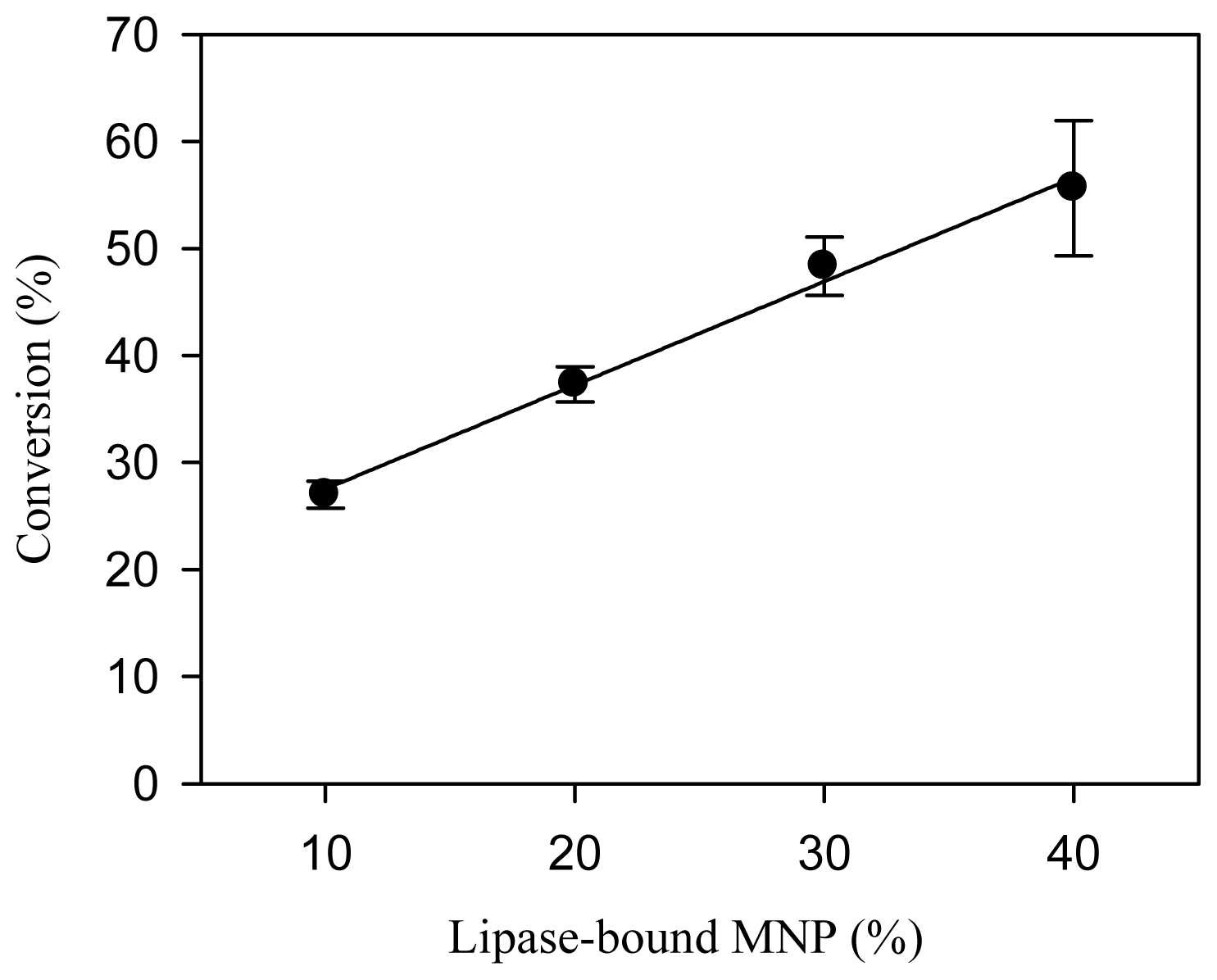
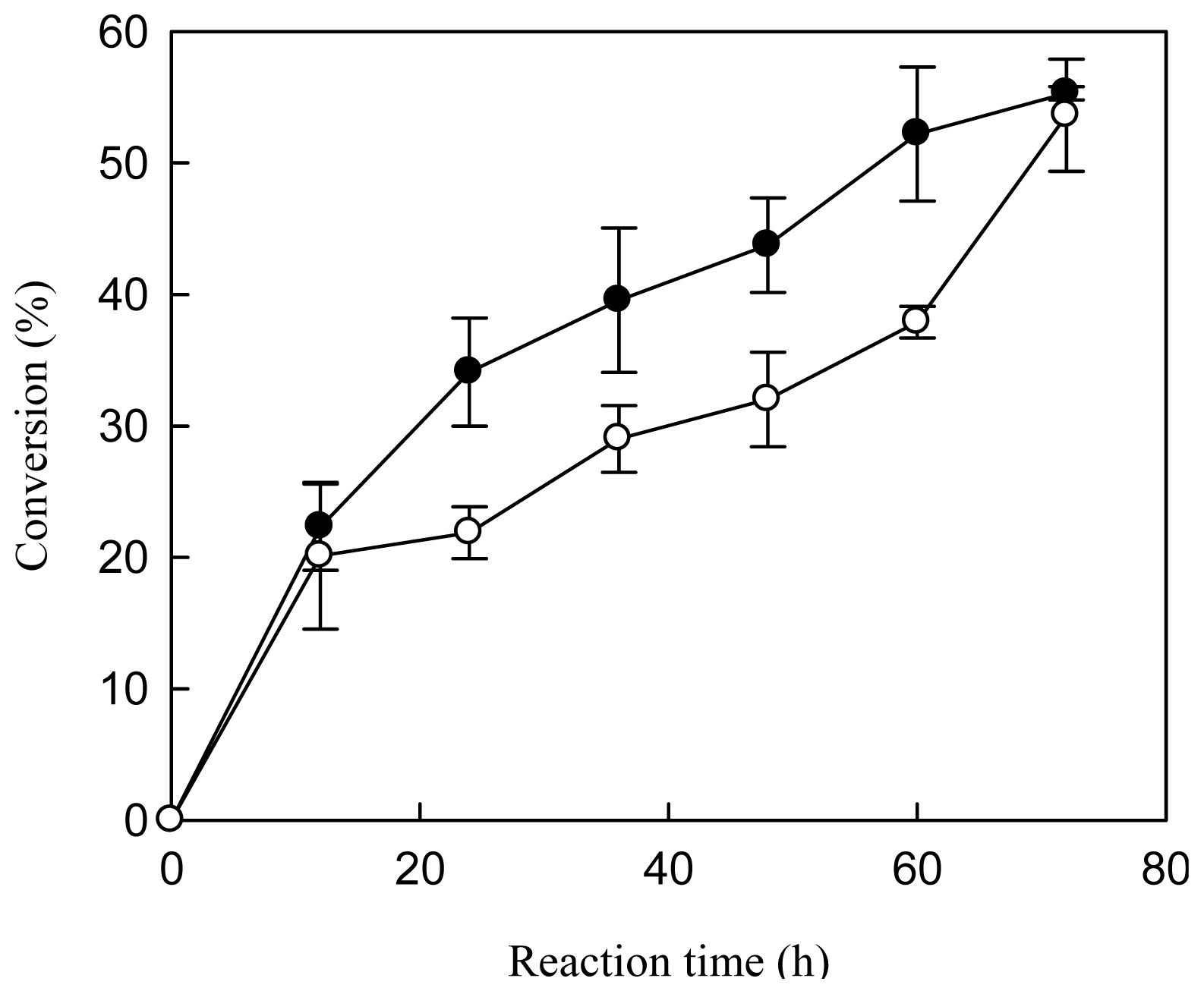
2.2. Effects of Dosage of Lipase-Bound MNP and Stepwise Addition of Methanol on the Conversion of FAME
2.3. Model Fitting and Analysis of Variance
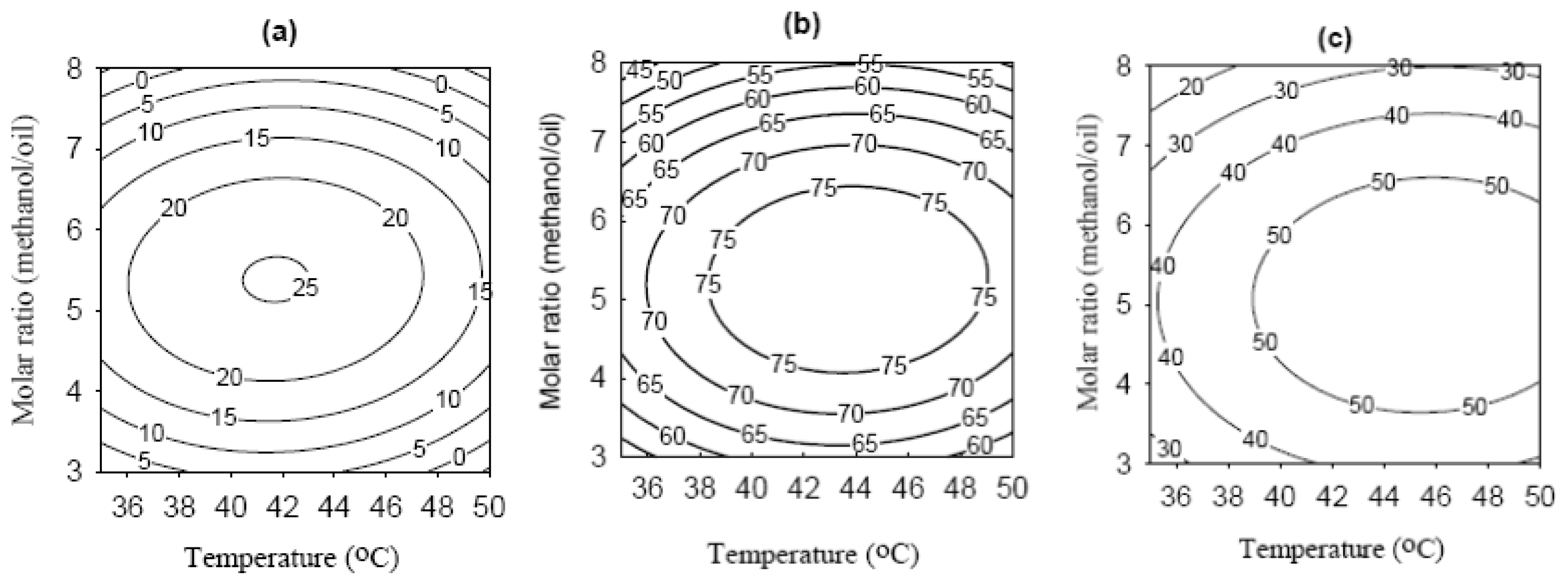
2.4. Effects of Variables and Their Optimization
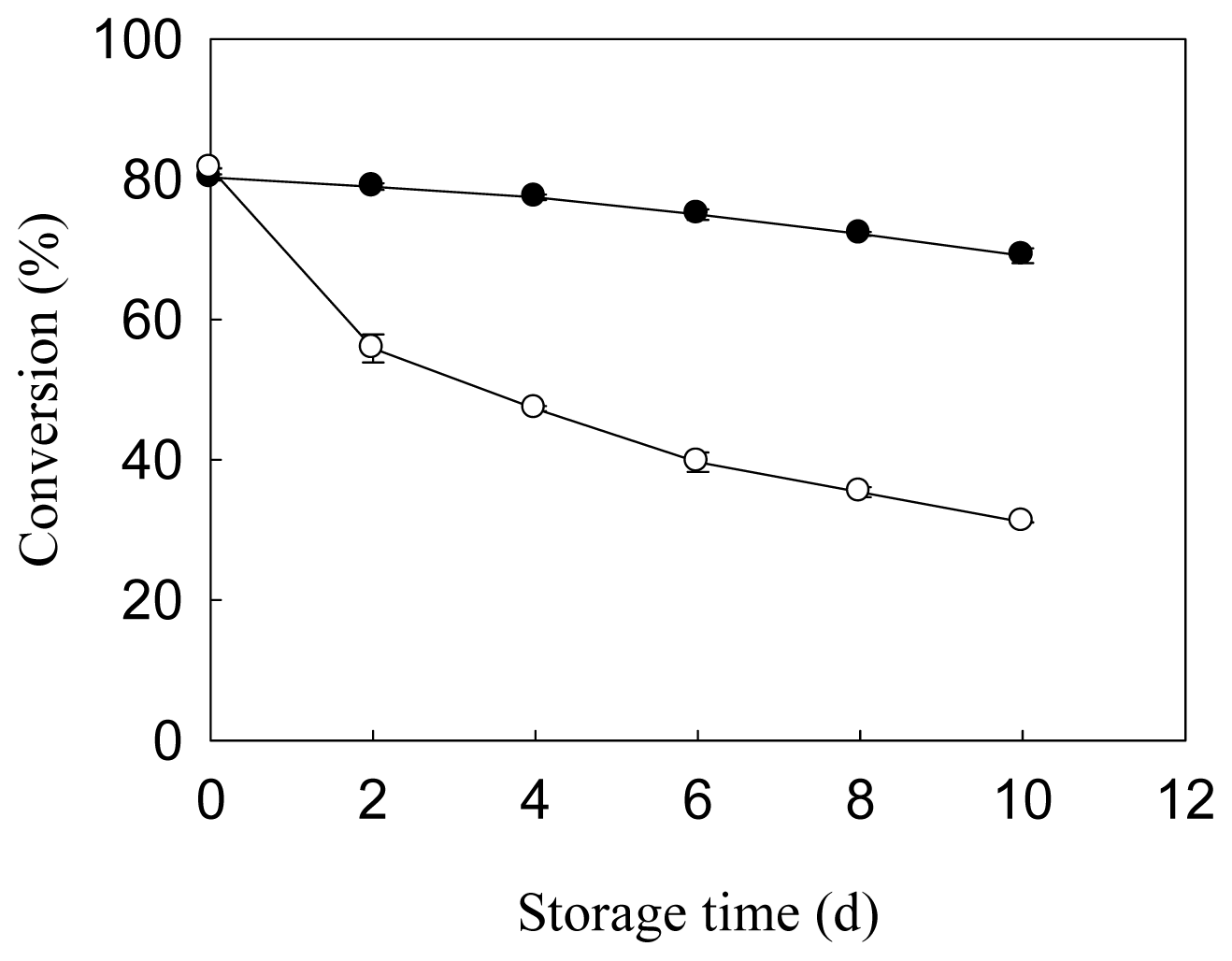
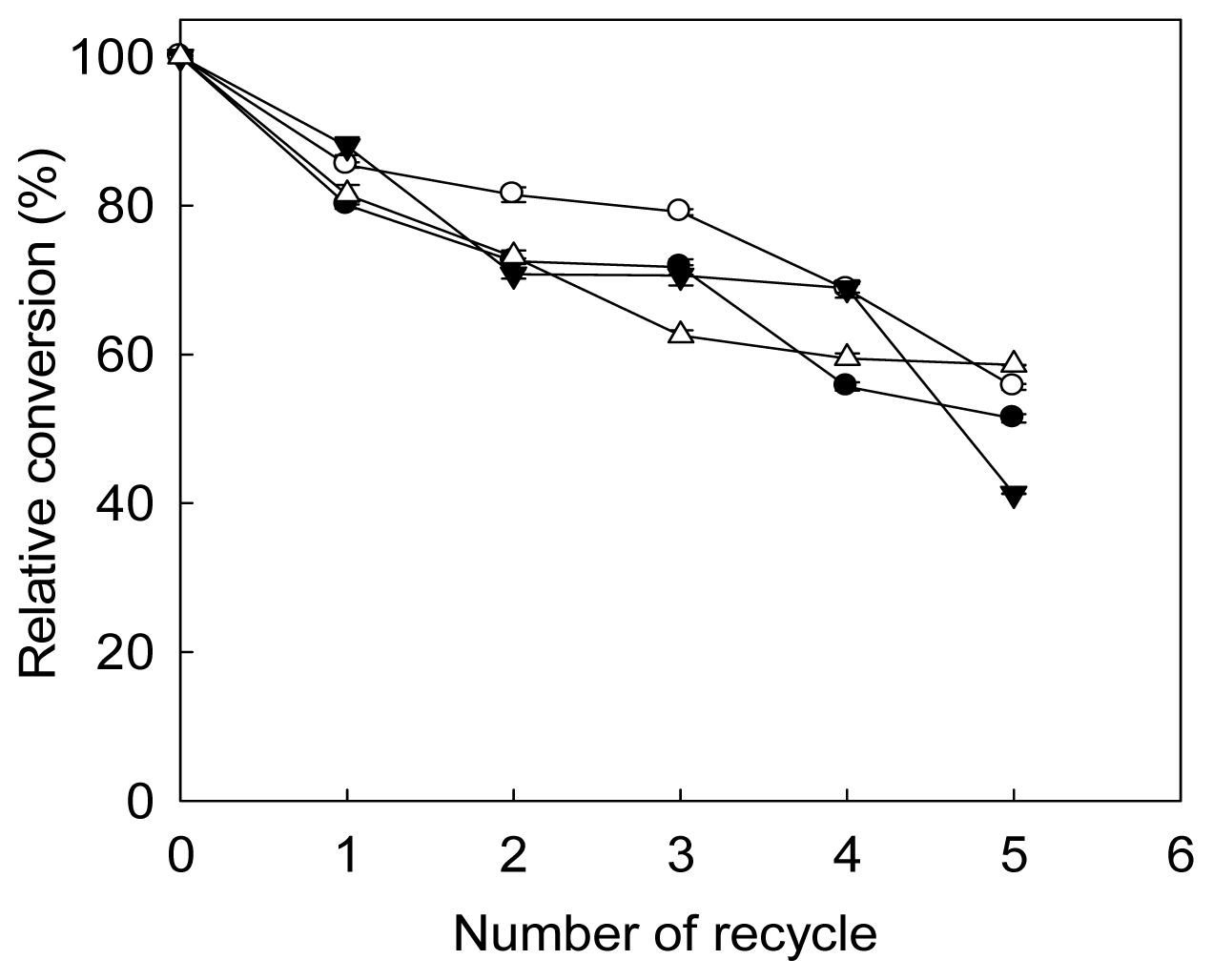
2.5. Storage Stability and Reusability of Immobilized Lipase
3. Experimental Section
3.1. Preparation of MNP
3.2. Immobilization of Lipase
3.3. Assay for Lipase Activity
3.4. Experimental Design
3.5. Transesterification of Oil to Biodiesel
3.6. Analysis of FAME
3.7. Data Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Canakci, M.; Sanli, H. Biodiesel production from various feedstocks and their effects on the fuel properties. J. Ind. Microbiol. Biotechnol 2008, 35, 431–441. [Google Scholar]
- Canakci, M.; Gerpen, J.V. Biodiesel production from oils and fats with high free fatty acids. Trans. ASAE 2001, 44, 1429–1436. [Google Scholar]
- Kulkarni, M.G.; Dalai, A.K. Waste cooking oil-an economical source for biodiesel: A review. Ind. Eng. Chem. Res 2006, 45, 2901–2913. [Google Scholar]
- Escobar, J.C.; Lora, E.S.; Venturini, O.J.; Yáñez, E.E.; Castillo, E.F.; Almazan, O. Biofuels: Environment, technology and food security. Renew. Sustain. Energy Rev 2009, 13, 1275–1287. [Google Scholar]
- Hasan, F.; Shah, A.A.; Hameed, A. Industrial applications of microbial lipases. Enzyme Microbial. Technol 2006, 39, 235–251. [Google Scholar]
- Bisen, P.; Sanodiya, B.; Thakur, G.; Baghel, R.; Prasad, G. Biodiesel production with special emphasis on lipase-catalyzed transesterification. Biotechnol. Lett 2010, 32, 1019–1030. [Google Scholar]
- Jegannathan, K.R.; Abang, S.; Poncelet, D.; Chan, E.S.; Ravindra, P. Production of biodiesel using immobilized lipase—A critical review. Crit. Rev. Biotechnol 2008, 28, 253–264. [Google Scholar]
- Shah, S.; Sharma, S.; Gupta, M.N. Biodiesel preparation by lipase-catalyzed transesterification of jatropha oil. Energy Fuels 2004, 18, 154–159. [Google Scholar]
- Shaw, J.F.; Chang, S.W.; Lin, S.C.; Wu, T.T.; Ju, H.Y.; Akoh, C.C.; Chang, R.H.; Shieh, C.J. Continuous enzymatic synthesis of biodiesel with Novozym 435. Energy Fuels 2008, 22, 840–844. [Google Scholar]
- Oliveira, D.; Oliveira, J.V. Enzymatic alcoholysis of palm kernel oil in n-hexane and SCCO2. J. Supercrit. Fluids 2001, 19, 141–148. [Google Scholar]
- Mittelbach, M. Lipase catalyzed alcoholysis of sunflower oil. J. Am. Oil Chem. Soc 1990, 67, 168–170. [Google Scholar]
- Li, S.-F.; Fan, Y.-H.; Hu, R.-F.; Wu, W.-T. Pseudomonas cepacia lipase immobilized onto the electrospun PAN nanofibrous membranes for biodiesel production from soybean oil. J. Mol. Catal. B 2011, 72, 40–45. [Google Scholar]
- Kumari, V.; Shah, S.; Gupta, M.N. Preparation of biodiesel by lipase-catalyzed transesterification of high free fatty acid containing oil from Madhuca indica. Energy Fuels 2006, 21, 368–372. [Google Scholar]
- Hsu, A.-F.; Jones, K.; Marmer, W.; Foglia, T. Production of alkyl esters from tallow and grease using lipase immobilized in a phyllosilicate sol-gel. J. Am. Oil Chem. Soc 2001, 78, 585–588. [Google Scholar]
- Noureddini, H.; Gao, X.; Philkana, R.S. Immobilized Pseudomonas cepacia lipase for biodiesel fuel production from soybean oil. Bioresour. Technol 2005, 96, 769–777. [Google Scholar]
- Orçaire, O.; Buisson, P.; Pierre, A.C. Application of silica aerogel encapsulated lipases in the synthesis of biodiesel by transesterification reactions. J. Mol. Catal. B 2006, 42, 106–113. [Google Scholar]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar]
- Ito, A.; Shinkai, M.; Honda, H.; Kobayashi, T. Medical application of functionalized magnetic nanoparticles. J. Biosci. Bioeng 2005, 100, 1–11. [Google Scholar]
- Huang, S.-H.; Liao, M.-H.; Chen, D.-H. Direct binding and characterization of lipase onto magnetic nanoparticles. Biotechnol. Prog 2003, 19, 1095–1100. [Google Scholar]
- Mak, K.-H. Immobilization of Lipase from Pseudomonas cepacia onto Magnetic Nanoparticles. Master’s Thesis, Tatung University, Taipei, Taiwan, June 2008. [Google Scholar]
- Mak, K.-H.; Yu, C.-Y.; Kuan, I.-C.; Lee, S.-L. Immobilization of Pseudomonas cepecia Lipase onto Magnetic Nanoparticles for Biodiesel Production. In Progress in Development and Applications of Renewable Energy; Yang, S.-S., Sayigh, A.A.M., Lai, C.-M., Chen, S., Eds.; National Taiwan University: Taipei, Taiwan, 2009; pp. 51–58. [Google Scholar]
- Montgomery, D.C. Response Surface Methods and Designs. In Design and Analysis of Experiments, 6th ed; John Wiley & Sons: Hoboken, NJ, USA, 2005; pp. 405–463. [Google Scholar]
- Kuan, I.-C.; Lee, C.-C.; Tsai, B.-H.; Lee, S.-L.; Lee, W.-T.; Yu, C.-Y. Optimizing the production of biodiesel using lipase entrapped in biomimetic silica. Energies 2013, 6, 2052–2064. [Google Scholar]
- Chen, H.-C.; Ju, H.-Y.; Wu, T.-T.; Liu, Y.-C.; Lee, C.-C.; Chang, C.; Chung, Y.-L.; Shieh, C.-J. Continuous production of lipase-catalyzed biodiesel in a packed-bed reactor: Optimization and enzyme reuse study. J. Biomed. Biotechnol 2011, 2011, 1–6. [Google Scholar]
- Mears, D.E. Tests for transport limitations in experimental catalytic reactors. Ind. Eng. Chem. Process Des. Dev 1971, 10, 541–547. [Google Scholar]
- Mineralogy Database. Available online: http://webmineral.com/ (accessed on 19 November 2013).
- Cussler, E.L. Fundamentals of Mass Transfer. In Diffusion: Mass Transfer in Fluid Systems, 3rd ed; Cambridge University Press: New York, NY, USA, 2009; pp. 237–273. [Google Scholar]
- Wilke, C.R.; Chang, P. Correlation of diffusion coefficients in dilute solutions. AIChE J 1955, 1, 264–270. [Google Scholar]
- Bailey, J.E.; Ollis, D.F. The Kinetics of Enzyme-Catalyzed Reactions. In Biochemical Engineering Fundamentals, 2nd ed; McGraw-Hill Inc: Columbus, OH, USA, 1986; pp. 86–156. [Google Scholar]
- Watanabe, Y.; Shimada, Y.; Sugihara, A.; Tominaga, Y. Enzymatic conversion of waste edible oil to biodiesel fuel in a fixed-bed bioreactor. J. Am. Oil Chem. Soc 2001, 78, 703–707. [Google Scholar]
- Shimada, Y.; Watanabe, Y.; Sugihara, A.; Tominaga, Y. Enzymatic alcoholysis for biodiesel fuel production and application of the reaction to oil processing. J. Mol. Catal. B 2002, 17, 133–142. [Google Scholar]
- Shah, S.; Gupta, M.N. Lipase catalyzed preparation of biodiesel from Jatropha oil in a solvent free system. Process Biochem 2007, 42, 409–414. [Google Scholar]
- Tran, D.-T.; Yeh, K.-L.; Chen, C.-L.; Chang, J.-S. Enzymatic transesterification of microalgal oil from Chlorella vulgaris ESP-31 for biodiesel synthesis using immobilized Burkholderia lipase. Bioresour. Technol 2012, 108, 119–127. [Google Scholar]
- Hsu, A.-F.; Jones, K.; Foglia, T.A.; Marmer, W.N. Immobilized lipase-catalysed production of alkyl esters of restaurant grease as biodiesel. Biotechnol. Appl. Biochem 2002, 36, 181–186. [Google Scholar]
- Chen, J.-W.; Wu, W.-T. Regeneration of immobilized Candida antarctica lipase for transesterification. J. Biosci. Bioeng 2003, 95, 466–469. [Google Scholar]
- Li, L.; Du, W.; Liu, D.; Wang, L.; Li, Z. Lipase-catalyzed transesterification of rapeseed oils for biodiesel production with a novel organic solvent as the reaction medium. J. Mol. Catal. B 2006, 43, 58–62. [Google Scholar]
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of protein using bicinchoninic acid. Anal. Biochem 1985, 150, 76–85. [Google Scholar]
- Pencreac'h, G.; Leullier, M.; Baratti, J.C. Properties of free and immobilized lipase from Pseudomonas cepacia. Biotechnol. Bioeng 1997, 56, 181–189. [Google Scholar]
- Palomo, J.M.; Segura, R.L.; Fernández-Lorente, G.; Pernas, M.; Rua, M.L.; Guisán, J.M.; Fernández-Lafuente, R. Purification, immobilization, and stabilization of a lipase from Bacillus thermocatenulatus by interfacial adsorption on hydrophobic supports. Biotechnol. Prog 2004, 20, 630–635. [Google Scholar]
- Hosseini, M.; Karkhane, A.; Yakhchali, B.; Shamsara, M.; Aminzadeh, S.; Morshedi, D.; Haghbeen, K.; Torktaz, I.; Karimi, E.; Safari, Z. In silico and experimental characterization of chimeric Bacillus thermocatenulatus lipase with the complete conserved pentapeptide of Candida rugosa lipase. Appl. Biochem. Biotechnol 2013, 169, 773–785. [Google Scholar]
| Treatment No. a | Variable b | Conversion (%) | ||
|---|---|---|---|---|
| Temperature (°C) | Molar ratio (methanol/oil) | Water content (%, w/w of oil) | ||
| 1 | 35 (−1) | 3 (−1) | 10.5 (0) | 48 ± 1 |
| 2 | 50 (1) | 3 (−1) | 10.5 (0) | 54.1 ± 0.3 |
| 3 | 35 (−1) | 8 (1) | 10.5 (0) | 42.70 ± 0.01 |
| 4 | 50 (1) | 8 (1) | 10.5 (0) | 52.6 ± 0.4 |
| 5 | 35 (−1) | 5.5 (0) | 1 (−1) | 13.5 ± 0.1 |
| 6 | 50 (1) | 5.5 (0) | 1 (−1) | 8 ± 1 |
| 7 | 35 (−1) | 5.5 (0) | 20 (1) | 44.6 ± 0.9 |
| 8 | 50 (1) | 5.5 (0) | 20 (1) | 58.13 ± 0.06 |
| 9 | 42.5 (0) | 3 (−1) | 1 (−1) | 14.0 ± 0.9 |
| 10 | 42.5 (0) | 8 (1) | 1 (−1) | 5.49 ± 0.02 |
| 11 | 42.5 (0) | 3 (−1) | 20 (1) | 38.2 ± 0.5 |
| 12 | 42.5 (0) | 8 (1) | 20 (1) | 20.0 ± 0.9 |
| 13 | 42.5 (0) | 5.5 (0) | 10.5 (0) | 75.1 ± 0.8 |
| 14 | 42.5 (0) | 5.5 (0) | 10.5 (0) | 76 ± 1 |
| 15 | 42.5 (0) | 5.5 (0) | 10.5 (0) | 79.0 ± 0.2 |
| 16 | 42.5 (0) | 5.5 (0) | 10.5 (0) | 82.1 ± 0.3 |
| 17 | 42.5 (0) | 5.5 (0) | 10.5 (0) | 84.0 ± 0.8 |
| Source | Sum of squares | Degree of freedom | Mean square | F-value | Prob > Fa,b |
|---|---|---|---|---|---|
| Model | 11398.83 | 9 | 1266.537 | 25.92057 | 0.0001 |
| Temperature (A) | 69.32531 | 1 | 69.32531 | 1.418791 | 0.2724 |
| Substrate molar ratio (B) | 142.1298 | 1 | 142.1298 | 2.908786 | 0.1319 |
| Water content (C) | 1799.7 | 1 | 1799.7 | 36.83213 | 0.0005 |
| A2 | 346.665 | 1 | 346.665 | 7.094744 | 0.0323 |
| B2 | 1805.258 | 1 | 1805.258 | 36.94588 | 0.0005 |
| C2 | 6430.1 | 1 | 6430.1 | 131.5965 | <0.0001 |
| AB | 4.2849 | 1 | 4.2849 | 0.087693 | 0.7757 |
| AC | 91.48922 | 1 | 91.48922 | 1.872391 | 0.2135 |
| BC | 23.3289 | 1 | 23.3289 | 0.477442 | 0.5118 |
| Residual | 342.0356 | 7 | 48.86223 | ||
| Lack of fit | 283.2356 | 3 | 94.41188 | 6.422577 | 0.0521 |
| Pure error | 58.8 | 4 | 14.7 | ||
| Cor total | 11740.87 | 16 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yu, C.-Y.; Huang, L.-Y.; Kuan, I.-C.; Lee, S.-L. Optimized Production of Biodiesel from Waste Cooking Oil by Lipase Immobilized on Magnetic Nanoparticles. Int. J. Mol. Sci. 2013, 14, 24074-24086. https://doi.org/10.3390/ijms141224074
Yu C-Y, Huang L-Y, Kuan I-C, Lee S-L. Optimized Production of Biodiesel from Waste Cooking Oil by Lipase Immobilized on Magnetic Nanoparticles. International Journal of Molecular Sciences. 2013; 14(12):24074-24086. https://doi.org/10.3390/ijms141224074
Chicago/Turabian StyleYu, Chi-Yang, Liang-Yu Huang, I-Ching Kuan, and Shiow-Ling Lee. 2013. "Optimized Production of Biodiesel from Waste Cooking Oil by Lipase Immobilized on Magnetic Nanoparticles" International Journal of Molecular Sciences 14, no. 12: 24074-24086. https://doi.org/10.3390/ijms141224074




