Molecular Research in Penile Cancer—Lessons Learned from the Past and Bright Horizons of the Future?
Abstract
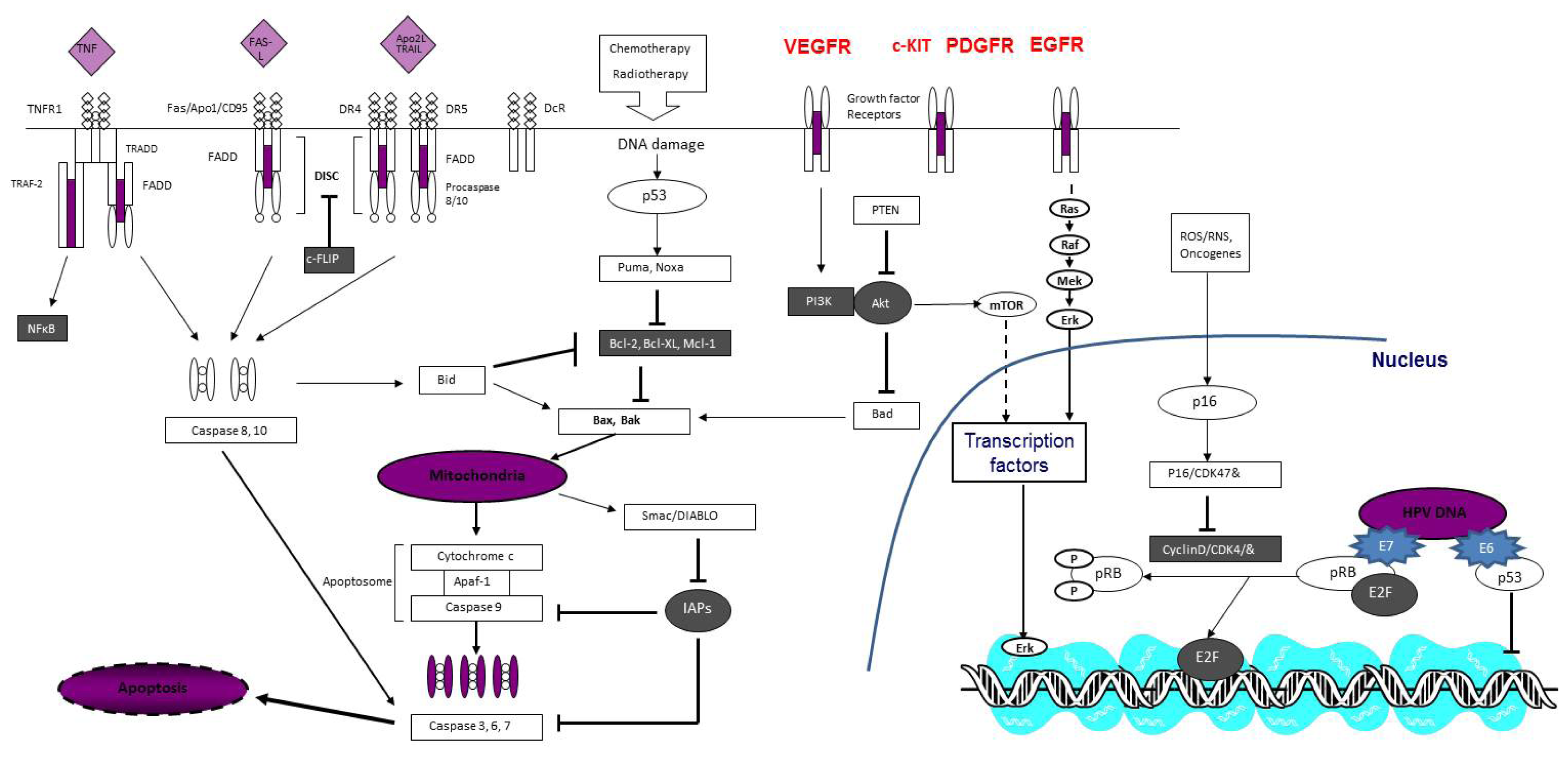
:1. Introduction
2. What Do We Focus on?
- Carcinogenesis (escape from apoptosis, tumor suppressor genes, and immune defense mechanisms),
- Tumor progression (invasion, transformation),
- Metastatic spread and metastatic seeding (resistance to environmental influences i.e., chemoresistance).
3. Molecular Mechanisms of Penile Cancer Progression
3.1. Carcinogenesis
3.2. Progression and Invasion
3.3. Metastasis and Resistance to Environmental Influences
4. Conclusions
| Carcinogenesis | Proliferation/Invasion | Metastases |
|---|---|---|
| Inflammation | Growth factors/receptors | Metastases suppressor genes |
| COX-2 | EGFR | KAI1 |
| PGE-2 | HER-3/HER-4 | Nm23H1 |
| Tumor suppressor genes | VEGF | |
| p53 | PI3K/PTEN/AKT | |
| p16 | EMT | |
| PTEN | MMP2/MMP9 | |
| Oncogenes | E-cadherin | |
| HPV E6/E7 | TenascinC | |
| MYC | Annexins | |
| Apoptosis/cell death | Glut1 | |
| DR4/DR5 | ||
| Bcl-2/BAX | ||
| p53 | ||
| Telomerases |
Conflicts of Interest
References
- Cancer Incidence in Five Continents Vol VIII; Parkin, D.M.; Whelan, S.L.; Ferlay, J.; Teppo, L.; Thomas, D.B (Eds.) IARC Scientific Publications No 155; Lyon, France, 2002.
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar]
- Dillner, J.; von Krogh, G.; Horenblas, S.; Meijer, C.J. Etiology of squamous cell carcinoma of the penis. Scand. J. Urol. Nephrol. Suppl 2000, 205, 189–193. [Google Scholar]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar]
- Lee, H.N.; Na, H.K.; Surh, Y.J. Resolution of inflammation as a novel chemopreventive strategy. Semin. Immunopathol 2013, 35, 151–161. [Google Scholar]
- Poetsch, M.; Hemmerich, M.; Kakies, C.; Kleist, B.; Wolf, E.; vom Dorp, F.; Hakenberg, O.W.; Protzel, C. Alterations in the tumor suppressor gene p16(INK4A) are associated with aggressive behavior of penile carcinomas. Virchows Arch 2011, 458, 221–229. [Google Scholar]
- Castellone, M.D.; Teramoto, H.; Williams, B.O.; Druey, K.M.; Gutkind, J.S. Prostaglandin E2 promotes colon cancer cell growth through a Gs-axin-beta-catenin signaling axis. Science 2005, 310, 1504–1510. [Google Scholar]
- Golijanin, D.; Tan, J.Y.; Kazior, A.; Cohen, E.G.; Russo, P.; Dalbagni, G.; Auborn, K.J.; Subbaramaiah, K.; Dannenberg, A.J. Cyclooxygenase-2 and microsomal prostaglandin E synthase-1 are overexpressed in squamous cell carcinoma of the penis. Clin. Cancer Res 2004, 10, 1024–1031. [Google Scholar]
- Madsen, B.S.; van den Brule, A.J.; Jensen, H.L.; Wohlfahrt, J.; Frisch, M. Risk factors for squamous cell carcinoma of the penis—Population-based case-control study in Denmark. Cancer Epidemiol. Biomark. Prev 2008, 17, 2683–2691. [Google Scholar]
- Briitebo, E.B.; Loefberg, B.; Tjaelve, H. Sites of metabolism of N-Nitrosodiethylamine in mice. Chem. Biol. Interact 1981, 34, 209–221. [Google Scholar]
- Masferrer, E.; Ferrandiz-Pulido, C.; Lloveras, B.; Masferrer-Niubò, M.; Espinet, B.; Salido, M.; Rodríguez-Rivera, M.; Alemany, L.; Placer, J.; Gelabert, A.; et al. MYC copy number gains are associated with poor outcome in penile squamous cell carcinoma. J. Urol 2012, 188, 1965–1971. [Google Scholar]
- zur Hausen, H. Papillomaviruses in the causation of human cancers—A brief historical account. Virology 2009, 384, 260–265. [Google Scholar]
- Rubin, M.A.; Kleter, B.; Zhou, M.; Ayala, G.; Cubilla, A.L.; Quint, W.G.; Pirog, E.C. Detection and typing of human papillomavirus DNA in penile carcinoma: Evidence for multiple independent pathways of penile carcinogenesis. Am. J. Pathol 2001, 159, 1211–1218. [Google Scholar]
- Prowse, D.M.; Ktori, E.N.; Chandrasekaran, D.; Prapa, A.; Baithun, S. Human papillomavirus-associated increase in p16INK4A expression in penile lichen sclerosus and squamous cell carcinoma. Br. J. Dermatol 2008, 158, 261–265. [Google Scholar]
- Poetsch, M.; Schuart, B.J.; Schwesinger, G.; Kleist, B.; Protzel, C. Screening of microsatellite markers in penile cancer reveals differences between metastatic and nonmetastatic carcinomas. Mod. Pathol 2007, 20, 1069–1077. [Google Scholar]
- Alves, G.; Heller, A.; Fiedler, W.; Campos, M.M.; Claussen, U.; Ornellas, A.A.; Liehr, T. Genetic imbalances in 26 cases of penile squamous cell carcinoma. Genes Chromosomes Cancer 2001, 31, 48–53. [Google Scholar]
- Peter, M.; Rosty, C.; Couturier, J.; Radvanyi, F.; Teshima, H.; Sastre-Garau, X. MYC activation associated with the integration of HPV DNA at the MYC locus in genital tumors. Oncogene 2006, 25, 5985–5993. [Google Scholar]
- Tobian, A.A.; Serwadda, D.; Quinn, T.C.; Kigozi, G.; Gravitt, P.E.; Laeyendecker, O.; Charvat, B.; Ssempijja, V.; Riedesel, M.; Oliver, A.E.; et al. Male circumcision for the prevention of HSV-2 and HPV infections and syphilis. N. Engl. J. Med 2009, 360, 1298–1309. [Google Scholar]
- Protzel, C.; Hakenberg, O.W. Emerging apoptosis agonists for bladder cancer. Expert Opin. Emerg. Drugs 2009, 14, 607–618. [Google Scholar]
- Ashkenazi, A. Directing cancer cells to self-destruct with pro-apoptotic receptor agonists. Nat. Rev. Drug Discov 2008, 7, 1001–1012. [Google Scholar]
- Jorg, M.; Becker, U.; Hartmann, A.; Knoell, A.; Burger, M.; Junker, K. Correlation of genomic alterations and hpv infetion in penile carcinomas. J. Urol 2013, 189, e389. [Google Scholar]
- Martins, A.C.; Faria, S.M.; Cologna, A.J.; Suaid, H.J.; Tucci, S., Jr. Immunoexpression of p53 protein and proliferating cell nuclear antigen in penile carcinoma. J. Urol 2002, 167, 89–92, discussion 92–83. [Google Scholar]
- Lopes, A.; Bezerra, A.L.; Pinto, C.A.; Serrano, S.V.; de Mell, O.C.; Villa, L.L. p53 as a new prognostic factor for lymph node metastasis in penile carcinoma: Analysis of 82 patients treated with amputation and bilateral lymphadenectomy. J. Urol 2002, 168, 81–86. [Google Scholar]
- Saeed, S.; Keehn, C.A.; Khalil, F.K.; Morgan, M.B. Immunohistochemical expression of Bax and Bcl-2 in penile carcinoma. Ann. Clin. Lab. Sci 2005, 35, 91–96. [Google Scholar]
- Blasco, M.A. Telomeres and human disease: Ageing, cancer and beyond. Nat. Rev. Genet 2005, 6, 611–622. [Google Scholar]
- Alves, G.; Fiedler, W.; Guenther, E.; Nascimento, P.; Campos, M.M.; Ornellas, A.A. Determination of telomerase activity in squamous cell carcinoma of the penis. Int. J. Oncol 2001, 18, 67–70. [Google Scholar]
- Protzel, C.; Knoedel, J.E.; Zimmermann, U.; Klebingat, K.J.; Giebel, J.; Woenckhaus, C. Expression of proliferation marker Ki67 correlates to occurrence of metastasis and prognosis, histological subtypes and HPV DNA detection in penile carcinomas. Histol. Histopathol 2007, 22, 1197–1204. [Google Scholar]
- Borgermann, C.; Schmitz, K.J.; Sommer, S.; Rubben, H.; Krege, S. Characterization of the EGF receptor status in penile cancer: Retrospective analysis of the course of the disease in 45 patients. Urol. A 2009, 48, 1483–1489. [Google Scholar]
- Necchi, A.; Nicolai, N.; Colecchia, M.; Catanzaro, M.; Torelli, T.; Piva, L.; Salvioni, R. Proof of activity of anti-epidermal growth factor receptor-targeted therapy for relapsed squamous cell carcinoma of the penis. J. Clin. Oncol 2011, 29, e650–e652. [Google Scholar]
- Giannatempo, P.; Nicolai, N.; Raggi, D.; Farè, E.; Colecchiaet, M.; Catanzaro, M.; Torelli, T.; Biasoni, D.; Stagni, S.; Piva, L.; et al. Early results of the pilot study with the antiepidermal growth-factor receptor (EGFR) monoclonal antibody panitumumab in patients with multi-relapsed or refractory squamous cell carcinoma of the penis. J. Urol 2013, 189, e309. [Google Scholar]
- Gou, H.F.; Li, X.; Qiu, M.; Cheng, K.; Li, L.H.; Dong, H.; Chen, Y.; Tang, Y.; Gao, F.; Zhao, F.; et al. Epidermal growth factor receptor (EGFR)-RAS signaling pathway in penile squamous cell carcinoma. PLoS One 2013, 8, e62175. [Google Scholar]
- Stankiewicz, E.; Prowse, D.M.; Ng, M.; Cuzick, J.; Mesher, D.; Hiscock, F.; Lu, Y.J.; Watkin, N.; Corbishley, C.; Lam, W.; et al. Alternative HER/PTEN/Akt pathway activation in HPV positive and negative penile carcinomas. PLoS One 2011, 6, e17517. [Google Scholar]
- Andersson, P.; Kolaric, A.; Windahl, T.; Kirrander, P.; Soderkvist, P.; Karlsson, M.G. PIK3CA, HRAS and KRAS gene mutations in human penile cancer. J. Urol 2008, 179, 2030–2034. [Google Scholar]
- Sanchez-Tillo, E.; Liu, Y.; de Barrios, O.; Siles, L.; Fanlo, L.; Cuatrecasas, M.; Darling, D.S.; Dean, D.C.; Castells, A.; Postigo, A. EMT-activating transcription factors in cancer: Beyond EMT and tumor invasiveness. Cell. Mol. Life Sci 2012, 69, 3429–3456. [Google Scholar]
- Bullock, M.D.; Sayan, A.E.; Packham, G.K.; Mirnezami, A.H. MicroRNAs: Critical regulators of epithelial to mesenchymal (EMT) and mesenchymal to epithelial transition (MET) in cancer progression. Biol. Cell 2012, 104, 3–12. [Google Scholar]
- Bose, P.; Brockton, N.T.; Dort, J.C. Head and neck cancer: From anatomy to biology. Int. J. Cancer 2013, 133, 2013–2023. [Google Scholar]
- Campos, R.S.; Lopes, A.; Guimaraes, G.C.; Carvalho, A.L.; Soares, F.A. E-cadherin, MMP-2, and MMP-9 as prognostic markers in penile cancer: Analysis of 125 patients. Urology 2006, 67, 797–802. [Google Scholar]
- Zhu, Y.; Zhou, X.Y.; Yao, X.D.; Dai, B.; Ye, D.W. The prognostic significance of p53, Ki-67, epithelial cadherin and matrix metalloproteinase-9 in penile squamous cell carcinoma treated with surgery. BJU Int 2007, 100, 204–208. [Google Scholar]
- Protzel, C.; Richter, M.; Poetsch, M.; Kakies, C.; Zimmermann, U.; Woenckhaus, C.; Klebingat, K.J.; Hakenberg, O.W.; Giebel, J. The role of annexins I, II and IV in tumor development, progression and metastasis of human penile squamous cell carcinomas. World J. Urol 2011, 29, 393–398. [Google Scholar]
- Protzel, C.; Kakies, C.; Erbersdobler, A.; Spiess, P.E.; Hakenberg, O.W. Strong expression of glucosetransporter 1 in the invasion front correlates with aggressive behaviour in penile cancer. J. Urol 2013, 189, e390–e391. [Google Scholar]
- Qian, B.Z.; Pollard, J.W. Macrophage diversity enhances tumor progression and metastasis. Cell 2010, 141, 39–51. [Google Scholar]
- Valastyan, S.; Weinberg, R.A. Tumor metastasis: Molecular insights and evolving paradigms. Cell 2011, 147, 275–292. [Google Scholar]
- De Paula, A.A.; Motta, E.D.; de Alencar, R. C.; Saddi, V.A.; da Silva, R.C.; Caixeta, G.N.; Almeida Netto, J.C.; Carneiro, M.A. The impact of cyclooxygenase-2 and vascular endothelial growth factor C immunoexpression on the prognosis of penile carcinoma. J. Urol 2012, 187, 134–140. [Google Scholar]
- Rinker-Schaeffer, C.W.; Hickson, J.A. Stopping cancer before it colonizes. Nat. Med 2006, 12, 887–888. [Google Scholar]
- Protzel, C.; Kakies, C.; Kleist, B.; Poetsch, M.; Giebel, J. Down-regulation of the metastasis suppressor protein KAI1/CD82 correlates with occurrence of metastasis, prognosis and presence of HPV DNA in human penile squamous cell carcinoma. Virchows Arch 2008, 452, 369–375. [Google Scholar]
- Protzel, C.; Kakies, C.; Poetsch, M.; Giebel, J.; Wolf, E.; Hakenberg, O.W. Down-Regulation of metastasis suppressor gene nm23-H1 correlates with the occurence of metastases and poor prognosis in penile squamous cell carcinoma. J. Urol 2009, 181, e202. [Google Scholar]
- Lim, J.; Jang, G.; Kang, S.; Lee, G.; Nga do, T.T.; Phuong do, T.L.; Kim, H.; El-Rifai, W.; Ruley, H.E.; Jo, D. Cell-permeable NM23 blocks the maintenance and progression of established pulmonary metastasis. Cancer Res 2011, 71, 7216–7225. [Google Scholar]
- Zahreddine, H.; Borden, K.L. Mechanisms and insights into drug resistance in cancer. Front. Pharmacol 2013, 4. [Google Scholar] [CrossRef]
- Park, Y.; Kim, D.S.; Park, K.H.; Baek, S.K.; Kwon, S.Y.; Shin, S.W.; Jung, K.Y.; Kim, C.Y.; Kim, Y.H.; Lee, N.J.; et al. RASSF1A and ERCC1 expression levels might be predictive of prognosis in advanced, recurrent, and metastatic squamous cell carcinoma of the head and neck treated with docetaxel and cisplatin. Onkologie 2012, 35, 673–682. [Google Scholar]
- Sun, J.M.; Sung, J.Y.; Park, S.H.; Kwon, G.Y.; Jeong, B.C.; Seo, S.I.; Jeon, S.S.; Lee, H.M.; Jo, J.; Choi, H.Y.; et al. ERCC1 as a biomarker for bladder cancer patients likely to benefit from adjuvant chemotherapy. BMC Cancer 2012, 12. [Google Scholar] [CrossRef]
- Tiseo, M.; Bordi, P.; Bortesi, B.; Boni, L.; Boni, C.; Baldini, E.; Grossi, F.; Recchia, F.; Zanelli, F.; Fontanini, G.; et al. ERCC1/BRCA1 expression and gene polymorphisms as prognostic and predictive factors in advanced NSCLC treated with or without cisplatin. Br. J. Cancer 2013, 108, 1695–1703. [Google Scholar]
- Prislei, S.; Martinelli, E.; Mariani, M.; Raspaglio, G.; Sieber, S.; Ferrandina, G.; Shahabi, S.; Scambia, G.; Ferlini, C. MiR-200c and HuR in ovarian cancer. BMC Cancer 2013, 13. [Google Scholar] [CrossRef]
- Bonnet, D.; Dick, J.E. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat. Med 1997, 3, 730–737. [Google Scholar]
- Dick, J.E. Stem cell concepts renew cancer research. Blood 2008, 112, 4793–4807. [Google Scholar]
- Leal, J.A.; Lleonart, M.E. MicroRNAs and cancer stem cells: Therapeutic approaches and future perspectives. Cancer Lett. 2013, 338, 174–183. [Google Scholar]
- Chen, Y.C.; Chen, Y.W.; Hsu, H.S.; Tseng, L.M.; Huang, P.I.; Lu, K.H.; Chen, D.T.; Tai, L.K.; Yung, M.C.; Chang, S.C.; et al. Aldehyde dehydrogenase 1 is a putative marker for cancer stem cells in head and neck squamous cancer. Biochem. Biophys. Res. Commun 2009, 385, 307–313. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Protzel, C.; Spiess, P.E. Molecular Research in Penile Cancer—Lessons Learned from the Past and Bright Horizons of the Future? Int. J. Mol. Sci. 2013, 14, 19494-19505. https://doi.org/10.3390/ijms141019494
Protzel C, Spiess PE. Molecular Research in Penile Cancer—Lessons Learned from the Past and Bright Horizons of the Future? International Journal of Molecular Sciences. 2013; 14(10):19494-19505. https://doi.org/10.3390/ijms141019494
Chicago/Turabian StyleProtzel, Chris, and Philippe E. Spiess. 2013. "Molecular Research in Penile Cancer—Lessons Learned from the Past and Bright Horizons of the Future?" International Journal of Molecular Sciences 14, no. 10: 19494-19505. https://doi.org/10.3390/ijms141019494




