Isolation and Molecular Characterization of Thirteen R2R3-MYB Transcription Factors from Epimedium sagittatum
Abstract
:1. Introduction
2. Results
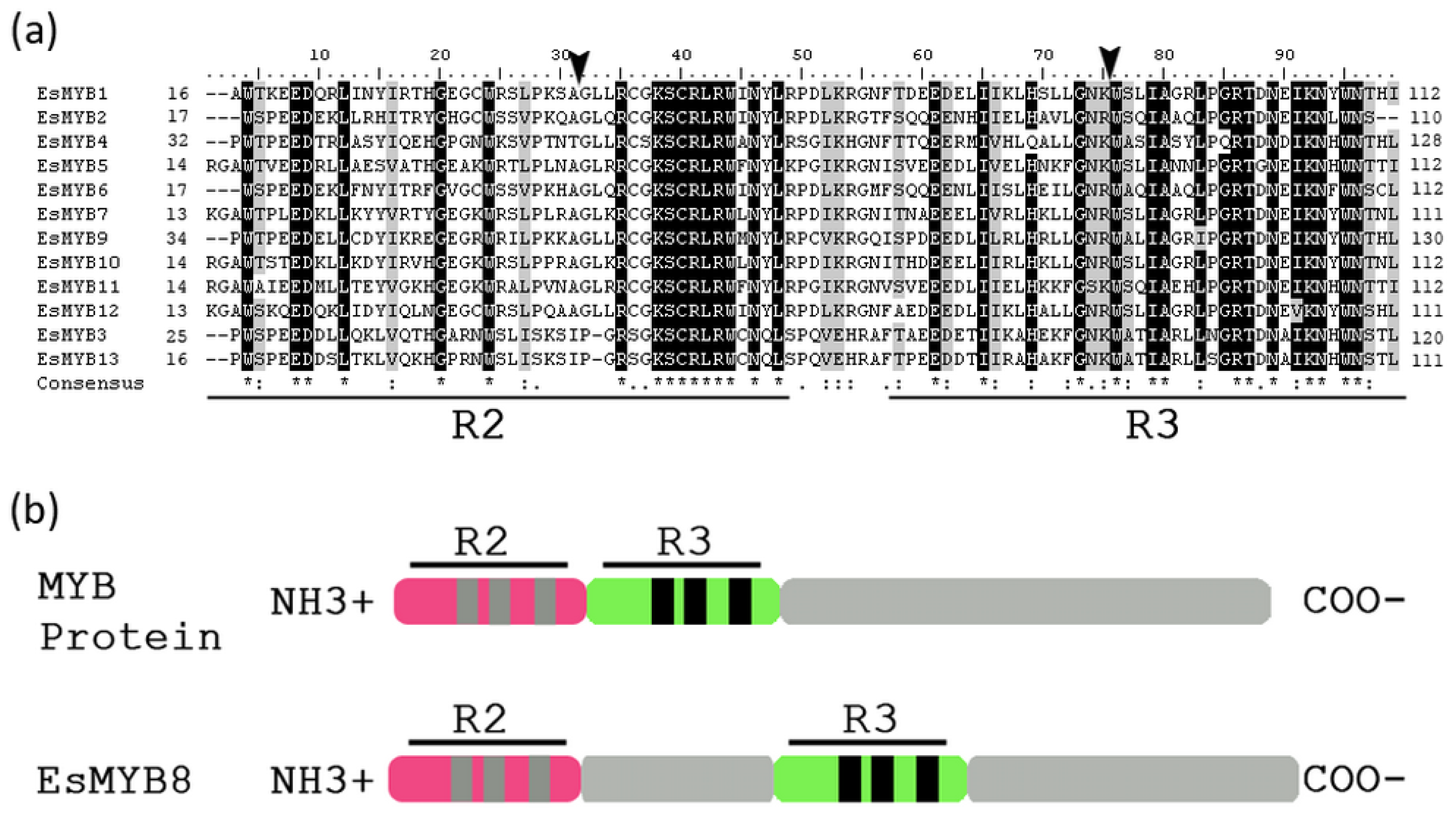
2.1. Isolation and Sequence Analysis of 13 R2R3-MYB Genes from Epimedium
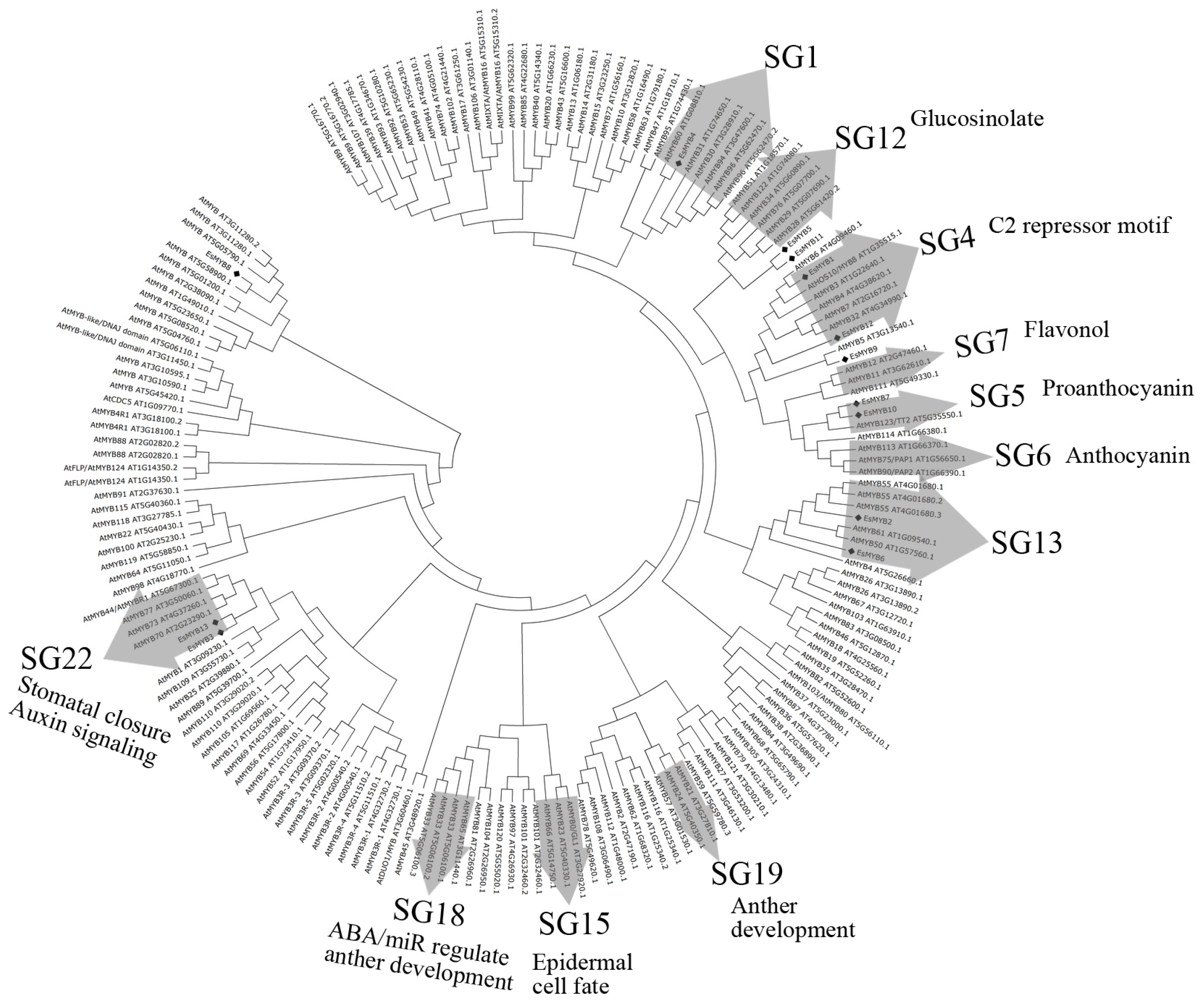
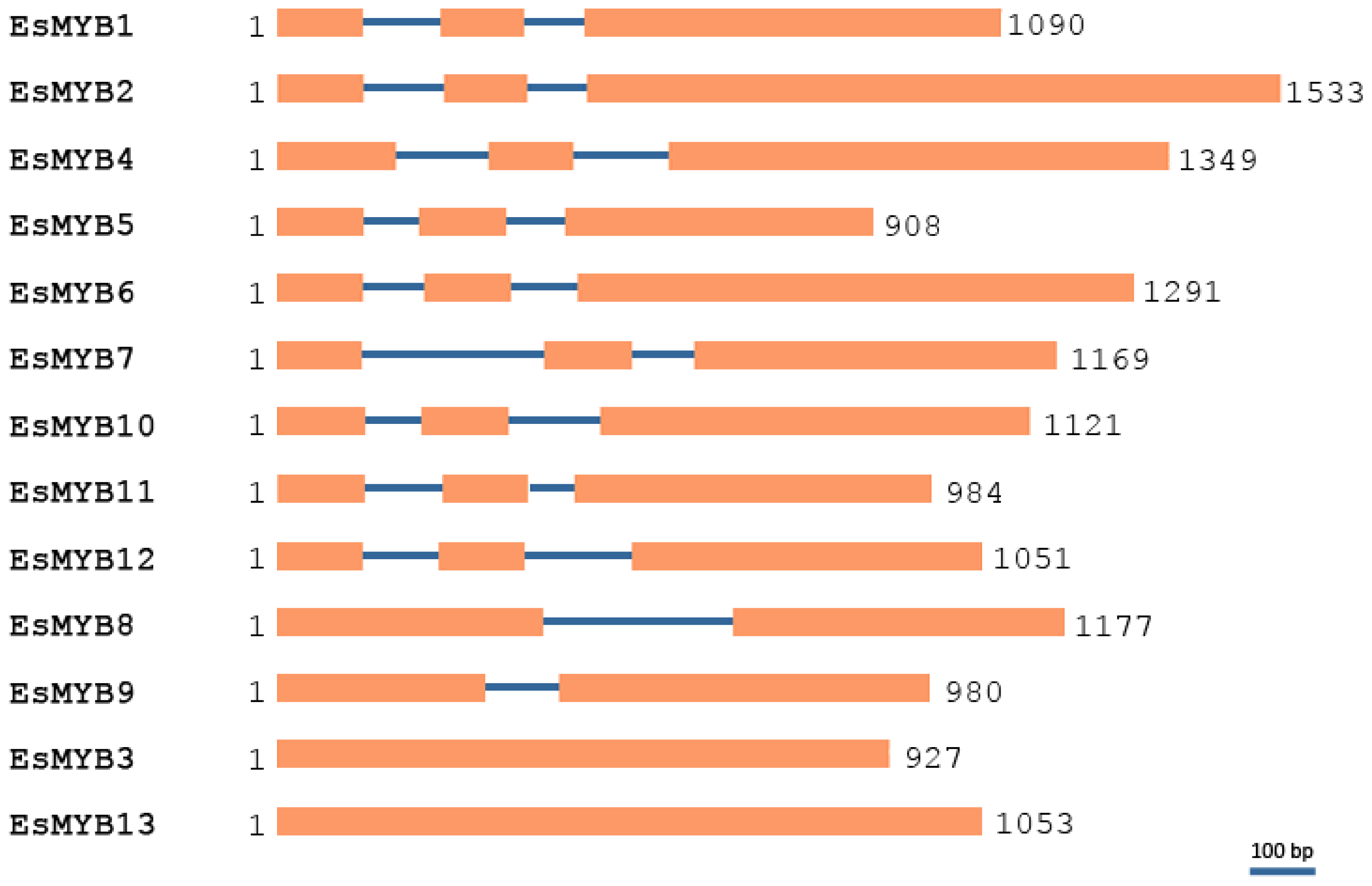
2.2. Phylogenetic Relationships and Genomic Structure Analysis of Epimedium R2R3-MYB Genes
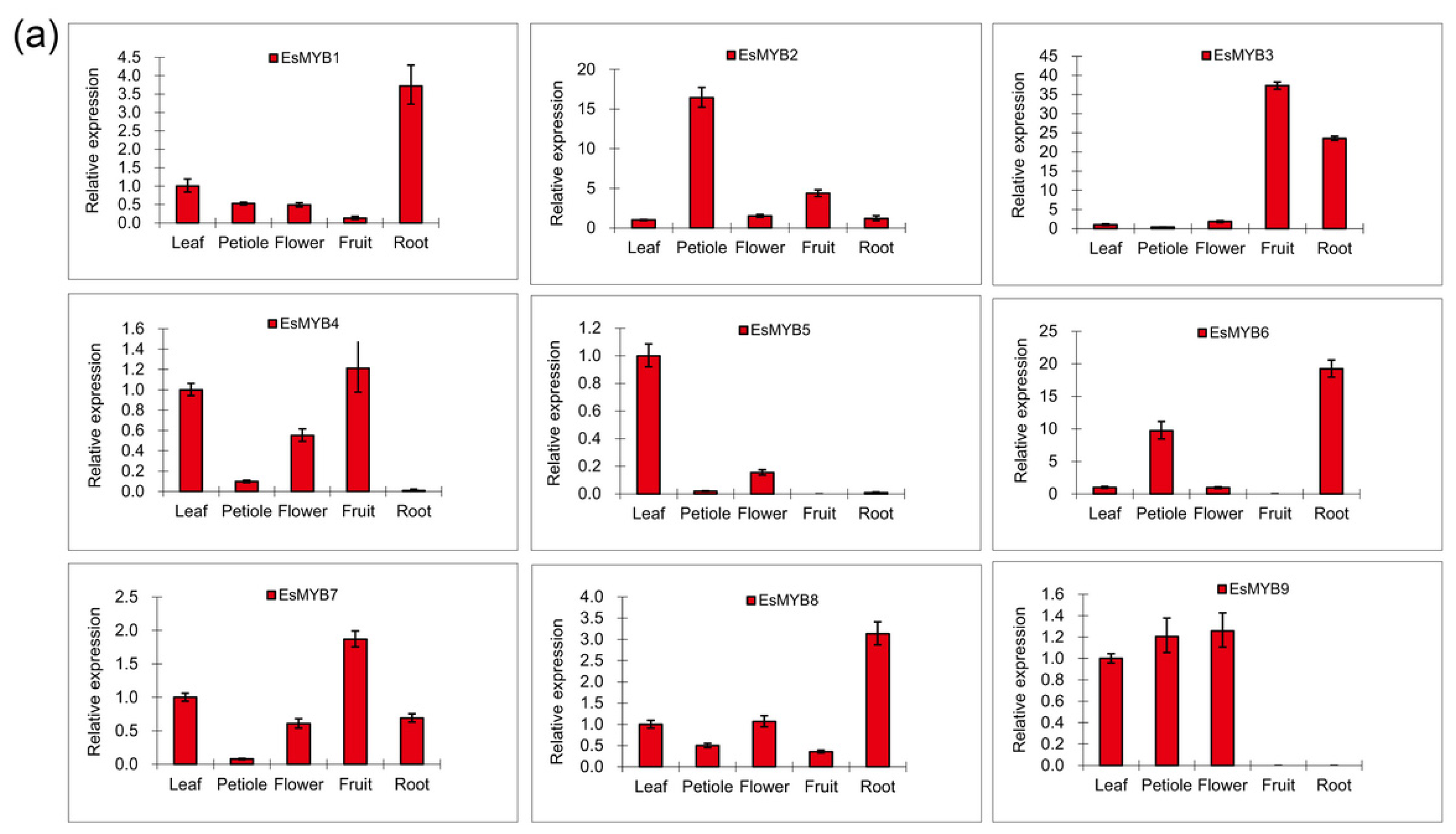
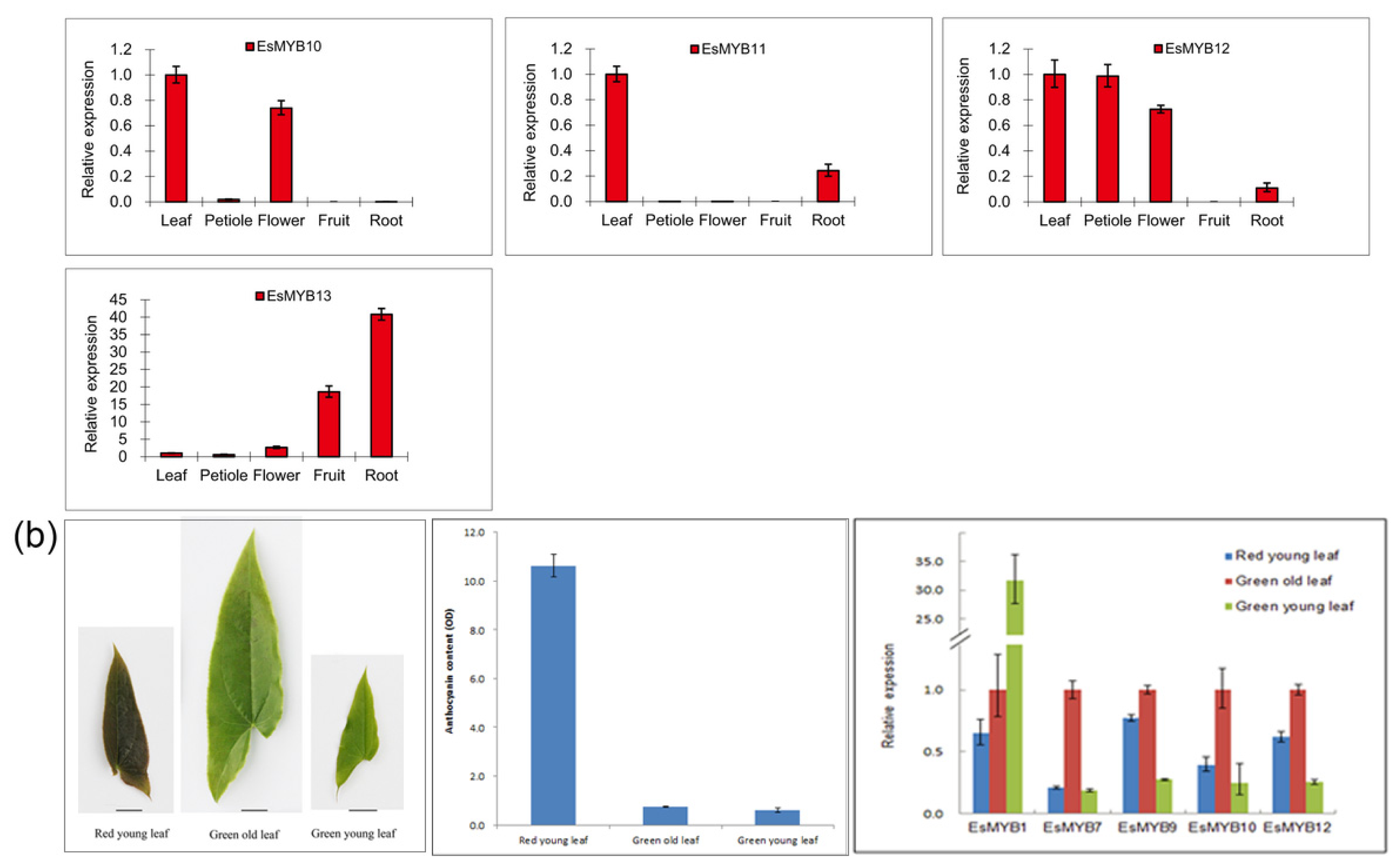
2.3. Expression Patterns of Epimedium R2R3-MYB Genes in Various Tissues
3. Discussion
4. Experimental Section
4.1. Plant Materials
4.2. DNA and RNA Extraction
4.3. Reverse Transcription and Isolation of R2R3-MYB cDNA Clones
4.4. Sequence and Phylogenetic Analysis
4.5. Quantitative RT-PCR
4.6. Total Anthocyanin Content Determination
5. Conclusions
Supplementary Information
ijms-14-00594-s001.pdfSupplementary Information
ijms-14-00594-s002.pdfAcknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Riechmann, J.L.; Heard, J.; Martin, G.; Reuber, L. Arabidopsis transcription factors: Genome-wide comparative analysis among eukaryotes. Science 2000, 290, 2105–2110. [Google Scholar]
- Dubos, C.; Stracke, R.; Grotewold, E.; Weisshaar, B.; Martin, C.; Lepiniec, L. MYB transcription factors in Arabidopsis. Trends Plant Sci 2010, 15, 573–581. [Google Scholar]
- Lipsick, J.S. One billion years of Myb. Oncogene 1996, 13, 223–235. [Google Scholar]
- Ogata, K.; Hojo, H.; Aimoto, S.; Nakai, T.; Nakamura, H.; Sarai, A.; Ishii, S.; Nishimura, Y. Solution structure of a DNA-binding unit of Myb: A helix-turn-helix-related motif with conserved tryptophans forming a hydrophobic core. Proc. Natl. Aca. Sci. USA 1992, 89, 6428–6432. [Google Scholar]
- Stracke, R.; Werber, M.; Weisshaar, B. The R2R3-MYB gene family in Arabidopsis thaliana. Curr. Opin. Plant Biol 2001, 4, 447–456. [Google Scholar]
- Jiang, C.; Gu, X.; Peterson, T. Identification of conserved gene structures and carboxy-terminal motifs in the Myb gene family of Arabidopsis and Oryza sativa L. ssp. indica. Genome Biol 2004, 5, R46. [Google Scholar]
- Wilkins, O.; Nahal, H.; Foong, J.; Provart, N.J.; Campbell, M.M. Expansion and diversification of the Populus R2R3-MYB family of transcription factors. Plant Physiol 2009, 149, 981–993. [Google Scholar]
- Matus, J.T.; Aquea, F.; Arce-Johnson, P. Analysis of the grape MYB R2R3 subfamily reveals expanded wine quality-related clades and conserved gene structure organization across Vitis and Arabidopsis genomes. BMC Plant Biol 2008, 8, 83. [Google Scholar]
- Deluc, L.; Bogs, J.; Walker, A.R.; Ferrier, T.; Decendit, A.; Merillon, J.M.; Robinson, S.P.; Barrieu, F. The transcription factor VvMYB5b contributes to the regulation of anthocyanin and proanthocyanidin biosynthesis in developing grape berries. Plant Physiol 2008, 147, 2041–2053. [Google Scholar]
- Jin, H.; Martin, C. Multifunctionality and diversity within the plant MYB-gene family. Plant Mol. Biol 1999, 41, 577–585. [Google Scholar]
- Paz-Ares, J.; Ghosal, D.; Wienand, U.; Peterson, P.; Saedler, H. The regulatory c1 locus of Zea mays encodes a protein with homology to myb proto-oncogene products and with structural similarities to transcriptional activators. EMBO J 1987, 6, 3553–3558. [Google Scholar]
- Hichri, I.; Barrieu, F.; Bogs, J.; Kappel, C.; Delrot, S.; Lauvergeat, V. Recent advances in the transcriptional regulation of the flavonoid biosynthetic pathway. J. Exp. Botany 2011, 62, 2465–2483. [Google Scholar]
- Nesi, N.; Jond, C.; Debeaujon, I.; Caboche, M.; Lepiniec, L. The Arabidopsis TT2 gene encodes an R2R3 MYB domain protein that acts as a key determinant for proanthocyanidin accumulation in developing seed. Plant Cell Online 2001, 13, 2099–2114. [Google Scholar]
- Borevitz, J.O.; Xia, Y.; Blount, J.; Dixon, R.A.; Lamb, C. Activation tagging identifies a conserved MYB regulator of phenylpropanoid biosynthesis. Plant Cell Online 2000, 12, 2383–2393. [Google Scholar]
- Bogs, J.; Jaffé, F.W.; Takos, A.M.; Walker, A.R.; Robinson, S.P. The grapevine transcription factor VvMYBPA1 regulates proanthocyanidin synthesis during fruit development. Plant Physiol 2007, 143, 1347–1361. [Google Scholar]
- Czemmel, S.; Stracke, R.; Weisshaar, B.; Cordon, N.; Harris, N.N.; Walker, A.R.; Robinson, S.P.; Bogs, J. The grapevine R2R3-MYB transcription factor VvMYBF1 regulates flavonol synthesis in developing grape berries. Plant Physiol 2009, 151, 1513–1530. [Google Scholar]
- Jeong, S.T.; Goto-Yamamoto, N.; Hashizume, K.; Kobayashi, S.; Esaka, M. Expression of VvmybA1 gene and anthocyanin accumulation in various grape organs. Am. J. Enol. Viticulture 2006, 57, 507–510. [Google Scholar]
- Gonzalez, A.; Zhao, M.; Leavitt, J.M.; Lloyd, A.M. Regulation of the anthocyanin biosynthetic pathway by the TTG1/bHLH/Myb transcriptional complex in Arabidopsis seedlings. Plant J 2008, 53, 814–827. [Google Scholar]
- Broun, P. Transcriptional control of flavonoid biosynthesis: A complex network of conserved regulators involved in multiple aspects of differentiation in Arabidopsis. Curr. Opin. Plant Biol 2005, 8, 272–279. [Google Scholar]
- Ramsay, N.A.; Glover, B.J. MYB-bHLH-WD40 protein complex and the evolution of cellular diversity. Trends Plant Sci 2005, 10, 63–70. [Google Scholar]
- Guo, B.; Xiao, P. Comment on main species of herba epimedii. China J. Chin. Mater. Medica 2003, 28, 303–307. [Google Scholar]
- Ma, H.; He, X.; Yang, Y.; Li, M.; Hao, D.; Jia, Z. The genus Epimedium: An ethnopharmacological and phytochemical review. J. Ethnopharmacol 2011, 134, 519–541. [Google Scholar]
- Wu, H.; Lien, E.J.; Lien, L.L. Chemical and Pharmacological Investigations of Epimedium Species: A Survey. In Progress in Drug Research; Birkhäuser Verlag: Berlin, Germany, 2003; Volume 60, pp. 1–57. [Google Scholar]
- Commission, P. Pharmacopoeia of the People’s Republic of China; Chemical Industry Press: Beijing, China, 2005; Volume 1, pp. 229–229. [Google Scholar]
- Zeng, S.; Xiao, G.; Guo, J.; Fei, Z.; Xu, Y.; Roe, B.A.; Wang, Y. Development of a EST dataset and characterization of EST-SSRs in a traditional Chinese medicinal plant, Epimedium sagittatum (Sieb. Et Zucc.) Maxim. BMC Genomics 2010, 11, 94. [Google Scholar]
- Shen, P.; Guo, B.; Gong, Y.; Hong, D.Y.Q.; Hong, Y.; Yong, E. Taxonomic, genetic, chemical and estrogenic characteristics of Epimedium species. Phytochemistry 2007, 68, 1448–1458. [Google Scholar]
- Xie, P.S.; Yan, Y.Z.; Guo, B.L.; Lam, C.; Chui, S.; Yu, Q.X. Chemical pattern-aided classification to simplify the intricacy of morphological taxonomy of Epimedium species using chromatographic fingerprinting. J. Pharmaceutical Biomed. Anal 2010, 52, 452–460. [Google Scholar]
- Li, Z.; Xu, Y.; Wang, Y.; Huang, H. Status and prospect of research on medicinal plants of Epimedium L. Chin. Tradit. Herbal Drugs 2005, 36, 289–295. [Google Scholar]
- Kranz, H.D.; Denekamp, M.; Greco, R.; Jin, H.; Leyva, A.; Meissner, R.C.; Petroni, K.; Urzainqui, A.; Bevan, M.; Martin, C. Towards functional characterisation of the members of the R2R3-MYB gene family from Arabidopsis thaliana. Plant J 1998, 16, 263–276. [Google Scholar]
- Jin, H.; Cominelli, E.; Bailey, P.; Parr, A.; Mehrtens, F.; Jones, J.; Tonelli, C.; Weisshaar, B.; Martin, C. Transcriptional repression by AtMYB4 controls production of UV-protecting sunscreens in Arabidopsis. EMBO J 2000, 19, 6150–6161. [Google Scholar]
- Terrier, N.; Torregrosa, L.; Ageorges, A.; Vialet, S.; Verries, C.; Cheynier, V.; Romieu, C. Ectopic expression of VvMybPA2 promotes proanthocyanidin biosynthesis in grapevine and suggests additional targets in the pathway. Plant Physiol 2009, 149, 1028–1041. [Google Scholar]
- Galego, L.; Almeida, J. Role of DIVARICATA in the control of dorsoventral asymmetry in Antirrhinum flowers. Genes Dev 2002, 16, 880–891. [Google Scholar]
- Howarth, D.G.; Donoghue, M.J. Duplications and expression of DIVARICATA-like genes in Dipsacales. Mol. Biol. Evolut 2009, 26, 1245–1258. [Google Scholar]
- Zimmermann, I.M.; Heim, M.A.; Weisshaar, B.; Uhrig, J.F. Comprehensive identification of Arabidopsis thaliana MYB transcription factors interacting with R/B like BHLH proteins. Plant J 2004, 40, 22–34. [Google Scholar]
- Deluc, L.; Barrieu, F.; Marchive, C.; Lauvergeat, V.; Decendit, A.; Richard, T.; Carde, J.P.; Mérillon, J.M.; Hamdi, S. Characterization of a grapevine R2R3-MYB transcription factor that regulates the phenylpropanoid pathway. Plant Physiol 2006, 140, 499–511. [Google Scholar]
- Zhou, X.R.; Wang, Y.Z.; Smith, J.F.; Chen, R. Altered expression patterns of TCP and MYB genes relating to the floral developmental transition from initial zygomorphy to actinomorphy in Bournea (Gesneriaceae). New Phytol 2008, 178, 532–543. [Google Scholar]
- Tamagnone, L.; Merida, A.; Parr, A.; Mackay, S.; Culianez-Macia, F.A.; Roberts, K.; Martin, C. The AmMYB308 and AmMYB330 transcription factors from Antirrhinum regulate phenylpropanoid and lignin biosynthesis in transgenic tobacco. Plant Cell Online 1998, 10, 135–154. [Google Scholar]
- Aharoni, A.; de Vos, C.; Wein, M.; Sun, Z.; Greco, R.; Kroon, A.; Mol, J.N.M.; O’Connell, A.P. The strawberry FaMYB1 transcription factor suppresses anthocyanin and flavonol accumulation in transgenic tobacco. Plant J 2001, 28, 319–332. [Google Scholar]
- Yoshida, K.; Iwasaka, R.; Kaneko, T.; Sato, S.; Tabata, S.; Sakuta, M. Functional differentiation of Lotus japonicus TT2s, R2R3-MYB transcription factors comprising a multigene family. Plant Cell Physiol 2008, 49, 157–169. [Google Scholar]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol. Biol. Evolution 2007, 24, 1596–1599. [Google Scholar]
- Pérez-Rodríguez, P.; Riaño-Pachón, D.M.; Corrêa, L.G.G.; Rensing, S.A.; Kersten, B.; Mueller-Roeber, B. PlnTFDB: Updated content and new features of the plant transcription factor database. Nucleic Acids Res 2010, 38, D822–D827. [Google Scholar]
- Grotewold, E.; Sainz, M.B.; Tagliani, L.; Hernandez, J.M.; Bowen, B.; Chandler, V.L. Identification of the residues in the Myb domain of maize C1 that specify the interaction with the bHLH cofactor R. Proc. Natl. Aca. Sci. USA 2000, 97, 13579–13584. [Google Scholar]
- Yanhui, C.; Xiaoyuan, Y.; Kun, H.; Meihua, L.; Jigang, L.; Zhaofeng, G.; Zhiqiang, L.; Yunfei, Z.; Xiaoxiao, W.; Xiaoming, Q. The MYB transcription factor superfamily of Arabidopsis: Expression analysis and phylogenetic comparison with the rice MYB family. Plant Mol. Biol 2006, 60, 107–124. [Google Scholar]
- Legay, S.; Lacombe, E.; Goicoechea, M.; Brière, C.; Séguin, A.; Mackay, J.; Grima-Pettenati, J. Molecular characterization of EgMYB1, a putative transcriptional repressor of the lignin biosynthetic pathway. Plant Sci 2007, 173, 542–549. [Google Scholar]
- Doyle, J.; Doyle, J. Isolation of plant DNA from fresh tissue. Focus 1990, 12, 13–15. [Google Scholar]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol 1990, 215, 403–410. [Google Scholar]
- Wheeler, D.L.; Church, D.M.; Federhen, S.; Lash, A.E.; Madden, T.L.; Pontius, J.U.; Schuler, G.D.; Schriml, L.M.; Sequeira, E.; Tatusova, T.A.; et al. Database resources of the National Center for Biotechnology. Nucleic Acids Res 2003, 31, 28–33. [Google Scholar]
- Wheelan, S.J.; Church, D.M.; Ostell, J.M. Spidey: A tool for mRNA-to-genomic alignments. Genome Res 2001, 11, 1952–1957. [Google Scholar]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; Thompson, J.D.; Gibson, T.J.; Higgins, D.G. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar]
- Mancinelli, A. Interaction between light quality and light quantity in the photoregulation of anthocyanin production. Plant Physiol 1990, 92, 1191–1195. [Google Scholar]
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, W.; Sun, W.; Lv, H.; Xiao, G.; Zeng, S.; Wang, Y. Isolation and Molecular Characterization of Thirteen R2R3-MYB Transcription Factors from Epimedium sagittatum. Int. J. Mol. Sci. 2013, 14, 594-610. https://doi.org/10.3390/ijms14010594
Huang W, Sun W, Lv H, Xiao G, Zeng S, Wang Y. Isolation and Molecular Characterization of Thirteen R2R3-MYB Transcription Factors from Epimedium sagittatum. International Journal of Molecular Sciences. 2013; 14(1):594-610. https://doi.org/10.3390/ijms14010594
Chicago/Turabian StyleHuang, Wenjun, Wei Sun, Haiyan Lv, Gong Xiao, Shaohua Zeng, and Ying Wang. 2013. "Isolation and Molecular Characterization of Thirteen R2R3-MYB Transcription Factors from Epimedium sagittatum" International Journal of Molecular Sciences 14, no. 1: 594-610. https://doi.org/10.3390/ijms14010594



