Blocking Autophagic Flux Enhances Matrine-Induced Apoptosis in Human Hepatoma Cells
Abstract
:1. Introduction
2. Results
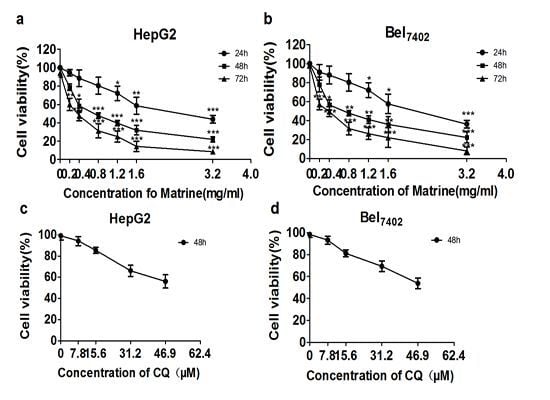
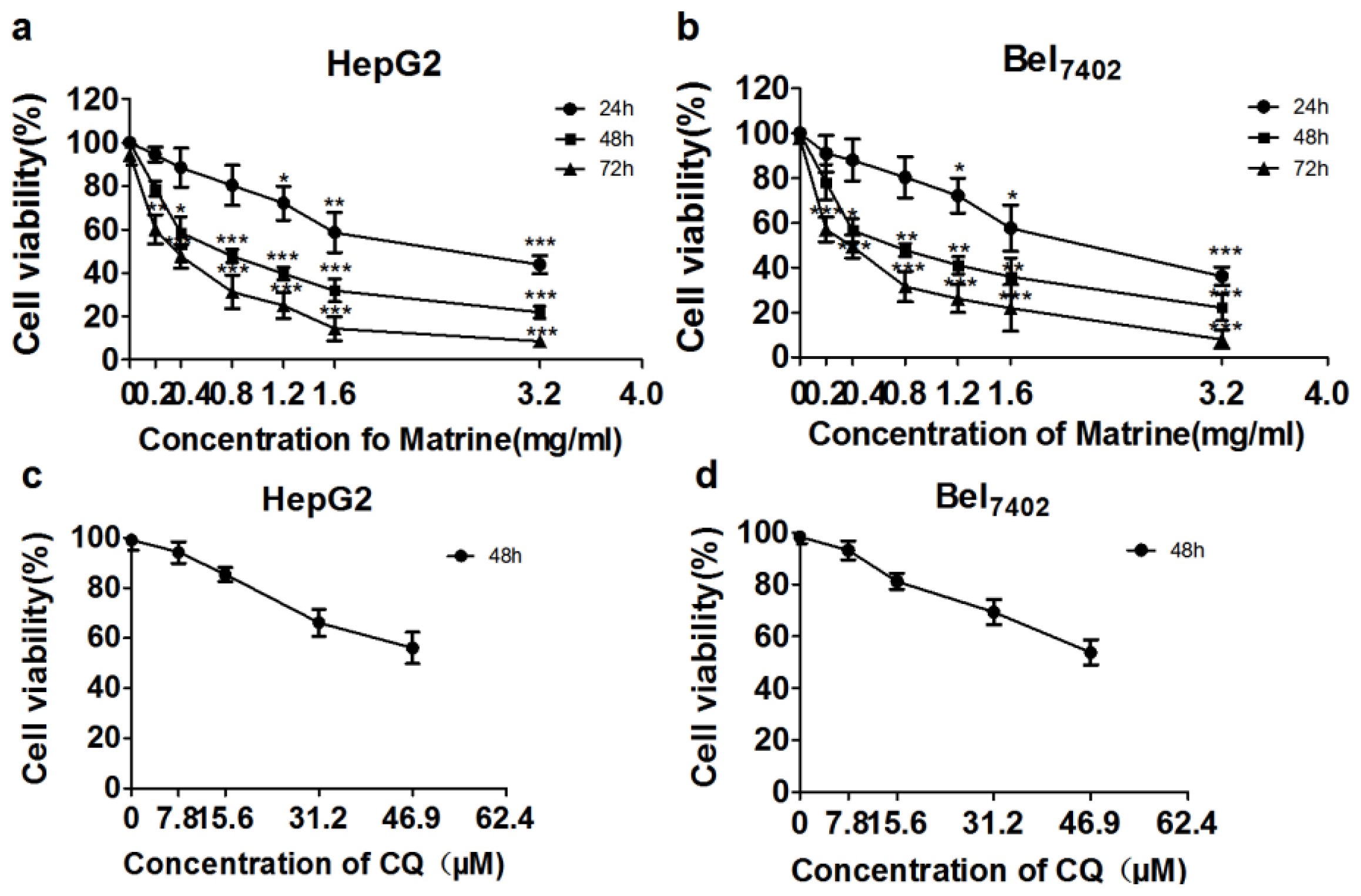
2.1. Matrine Inhibited Viability of HepG2 and Bel7402 Cells
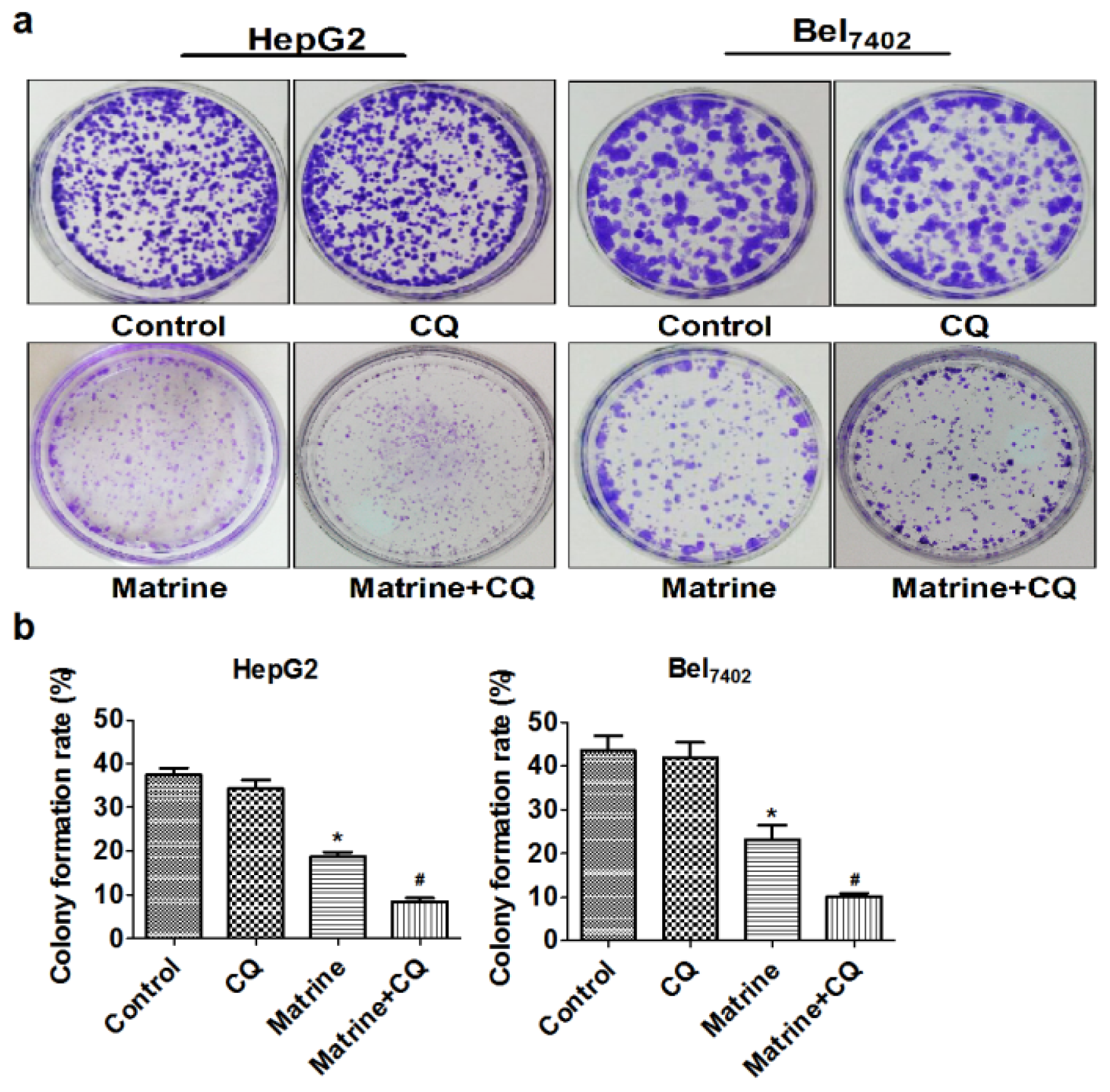
2.2. CQ Enhanced Suppressive Effect of Matrine on Proliferation of Hepatoma Cells
2.3. CQ Blocked Matrine-Induced Autophagic Flux in Hepatoma Cells
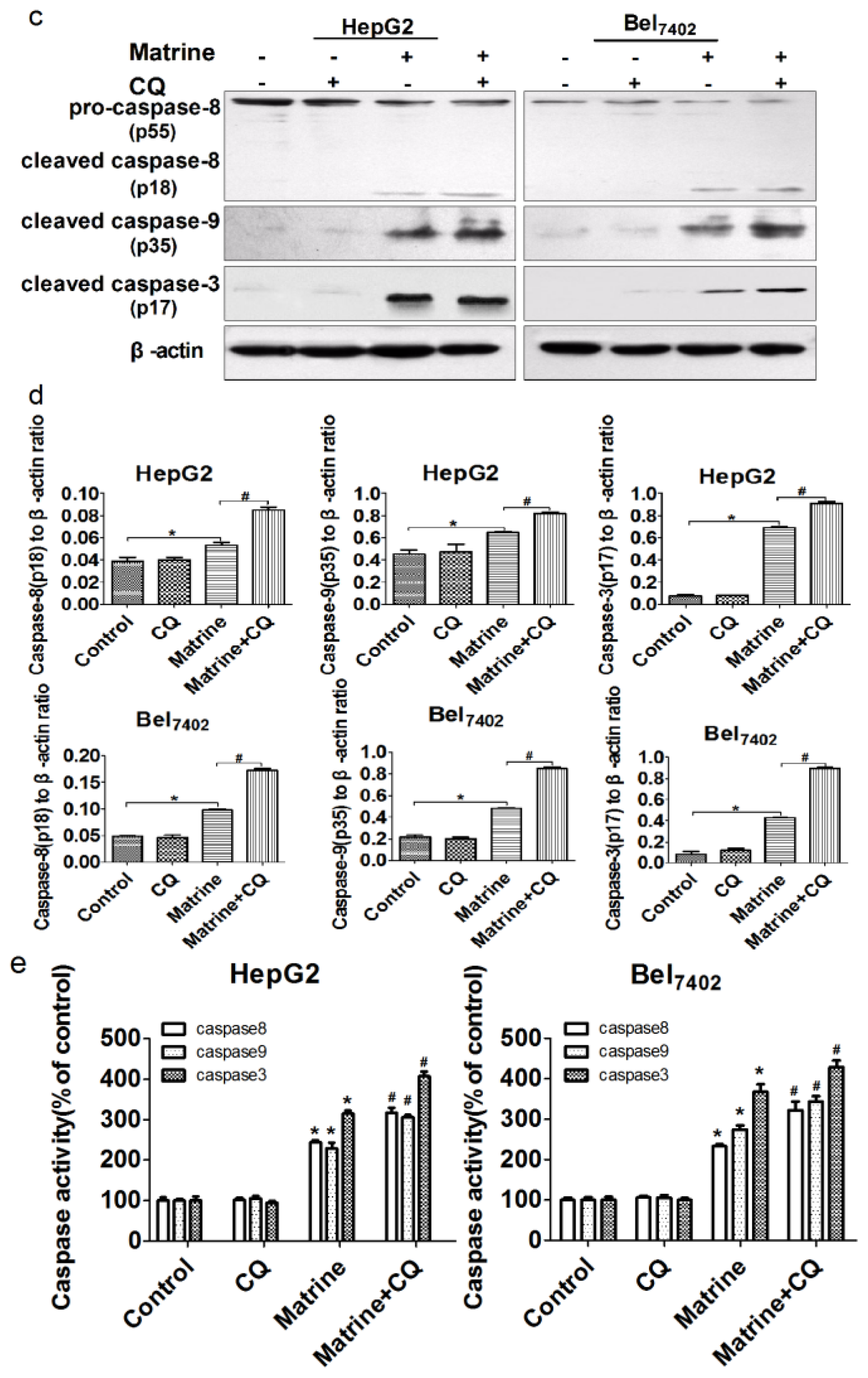
2.4. Combined Treatment with the Autophagic Inhibitor CQ Enhanced Matrine-Induced Apoptosis in a Caspase-Dependent Manner
2.5. Matrine-Induced Autophagy via Inhibition of the PI3K/AKT/mTOR Pathway and Up-Regulation of Beclin-1
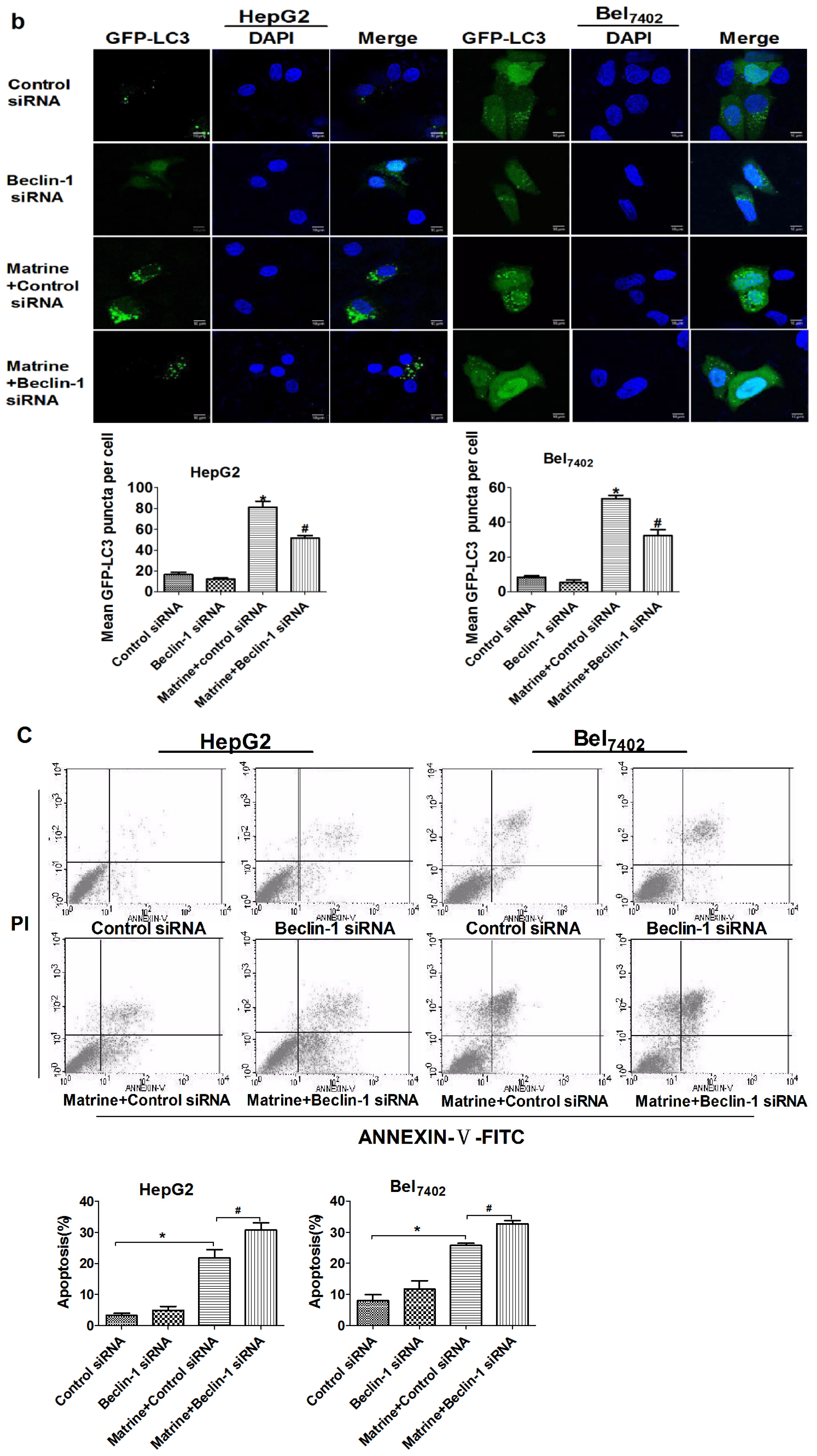
2.6. Silencing Beclin-1 Promoted Matrine-Induced Apoptotic Cell Death through Blockage of Autophagic Flux
3. Discussion
4. Experimental Section
4.1. Reagents and Antibodies
4.2. Cell Lines and Cell Culture
4.3. Plate Colony Forming Assay
4.4. Measurement of Cell Viability
4.5. Flow Cytometry Analysis of Apoptosis
4.6. Caspase Activity
4.7. Cell Lysis and Western Blot Analysis
4.8. GFP-LC3 Plasmid Transfection
4.9. Transmission Electron Microscopy
4.10. siRNA Transfection
4.11. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Chen, W.Q.; Zheng, R.S.; Zhang, S.W. Liver cancer incidence and mortality in China, 2009. Chin. J. Cancer 2013, 32, 162–169. [Google Scholar]
- Llovet, J.M.; Bruix, J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology 2003, 37, 429–442. [Google Scholar]
- El-Serag, H.B.; Siegel, A.B.; Davila, J.A.; Shaib, Y.H.; Cayton-Woody, M.; McBride, R.; McGlynn, K.A. Treatment and outcomes of treating of hepatocellular carcinoma among medicare recipients in the United States: A population-based study. J. Hepatol 2006, 44, 158–166. [Google Scholar]
- Samuel, M.; Chow, P.K.; Chan, S.E.; Machin, D.; Soo, K.C. Neoadjuvant and adjuvant therapy for surgical resection of hepatocellular carcinoma. Cochrane Database Syst. Rev 2009. [Google Scholar] [CrossRef]
- Davila, J.A.; Duan, Z.; McGlynn, K.A.; El-Serag, H.B. Utilization and outcomes of palliative therapy for hepatocellular carcinoma: A population-based study in the United States. J. Clin. Gastroenterol 2012, 46, 71–77. [Google Scholar]
- Thomas, M.B.; O’Beirne, J.P.; Furuse, J.; Chan, A.T.; Abou-Alfa, G.; Johnson, P. Systemic therapy for hepatocellular carcinoma: Cytotoxic chemotherapy, targeted therapy and immunotherapy. Ann. Surg. Oncol 2008, 15, 1008–1014. [Google Scholar]
- Ang, C.; O’Reilly, E.M.; Abou-Alfa, G.K. Targeted agents and systemic therapy in hepatocellular carcinoma. Recent Results Cancer Res 2013, 190, 225–246. [Google Scholar]
- Zhang, J.P.; Zhang, M.; Zhou, J.P.; Liu, F.T.; Zhou, B.; Xie, W.F.; Guo, C. Antifibrotic effects of matrine on in vitro and in vivo models of liver fibrosis in rats. Acta Pharmacol. Sin 2001, 22, 183–186. [Google Scholar]
- Zhang, S.; Zhang, Y.; Zhuang, Y.; Wang, J.; Ye, J.; Zhang, S.; Wu, J.; Yu, K.; Han, Y. Matrine induces apoptosis in human acute myeloid leukemia cells via the mitochondrial pathway and Akt inactivation. PLoS One 2012, 7, e46853. [Google Scholar]
- Li, Y.; Zhang, J.; Ma, H.; Chen, X.; Liu, T.; Jiao, Z.; He, W.; Wang, F.; Liu, X.; Zeng, X. Protective role of autophagy in matrineinduced gastric cancer cell death. Int. J. Oncol 2013, 42, 1417–1426. [Google Scholar]
- Ma, L.; Wen, S.; Zhan, Y.; He, Y.; Liu, X.; Jiang, J. Anticancer effects of the Chinese medicine matrine on murine hepatocellular carcinoma cells. Planta Med 2008, 74, 245–251. [Google Scholar]
- Qin, X.G.; Hua, Z.; Shuang, W.; Wang, Y.H.; Cui, Y.D. Effects of matrine on HepG2 cell proliferation and expression of tumor relevant proteinsin vitro. Pharm. Biol. 2010, 48, 275–281. [Google Scholar]
- Liu, T.; Song, Y.; Chen, H.; Pan, S.; Sun, X. Matrine inhibits proliferation and induces apoptosis of pancreatic cancer cells in vitro and in vivo. Biol. Pharm. Bull. 2010, 33, 1740–1745. [Google Scholar]
- Zhang, J.Q.; Li, Y.M.; Liu, T.; He, W.T.; Chen, Y.T.; Chen, X.H.; Li, X.; Zhou, W.C.; Yi, J.F.; Ren, Z.J. Antitumor effect of matrine in human hepatoma G2 cells by inducing apoptosis and autophagy. World J. Gastroenterol 2010, 16, 4281–4290. [Google Scholar]
- Levine, B.; Kroemer, G. Autophagy in the pathogenesis of disease. Cell 2008, 132, 27–42. [Google Scholar]
- Chen, S.; Rehman, S.K.; Zhang, W.; Wen, A.; Yao, L.; Zhang, J. Autophagy is a therapeutic target in anticancer drug resistance. Biochim. Biophys. Acta 2010, 1806, 220–229. [Google Scholar]
- Janku, F.; McConkey, D.J.; Hong, D.S.; Kurzrock, R. Autophagy as a target for anticancer therapy. Nat. Rev. Clin. Oncol 2011, 8, 528–539. [Google Scholar]
- Zhang, X.J.; Chen, S.; Huang, K.X.; Le, W.D. Why should autophagic flux be assessed? Acta Pharmacol. Sin 2013, 34, 595–599. [Google Scholar]
- Fan, C.; Wang, W.; Zhao, B.; Zhang, S.; Miao, J. Chloroquine inhibits cell growth and induces cell death in A549 lung cancer cells. Bioorg. Med. Chem 2006, 14, 3218–3222. [Google Scholar]
- Kabeya, Y.; Mizushima, N.; Ueno, T.; Yamamoto, A.; Kirisako, T.; Noda, T.; Kominami, E.; Ohsumi, Y.; Yoshimori, T. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 2000, 19, 5720–5728. [Google Scholar]
- Komatsu, M.; Waguri, S.; Koike, M.; Sou, Y.S.; Ueno, T.; Hara, T.; Mizushima, N.; Iwata, J.; Ezaki, J.; Murata, S.; et al. Homeostatic levels of p62 control cytoplasmic inclusion body formation in autophagy-deficient mice. Cell 2007, 131, 1149–1163. [Google Scholar]
- Amaravadi, R.K.; Thompson, C.B. The roles of therapy-induced autophagy and necrosis in cancer treatment. Clin. Cancer Res 2007, 13, 7271–7279. [Google Scholar]
- Wirawan, E.; Lippens, S.; Vanden, B.T.; Romagnoli, A.; Fimia, G.M.; Piacentini, M.; Vandenabeele, P. Beclin-1: A role in membrane dynamics and beyond. Autophagy 2012, 8, 6–17. [Google Scholar]
- Kruidering, M.; Evan, G.I. Caspase-8 in apoptosis: The beginning of “the end”? IUBMB Life 2000, 50, 85–90. [Google Scholar]
- Porter, A.G.; Janicke, R.U. Emerging roles of caspase-3 in apoptosis. Cell Death Differ 1999, 6, 99–104. [Google Scholar]
- Shi, Y.H.; Ding, Z.B.; Zhou, J.; Hui, B.; Shi, G.M.; Ke, A.W.; Wang, X.Y.; Dai, Z.; Peng, Y.F.; Gu, C.Y.; et al. Targeting autophagy enhances sorafenib lethality for hepatocellular carcinoma via ER stress-related apoptosis. Autophagy 2011, 7, 1159–1172. [Google Scholar]
- Xie, B.S.; Zhao, H.C.; Yao, S.K.; Zhuo, D.X.; Jin, B.; Lv, D.C.; Wu, C.L.; Ma, D.L.; Gao, C.; Shu, X.M.; et al. Autophagy inhibition enhances etoposide-induced cell death in human hepatoma G2 cells. Int. J. Mol. Med 2011, 27, 599–606. [Google Scholar]
- Wu, J.; Hu, D.; Zhang, R. Depletion of Bmi-1 enhances 5-fluorouracil-induced apoptosis and autophagy in hepatocellular carcinoma cells. Oncol. Lett 2012, 4, 723–726. [Google Scholar]
- Klionsky, D.J.; Abdalla, F.C.; Abeliovich, H.; Abraham, R.T.; Acevedo-Arozena, A.; Adeli, K.; Agholme, L.; Agnello, M.; Agostinis, P.; Aguirre-Ghiso, J.A.; et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 2012, 8, 445–544. [Google Scholar]
- Shingu, T.; Fujiwara, K.; Bogler, O.; Akiyama, Y.; Moritake, K.; Shinojima, N.; Tamada, Y.; Yokoyama, T.; Kondo, S. Stage-specific effect of inhibition of autophagy on chemotherapy-induced cytotoxicity. Autophagy 2009, 5, 537–539. [Google Scholar]
- Amaravadi, R.K.; Lippincott-Schwartz, J.; Yin, X.M.; Weiss, W.A.; Takebe, N.; Timmer, W.; di Paola, R.S.; Lotze, M.T.; White, E. Principles and current strategies for targeting autophagy for cancer treatment. Clin. Cancer Res 2011, 17, 654–666. [Google Scholar]
- Gao, L.; Song, J.R.; Zhang, J.W.; Zhao, X.; Zhao, Q.D.; Sun, K.; Deng, W.J.; Li, R.; Lv, G.; Cheng, H.Y.; et al. Chloroquine promotes the anticancer effect of TACE in a rabbit VX2 liver tumor model. Int. J. Biol. Sci 2013, 9, 322–330. [Google Scholar]
- Cui, J.; Gong, Z.; Shen, H.M. The role of autophagy in liver cancer: Molecular mechanisms and potential therapeutic targets. Biochim. Biophys. Acta 2013, 1836, 15–26. [Google Scholar]
- Kang, R.; Zeh, H.J.; Lotze, M.T.; Tang, D. The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 2011, 18, 571–580. [Google Scholar]
- Zhou, L.; Huang, Y.; Li, J.; Wang, Z. The mTOR pathway is associated with the poor prognosis of human hepatocellular carcinoma. Med. Oncol 2010, 27, 255–261. [Google Scholar]
- Villanueva, A.; Chiang, D.Y.; Newell, P.; Peix, J.; Thung, S.; Alsinet, C.; Tovar, V.; Roayaie, S.; Minguez, B.; Sole, M.; et al. Pivotal role of mTOR signaling in hepatocellular carcinoma. Gastroenterology 2008, 135. [Google Scholar] [CrossRef]
- Zhou, Q.; Lui, V.W.; Yeo, W. Targeting the PI3K/Akt/mTOR pathway in hepatocellular carcinoma. Future Oncol 2011, 7, 1149–1167. [Google Scholar]
- Li, L.Q.; Li, X.L.; Wang, L.; Du, W.J.; Guo, R.; Liang, H.H.; Liu, X.; Liang, D.S.; Lu, Y.J.; Shan, H.L.; et al. Matrine inhibits breast cancer growth via miR-21/PTEN/Akt pathway in MCF-7 cells. Cell Physiol. Biochem 2012, 30, 631–641. [Google Scholar]
- Lamoureux, F.; Zoubeidi, A. Dual inhibition of autophagy and the AKT pathway in prostate cancer. Autophagy 2013, 9, 1119–1120. [Google Scholar]
- Li, J.R.; Cheng, C.L.; Yang, C.R.; Ou, Y.C.; Wu, M.J.; Ko, J.L. Dual inhibitor of phosphoinositide 3-kinase/mammalian target of rapamycin NVP-BEZ235 effectively inhibits cisplatin-resistant urothelial cancer cell growth through autophagic flux. Toxicol. Lett 2013, 220, 267–276. [Google Scholar]
- Chang, Z.; Shi, G.; Jin, J.; Guo, H.; Guo, X.; Luo, F.; Song, Y.; Jia, X. Dual PI3K/mTOR inhibitor NVP-BEZ235-induced apoptosis of hepatocellular carcinoma cell lines is enhanced by inhibitors of autophagy. Int. J. Mol. Med 2013, 31, 1449–1456. [Google Scholar]





© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, L.; Gao, C.; Yao, S.; Xie, B. Blocking Autophagic Flux Enhances Matrine-Induced Apoptosis in Human Hepatoma Cells. Int. J. Mol. Sci. 2013, 14, 23212-23230. https://doi.org/10.3390/ijms141223212
Wang L, Gao C, Yao S, Xie B. Blocking Autophagic Flux Enhances Matrine-Induced Apoptosis in Human Hepatoma Cells. International Journal of Molecular Sciences. 2013; 14(12):23212-23230. https://doi.org/10.3390/ijms141223212
Chicago/Turabian StyleWang, Li, Chun Gao, Shukun Yao, and Bushan Xie. 2013. "Blocking Autophagic Flux Enhances Matrine-Induced Apoptosis in Human Hepatoma Cells" International Journal of Molecular Sciences 14, no. 12: 23212-23230. https://doi.org/10.3390/ijms141223212