Sorption, Solubility, Bond Strength and Hardness of Denture Soft Lining Incorporated with Silver Nanoparticles
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.2. Discussion
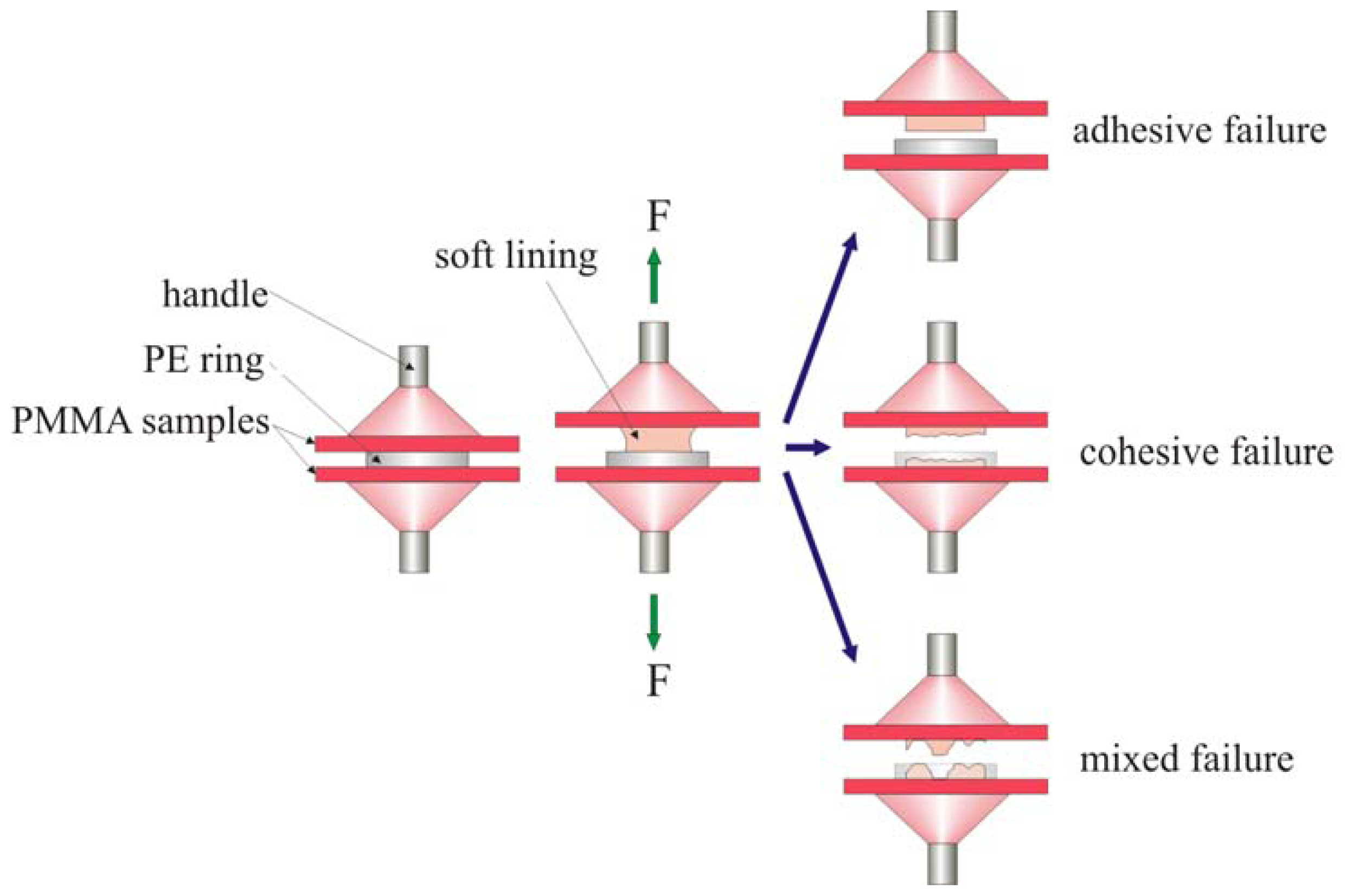
3. Experimental Section
4. Conclusions
Acknowledgements
- Conflict of InterestThe authors declare no conflict of interest.
References
- Bulad, K.; Taylor, R.L.; Verran, J.; McCord, J.F. Colonization and penetration of denture soft lining materials by Candida albicans. Dent. Mater 2004, 20, 167–175. [Google Scholar]
- Pavan, S.; dos Santos, P.H.; Filho, J.N.; Spolidorio, D.M. Colonisation of soft lining materials by micro-organisms. Gerodontology 2010, 27, 211–216. [Google Scholar]
- Taylor, R.L.; Bulad, K.; Verran, J.; McCord, J.F. Colonization and deterioration of soft denture lining materials in vivo. Eur. J. Prosthodont. Restor. Dent 2008, 16, 50–55. [Google Scholar]
- Nikawa, H.; Hamada, T.; Yamamoto, T.; Kumagai, H. Effects of salivary or serum pellicles on the Candida albicans growth and biofilm formation on soft lining materials in vitro. J. Oral. Rehabil 1997, 24, 594–604. [Google Scholar]
- Fan, C.; Chu, L.; Rawls, H.R.; Norling, B.K.; Cardenas, H.L.; Whang, K. Development of an antimicrobial resin—A pilot study. Dent. Mater 2011, 27, 322–328. [Google Scholar]
- Nam, K.Y.; Lee, C.H.; Lee, C.J. Antifungal and physical characteristics of modified denture base acrylic incorporated with silver nanoparticles. Gerodontology 2012, 29, 413–419. [Google Scholar]
- Nam, K.Y. In vitro antimicrobial effect of the tissue conditioner containing silver nanoparticles. J. Adv. Prosthodont 2011, 3, 20–24. [Google Scholar]
- Abe, Y.; Ishii, M.; Takeuchi, M.; Ueshige, M.; Tanaka, S.; Akagawa, Y. Effect of saliva on an antimicrobial tissue conditioner containing silver-zeolite. J. Oral. Rehabil 2004, 31, 568–573. [Google Scholar]
- Chladek, G.; Mertas, A.; Barszczewska–Rybarek, I.; Nalewajek, T.; Zmudzki, J.; Król, W.; Lukaszczyk, J. Antifungal activity of denture soft lining material modified by silver nanoparticles—A pilot study. Int. J. Mol. Sci 2011, 12, 4735–4744. [Google Scholar]
- Jones, D.W.; Sutow, E.J.; Hall, G.C.; Tobin, W.M.; Graham, B.S. Dental soft polymers, plasticizer composition and leachability. Dent. Mater 1988, 4, 1–7. [Google Scholar]
- Braden, M.; Wright, P.S. Water absorption and water solubility of soft lining materials for acrylic dentures. J. Dent. Res 1983, 62, 764–768. [Google Scholar]
- Kawano, F.; Dootz, E.R.; Koran, A., 3rd; Craig, R.G. Sorption and solubility of 12 soft denture liners. J. Prosthet. Dent. 1994, 72, 393–398. [Google Scholar]
- Mancuso, D.N.; Goiato, M.C.; Zuccolotti, B.C.; Moreno, A.; dos Santos, D.M.; Pesqueira, A.A. Effect of thermocycling on hardness, absorption, solubility and colour change of soft liners. Gerodontology 2012, 29, 215–219. [Google Scholar]
- Aydin, A.K.; Terzioğlu, H.; Akinay, A.E.; Ulubayram, K.; Hasirci, N. Bond strength and failure analysis of lining materials to denture resin. Dent. Mater 1999, 15, 211–218. [Google Scholar]
- Kvitek, L.; Panacek, A.; Prucek, R.; Soukupova, J.; Vanickova, M.; Kolar, M.; Zboril, R. Antibacterial activity and toxicity of silver—Nanosilver versus ionic silver. J. Phys. Conf. Ser. 2011, 304, 012029:1–012029:8. [Google Scholar] [CrossRef]
- Soriano-Corral, F.; Morales, G. Nanocomposites based on high impact polystyrene/silver nanoparticles: Effect of silver nanoparticles concentration on the reaction evolution, morphology, and impact strength. Polym. Eng. Sci 2011, 51, 1866–1874. [Google Scholar]
- Yeum, J.H.; Sun, Q.; Deng, Y. Poly(vinyl acetate)/silver nanocomposite microspheres prepared by suspension polymerization at low temperature. Macromol. Mater. Eng 2005, 290, 78–84. [Google Scholar]
- Yeum, J.H.; Ghim, H.D.; Deng, Y. Low temperature suspension polymerization of methyl methacrylate for the preparation of high molecular weight poly(methyl methacrylate)/silver nanocomposite microspheres. Fiber. Polym 2005, 6, 277–283. [Google Scholar]
- ISO International Organization for Standardization. EN ISO 10139-2:2009 Dentistry—Soft Lining Materials for Removable Dentures— Part 2: Materials for Long-Term Use, 2009
- Yoeli, Z.; Miller, V.; Zeltser, C. Consistency and softness of soft liners. J. Prosthet. Dent 1996, 75, 412–418. [Google Scholar]
- Mese, A.; Guzel, K.G. Effect of storage duration on the hardness and tensile bond strength of silicone and acrylic resin-based resilient denture liners to a processed denture base acrylic resin. J. Prosthet. Dent 2008, 99, 153–159. [Google Scholar]
- Polyzois, G.L.; Frangou, M.J. Influence of curing method, sealer, and water storage on the hardness of a soft lining material over time. J. Prosthodont 2001, 10, 42–45. [Google Scholar]
- Dinçkal Yanikoglu, N.; Yeşil Duymuş, Z. Comparative study of water sorption and solubility of soft lining materials in the different solutions. Dent. Mater. J 2004, 23, 233–239. [Google Scholar]
- Al-Athel, M.S.; Jagger, R.G. Effect of test method on the bond strength of a silicone resilient denture lining material. J. Prosthet. Dent 1996, 76, 535–540. [Google Scholar]
- Elias, C.N.; Henriques, F.Q. Effect of thermocycling on the tensile and shear bond strengths of three soft liners to a denture base resin. J. Appl. Oral. Sci 2007, 15, 18–23. [Google Scholar]
- Mutluay, M.M.; Ruyter, I.E. Evaluation of bond strength of soft relining materials to denture base polymers. Dent. Mater 2007, 23, 1373–1381. [Google Scholar]
- Lassila, L.V.; Mutluay, M.M.; Tezvergil-Mutluay, A.; Vallittu, P.K. Bond strength of soft liners to fiber reinforced denture base resin. J. Prosthodont 2010, 19, 620–624. [Google Scholar]
- Yanikoglu, N.; Denizoglu, S. The effect of different solutions on the bond strength of soft lining materials to acrylic resin. Dent. Mater. J 2006, 2, 39–44. [Google Scholar]

| Silver nanoparticle concentration, ppm | Hardness, Shore A units | ||
|---|---|---|---|
| 24 h | 7 days | 28 days | |
| 0 | 31.2 (0.6) A,a | 31.3 (0.5) A,a | 31.1 (0.5) A,a |
| 10 | 28.9 (0.5) B,b | 28.8 (0.7) B,b | 28.9 (0.8) B,b |
| 20 | 28.2 (0.6) C,bd | 28.2 (0.6) C,bd | 28.1 (0.7) C,d |
| 40 | 27.5 (0.7) D,d | 27.5 (0.7) D,d | 27.6 (0.7) D,d |
| 80 | 25.9 (0.4) E,e | 26.3 (0.5) E,e | 26.2 (0.8) E,e |
| 120 | 22.8 (0.4) F,f | 22.9 (0.5) F,f | 22.8 (0.5) F,f |
| 200 | 21.4 (1.0) G,g | 21.3 (1.1) G,g | 21.3 (0.8) G,g |
| Silver nanoparticle concentration, ppm | Sorption, % | Solubility, % | ||
|---|---|---|---|---|
| 7 days | 28 days | 7 days | 28 days | |
| 0 | 0.27 (0.05) A,a | 0.37 (0.06) A,a | 0.09 (0.01) A,a | 0.10 (0.02) A,a |
| 10 | 0.32 (0.06) A,ab | 0.41 (0.06) A,ab | 0.09 (0.02) A,a | 0.11 (0.02) A,a |
| 20 | 0.41 (0.06) A,ab | 0.46 (0.08) A,ab | 0.11 (0.03) A,a | 0.10 (0.03) A,a |
| 40 | 0.37 (0.06) A,ab | 0.51 (0.09) A,ab | 0.09 (0.02) A,a | 0.09 (0.02) A,a |
| 80 | 0.38 (0.08) A,ab | 0.59 (0.09) B,b | 0.11 (0.02) A,a | 0.14 (0.02) A,ab |
| 120 | 0.51 (0.09) A,b | 0.79 (0.14) B,c | 0.15 (0.03) A,a | 0.19 (0.04) A,b |
| 200 | 0.72 (0.12) A,c | 1.24 (0.18) B,d | 0.22 (0.05) A,b | 0.30 (0.05) B,c |
| Silver nanoparticle concentration, ppm | Bond strength, MPa | ||
|---|---|---|---|
| 24 h | 7 days | 28 days | |
| 0 | 1.18 (0.17) A,a | 1.53 (0.17) B,a | 1.48 (0.29) B,a |
| 10 | 1.29 (0.24) A,a | 1.62 (0.23) B,a | 1.51 (0.31) AB,a |
| 20 | 1.28 (0.16) A,a | 1.61 (0.30) B,a | 1.55 (0.26) AB,a |
| 40 | 1.33 (0.22) A,a | 1.59 (0.36) A,a | 1.58 (0.37) A,a |
| 80 | 0.91 (0.15) A,b | 0.96 (0.14) A,b | 0.93 (0.11) A,b |
| 120 | 0.51 (0.06) A,c | 0.54 (0.02) A,c | 0.52 (0.02) A,c |
| 200 | 0.22 (0.03) A,d | 0.25 (0.02) A,d | 0.21 (0.02) A,d |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chladek, G.; Kasperski, J.; Barszczewska-Rybarek, I.; Żmudzki, J. Sorption, Solubility, Bond Strength and Hardness of Denture Soft Lining Incorporated with Silver Nanoparticles. Int. J. Mol. Sci. 2013, 14, 563-574. https://doi.org/10.3390/ijms14010563
Chladek G, Kasperski J, Barszczewska-Rybarek I, Żmudzki J. Sorption, Solubility, Bond Strength and Hardness of Denture Soft Lining Incorporated with Silver Nanoparticles. International Journal of Molecular Sciences. 2013; 14(1):563-574. https://doi.org/10.3390/ijms14010563
Chicago/Turabian StyleChladek, Grzegorz, Jacek Kasperski, Izabela Barszczewska-Rybarek, and Jarosław Żmudzki. 2013. "Sorption, Solubility, Bond Strength and Hardness of Denture Soft Lining Incorporated with Silver Nanoparticles" International Journal of Molecular Sciences 14, no. 1: 563-574. https://doi.org/10.3390/ijms14010563





