Boronic Acid-Based Approach for Separation and Immobilization of Glycoproteins and Its Application in Sensing
Abstract
:1. Introduction
2. Enrichment of Glycoproteins with Boronic Acid-Functionalized Materials
2.1. Boronic Acid-Functionalized Monoliths
2.2. Boronic Acid-Functionalized Magnetic Particles
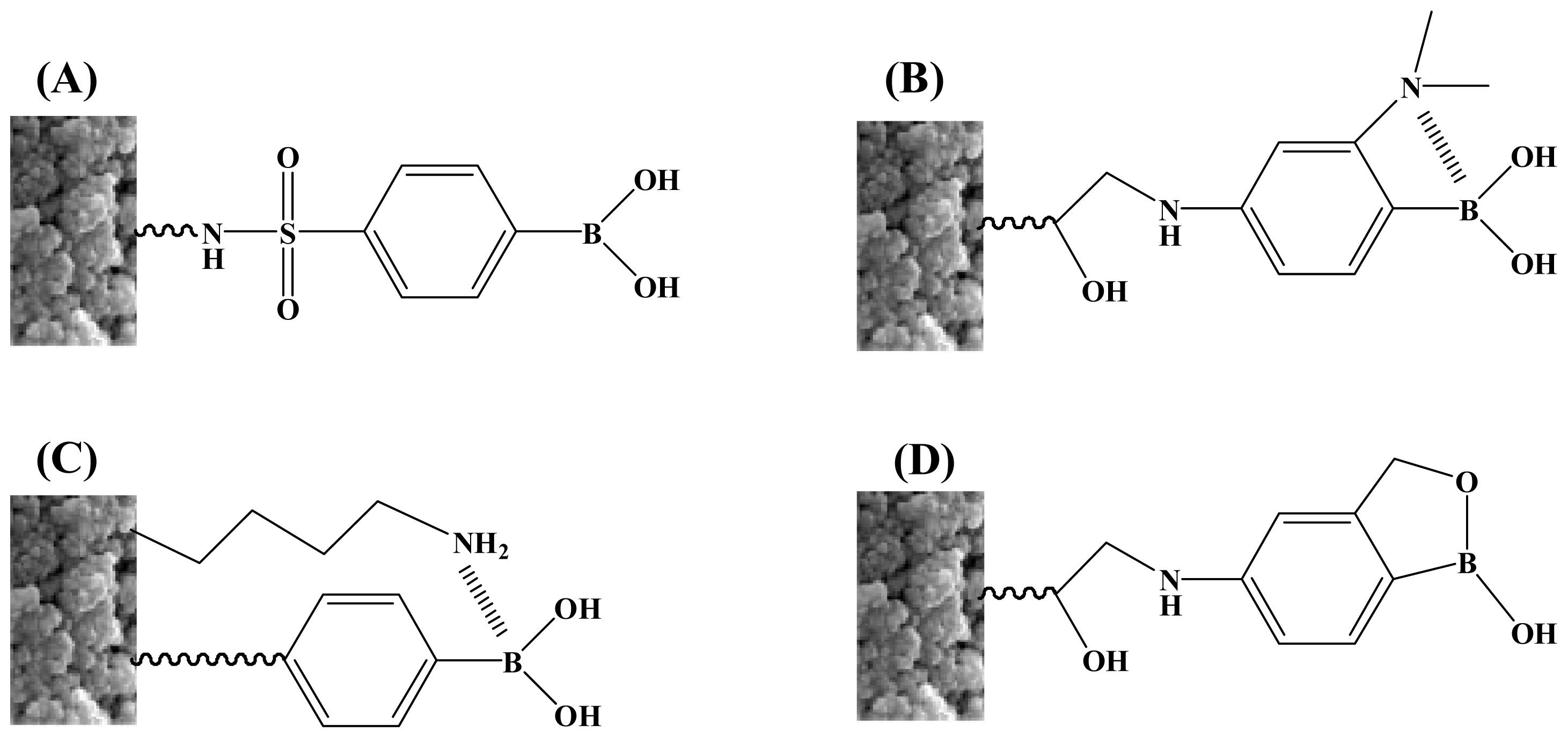
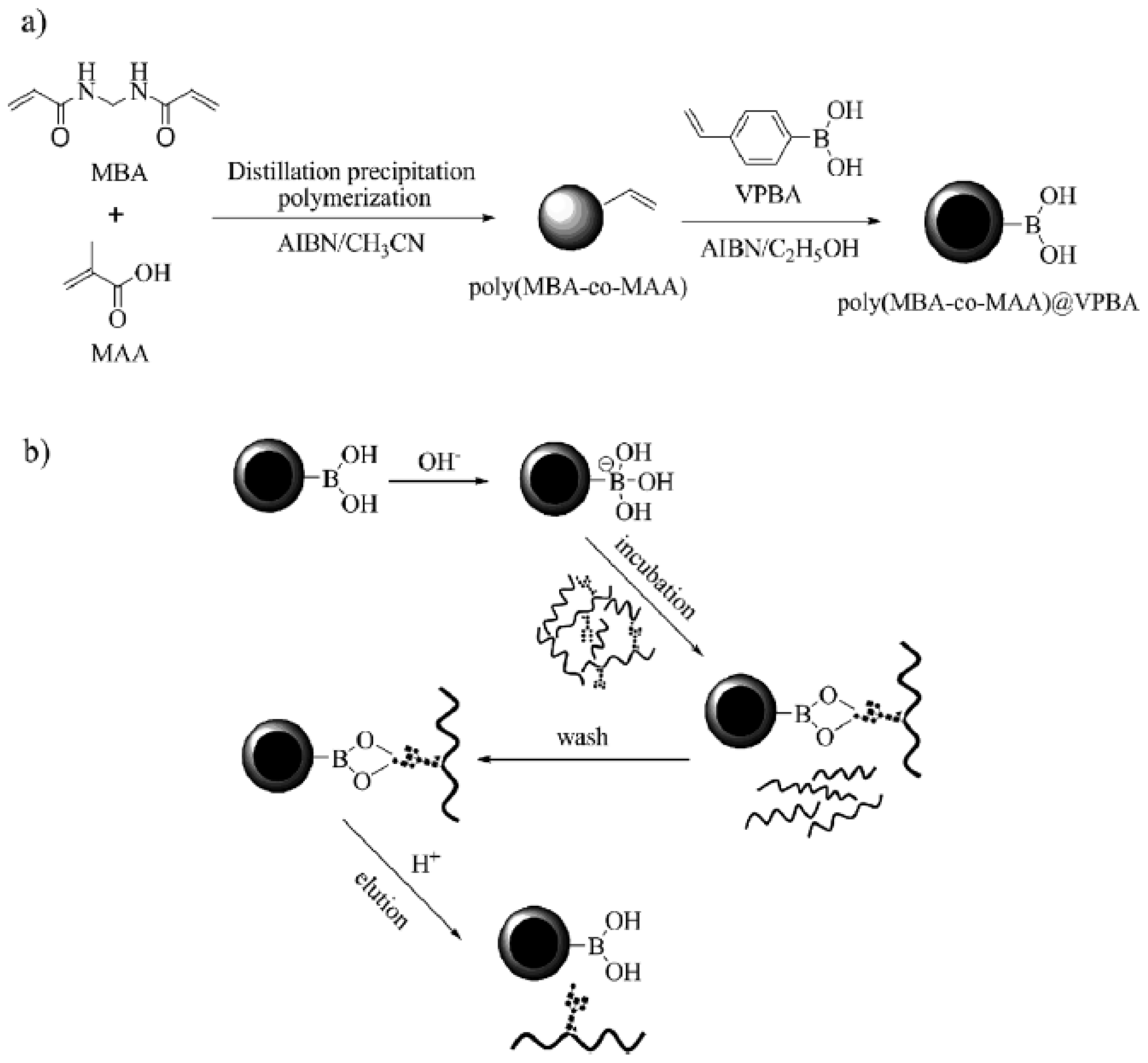
2.3. Other Boronic Acid-Based Materials
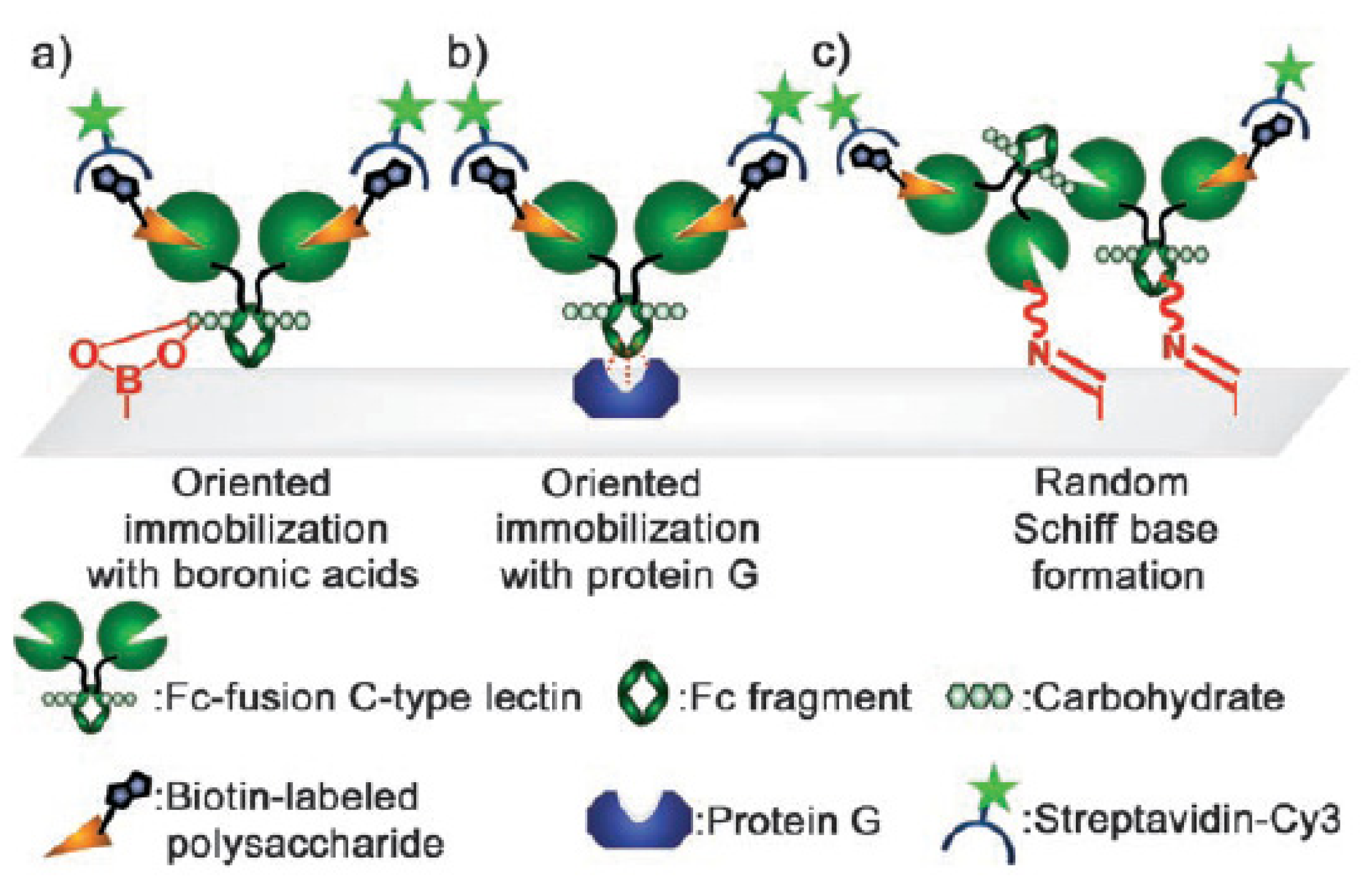
3. Immobilization of Antibodies and Enzymes on Boronic Acid-Modified Substrates for Sensing
3.1. Immobilization of Antibodies for Sensing
3.2. Immobilization of Enzymes for Sensing
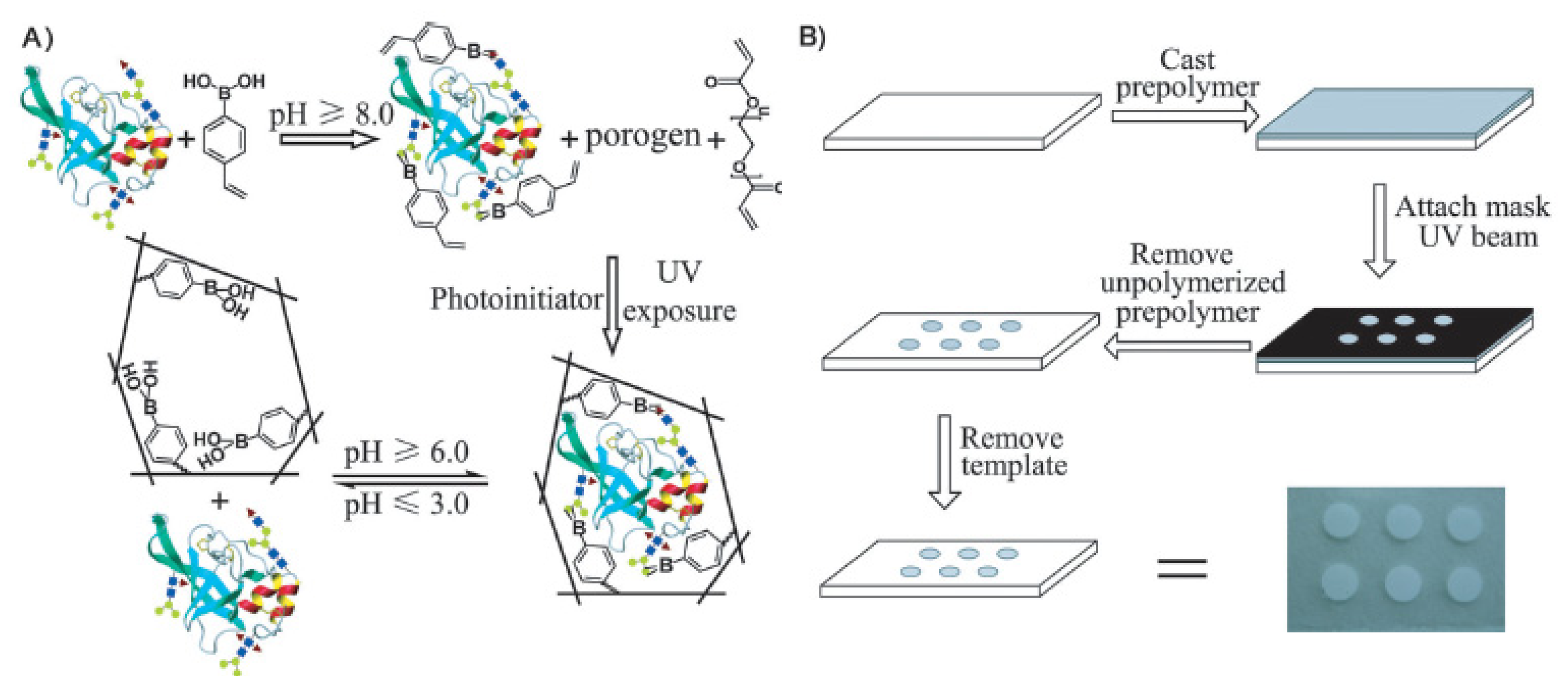
4. Detection of Glycoproteins by the Recognition of Boronic Acid-Functionalized Materials
5. Conclusions



Acknowledgments
Conflicts of Interest
References
- Roth, J. Protein N-glycosylation along the secretory pathway: Relationship to organelle topography and function, protein quality control, and cell interactions. Chem. Rev 2002, 102, 285–303. [Google Scholar]
- Muniyan, S.; Chaturvedi, N.K.; Dwyer, J.G.; LaGrange, C.A.; Chaney, W.G.; Lin, M.F. Human prostatic acid phosphatase: Structure, function and regulation. Int. J. Mol. Sci 2013, 14, 10438–10464. [Google Scholar]
- Burnham-Marusich, A.R.; Berninsone, P.M. Multiple proteins with essential mitochondrial functions have glycosylated isoforms. Mitochondrion 2012, 12, 423–427. [Google Scholar]
- Mu, A.K.W.; Lim, B.K.; Hashim, O.H.; Shuib, A.S. Identification of O-glycosylated proteins that are aberrantly excreted in the urine of patients with early stage ovarian cancer. Int. J. Mol. Sci 2013, 14, 7923–7931. [Google Scholar]
- Dell, A.; Morris, H.R. Glycoprotein structure determination by mass spectrometry. Science 2001, 291, 2351–2356. [Google Scholar]
- Nagae, M.; Yamaguchi, Y. Function and 3D structure of the N-glycans on glycoproteins. Int. J. Mol. Sci 2012, 13, 8398–8429. [Google Scholar]
- Feng, S.; Yang, N.; Pennathur, S.; Goodison, S.; Lubman, M. Enrichment of glycoproteins using nano-scale chelating Con A monolithic capillary chromatography. Anal. Chem 2009, 81, 3776–3783. [Google Scholar]
- Madera, M.; Mechref, Y.; Klouckova, I.; Novotny, M.V. Semiautomated high-sensitivity profiling of human blood serum glycoproteins through lectin preconcentration and multidimensional chromatography/tandem mass spectrometry. J. Proteome Res 2006, 5, 2348–2363. [Google Scholar]
- Zhang, H.; Li, X.J.; Martin, D.B.; Aebersold, R. Identification and quantification of N-linked glycoproteins using hydrazide chemistry, stable isotope labeling and mass spectrometry. Nat. Biotechnol 2003, 21, 660–666. [Google Scholar]
- Chen, R.; Jiang, X.N.; Sun, D.G.; Han, G.H.; Wang, F.J.; Ye, M.L.; Wang, L.M.; Zou, H.F. Glycoproteomics analysis of human liver tissue by combination of multiple enzyme digestion and hydrazide chemistry. J. Proteome Res 2009, 8, 651–661. [Google Scholar]
- Hägglund, P.; Bunkenborg, J.; Elortza, F.; Nørregaard, J.O.; Roepstorff, P. A new strategy for identification of N-glycosylated proteins and unambiguous assignment of their glycosylation sites using HILIC enrichment and partial deglycosylation. J. Proteome Res 2004, 3, 556–566. [Google Scholar]
- Wada, Y.; Tajiri, M.; Yoshida, S. Hydrophilic affinity isolation and MALDI multiple-stage tandem mass spectrometry of glycopeptides for glycoproteomics. Anal. Chem 2004, 76, 6560–6565. [Google Scholar]
- Bykova, N.V.; Rampitsch, C.; Krokhin, O.; Standing, K.G.; Ens, W. Determination and characterization of site-specific N-glycosylation using MALDI-Qq-TOF tandem mass spectrometry: Case study with a plant protease. Anal. Chem 2006, 78, 1093–1103. [Google Scholar]
- Imre, T.; Schlosser, G.; Pocsfalvi, G.; Siciliano, R.; Molnár-Szöllosi, E.; Kremmer, T.; Malorni, A.; Vékey, K. Glycosylation site analysis of human alpha-1-acid glycoprotein (AGP) by capillary liquid chromatography-electrospray mass spectrometry. J. Mass Spectrom 2005, 40, 1472–1483. [Google Scholar]
- Burnham-Marusich, A.R.; Snodgrass, C.J.; Johnson, A.M.; Kiyoshi, C.M.; Buzby, S.E.; Gruner, M.R.; Berninsone, P.M. Metabolic labeling of caenorhabditis elegans primary embryonic cells with azido-sugars as a tool for glycoprotein discovery. PLoS One 2012, 7, e49020. [Google Scholar]
- Zhang, Q.; Schepmoes, A.A.; Brock, J.W.C.; Wu, S.; Moore, R.J.; Purvine, S.O.; Baynes, J.W.; Smith, R.D.; Metz, T.O. Improved methods for the enrichment and analysis of glycated peptides. Anal. Chem 2008, 80, 9822–9829. [Google Scholar]
- Zhang, Q.; Tang, N.; Brock, J.W.C.; Mottaz, H.M.; Ames, J.M.; Baynes, J.W.; Smith, R.D.; Metz, T.O. Enrichment and analysis of nonenzymatically glycated peptides: Boronate affinity chromatography coupled with electron-transfer dissociation mass spectrometry. J. Proteome Res 2007, 6, 2323–2330. [Google Scholar]
- Zhang, Q.; Tang, N.; Schepmoes, A.A.; Phillips, L.S.; Smith, R.D.; Metz, T.O. Proteomic profiling of nonenzymatically glycated proteins in human plasma and erythrocyte membranes. J. Proteome Res 2008, 7, 2025–2032. [Google Scholar]
- Takátsy, A.; Böddi, K.; Nagy, L.; Nagy, G.; Szabó, S.; Markó, L.; Wittmann, I.; Ohmacht, R.; Ringer, T.; Bonn, G.K.; et al. Enrichment of Amadori products derived from the nonenzymatic glycation of proteins using microscale boronate affinity chromatography. Anal. Biochem 2009, 393, 8–22. [Google Scholar]
- Potter, O.G.; Breadmore, M.C.; Hilder, E.F. Boronate functionalised polymer monoliths for microscale affinity chromatography. Analyst 2006, 131, 1094–1096. [Google Scholar]
- Chen, M.; Lu, Y.; Ma, Q.; Guo, L.; Feng, Y.-Q. Boronate affinity monolith for highly selective enrichment of glycopeptides and glycoproteins. Analyst 2009, 134, 2158–2164. [Google Scholar]
- Ren, L.; Liu, Z.; Dong, M.; Ye, M.; Zou, H. Synthesis and characterization of a new boronate affinity monolithic capillary for specific capture of cis-diol-containing compounds. J. Chromatogr. A 2009, 1216, 4768–4774. [Google Scholar]
- Ren, L.; Liu, Y.; Dong, M.; Liu, Z. Synthesis of hydrophilic boronate affinity monolithic capillary for specific capture of glycoproteins by capillary liquid chromatography. J. Chromatogr. A 2009, 1216, 8421–8425. [Google Scholar]
- Lin, Z.A.; Pang, J.L.; Lin, Y.; Huang, H.; Cai, Z.W.; Zhang, L.; Chen, G.N. Preparation and evaluation of a phenylboronate affinity monolith for selective capture of glycoproteins by capillary liquid chromatography. Analyst 2011, 136, 3281–3288. [Google Scholar]
- Singhal, R.P.; Ramamurhy, B.; Govindraj, N.; Sarwar, Y. New ligands for boronate affinity chromatography. Synthesis and properties. J. Chromatogr. A 1991, 543, 17–38. [Google Scholar]
- Li, X.; Pennington, J.; Stobaugh, J.F.; Schöneich, C. Synthesis of sulfonamide- and sulfonyl-phenylboronic acid-modified silica phases for boronate affinity chromatography at physiological pH. Anal. Biochem 2008, 372, 227–236. [Google Scholar]
- Matsumoto, A.; Yoshida, R.; Kataoka, K. Glucose-responsive polymer gel bearing phenylborate derivative as a glucose-sensing moiety operating at the physiological pH. Biomacromolecules 2004, 5, 1038–1045. [Google Scholar]
- Liu, Y.; Ren, L.; Liu, Z. A unique boronic acid functionalized monolithic capillary for specific capture, separation and immobilization of cis-diol biomolecules. Chem. Commun 2011, 47, 5067–5069. [Google Scholar]
- Wulff, G.; Lauer, M.; Böhnke, H. Rapid proton transfer as cause of an unusually large neighboring group effect. Angew. Chem. Int. Ed 1984, 23, 741–742. [Google Scholar]
- Wulff, G. Molecular imprinting in cross-linked materials with the aid of molecular templates—A way towards artificial antibodies. Angew. Chem. Int. Ed 1995, 34, 1812–1832. [Google Scholar]
- Tuytten, R.; Lemiere, F.; Esmans, E.L.; Herrebout, W.A.; van der Veken, B.J.; Maes, B.U.W.; Witters, E.; Newton, R.P.; Dudley, E. Role of nitrogen lewis basicity in boronate affinity chromatography of nucleosides. Anal. Chem 2007, 79, 6662–6669. [Google Scholar]
- Ren, L.; Liu, Z.; Liu, Y.; Dou, P.; Chen, H.-Y. Ring-upening polymerization with synergistic co-monomers: Access to a boronate-functionalized polymeric monolith for the specific capture of cis-diol-containing biomolecules under neutral conditions. Angew. Chem. Int. Ed 2009, 48, 6704–6707. [Google Scholar]
- Liu, Y.; Lu, Y.; Liu, Z. Restricted access boronate affinity porous monolith as a protein A mimetic for the specific capture of immunoglobulin G. Chem. Sci 2012, 3, 1467–1471. [Google Scholar]
- Dowlut, M.; Hall, D.G. An improved class of sugar-binding boronic acids, soluble and capable of complexing glycosides in neutral water. J. Am. Chem. Soc 2006, 128, 4226–4227. [Google Scholar]
- Li, H.; Wang, H.; Liu, Y.; Liu, Z. A benzoboroxole-functionalized monolithic column for the selective enrichment and separation of cis-diol containing biomolecules. Chem. Commun 2012, 48, 4115–4117. [Google Scholar]
- Yang, F.; Lin, Z.; He, X.; Chen, L.; Zhang, Y. Synthesis and application of a macroporous boronate affinity monolithic column using a metal-organic gel as a porogenic template for the specific capture of glycoproteins. J. Chromatogr. A 2011, 1218, 9194–9201. [Google Scholar]
- Lin, Z.; Pang, J.; Yang, H.; Cai, Z.; Zhang, L.; Chen, G. One-pot synthesis of an organic–inorganic hybrid affinity monolithic column for specific capture of glycoproteins. Chem. Commun 2011, 47, 9675–9677. [Google Scholar]
- Lu, Y.; Bie, Z.; Liu, Y.; Liu, Z. Fine-tuning the specificity of boronate affinity monoliths toward glycoproteins through pH manipulation. Analyst 2013, 138, 290–298. [Google Scholar]
- Lee, J.H.; Kim, Y.; Ha, M.Y.; Lee, E.K.; Choo, J. Immobilization of aminophenylboronic acid on magnetic beads for the direct determination of glycoproteins by matrix assisted laser desorption ionization mass spectrometry. J. Am. Soc. Mass Spectrom 2005, 16, 1456–1460. [Google Scholar]
- Sparbier, K.; Koch, S.; Kessler, I.; Wenzel, T.; Kostrzewa, M. Selective isolation of glycoproteins and glycopeptides for MALDI-TOF MS detection supported by magnetic particles. J. Biomol. Tech 2005, 4, 407–413. [Google Scholar]
- Zhou, W.; Yao, N.; Yao, G.; Deng, C.; Zhang, X.; Yang, P. Facile synthesis of aminophenylboronic acid-functionalized magnetic nanoparticles for selective separation of glycopeptides and glycoproteins. Chem. Commun 2008, 5577–5579. [Google Scholar]
- Bao, J.; Chen, W.; Liu, T.; Zhu, Y.; Jin, P.; Wang, L.; Liu, J.; Wei, Y.; Li, Y. Bifunctional Au-Fe3O4 nanoparticles for protein separation. ACS Nano 2007, 1, 293–298. [Google Scholar]
- Lyon, J.L.; Fleming, D.A.; Stone, M.B.; Schiffer, P.; Williams, M.E. Synthesis of Fe oxide core/Au shell nanoparticles by iterative hydroxylamine seeding. Nano Lett 2004, 4, 719–723. [Google Scholar]
- Stoeva, S.I.; Huo, F.; Lee, J.-S.; Mirkin, C.A. Three-layer composite magnetic nanoparticle probes for DNA. J. Am. Chem. Soc 2005, 127, 15362–15363. [Google Scholar]
- Xu, C.; Xie, J.; Ho, D.; Wang, C.; Kohler, N.; Walsh, E.G.; Morgan, J.R.; Chin, Y.E.; Sun, S. Au-Fe3O4 dumbbell nanoparticles as dual-functional probes. Angew. Chem. Int. Ed 2008, 47, 173–176. [Google Scholar]
- Yu, H.; Chen, M.; Rice, P.M.; Wang, S.X.; White, R.L.; Sun, S. Dumbbell-like bifunctional Au-Fe3O4 nanoparticles. Nano Lett 2005, 5, 379–382. [Google Scholar]
- Zhao, X.; Cai, Y.; Wang, T.; Shi, Y.; Jiang, G. Preparation of alkanethiolate-functionalized core/shell Fe3O4@Au nanoparticles and its interaction with several typical target molecules. Anal. Chem 2008, 80, 9091–9096. [Google Scholar]
- Kouassi, G.K.; Irudayaraj, J. Magnetic and gold-coated magnetic nanoparticles as a DNA sensor. Anal. Chem 2006, 78, 3234–3241. [Google Scholar]
- Zhang, H.; Meyerhoff, M.E. Gold coated magnetic particles for solid phase immunoassays: Enhancing immobilized antibody binding efficiency and analytical performance. Anal. Chem 2006, 78, 609–616. [Google Scholar]
- Qi, D.; Zhang, H.; Tang, J.; Deng, C.; Zhang, X. Facile synthesis of mercaptophenylboronic acid-functionalized core-shell structure Fe3O4@C@Au magnetic microspheres for selective enrichment of glycopeptides and glycoproteins. J. Phys. Chem. C 2010, 114, 9221–9226. [Google Scholar]
- Liang, L.; Liu, Z. A self-assembled molecular team of boronic acids at the gold surface for specific capture of cis-diol biomolecules at neutral pH. Chem. Commun 2011, 47, 2255–2257. [Google Scholar]
- Zhang, X.; He, X.; Chen, L.; Zhang, Y. Boronic acid modified magnetic nanoparticles for enrichment of glycoproteins via azide and alkyne click chemistry. J. Mater. Chem 2012, 22, 16520–16526. [Google Scholar]
- Lin, Z.-A.; Zheng, J.-N.; Lin, F.; Zhang, L.; Cai, Z.; Chen, G.-N. Synthesis of magnetic nanoparticles with immobilized aminophenylboronic acid for selective capture of glycoproteins. J. Mater. Chem 2011, 21, 518–524. [Google Scholar]
- Shen, W.-W.; Ma, C.-N.; Wang, S.-F.; Xiong, H.-M.; Lu, H.-J.; Yang, P.-Y. SnO2@poly(HEMA-co-St-co-VPBA) core-shell nanoparticles designed for selectively enriching glycopeptides followed by MALDI-MS analysis. Chem. Eur. J 2010, 5, 1185–1191. [Google Scholar]
- Qu, Y.; Liu, J.; Yang, K.; Liang, Z.; Zhang, L.; Zhang, Y. Boronic acid functionalized core–shell polymer nanoparticles prepared by distillation precipitation polymerization for glycopeptide enrichment. Chem. Eur. J 2012, 18, 9056–9062. [Google Scholar]
- Xu, Y.; Zhang, L.; Lu, H.; Yang, P. On-plate enrichment of glycopeptides by using boronic acid functionalized gold-coated Si wafer. Proteomics 2010, 10, 1079–1086. [Google Scholar]
- Tang, J.; Liu, Y.; Qi, D.; Yao, G.; Deng, C.; Zhang, X. On-plate-selective enrichment of glycopeptides using boronic acid-modified gold nanoparticles for direct MALDI-QIT-TOF MS analysis. Proteomics 2009, 9, 5046–5055. [Google Scholar]
- Zeng, Z.; Wang, Y.; Guo, X.; Wang, L.; Lu, N. On-plate glycoproteins/glycopeptides selective enrichment and purification based on surface pattern for direct MALDI MS analysis. Analyst 2013, 138, 3032–3037. [Google Scholar]
- Xu, Y.; Wu, Z.; Zhang, L.; Lu, H.; Yang, P.; Webley, P.A.; Zhao, D. Highly specific enrichment of glycopeptides using boronic acid-functionalized mesoporous silica. Anal. Chem 2009, 81, 503–508. [Google Scholar]
- Chen, M.-L.; Adak, A.K.; Yeh, N.-C.; Yang, W.-B.; Chuang, Y.-J.; Wong, C.-H.; Hwang, K.-C.; Hwu, J.-R.R.; Hsieh, S.-L.; Lin, C.-C. Fabrication of an oriented Fc-fused lectin microarray through boronate formation. Angew. Chem. Int. Ed 2008, 45, 8627–8630. [Google Scholar]
- Lin, P.-C.; Chen, S.-H.; Wang, K.-Y.; Chen, M.-L.; Adak, A.K.; Hwu, J.-R.R.; Chen, Y.-J.; Lin, C.-C. Fabrication of oriented antibody-conjugated magnetic nanoprobes and their immunoaffinity application. Anal. Chem 2009, 81, 8774–8782. [Google Scholar]
- Zhang, X.; Wu, Y.; Tu, Y.; Liu, S. A reusable electrochemical immunosensor for carcinoembryonic antigen via molecular recognition of glycoprotein antibody by phenylboronic acid self-assembly layer on gold. Analyst 2008, 133, 485–492. [Google Scholar]
- Wang, Z.; Tu, Y.; Liu, S. Electrochemical immunoassay for α-fetoprotein through a phenylboronic acid monolayer on gold. Talanta 2008, 77, 815–821. [Google Scholar]
- Liu, S.; Zhang, X.; Wu, Y.; Tu, Y.; He, L. Prostate-specific antigen detection by using a reusable amperometric immunosensor based on reversible binding and leasing of HRP-anti-PSA from phenylboronic acid modified electrode. Clin. Chim. Acta 2008, 395, 51–56. [Google Scholar]
- Ho, J.A.; Hsu, W.-L.; Liao, W.-C.; Chiu, J.-K.; Chen, M.-L.; Chang, H.-C.; Li, C.-C. Ultrasensitive electrochemical detection of biotin using electrically addressable site-oriented antibody immobilization approach via aminophenyl boronic acid. Biosens. Bioelectron 2010, 26, 1021–1027. [Google Scholar]
- Moreno-Guzmán, M.; González-Cortés, A.; Yáñez-Sedeño, P.; Pingarrón, J.M. Multiplexed ultrasensitive determination of adrenocorticotropin and cortisol hormones at a dual electrochemical immunosensor. Electroanalysis 2012, 24, 1100–1108. [Google Scholar]
- Moreno-Guzmán, M.; Ojeda, I.; Villalonga, R.; González-Cortés, A.; Yáñez-Sedeño, P.; Pingarrón, J.M. Ultrasensitive detection of adrenocorticotropin hormone (ACTH) using disposable phenylboronic-modified electrochemical immunosensors. Biosens. Bioelectron 2012, 35, 82–86. [Google Scholar]
- Abad, J.M.; Vélez, M.; Santamaría, C.; Guisán, J.M.; Matheus, P.R.; Vázquez, L.; Gazaryan, I.; Gorton, L.; Gibson, T.; Fernández, V.M. Immobilization of peroxidase glycoprotein on gold electrodes modified with mixed epoxy-boronic acid monolayers. J. Am. Chem. Soc 2002, 124, 12845–12853. [Google Scholar]
- Zayats, M.; Katz, E.; Willner, I. Electrical contacting of glucose oxidase by surface-reconstitution of the apo-protein on a relay-boronic acid-FAD cofactor monolayer. J. Am. Chem. Soc 2002, 124, 2120–2121. [Google Scholar]
- Zayats, M.; Katz, E.; Willner, I. Electrical contacting of flavoenzymes and NAD(P)+-dependent enzymes by reconstitution and affinity interactions on phenylboronic acid monolayers associated with Au-electrodes. J. Am. Chem. Soc 2002, 124, 14724–14735. [Google Scholar]
- Ma, Y.; Gao, Q.; Yang, X. Immobilization of glycosylated enzymes on carbon electrodes, and its application in biosensors. Microchim. Acta 2005, 150, 21–26. [Google Scholar]
- Liu, S.; Miller, B.; Chen, A. Phenylboronic acid self-assembled layer on glassy carbon electrode for recognition of glycoprotein peroxidase. Electrochem. Commun 2005, 7, 1232–1236. [Google Scholar]
- Liu, S.; Bakovic, L.; Chen, A. Specific binding of glycoproteins with poly(aniline boronic acid) thin film. J. Electroanal. Chem 2006, 591, 210–216. [Google Scholar]
- Villalonga, R.; Díez, P.; Yáñez-Sedeño, P.; Pingarrón, J.M. Wiring horseradish peroxidase on gold nanoparticles-based nanostructured polymeric network for the construction of mediatorless hydrogen peroxide biosensor. Electrochim. Acta 2011, 56, 4672–4677. [Google Scholar]
- Dong, J.; Liu, T.; Meng, X.; Zhu, J.; Shang, K.; Ai, S.; Cui, S. Amperometric biosensor based on immobilization of acetylcholinesterase via specific binding on biocompatible boronic acid-functionalized Fe@Au magnetic nanoparticles. J. Solid State Electrochem 2012, 16, 3783–3790. [Google Scholar]
- Nie, H.; Chen, Y.; Lü, C.; Liu, Z. Efficient selection of glycoprotein-binding DNA aptamers via boronate affinity monolithic capillary. Anal. Chem 2013, 85, 8277–8283. [Google Scholar]
- Gornik, O.; Lauc, G. Glycosylation of serum proteins in inflammatory diseases. Dis. Markers 2008, 25, 267–278. [Google Scholar]
- Mu, A.K.W.; Lim, B.K.; Hashim, O.H.; Shuib, A.S. Detection of differential levels of proteins in the urine of patients with endometrial cancer: Analysis using two-dimensional gel electrophoresis and O-glycan binding lectin. Int. J. Mol. Sci 2012, 13, 9489–9501. [Google Scholar]
- De Guzman, J.M.; Soper, S.A.; McCarley, R.L. Assessment of glycoprotein interactions with 4-[(2-aminoethyl)carbamoyl]phenylboronic acid surfaces using surface plasmon resonance spectroscopy. Anal. Chem 2010, 82, 8970–8977. [Google Scholar]
- Park, J.Y.; Chang, B.-Y.; Nam, H.; Park, S.-M. Selective electrochemical sensing of glycated hemoglobin (HbA1c) on thiophene-3-boronic acid self-assembled monolayer covered gold electrodes. Anal. Chem 2008, 80, 8035–8044. [Google Scholar]
- Chuang, Y.-C.; Lan, K.-C.; Hsieh, K.-M.; Jang, L.-S.; Chen, M.-K. Detection of glycated hemoglobin (HbA1c) based on impedance measurement with parallel electrodes integrated into a microfluidic device. Sensor. Actuat. B 2012, 171–172, 1222–1230. [Google Scholar]
- Song, S.Y.; Han, Y.D.; Park, Y.M.; Jeong, C.Y.; Yang, Y.J.; Kim, M.S.; Ku, Y.; Yoon, H.C. Bioelectrocatalytic detection of glycated hemoglobin (HbA1c) based on the competitive binding of target and signaling glycoproteins to a boronate-modified surface. Biosens. Bioelectron 2012, 35, 355–362. [Google Scholar]
- Song, S.Y.; Yoon, H.C. Boronic acid-modified thin film interface for specific binding of glycated hemoglobin (HbA1c) and electrochemical biosensing. Sensor. Actuat. B 2009, 140, 233–239. [Google Scholar]
- Ivanov, A.E.; Solodukhina, N.; Wahlgren, M.; Nilsson, L.; Vikhrov, A.A.; Nikitin, M.P.; Orlov, A.V.; Nikitin, P.I.; Kuzimenkova, M.V.; Zubov, V.P. Reversible conformational transitions of a polymer brush containing boronic acid and its interaction with mucin glycoprotein. Macromol. Biosci 2011, 11, 275–284. [Google Scholar]
- Nguyena, Q.H.; Ali, M.; Neumannb, R.; Ensingera, W. Saccharide/glycoprotein recognition inside synthetic ion channels modified with boronic acid. Sensor. Actuat. B 2012, 162, 216–222. [Google Scholar]
- Sun, Z.; Han, C.; Wen, L.; Tian, D.; Li, H.; Jiang, L. pH gated glucose responsive biomimetic single nanochannels. Chem. Commun 2012, 48, 3282–3284. [Google Scholar]
- Liu, M.; Zhang, L.; Xu, Y.; Yang, P.; Lu, H. Mass spectrometry signal amplification for ultrasensitive glycoprotein detection using gold nanoparticle as mass tag combined with boronic acid based isolation strategy. Anal. Chim. Acta 2013, 788, 129–134. [Google Scholar]
- Baker, G.A.; Desikan, R.; Thundat, T. Label-free sugar detection using phenylboronic acid-functionalized piezoresistive microcantilevers. Anal. Chem 2008, 80, 4860–4865. [Google Scholar]
- Rohovec, J.; Maschmeyer, T.; Aime, S.; Peters, J.A. The structure of the sugar residue in glycated human serum albumin and its molecular recognition by phenylboronate. Chem. Eur. J 2003, 9, 2193–2199. [Google Scholar]
- Kanayama, N.; Kitano, H. Interfacial recognition of sugars by boronic acid-carrying self-assembled monolayer. Langmuir 2000, 16, 577–583. [Google Scholar]
- Xia, N.; Deng, D.; Zhang, L.; Yuan, B.; Jing, M.; Du, J.; Liu, L. Sandwich-type electrochemical biosensor for glycoproteins detection based on dual-amplification of boronic acid-gold nanoparticles and dopamine-gold nanoparticles. Biosens. Bioelectron 2013, 43, 155–159. [Google Scholar]
- Bossi, A.; Piletsky, S.A.; Piletska, E.V.; Righetti, P.G.; Turner, A.P.F. Surface-grafted molecularly imprinted polymers for protein recognition. Anal. Chem 2001, 73, 5281–5286. [Google Scholar]
- Li, L.; Lu, Y.; Bie, Z.; Chen, H.-Y.; Liu, Z. Photolithographic boronate affinity molecular imprinting: A general and facile approach for glycoprotein imprinting. Angew. Chem. Int. Ed 2013, 52, 7451–7454. [Google Scholar]
- Li, Y.; Hong, M.; Miaomiao Bin, Q.; Lin, Z.; Cai, Z.; Chen, G. Novel composites of multifunctional Fe3O4@Au nanofibers for highly efficient glycoprotein imprinting. J. Mater. Chem. B 2013, 1, 1044–1051. [Google Scholar]
- Tiralongo, J.; Fujita, A.; Sato, C.; Kitajima, K.; Lehmann, F.; Oschlies, M.; Gerardy-Schahn, R.; Munster-Kuhnel, A.K. The rainbow trout CMP-sialic acid synthetase utilises a nuclear localization signal different from that identified in the mouse enzyme. Glycobiology 2007, 17, 945–954. [Google Scholar]
- Schaper, W.; Bentrop, J.; Ustinova, J.; Blume, L.; Kats, E.; Tiralongo, J.; Weinhold, B.; Bastmeyer, M.; Munster-Kuhnel, A.K. Identification and biochemical characterization of two functional CMP-sialic acid synthetases in danio rerio. J. Biol. Chem 2012, 287, 13239–13248. [Google Scholar]
- Kaneko, Y.; Nimmerjahn, F.; Ravetch, J.V. Anti-inflammatory activity of immunoglobulin G resulting from Fc sialylation. Science 2006, 313, 670–673. [Google Scholar]
- Kannagi, R.; Izawa, M.; Koike, T.; Miyazaki, K.; Kimura, N. Carbohydrate-mediated cell adhesion in cancer metastasis and angiogenesis. Cancer Sci 2004, 95, 377–384. [Google Scholar]
- Duncker, I.M.; Salinas-Marin, R.; Martinez-Duncker, C. Towards in vivo imaging of cancer sialylation. Int. J. Mol. Imaging 2011, 2011, 1–10. [Google Scholar]
- Fernández-Briera, A.; Garzia-Parcerio, I.; Cuevas, E.; Gili-Martin, E. Effect of human colorectal carcinogenesis on the neural cell adhesion molecule expression and polysialylation. Oncology 2010, 78, 196–204. [Google Scholar]
- Otsuka, H.; Uchimura, E.; Koshino, H.; Okano, T.; Kataoka, K. Anomalous binding profile of phenylboronic acid with N-acetylneuraminic acid (Neu5Ac) in aqueous solution with varying pH. J. Am. Chem. Soc 2003, 125, 3493–3502. [Google Scholar]
- Liu, A.; Peng, S.; Soo, J.C.; Kuang, M.; Chen, P.; Duan, H. Quantum dots with phenylboronic acid tags for specific labeling of sialic acids on living cells. Anal. Chem 2011, 83, 1124–1130. [Google Scholar]
- Crich, S.G.; Alberti, D.; Szabo, I.; Aime, S.; Djanashvili, K. MRI visualization of melanoma cells by targeting overexpressed sialic acid with a GdIII-dota-en-pba imaging reporter. Angew. Chem. Int. Ed 2013, 52, 1161–1164. [Google Scholar]
- Liu, H.; Li, Y.; Sun, K.; Fan, J.; Zhang, P.; Meng, J.; Wang, S.; Jiang, L. Dual-responsive surfaces modified with phenylboronic acid-containing polymer brush to reversibly capture and release cancer cells. J. Am. Chem. Soc 2013, 135, 7603–7609. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, X.; Xia, N.; Liu, L. Boronic Acid-Based Approach for Separation and Immobilization of Glycoproteins and Its Application in Sensing. Int. J. Mol. Sci. 2013, 14, 20890-20912. https://doi.org/10.3390/ijms141020890
Wang X, Xia N, Liu L. Boronic Acid-Based Approach for Separation and Immobilization of Glycoproteins and Its Application in Sensing. International Journal of Molecular Sciences. 2013; 14(10):20890-20912. https://doi.org/10.3390/ijms141020890
Chicago/Turabian StyleWang, Xiaojin, Ning Xia, and Lin Liu. 2013. "Boronic Acid-Based Approach for Separation and Immobilization of Glycoproteins and Its Application in Sensing" International Journal of Molecular Sciences 14, no. 10: 20890-20912. https://doi.org/10.3390/ijms141020890



