Ultrastructural Analysis of Nanogold-Labeled Cell Surface Microvilli in Liquid by Atmospheric Scanning Electron Microscopy and Their Relevance in Cell Adhesion
Abstract
:1. Introduction
2. Results and Discussion
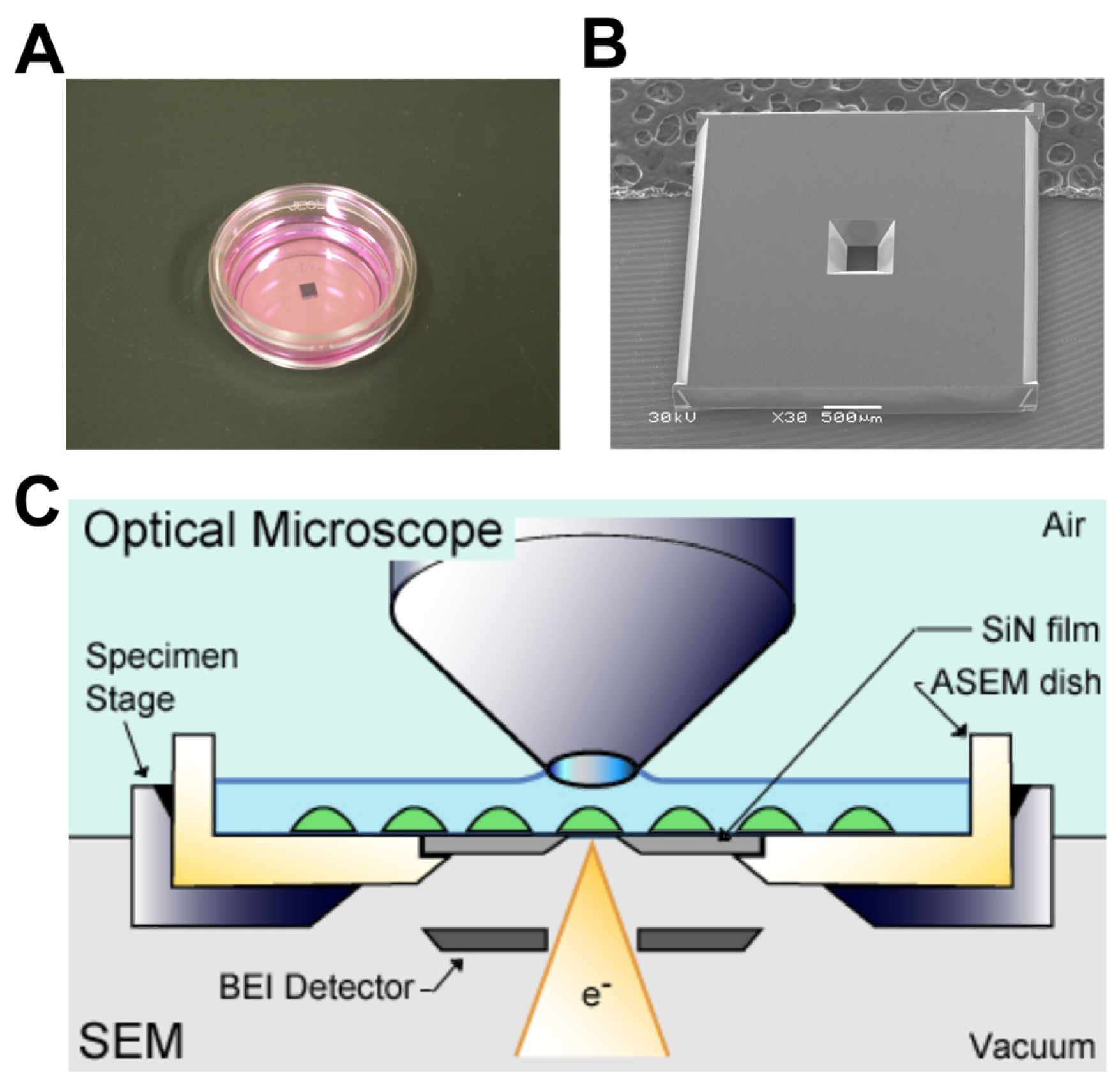
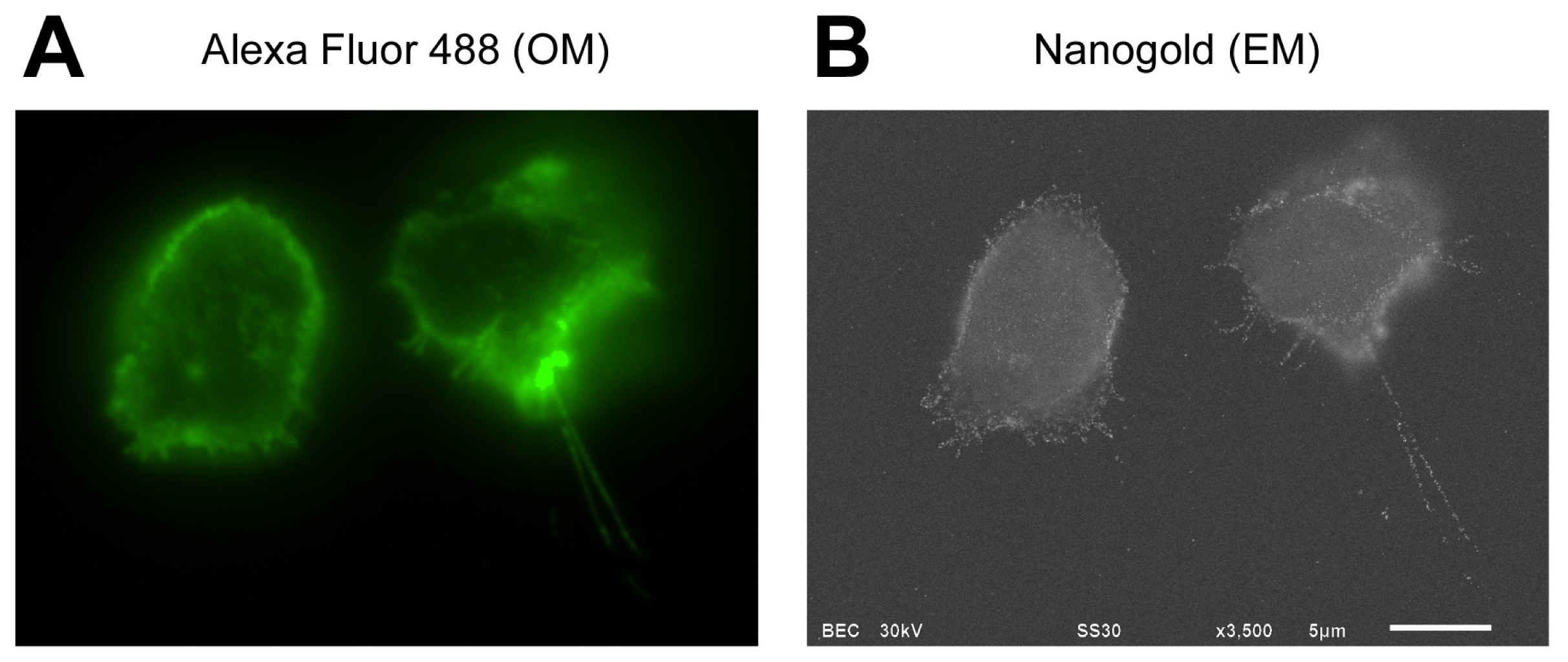
2.1. Correlative Observation of CD44 Distribution on Lymphocyte Cell Surface by ASEM
2.2. Disruption of Cytoskeletal Structure with Cytochalasin D Abrogates Microvilli Formation and CD44 Activity
2.3. Ultrastructure of Lymphocyte Cell Surface Visualized by Positively Charged Gold Particles
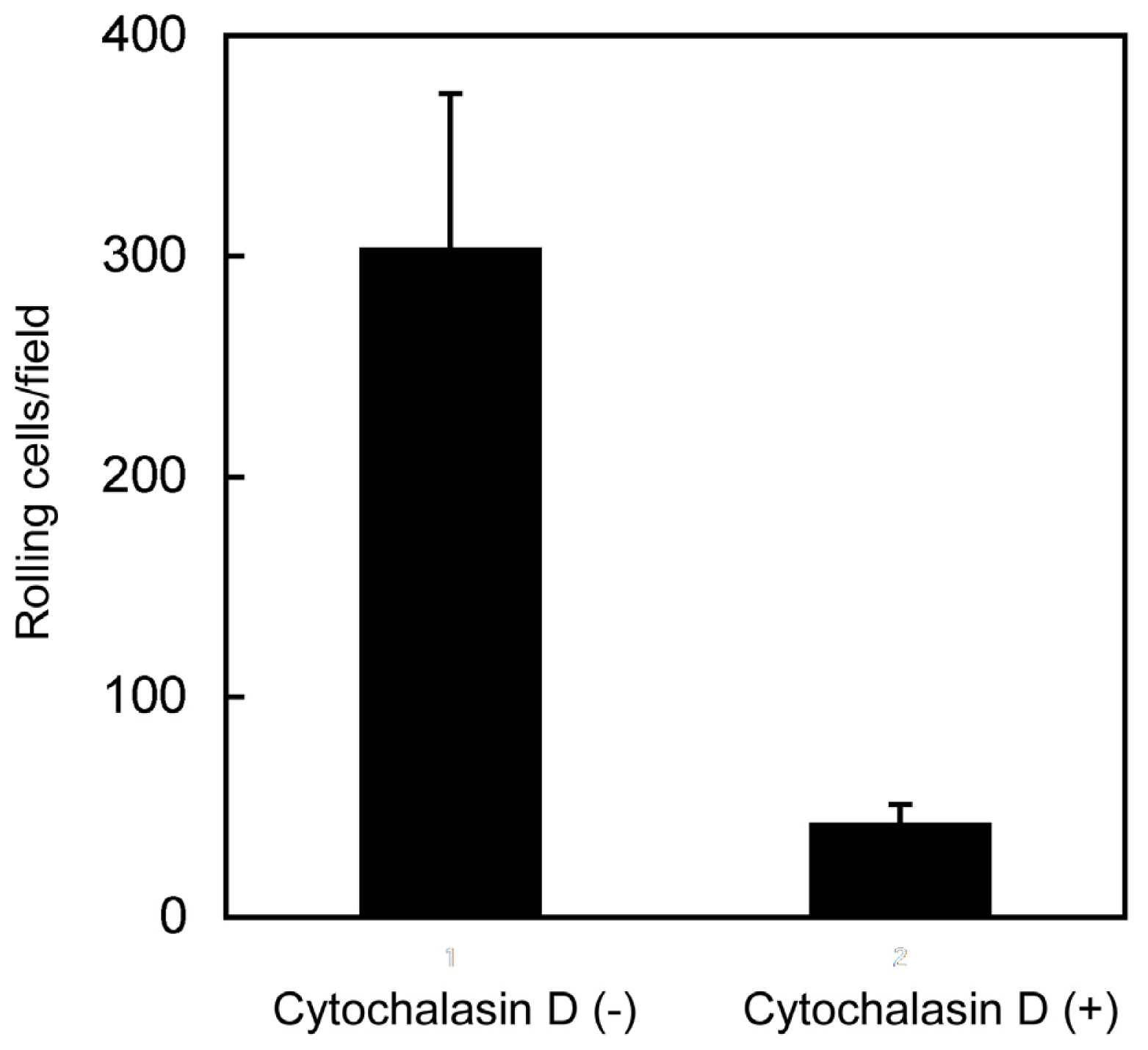
2.4. Disruption of Cytoskeletal Structure with Cytochalasin D Abrogates CD44-Mediated Rolling under Flow Conditions
3. Experimental Section
3.1. Reagents
3.2. Cell Culture
3.3. Cell Staining
3.4. ASEM
3.5. Flow Cytometry
3.6. Shear Flow Assay
4. Conclusions



Acknowledgments
Conflicts of Interest
References
- Springer, T.A. Traffic signals for lymphocyte recirculation and leukocyte emigration: The multistep paradigm. Cell 1994, 76, 301–314. [Google Scholar]
- Clark, R.A.; Alon, R.; Springer, T.A. CD44 and hyaluronan-dependent rolling interactions of lymphocytes on tonsillar stroma. J. Cell Biol 1996, 134, 1075–1087. [Google Scholar]
- Lesley, J.; Hyman, R.; Kincade, P.W. CD44 and its interaction with extracellular matrix. Adv. Immunol 1993, 54, 271–335. [Google Scholar]
- Maiti, A.; Maki, G.; Johnson, P. TNF-α induction of CD44-mediated leukocyte adhesion by sulfation. Science 1998, 282, 941–943. [Google Scholar]
- Brown, K.L.; Maiti, A.; Johnson, P. Role of sulfation in CD44-mediated hyaluronan binding induced by inflammatory mediators in human CD14+ peripheral blood monocytes. J. Immunol 2001, 167, 5367–5374. [Google Scholar]
- Murai, T.; Miyauchi, T.; Yanagida, T.; Sako, Y. Epidermal growth factor-regulated activation of Rac GTPase enhances CD44 cleavage by metalloproteinase disintegrin ADAM10. Biochem. J 2006, 395, 65–71. [Google Scholar]
- Ruffell, B.; Poon, G.F.T.; Lee, S.S.M.; Brown, K.L.; Tjew, S.L.; Cooper, J.; Johnson, P. Differential use of chondroitin sulfate to regulate hyaluronan binding by receptor CD44 in inflammatory and interleukin 4-activated macrophages. J. Biol. Chem 2011, 286, 19179–19190. [Google Scholar]
- Pospieszalska, M.K.; Ley, K. Dynamics of microvillus extension and tether formation in rolling leukocytes. Cell. Mol. Bioeng 2009, 2, 207–217. [Google Scholar]
- Gustafsson, M.G. Nonlinear structured-illumination microscopy: Wide-field fluorescence imaging with theoretically unlimited resolution. Proc. Natl. Acad. Sci. USA 2005, 102, 13081–13086. [Google Scholar]
- Betzig, E.; Patterson, G.H.; Sougrat, R.; Lindwasser, O.W.; Olenych, S.; Bonifacino, J.S.; Davidson, M.W.; Lippincott-Schwartz, J.; Hess, H.F. Imaging intracellular fluorescent proteins at nanometer resolution. Science 2006, 313, 1642–1645. [Google Scholar]
- Hell, S.W. Far-field optical nanoscopy. Science 2007, 316, 1153–1158. [Google Scholar]
- Beesley, J.E.; Pearson, J.D.; Hutchings, A.; Carleton, J.S.; Gordon, J.L. Granulocyte migration through endothelium in culture. J. Cell Sci 1979, 38, 237–248. [Google Scholar]
- Picker, L.J.; Warnock, R.A.; Burns, A.R.; Doerschuk, C.M.; Berg, E.L.; Butcher, E.C. The neutrophil selectin LECAM-1 presents carbohydrate ligands to the vascular selectins ELAM-1 and GMP-140. Cell 1991, 66, 921–933. [Google Scholar]
- Erlandsen, S.L.; Hasslen, S.R.; Nelson, R.D. Detection and spatial distribution of the 2 integrin (Mac-1) and L-selectin (LECAM-1) adherence receptors on human neutrophils by high-resolution field emission SEM. J. Histochem. Cytochem 1993, 41, 327–333. [Google Scholar]
- Hasslen, S.R.; von Andrian, U.H.; Butcher, E.C.; Nelson, R.D.; Erlandsen, S.L. Spatial distribution of L-selectin (CD62L) on human lymphocytes and transfected murine L1–2 cells. Histochem. J 1995, 27, 547–554. [Google Scholar]
- Bruehl, R.E.; Springer, T.A.; Bainton, D.F. Quantitation of l-selectin distribution on human leukocyte microvilli by immunogold labeling and electron microscopy. J. Histochem. Cytochem 1996, 44, 835–844. [Google Scholar]
- Abrams, I.M.; McBrain, J.W. A closed cell for electron microscopy. J. Appl. Phys. 1944, 15, 607–609. [Google Scholar]
- Thiberge, S.; Nechushtan, A.; Sprinzak, D.; Gileadi, O.; Behar, V.; Zik, O.; Chowers, Y.; Michaeli, S.; Schlessinger, J.; Moses, E. Scanning electron microscopy of cells and tissues under fully hydrated conditions. Proc. Natl. Acad. Sci. USA 2004, 101, 3346–3351. [Google Scholar]
- De Jonge, N.; Peckys, D.B.; Kremers, G.J.; Piston, D.W. Electron microscopy of whole cells in liquid with nanometer resolution. Proc. Natl. Acad. Sci. USA 2009, 106, 2159–2164. [Google Scholar]
- De Jonge, N.; Ross, F.M. Electron microscopy of specimens in liquid. Nat. Nanotechnol 2011, 6, 695–704. [Google Scholar]
- Evans, J.E.; Jungjohann, K.L.; Wong, P.C.K.; Chiu, P.L.; Dutrow, G.H.; Arslan, I.; Browning, N.D. Visualizing macromolecular complexes with in situ liquid scanning transmission electron microscopy. Micron 2012, 43, 1085–1090. [Google Scholar]
- Inami, W.; Nakajima, K.; Miyakawa, A.; Kawata, Y. Electron beam excitation assisted optical microscope with ultra-high resolution. Opt. Express 2010, 18, 12897–12902. [Google Scholar]
- Nishiyama, H.; Suga, M.; Ogura, T.; Maruyama, Y.; Koizumi, M.; Mio, K.; Kitamura, S.; Sato, C. Atmospheric scanning electron microscope observes cells and tissues in open medium through silicon nitride film. J. Struct. Biol 2010, 169, 438–449. [Google Scholar]
- Suga, M.; Nishiyama, H.; Konyuba, Y.; Iwamatsu, S.; Watanabe, Y.; Yoshiura, C.; Ueda, T.; Sato, C. The atmospheric scanning electron microscope with open sample space observes dynamic phenomena in liquid or gas. Ultramicroscopy 2011, 111, 1650–1658. [Google Scholar]
- Murai, T.; Maruyama, Y.; Mio, K.; Nishiyama, H.; Suga, M.; Sato, C. Low cholesterol triggers membrane microdomain-dependent CD44 shedding and suppresses tumor cell migration. J. Biol. Chem 2011, 286, 1999–2007. [Google Scholar]
- Murai, T.; Sato, C.; Sato, M.; Nishiyama, H.; Suga, M.; Mio, K.; Kawashima, H. Membrane cholesterol modulates the hyaluronan-binding ability of CD44 in T lymphocytes and controls rolling under shear flow. J. Cell Sci 2013, 126, 3284–3294. [Google Scholar]
- Sato, C.; Manaka, S.; Nakane, D.; Nishiyama, H.; Suga, M.; Nishizaka, T.; Miyata, M.; Maruyama, Y. Rapid imaging of mycoplasma in solution using atmospheric scanning electron microscopy (ASEM). Biochem. Biophys. Res. Commun 2012, 417, 1213–1218. [Google Scholar]
- Maruyama, Y.; Ebihara, T.; Nishiyama, H.; Konyuba, Y.; Senda, M.; Numaga-Tomita, T.; Senda, T.; Suga, M.; Sato, C. Direct observation of protein microcrystals in crystallization buffer by atmospheric scanning electron microscopy. Int. J. Mol. Sci 2012, 13, 10553–10567. [Google Scholar]
- Sartori, A.; Gatz, R.; Beck, F.; Rigort, A.; Baumeister, W.; Plitzko, J.M. Correlative microscopy: Bridging the gap between fluorescence light microscopy and cryo-electron tomography. J. Struct. Biol 2007, 160, 135–145. [Google Scholar]
- Agronskaia, A.V.; Valentijn, J.A.; van Driel, L.F.; Schneijdenberg, C.T.W.M.; Humbel, B.M.; van Bergen en Henegouwen, P.M.P.; Verkleij, A.J.; Koster, A.J.; Gerritsen, H.C. Integrated fluorescence and transmission electron microscopy. J. Struct. Biol 2008, 164, 183–189. [Google Scholar]
- Hwang, H.L.; Hwu, C.C.; Liue, J.C.; Lin, H.H. Analysis of low-pressure chemical vapor deposited silicon nitride by Rutherford backscattering spectrometry. Appl. Phys. Lett. 1982, 41, 844–846. [Google Scholar]
- Peckys, D.B.; Baun, J.P.; Eder, M.; Wermer, U.; de Jonge, N. Epidermal growth factor receptor subunit locations determined in hydrated cells with environmental scanning electron microscopy. Sci. Rep 2013. [Google Scholar] [CrossRef]
- Iancu, C.V.; Wright, E.R.; Heymann, J.B.; Jensen, G.J. A comparison of liquid nitrogen and liquid helium as cryogens for electron cryotomography. J. Struct. Biol 2006, 153, 231–240. [Google Scholar]
- Inaga, S.; Katsumoto, T.; Tanaka, K.; Kameie, T.; Nakane, H.; Naguro, T. Platinum blue as an alternative to uranyl acetate for staining in transmission electron microscopy. Arch. Histol. Cytol 2007, 70, 43–49. [Google Scholar]
- Inaga, S.; Hirashima, S.; Tanaka, K.; Katsumoto, T.; Kameie, T.; Nakane, H.; Naguro, T. Low vacuum scanning electron microscopy for paraffin sections utilizing the differential stainability of cells and tissues with platinum blue. Arch. Histol. Cytol 2009, 72, 101–106. [Google Scholar]
- Murai, T. The role of lipid rafts in cancer cell adhesion and migration. Int. J. Cell Biol 2012, 2012, 763283:1–763283:6. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Murai, T.; Sato, M.; Nishiyama, H.; Suga, M.; Sato, C. Ultrastructural Analysis of Nanogold-Labeled Cell Surface Microvilli in Liquid by Atmospheric Scanning Electron Microscopy and Their Relevance in Cell Adhesion. Int. J. Mol. Sci. 2013, 14, 20809-20819. https://doi.org/10.3390/ijms141020809
Murai T, Sato M, Nishiyama H, Suga M, Sato C. Ultrastructural Analysis of Nanogold-Labeled Cell Surface Microvilli in Liquid by Atmospheric Scanning Electron Microscopy and Their Relevance in Cell Adhesion. International Journal of Molecular Sciences. 2013; 14(10):20809-20819. https://doi.org/10.3390/ijms141020809
Chicago/Turabian StyleMurai, Toshiyuki, Mari Sato, Hidetoshi Nishiyama, Mitsuo Suga, and Chikara Sato. 2013. "Ultrastructural Analysis of Nanogold-Labeled Cell Surface Microvilli in Liquid by Atmospheric Scanning Electron Microscopy and Their Relevance in Cell Adhesion" International Journal of Molecular Sciences 14, no. 10: 20809-20819. https://doi.org/10.3390/ijms141020809




