Mutation in the pssA Gene Involved in Exopolysaccharide Synthesis Leads to Several Physiological and Symbiotic Defects in Rhizobium leguminosarum bv. trifolii
Abstract
:1. Introduction
2. Results and Discussion
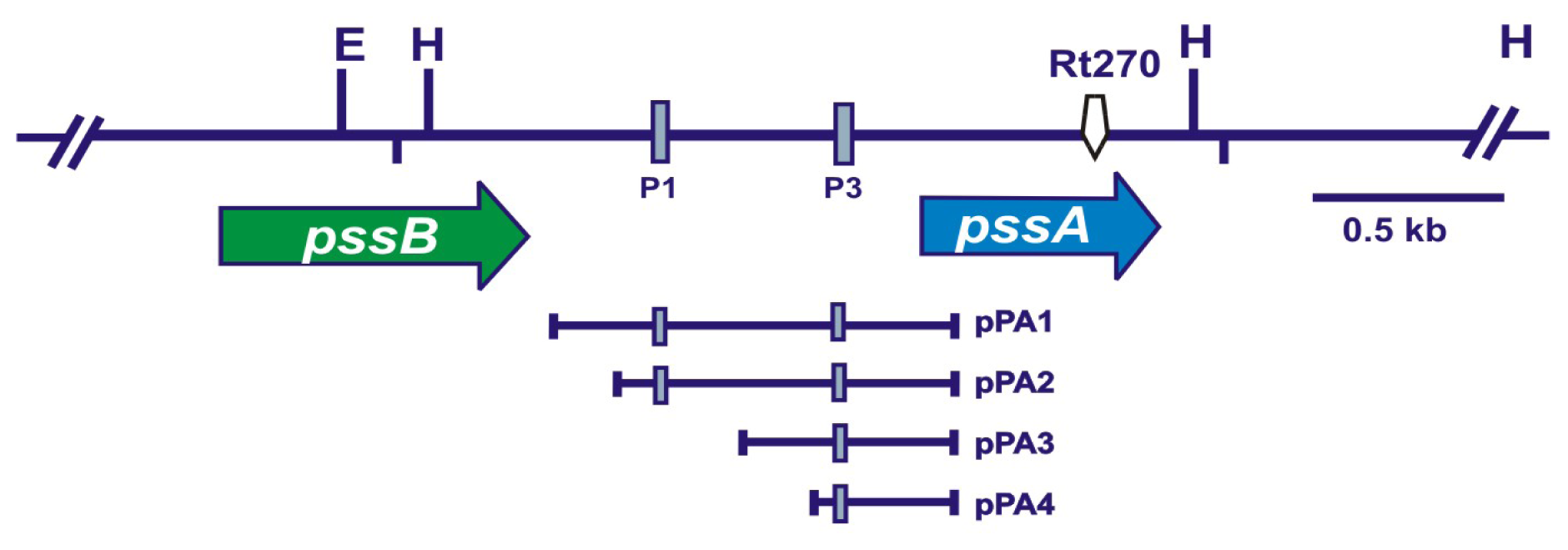
2.1. Mutagenesis of the 3′-End of pssA and the Influence of This Mutation on Exopolysaccharide Production and Symbiosis with Clover
2.2. Mutation in pssA Affects Bacterial Motility
2.3. Phenotype Analysis of the pssA Mutant Using Biolog Tests
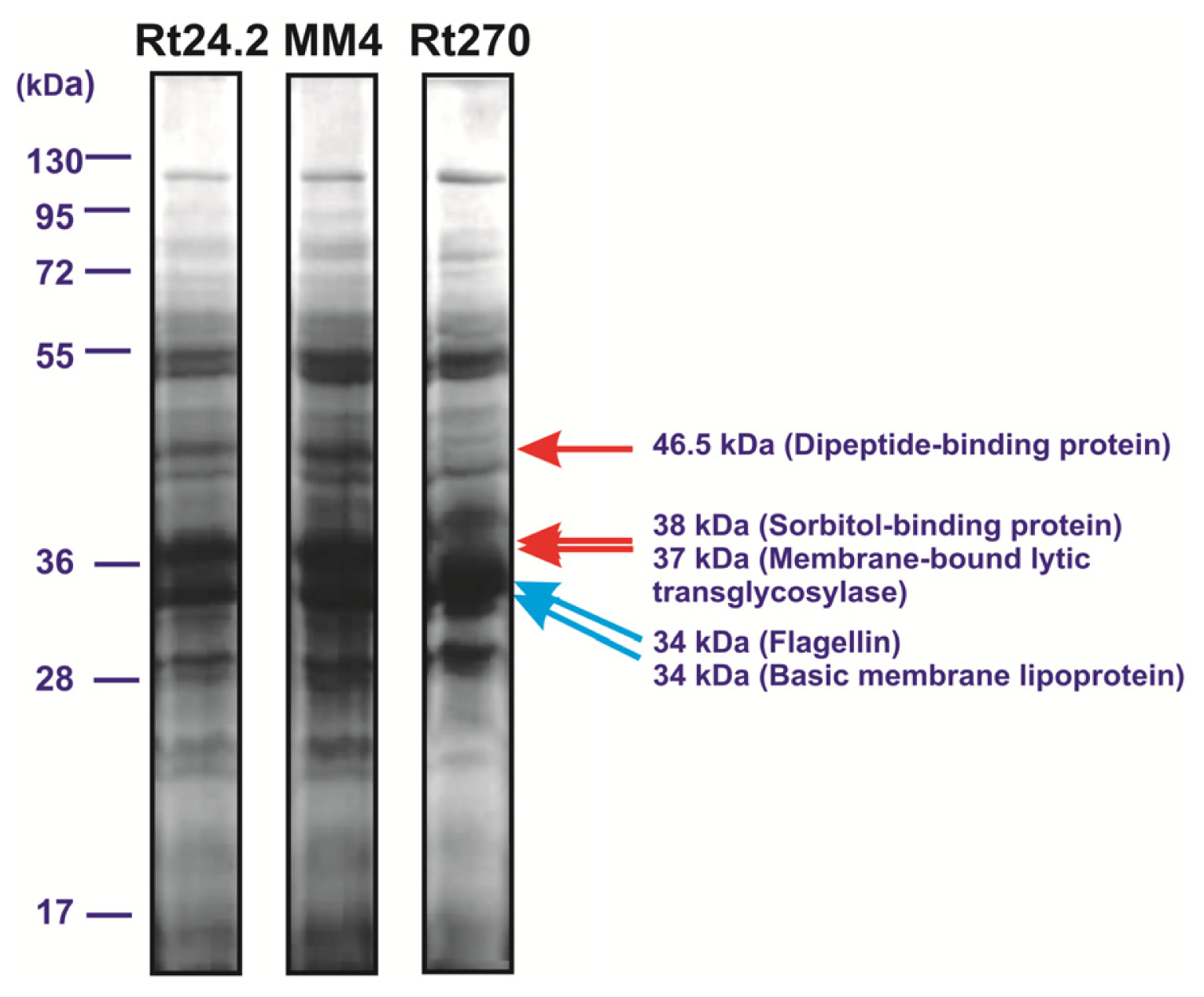
2.4. The Influence of pssA Mutation on the Profile of Extracellular Proteins
2.5. Sensitivity of the Wild-Type and pssA Mutant Strains to Detergents, Ethanol and Antibiotics
2.6. ExoR Negatively Affects the Expression of the pssA Gene and Exopolysaccharide Production
3. Experimental Section
3.1. Bacterial Strains, Plasmids and Growth Conditions
3.2. DNA Methods
3.3. Mutagenesis of the pssA Gene
3.4. Cloning of the exoR Gene into the pBBR1MCS-5 Plasmid
3.5. The Influence of Multiple exoR Copies on pssA Expression and EPS Production
3.6. β-Galactosidase Assay
3.7. β-Glucuronidase Assay
3.8. EPS Isolation and Quantification
3.9. Phenotype Analysis of the pssA Mutant Rt270 Using PM (Biolog) Test
3.10. Motility Assay
3.11. Isolation and Analysis of Extracellular Proteins
3.12. Plant Experiments
3.13. Gus Histochemistry
3.14. Assays for Sensitivity to Antibiotics and Stress Factors
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Spaink, H.P. Root nodulation and infection factors produced by rhizobial bacteria. Annu. Rev. Microbiol 2000, 54, 257–288. [Google Scholar]
- Gibson, K.E.; Kobayashi, H.; Walker, G.C. Molecular determinants of a symbiotic chronic infection. Annu. Rev. Genet 2008, 42, 413–441. [Google Scholar]
- van Workum, W.A.T.; van Slageren, S.; van Brussel, A.A.N.; Kijne, J.W. Role of exopolysaccharides of Rhizobium leguminosarum bv. viciae as host plant-specific molecules required for infection thread formation during nodulation of Vicia sativa. Mol. Plant Microbe Interact 1998, 11, 1233–1241. [Google Scholar]
- Cheng, H.P.; Walker, G.C. Succinoglycan is required for initiation and elongation of infection threads during nodulation of alfalfa by Rhizobium meliloti. J. Bacteriol 1998, 180, 5183–5191. [Google Scholar]
- Janczarek, M. Environmental signals and regulatory pathways that influence exopolysaccharide production in rhizobia. Int. J. Mol. Sci 2011, 12, 7898–7933. [Google Scholar]
- Ivashina, T.V.; Khmelnitsky, M.I.; Shlyapnikov, M.G.; Kanapin, A.A.; Ksenzenko, V.N. The pss4 gene from Rhizobium leguminosarum biovar viciae VF39: Cloning, sequence and the possible role in polysaccharide production and nodule formation. Gene 1994, 50, 111–116. [Google Scholar]
- Janczarek, M.; Jaroszuk-Œciseł, J.; Skorupska, A. Multiple copies of rosR and pssA genes enhance exopolysaccharide production, symbiotic competitiveness and clover nodulation in Rhizobium leguminosarum bv. trifolii. Antonie Van Leeuwenhoek 2009, 96, 471–486. [Google Scholar]
- Borthakur, D.; Barker, R.F.; Latchford, J.W.; Rossen, L.; Johnston, A.W.B. Analysis of pss genes of Rhizobium leguminosarum required for exopolysaccharide synthesis and nodulation of peas: their primary structure and their interaction with psi and other nodulation genes. Mol. Gen. Genet 1988, 213, 155–162. [Google Scholar]
- Ivashina, T.; Ksenzenko, V.N. Exopolysaccharide Biosynthesis in Rhizobium leguminosarum: From Genes to Functions. In The Complex World of Polysaccharides; InTech: Rijeka, Croatia, 2012; pp. 99–127. [Google Scholar]
- Pollock, T.J.; Workum, W.A.; Thorne, L.; Mikolajczak, M.J.; Yamazaki, M.; Kijne, J.W.; Armentrout, R.W. Assignment of biochemical functions to glycosyl transferase genes which are essential for biosynthesis of exopolysaccharides in Sphingomonas strain S88 and Rhizobium leguminosarum. J. Bacteriol 1998, 180, 586–593. [Google Scholar]
- Ksenzenko, V.N.; Ivashina, T.V.; Dubeĭkovskaia, Z.A.; Ivanov, S.G.; Nanazashvili, M.B.; Druzhinina, T.N.; Kalinchuk, N.A.; Shibaev, V.N. The pssA gene encodes UDP-glucose: Polyprenyl phosphate-glucosyl phosphotransferase initiating biosynthesis of Rhizobium leguminosarum exopolysaccharide Russ. J. Bioorg. Chem 2007, 33, 150–155. [Google Scholar]
- Król, J.E.; Mazur, A.; Marczak, M.; Skorupska, A. Syntenic arrangements of the surface polysaccharide biosynthesis genes in Rhizobium leguminosarum. Genomics 2007, 89, 237–247. [Google Scholar]
- Ivashina, T.V.; Fedorova, E.E.; Ashina, N.P.; Kalinchuk, N.A.; Druzhinina, T.N.; Shashkov, A.S.; Shibaev, V.N.; Ksenzenko, V.N. Mutation in the pssM gene encoding ketal pyruvate transferase leads to disruption of Rhizobium leguminosarum bv. viciae-Pisum sativum symbiosis. J. Appl. Microbiol 2010, 109, 731–742. [Google Scholar]
- Latchford, J.W.; Borthakur, D.; Johnston, A.W.B. The products of Rhizobium genes, psi and pss, which affect exopolysaccharide production, are associated with the bacterial cell surface. Mol. Microbiol 1991, 5, 2107–2114. [Google Scholar]
- Mimmack, M.L.; Borthakur, D.; Jones, M.A.; Downie, J.A.; Johnston, A.W.B. The psi operon of Rhizobium leguminosarum biovar phaseoli: Identification of two genes whose products are located at the bacterial cell surface. Microbiology 1994, 140, 1223–1229. [Google Scholar]
- Mimmack, M.L.; Hong, G.F.; Johnston, A.W.B. Sequence and regulation of psrA, a gene on the Sym plasmid of Rhizobium leguminosarum biovar phaseoli which inhibits transcription of the psi genes. Microbiology 1994, 140, 455–461. [Google Scholar]
- Reeve, W.G.; Dilworth, M.J.; Tiwari, R.P.; Glenn, A.R. Regulation of exopolysaccharide production in Rhizobium leguminosarum biovar viciae WSM710 involves exoR. Microbiology 1997, 143, 1951–1958. [Google Scholar]
- Ivashina, T.; Sadykov, M.P.; Thatujev, B.M.; Kanapin, A.A.; Shlapnikov, M.G.; Ksenzenko, W. Продукт гена pssB Rhizobium leguminosarum bv. viciae VF39 участвует в симбиот ической фиксации молекулярного азота. Докла∂ы Ака∂емuu Наук 1996, 350, 712–715. (in Russian). [Google Scholar]
- Janczarek, M.; Skorupska, A. The Rhizobium leguminosarum bv. trifolii pssB gene product is an inositol monophosphatase that influences exopolysaccharide synthesis. Arch. Microbiol 2001, 175, 143–151. [Google Scholar]
- Janczarek, M.; Skorupska, A. The Rhizobium leguminosarum bv. trifolii RosR: Transcriptional regulator involved in exopolysaccharide production. Mol. Plant Microbe Interact 2007, 20, 867–881. [Google Scholar]
- Janczarek, M.; Kutkowska, J.; Piersiak, T.; Skorupska, A. Rhizobium leguminosarum bv. trifolii rosR is required for interaction with clover, biofilm formation and adaptation to the environment. BMC Microbiol 2010, 10, 284. [Google Scholar]
- Borthakur, D.; Johnston, A.W.B. Sequence of psi, a gene of the symbiotic plasmid of Rhizobium phaseoli which inhibits exopolysaccharide synthesis and nodulation and demonstration that its transcription is inhibited by psr, another gene on the symbiotic plasmid. Mol. Gen. Genet 1987, 207, 149–154. [Google Scholar]
- Janczarek, M.; Kalita, M.; Skorupska, A. New taxonomic markers for identification of Rhizobium leguminosarum and discrimination between closely related species. Arch. Microbiol 2009, 191, 207–219. [Google Scholar]
- Janczarek, M.; Skorupska, A. Exopolysaccharide synthesis in R. leguminosarum bv. trifolii is related to various metabolic pathway. Res. Microbiol 2003, 154, 433–442. [Google Scholar]
- Van Workum, W.A.; Canter Cremers, H.C.J.; Wijfjes, A.H.M.; van der Kolk, C.; Wijffelman, C.A.; Kijne, J.W. Cloning and characterization of four genes of Rhizobium leguminosarum bv. trifolii involved in exopolysaccharide production and nodulation. Mol. Plant Microbe Interact 1997, 10, 290–301. [Google Scholar]
- Janczarek, M.; Urbanik-Sypniewska, T. Expression of the Rhizobium leguminosarum bv. trifolii pssA gene involved in exopolysaccharide synthesis is regulated by RosR, phosphate and the carbon source. J. Bacteriol 2013, 195, 3412–3423. [Google Scholar]
- Rolfe, B.G.; Carlson, R.W.; Ridge, R.W.; Dazzo, R.W.; Mateos, F.B.; Pankhurst, C.E. Defective infection and nodulation of clovers by exopolysaccharide mutants of Rhizobium leguminosarum bv. trifolii. Aust. J. Plant Physiol. 1996, 23, 285–303. [Google Scholar]
- Daniels, R.; Reynaert, S.; Hoekstra, H.; Verreth, C.; Janssens, J.; Braeken, K.; Fauvart, M.; Beullens, S.; Heusdens, C.; Lambrichts, I.; et al. Quorum signal molecules as biosurfactants affecting swarming in Rhizobium etli. Proc. Natl. Acad. Sci. USA 2006, 103, 14965–14970. [Google Scholar]
- Kohler, T.; Curty, L.K.; Barja, F.; van Delden, C.; Pechere, J.C. Swarming of Pseudomonas aeruginosa is dependent on cell-to-cell signaling and requires flagella and pili. J. Bacteriol 2000, 182, 5990–5996. [Google Scholar]
- Soto, M.J.; Fernández-Pascual, M.; Sanjuán, J.; Olivares, J. A fadD mutant of Sinorhizobium meliloti shows multicellular swarming migration and is impaired in nodulation efficiency on alfalfa roots. Mol. Microbiol 2002, 43, 371–382. [Google Scholar]
- Sule, S.; Cursino, L.; Zheng, D.; Hoch, H.C.; Burr, T.J. Surface motility and associated surfactant production in Agrobacterium vitis. Lett. Appl. Microbiol 2009, 49, 596–601. [Google Scholar]
- Allison, C.; Emody, L.; Coleman, N.; Hughes, C. The role of swarm cell differentiation and multicellular migration in the uropathogenicity of Proteus mirabilis. J. Infect. Dis 1994, 169, 1155–1158. [Google Scholar]
- Sánchez-Contreras, M.; Martin, M.; Villacieros, M.; O’Gara, F.; Bonilla, I.; Rivilla, R. Phenotypic selection and phase variation occur during alfalfa root colonization by Pseudomonas fluorescens F113. J. Bacteriol 2002, 184, 1587–1596. [Google Scholar]
- Tambalo, D.D.; Yost, C.K.; Hynes, M.F. Characterization of swarming motility in Rhizobium leguminosarum bv. viciae. FEMS Microbiol. Lett. 2010, 307, 165–174. [Google Scholar]
- Tambalo, D.D.; Del Bel, K.L.; Bustard, D.E.; Greenwood, P.R.; Steedman, A.E.; Hynes, M.F. Regulation of flagellar, motility and chemotaxis genes in Rhizobium leguminosarum by the VisN/R-Rem cascade. Microbiology 2010, 156, 1673–1685. [Google Scholar]
- Sourjik, V.; Muschler, P.; Scharf, B.; Schmitt, R. VisN and VisR are global regulators of chemotaxis, flagellar, and motility genes in Sinorhizobium (Rhizobium) meliloti. J. Bacteriol. 2000, 182, 782–788. [Google Scholar]
- Rotter, C.; Muhlbacher, S.; Salamon, D.; Schmitt, R.; Scharf, B. Rem, a new transcriptional activator of motility and chemotaxis in Sinorhizobium meliloti. J. Bacteriol 2006, 188, 6932–6942. [Google Scholar]
- Gurich, N.; González, J.E. Role of quorum sensing in Sinorhizobium meliloti-alfalfa symbiosis. J. Bacteriol 2009, 191, 4372–4382. [Google Scholar]
- Vanderlinde, E.M.; Yost, C.K. Mutation of the sensor kinase chvG in Rhizobium leguminosarum negatively impacts cellular metabolism, outer membrane stability and symbiosis. J. Bacteriol 2012, 181, 768–777. [Google Scholar]
- Vanderlinde, E.M.; Muszyński, A.; Harrison, J.J.; Koval, S.F.; Foreman, D.L.; Ceri, H.; Kannenberg, E.L.; Carlson, R.W.; Yost, C.K. Rhizobium leguminosarum biovar viciae 3841, deficient in 27-hydroxyoctacosanoate-modified lipopolysaccharide, is impaired in desiccation tolerance, biofilm formation and motility. Microbiology 2009, 155, 3055–3069. [Google Scholar]
- Guerreiro, N.; Ksenzenko, V.; Djordjevic, M.A.; Ivashina, T.V.; Rolfe, B. Elevated levels of synthesis of over 20 proteins results after mutation of the Rhizobium leguminosarum exopolysaccharide synthesis gene pssA. J. Bacteriol. 2000, 182, 4521–4532. [Google Scholar]
- Bélanger, L.; Dimmick, K.A.; Fleming, J.S.; Charles, T.C. Null mutations in Sinorhizobium meliloti exoS and chvI demonstrate the importance of this two-component regulatory system for symbiosis. Mol. Microbiol 2009, 74, 1223–1237. [Google Scholar]
- Wang, C.; Kemp, J.; Da Fonseca, I.O.; Equi, R.C.; Sheng, X.; Charles, T.C.; Sobral, B.W.S. Sinorhizobium meliloti 1021 loss-of-function deletion mutation in chvI and its phenotypic characteristics. Mol. Plant Microbe Interact 2010, 23, 153–160. [Google Scholar]
- Krehenbrink, M.; Downie, J.A. Identification of protein secretion systems and novel secreted proteins in Rhizobium leguminosarum bv. viciae. BMC Genomics 2008, 9, 55. [Google Scholar]
- Finnie, C.; Hartley, N.M.; Findlay, K.C.; Downie, J.A. The Rhizobium leguminosarum prsDE genes are required for secretion of several proteins, some of which influence nodulation, symbiotic nitrogen fixation and exopolysaccharide modification. Mol. Microbiol 1997, 25, 135–146. [Google Scholar]
- Downie, J.A. The roles of extracellular proteins, polysaccharides and signals in the interactions of rhizobia with legume roots. FEMS Microbiol. Rev 2010, 34, 150–170. [Google Scholar]
- Dinh, T.; Paulsen, I.T.; Saier, M.H., Jr. A family of extracytoplasmic proteins that allow transport of large molecules across the outer membranes of gram-negative bacteria. J. Bacteriol 1994, 176, 3825–3831. [Google Scholar]
- Wielbo, J.; Mazur, A.; Król, J.; Marczak, M.; Kutkowska, J.; Skorupska, A. Complexity of phenotypes and symbiotic behaviour of Rhizobium leguminosarum biovar trifolii exopolysaccharide mutants. Arch. Microbiol 2004, 182, 331–336. [Google Scholar]
- Foreman, D.L.; Vanderlinde, E.M.; Bay, D.C.; Yost, C.K. Characterization of a gene family of outer membrane proteins (ropB) in Rhizobium leguminosarum bv. viciae VF39SM and the role of the sensor kinase ChvG in their regulation. J. Bacteriol 2010, 192, 975–983. [Google Scholar]
- Nikaido, H. Molecular basis of bacterial outer membrane permeability revisited. Microbiol. Mol. Biol. Rev 2003, 67, 593–656. [Google Scholar]
- Reed, J.W.; Glazebrook, J.; Walker, G.C. The exoR gene of Rhizobium meliloti affect RNA levels of other exo genes but lacks homology to known transcriptional regulators. J. Bacteriol 1991, 173, 3789–3794. [Google Scholar]
- Quester, I.; Becker, A. Four promoters subject to regulation by ExoR and PhoB direct transcription of the Sinorhizobium meliloti exoYFQ operon involved in the biosynthesis of succinoglycan. J. Mol. Microbiol. Biotechnol 2004, 7, 115–132. [Google Scholar]
- Lu, H.Y.; Cheng, H.P. Autoregulation of Sinorhizobium meliloti exoR gene expression. Microbiology 2010, 156, 2092–2101. [Google Scholar]
- Janczarek, M.; Skorupska, A. Regulation of pssA and pssB gene expression in R. leguminosarum bv. trifolii in response to environmental factors. Antonie Van Leeuwenhoek 2004, 85, 217–227. [Google Scholar]
- Yao, S.Y.; Luo, L.; Har, K.J.; Becker, A.; Rüberg, S.; Yu, G.Q.; Zhu, J.B.; Cheng, H.P. Sinorhizobium meliloti ExoR and ExoS proteins regulate both succinoglycan and flagellum production. J. Bacteriol 2004, 186, 6042–6049. [Google Scholar]
- Chen, E.J.; Sabio, E.A.; Long, S.R. The periplasmic regulator ExoR inhibits ExoS/ChvI two-component signalling in Sinorhizobium meliloti. Mol. Microbiol 2008, 69, 1290–1303. [Google Scholar]
- Doherty, D.; Leigh, J.A.; Glazebrook, J.; Walker, G.C. Rhizobium meliloti mutants that overproduce the R. meliloti acidic calcofluor-binding exopolysaccharide. J. Bacteriol 1988, 170, 4249–4256. [Google Scholar]
- Ozga, D.A.; Lara, J.C.; Leigh, J.A. The regulation of exopolysaccharide production is important at two levels of nodule development in Rhizobium meliloti. Mol. Plant Microbe Interact 1994, 7, 758–765. [Google Scholar]
- Vincent, J.M. A manual for the Practical Study of Root Nodule Bacteria. In International Biological Program Handbook no. 15; Blackwell Scientific Publications Ltd.: Oxford, UK, 1970. [Google Scholar]
- Sambrook, J.; Fitsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1989. [Google Scholar]
- Brown, C.M.; Dilworth, M.J. Ammonia assimilation by Rhizobium cultures and bacteroids. J. Gen. Microbiol 1975, 86, 39–48. [Google Scholar]
- Wilson, K.J.; Sessitsch, A.; Corbo, J.C.C.; Giller, K.E.; Akkerman, A.D.L.; Jefferson, R.A. Glucuronidase (GUS) transposons for ecological and genetic studies of rhizobia and other Gram-negative bacteria. Microbiology 1995, 141, 1691–1705. [Google Scholar]
- Simon, R.; Priefer, U.; Pühler, A. A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in gram-negative bacteria. Nat. BioTechnol 1983, 1, 784–791. [Google Scholar]
- Kovach, M.E.; Elzer, P.H.; Hill, D.S.; Robertson, G.T.; Farris, M.A.; Roop, R.M.; Peterson, K.M. Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene 1995, 166, 175–176. [Google Scholar]
- Wielbo, J.; Skorupska, A. Construction of improved vectors and cassettes containing gusA and antibiotic resistance genes for studies of transcriptional activity and bacterial localization. J. Microbiol. Meth 2001, 45, 197–205. [Google Scholar]
- Reeve, W.G.; Tiwari, R.P.; Worsley, P.S.; Dilworth, M.J.; Glenn, A.R.; Howieson, J.G. Constructs for insertional mutagenesis, transcriptional signal localization and gene regulation studies in root nodule and other bacteria. Microbiology 1999, 145, 1307–1316. [Google Scholar]
- Altschul, S.; Gish, W.; Miller, W.; Myers, E.; Lipman, D. Basic local alignment search tool. J. Mol. Biol 1990, 215, 403–410. [Google Scholar]
- Young, J.P.W.; Crossman, L.C.; Johnston, A.W.B.; Thomson, N.R.; Ghazoui, Z.F.; Hull, K.H.; Wexler, M.; Curson, A.R.; Todd, J.D.; Poole, P.S.; et al. The genome of Rhizobium leguminosarum has recognizable core and accessory components. Genome Biol 2006, 7, R34. [Google Scholar]
- Miller, J.H. Experiments in Molecular Genetics; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1972. [Google Scholar]
- Yost, C.K.; Del Bel, K.L.; Quandt, J.; Hynes, M.F. Rhizobium leguminosarum methyl-accepting chemotaxis protein genes are down-regulated in the pea nodule. Arch. Microbiol 2004, 182, 505–513. [Google Scholar]
- Loewus, F.A. Improvement in the anthrone method for determination of carbohydrates. Anal. Chem 1952, 24, 219. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantification of microgram quanties of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- Jaszek, M.; Janczarek, M.; Kuczyński, K.; Piersiak, T.; Grzywnowicz, K. The response of the Rhizobium leguminosarum bv. trifolii wild-type and exopolysaccharide-deficient mutants to oxidative stress. Plant Soil 2013. [Google Scholar] [CrossRef]



| Strain | Migration distance (mm) a | |||||
|---|---|---|---|---|---|---|
| M1 | 79CA | TY | ||||
| 0.3% | 0.7% | 0.3% | 0.7% | 0.3% | 0.7% | |
| Rt24.2 (wild-type) | 9.5 ± 1.0 | 3.5 ± 0.5 | 19 ± 2.0 | 6.5 ± 1.0 | 15 ± 2.0 | 4.0 ± 0.5 |
| Rt270 (pssA) | 4.5 ± 0.5 * | 2 ± 0.5 * | 11 ± 1.0 * | 1.5 ± 0.5 * | 7.0 ± 0.5 * | 1.5 ± 0.5 * |
| Fusion | Promoter activity in 79CA medium * | Promoter activity in M1 medium * | ||||
|---|---|---|---|---|---|---|
| Rt24.2 (wild-type) | Rt270 (pssA mutant) | Ratio Rt270/Rt24.2 | Rt24.2 (wild-type) | Rt270 (pssA mutant) | Ratio Rt270/Rt24.2 | |
| pVNVP (visN-gusA) | 5496 ± 393 a | 3021 ± 251 b | 0.55 | 3297 ± 225 b | 2326 ± 187 c | 0.7 |
| pSVP SUM (rem-gusA) | 12707 ± 689 a | 8015 ± 421 b | 0.63 | 8640 ± 496 b | 6493 ± 422 c | 0.75 |
| pAVP (flaA-gusA) | 8804 ± 498 a | 5746 ± 339 c | 0.65 | 6855 ± 376 b | 3512 ± 198 d | 0.51 |
| pFus1par (control) | 131 ± 16 a | 126 ± 11 a | 0.96 | 110 ± 12 a | 104 ± 15 a | 0.94 |
| Strain | Minimal inhibitory concentration * | |||
|---|---|---|---|---|
| SDS (% w/v) | DOC (% w/v) | Sarcosyl (% w/v) | Ethanol (% v/v) | |
| Rt24.2 (wt) | 0.025 ± 0.005 a | 0.11 ± 0.005 a | 0.05 ± 0.005 a | 5.5 ± 0.25 a |
| Rt270 (pssA) | 0.020 ± 0.005 a | 0.07 ± 0.005 c | 0.06 ± 0.005 a | 3.5 ± 0.25 b |
| MM3 (pssD) | 0.015 ± 0.005 a | 0.09 ± 0.005 b | 0.05 ± 0.005 a | 5.0 ± 0.25 a |
| MM4(pssJIHGF) | 0.015 ± 0.005 a | 0.085 ± 0.005 b | 0.045 ± 0.005 a | 5.0 ± 0.25 a |
| Antibiotics | Growth inhibition zone (mm) * | |||
|---|---|---|---|---|
| Rt24.2 (wt) | Rt270 (pssA) | MM3 (pssD) | MM4 (pssJIHGF) | |
| Gentamicin | 28 ± 3 b | 30 ± 2 b | 36 ± 3 a | 37 ± 3 a |
| Tetracycline | 52 ± 4 a | 49 ± 3 a | 58 ± 3 a | 54 ± 3 a |
| Chloramphenicol | 54 ± 4 a | 44 ± 3 b | 58 ± 2 a | 56 ± 4 a |
| Erythromycin | 18 ± 2 a | 12 ± 2 b | 16 ± 2 a | 17 ± 2 a |
| Neomycin | 21 ± 3 a | 11 ± 2 b | 24 ± 2 a | 26 ± 3 a |
| Ampicillin | 12 ± 2 b | 0 ± 0 c | 23 ± 2 a | 27 ± 2 a |
| Penicillin G | 6 ± 1 a | 0 ± 0 b | 7 ± 1 a | 6 ± 2 a |
| Amoxicillin | 11 ± 2 b | 0 ± 0 c | 16 ± 2 a | 17 ± 2 a |
| Bacitracin | 13 ± 2 a | 0 ± 0 b | 10 ± 3 a | 9 ± 3 a |
| Oxocillin | 10 ± 2 a | 0 ± 0 b | 7 ± 1 a | 6 ± 2 a |
| Polimyxin B | 10 ± 1 a | 0 ± 0 b | 11 ± 2 a | 12 ± 2 a |
| Type of pssA-lacZ fusion | β-galactosidase activity at different NH4Cl concentration (Miller units) * | |||||
|---|---|---|---|---|---|---|
| Rt24.2 (wild-type) | Rt24.2pRR1 (exoR) | Rt24.2 (pBBR1MCS-5) | ||||
| 0.1 mM | 10 mM | 0.1 mM | 10 mM | 0.1 mM | 10 mM | |
| pPA1 | 4954 ± 389 a | 4804 ± 352 a | 1676 ± 148 c | 2286 ± 186 b | 4869 ± 363 a | 4617 ± 327 a |
| pPA2 | 3773 ± 293 a | 3034 ± 276 b | 2029 ± 178 c | 2318 ± 201 c | 3702 ± 311 a | 2966 ± 284 b |
| pPA3 | 1878 ± 143 a | 1545 ± 131 b | 944 ± 89 c | 1329 ± 124 b | 1821 ± 139 a | 1457 ± 126 b |
| pPA4 | 1540 ± 121 a | 1389 ± 120 a | 827 ± 76 c | 1103 ± 96 b | 1498 ± 132 a | 1317 ± 115 a |
| Strains and plasmids | Relevant characteristics | Sources or reference |
|---|---|---|
| R. leguminosarum | ||
| Rt24.2 | Wild type, Rifr, Nxr | [20] |
| Rt270 | Rt24.2 with a mini-Tn5 transposon insertion in 412/413 bp position of pssA coding region, Kmr | This work |
| MM4 | Rt24.2 with mTn5SSgusA40 transposon in the EPS I region (pssJIHGF), Spr | This work |
| MM3 E. coli | Rt24.2 with mTn5SSgusA40 transposon, pssD, Spr | This work |
| DH5α | supE44ΔlacU169(ϕ80lacZΔM15)hsdR17 recA1endA1gyrA96 thi-1 relA1 | [60] |
| S17-1 | 294, thi, RP4-2-Tc::Mu-Km::Tn7 | [63] |
| Plasmids | ||
| pUC19 | Cloning and sequencing vector, Apr | [60] |
| pSUP202 | pBR325 derivative, mob, Cmr, Tcr, Apr | [63] |
| pBBR1MCS-5 | mob, lacZα, Gmr cloning vector | [64] |
| pM34 | pUC19 containing 3.4-kb EcoRI fragment with pssA | [8] |
| pMT27 | pUC19 containing 3.4-kb EcoRI fragment with mini-Tn5 in position 412/413 bp of pssA coding region | This work |
| pMSUP27 | pSUP202 containing 4.6-kb EcoRI fragment with mini-Tn5 inserted in 412/413 bp of pssA ORF | This work |
| pPA1 | pMP220 carrying the −750 bp to +152 bp fragment of the pssA regulatory region, Tcr | [26] |
| pPA2 | pMP220 carrying the −538 bp to +152 bp fragment of the pssA regulatory region, Tcr | [26] |
| pPA3 | pMP220 carrying the −374 bp to +152 bp fragment of the pssA regulatory region, Tcr | [26] |
| pPA4 | pMP220 carrying the −284 bp to +152 bp fragment of the pssA regulatory region, Tcr | [26] |
| pJBA21Tc | pJB321 derivative carrying constitutively expressed gusA, Tcr | [65] |
| pEXOR13 | pUC19 containing 1.3 kb EcoRI fragment with Rt24.2 exoR gene | This work |
| pRR1 | pBBR1MCS-5 containing 1.3 kb EcoRI fragment with Rt24.2 exoR gene | This work |
| pFUS1par | pMP220 derivative with promoterless gusA, par, Tcr | [66] |
| pAVP | pFUS1, flaA::gusA, Tcr | Michael Hynes, [40] |
| pVNVP | pFUS1P, visN::gusA, Tcr, par stabilized | Michael Hynes, [40] |
| pSVP SUM | pFUS1P, rem::gusA, Tcr, par stabilized | Michael Hynes, [40] |
| Primers (5′-3′) | ||
| pExoR1 | CGTTTGAATTCGGTCGTTTCGCTT | This work |
| pExoR3 | GAACAACGGAATTCGCATCGACCA | This work |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Janczarek, M.; Rachwał, K. Mutation in the pssA Gene Involved in Exopolysaccharide Synthesis Leads to Several Physiological and Symbiotic Defects in Rhizobium leguminosarum bv. trifolii. Int. J. Mol. Sci. 2013, 14, 23711-23735. https://doi.org/10.3390/ijms141223711
Janczarek M, Rachwał K. Mutation in the pssA Gene Involved in Exopolysaccharide Synthesis Leads to Several Physiological and Symbiotic Defects in Rhizobium leguminosarum bv. trifolii. International Journal of Molecular Sciences. 2013; 14(12):23711-23735. https://doi.org/10.3390/ijms141223711
Chicago/Turabian StyleJanczarek, Monika, and Kamila Rachwał. 2013. "Mutation in the pssA Gene Involved in Exopolysaccharide Synthesis Leads to Several Physiological and Symbiotic Defects in Rhizobium leguminosarum bv. trifolii" International Journal of Molecular Sciences 14, no. 12: 23711-23735. https://doi.org/10.3390/ijms141223711




