In Vitro Cytotoxicity of Fluorescent Silica Nanoparticles Hybridized with Aggregation-Induced Emission Luminogens for Living Cell Imaging
Abstract
:1. Introduction
2. Results and Discussion
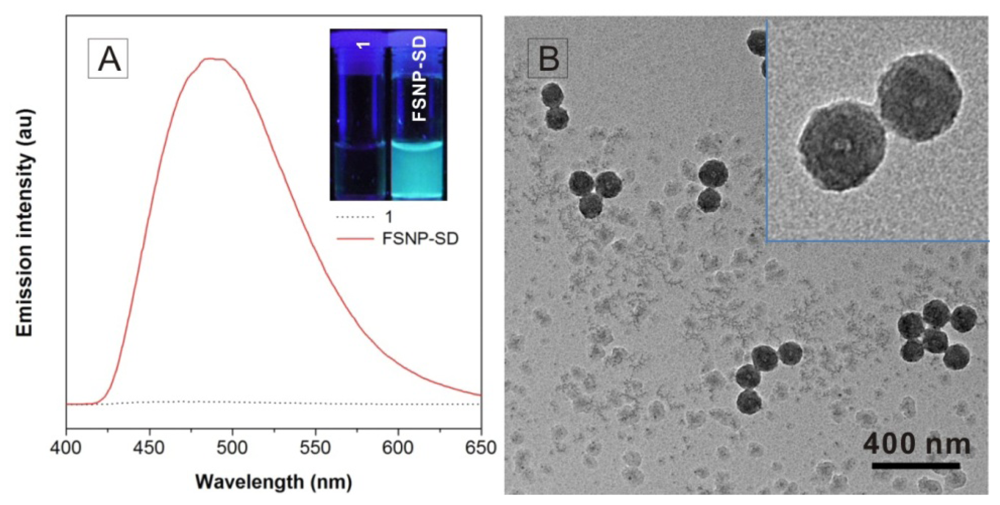
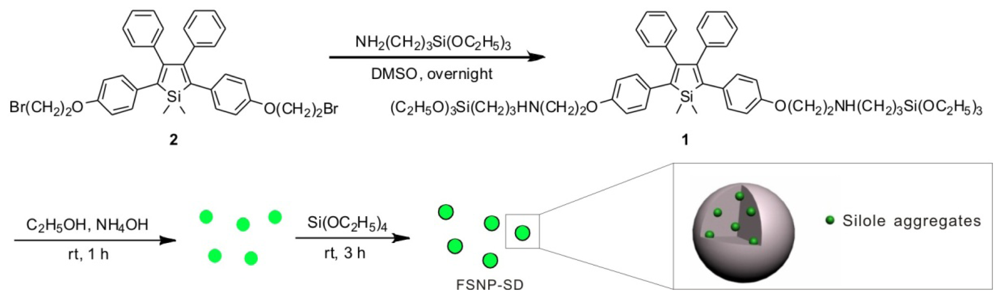
2.1. Fabrication and Characterization of FSNP-SD
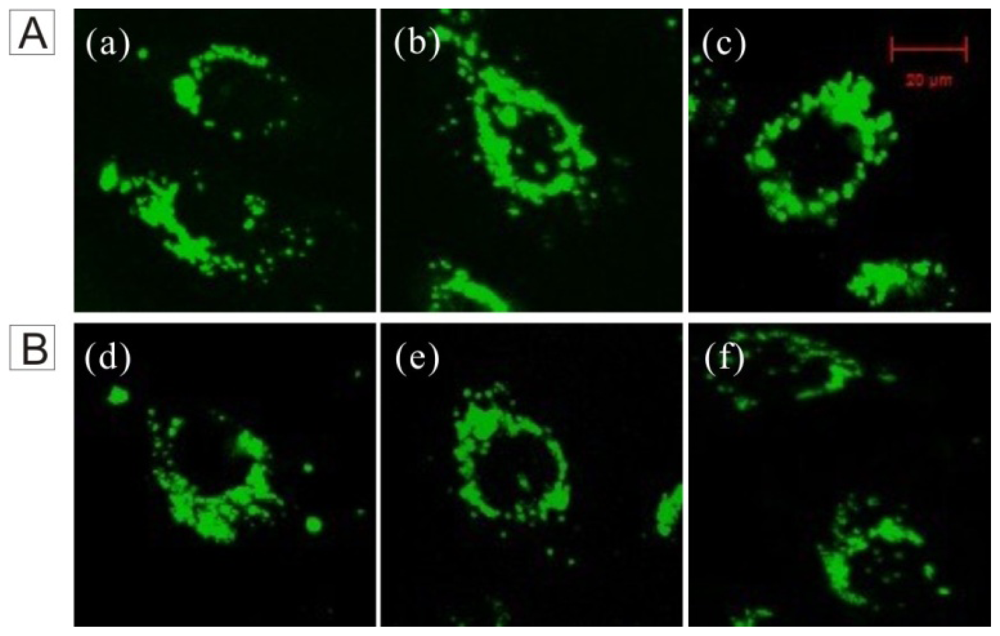
2.2. Cell Imaging
2.3. Distribution of FSNP-SD in Cells
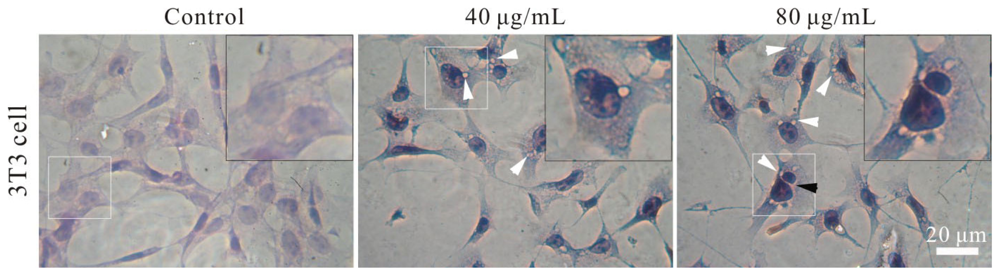
2.4. Effect of FSNP-SD on Cellular Morphology
2.5. Cell Viability and Survival
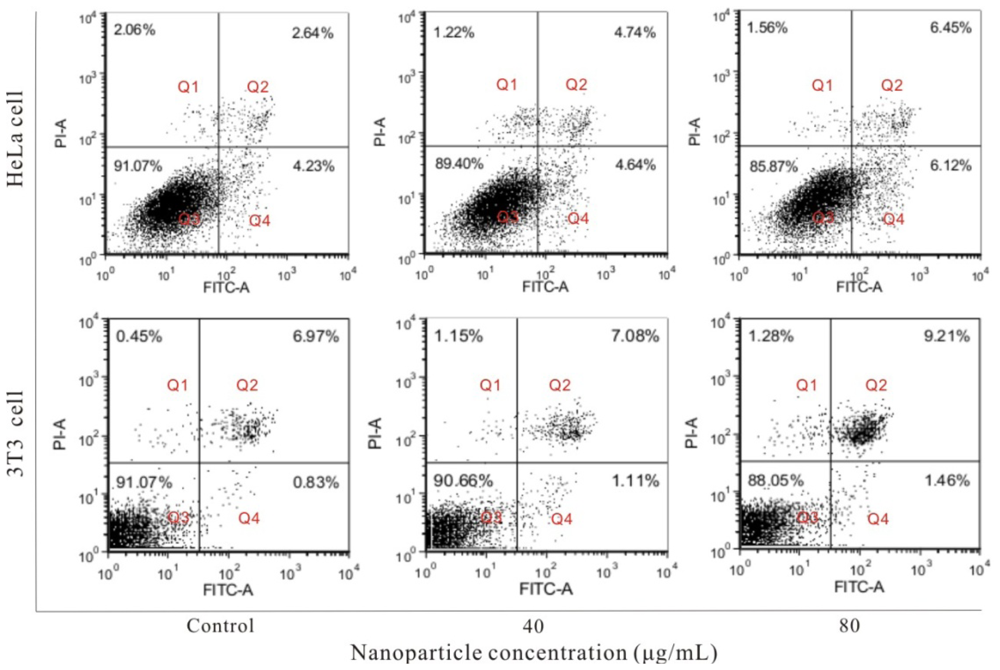
2.6. Cell Apoptosis Induced by FSNP-SD
2.7. Generation of Intracellular ROS
3. Experimental Section
3.1. Materials
3.2. Fabrication and Characterization of FSNP-SD
3.3. Cell Culture and Imaging
3.4. Transmission Electron Microscope
3.5. Observation of Cellular Morphology
3.6. Cell Viability Assay
3.7. Trypan Blue Exclusion Assay
3.8. Cell Apoptosis Assay
3.9. ROS Measurement
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Hong, Y.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission: Phenomenon, mechanism and applications. Chem. Commun. 2009, 4332–4353. [Google Scholar]
- Luo, J.; Xie, Z.; Lam, J.W.Y.; Cheng, L.; Chen, H.; Qiu, C.; Kwok, H.S.; Zhan, X.; Liu, Y.; Zhu, D.; Tang, B.Z. Aggregation-induced emission of 1-methyl-1,2,3,4,5-pentaphenylsilole. Chem. Commun. 2001, 1740–1741. [Google Scholar]
- Liu, J.Z.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission of silole molecules and polymers: Fundamental and applications. J. Inorg. Organomet. Polym 2009, 19, 249–285. [Google Scholar]
- Zhao, Z.J.; Lu, P.; Lam, J.W.Y.; Wang, Z.M.; Chan, C.Y.K.; Sung, H.H.Y.; Williams, I.D.; Ma, Y.G.; Tang, B.Z. Molecular anchors in the solid state: Restriction of intramolecular rotation boosts emission efficiency of luminogen aggregates to unity. Chem. Sci 2011, 2, 672–675. [Google Scholar]
- Santra, S. Fluorescent silica nanoparticles for cancer imaging. Methods Mol. Biol 2010, 624, 151–162. [Google Scholar]
- Taylor-Pashow, L.K.M.; Rocca, J.D.; Lin, W. Mesoporous silica nanoparticles with co-condensed gadolinium chelates for multimodal imaging. Nanomaterials 2012, 2, 1–14. [Google Scholar]
- Li, M.; Lam, J.W.Y.; Mahtab, F.; Chen, S.; Zhang, W.; Hong, Y.; Xiong, J.; Zheng, Q.; Tang, B.Z. Biotin-decorated fluorescent silica nanoparticles with aggregation-induced emission characteristics: Fabrication, cytotoxicity and biological applications. J. Mater. Chem. B 2013, 1, 676–684. [Google Scholar]
- Ow, H.; Larson, D.R.; Srivastava, M.; Baird, B.A.; Webb, W.W.; Wiesner, U. Bright and stable core-shell fluorescent silica nanoparticles. Nano Lett 2005, 5, 113–117. [Google Scholar]
- Bagwe, R.P.; Hilliard, L.R.; Tan, W.H. Surface modification of silica nanoparticles to reduce aggregation and nonspecific binding. Langmuir 2006, 22, 4357–4362. [Google Scholar]
- Cho, E.B.; Volkov, D.O.; Sokolov, I. Ultrabright fluorescent mesoporous silica nanoparticles. Small 2010, 6, 2314–2319. [Google Scholar]
- Michalet, X.; Pinaud, F.F.; Bentolila, L.A.; Tsay, J.M.; Doose, S.; Li, J.J.; Sundaresan, G.; Wu, A.M.; Gambhir, S.S.; Weiss, S. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 2005, 307, 538–544. [Google Scholar]
- Lin, S.; Xie, X.Y.; Patel, M.R.; Yang, Y.H.; Li, Z.J.; Cao, F.; Gheysens, O.; Zhang, Y.; Gambhir, S.S.; Rao, J.H.; Wu, J.C. Quantum dot imaging for embryonic stem cells. BMC Biotechnol 2007, 7, 67. [Google Scholar]
- Drummen, G.P. Quantum dots-from synthesis to applications in biomedicine and life sciences. Int. J. Mol. Sci 2010, 11, 154–163. [Google Scholar]
- Petrache, S.N.; Stanca, L.; Serban, A.I.; Sima, C.; Staicu, A.C.; Munteanu, M.C.; Costache, M.; Burlacu, R.; Zarnescu, O.; Dinischiotu, A. Structural and oxidative changes in the kidney of crucian carp induced by silicon-based quantum dots. Int. J. Mol. Sci 2012, 13, 10193–10211. [Google Scholar]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett 2004, 4, 11–18. [Google Scholar]
- Hsieh, M.S.; Shiao, N.H.; Chan, W.H. Cytotoxic effects of CdSe quantum dots on maturation of mouse oocytes, fertilization, and fetal development. Int. J. Mol. Sci 2009, 10, 2122–2135. [Google Scholar]
- Fan, H.Y.; Leve, E.W.; Scullin, C.; Gabaldon, J.; Tallant, D.; Bunge, S.; Boyle, T.; Wilson, M.C.; Brinker, C.J. Surfactant-assisted synthesis of water-soluble and biocompatible semiconductor quantum dot micelles. Nano Lett 2005, 5, 645–648. [Google Scholar]
- Sokolov, I.; Naik, S. Novel fluorescent silica nanoparticles: Towards ultrabright silica nanoparticles. Small 2008, 4, 934–939. [Google Scholar]
- Zhang, P.; Steelant, W.; Kumar, M.; Scholfield, M. Versatile photosensitizers for photodynamic therapy at infrared excitation. J. Am. Chem. Soc 2007, 129, 4526–4527. [Google Scholar]
- Nakamura, M.; Shono, M.; Ishimura, K. Synthesis, characterization, and biological applications of multifluorescent silica nanoparticles. Anal. Chem 2007, 79, 6507–6514. [Google Scholar]
- Chen, Y.; Parr, T.; Holmes, A.E.; Nakanishi, K. Porphyrinmaleimides: Towards thiol probes for cysteine residues in proteins. Bioconjug. Chem 2008, 19, 5–9. [Google Scholar]
- Birks, J.B. Photophysics of Aromatic Molecules; Wiley-Interscience: London, UK; New York, NY, USA, 1970. [Google Scholar]
- Faisal, M.; Hong, Y.; Liu, J.; Yu, Y.; Lam, J.W.Y.; Qin, A.; Lu, P.; Tang, B.Z. Fabrication of fluorescent silica nanoparticles hybridized with AIE luminogens and exploration of their applications as nanobiosensors in intracellular imaging. Chemistry 2010, 16, 4266–4272. [Google Scholar]
- Jose, J.; Loudet, A.; Ueno, Y.; Wu, L.; Chen, H.Y.; Son, D.H.; Barhoumi, R.; Burghardt, R.; Burgess, K. Energy transfer cassettes in silica nanoparticles target intracellular organelles. Org. Biomol. Chem 2011, 9, 3871–3877. [Google Scholar]
- Fernando, L.P.; Kandel, P.K.; Yu, J.; McNeill, J.; Ackroyd, P.C.; Christensen, K.A. Mechanism of cellular uptake of highly fluorescent conjugated polymer nanoparticles. Biomacromolecules 2010, 11, 2675–2682. [Google Scholar]
- Mahmoudi, M.; Azadmanesh, K.; Shokrgozar, M.A.; Journeay, W.S.; Laurent, S. Effect of nanoparticles on the cell life cycle. Chem. Rev 2011, 111, 3407–3432. [Google Scholar]
- Kirchner, C.; Liedl, T.; Kudera, S.; Pellegrino, T.; Munoz, J.A.; Gaub, H.E.; Stolzle, S.; Fertig, N.; Parak, W.J. Cytotoxicity of colloidal CdSe and CdSe/ZnS nanoparticles. Nano Lett 2005, 5, 331–338. [Google Scholar]
- Li, M.; Chen, S.; Xia, Y.; Xiong, J.; Peng, T.; Wu, Z.; Tang, B.Z.; Zheng, Q. Live cell imaging and cytotoxicity of tetraphenylethene with aggregation-induced emission characteristic. Acta Med. Univ. Sci. Technol. Huazhong 2011, 40, 279–282. [Google Scholar]
- Yu, Y.; Hong, Y.; Feng, C.; Liu, J.; Lam, J.W.Y.; Faisal, M.; Ng, K.M.; Luo, K.Q.; Tang, B.Z. Synthesis of an AIE-active fluorogen and its application in cell imaging. Sci. China Ser. B 2009, 52, 15–19. [Google Scholar]
- Yoshida, K.; Obata, S.; Ono, M.; Esaki, M.; Maejima, T.; Sawada, H. TPA-induced multinucleation of a mesenchymal stem cell-like clone is mediated primarily by karyokinesis without cytokinesis, although cell-cell fusion also occurs. Eur. J. Cell Biol 2007, 86, 461–471. [Google Scholar]
- Li, Y.; Sun, L.; Jin, M.; Du, Z.; Liu, X.; Guo, C.; Huang, P.; Sun, Z. Size-dependent cytotoxicity of amorphous silica nanoparticles in human hepatoma HepG2 cells. Toxicol. In Vitro 2011, 25, 1343–1352. [Google Scholar]
- Jin, Y.; Kannan, S.; Wu, M.; Zhao, J.X. Toxicity of luminescent silica nanoparticles to living cells. Chem. Res. Toxicol 2007, 20, 1126–1133. [Google Scholar]
- Park, E.J.; Yi, J.; Chung, K.H.; Ryu, D.Y.; Choi, J.; Park, K. Oxidative stress and apoptosis induced by titanium dioxide nanoparticles in cultured BEAS-2B cells. Toxicol. Lett 2008, 180, 222–229. [Google Scholar]




© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xia, Y.; Li, M.; Peng, T.; Zhang, W.; Xiong, J.; Hu, Q.; Song, Z.; Zheng, Q. In Vitro Cytotoxicity of Fluorescent Silica Nanoparticles Hybridized with Aggregation-Induced Emission Luminogens for Living Cell Imaging. Int. J. Mol. Sci. 2013, 14, 1080-1092. https://doi.org/10.3390/ijms14011080
Xia Y, Li M, Peng T, Zhang W, Xiong J, Hu Q, Song Z, Zheng Q. In Vitro Cytotoxicity of Fluorescent Silica Nanoparticles Hybridized with Aggregation-Induced Emission Luminogens for Living Cell Imaging. International Journal of Molecular Sciences. 2013; 14(1):1080-1092. https://doi.org/10.3390/ijms14011080
Chicago/Turabian StyleXia, Yun, Min Li, Tao Peng, Weijie Zhang, Jun Xiong, Qinggang Hu, Zifang Song, and Qichang Zheng. 2013. "In Vitro Cytotoxicity of Fluorescent Silica Nanoparticles Hybridized with Aggregation-Induced Emission Luminogens for Living Cell Imaging" International Journal of Molecular Sciences 14, no. 1: 1080-1092. https://doi.org/10.3390/ijms14011080



