Purification and Characterization of a Ginsenoside Rb1-Hydrolyzing β-Glucosidase from Aspergillus niger KCCM 11239
Abstract
:1. Introduction
2. Results and Discussion
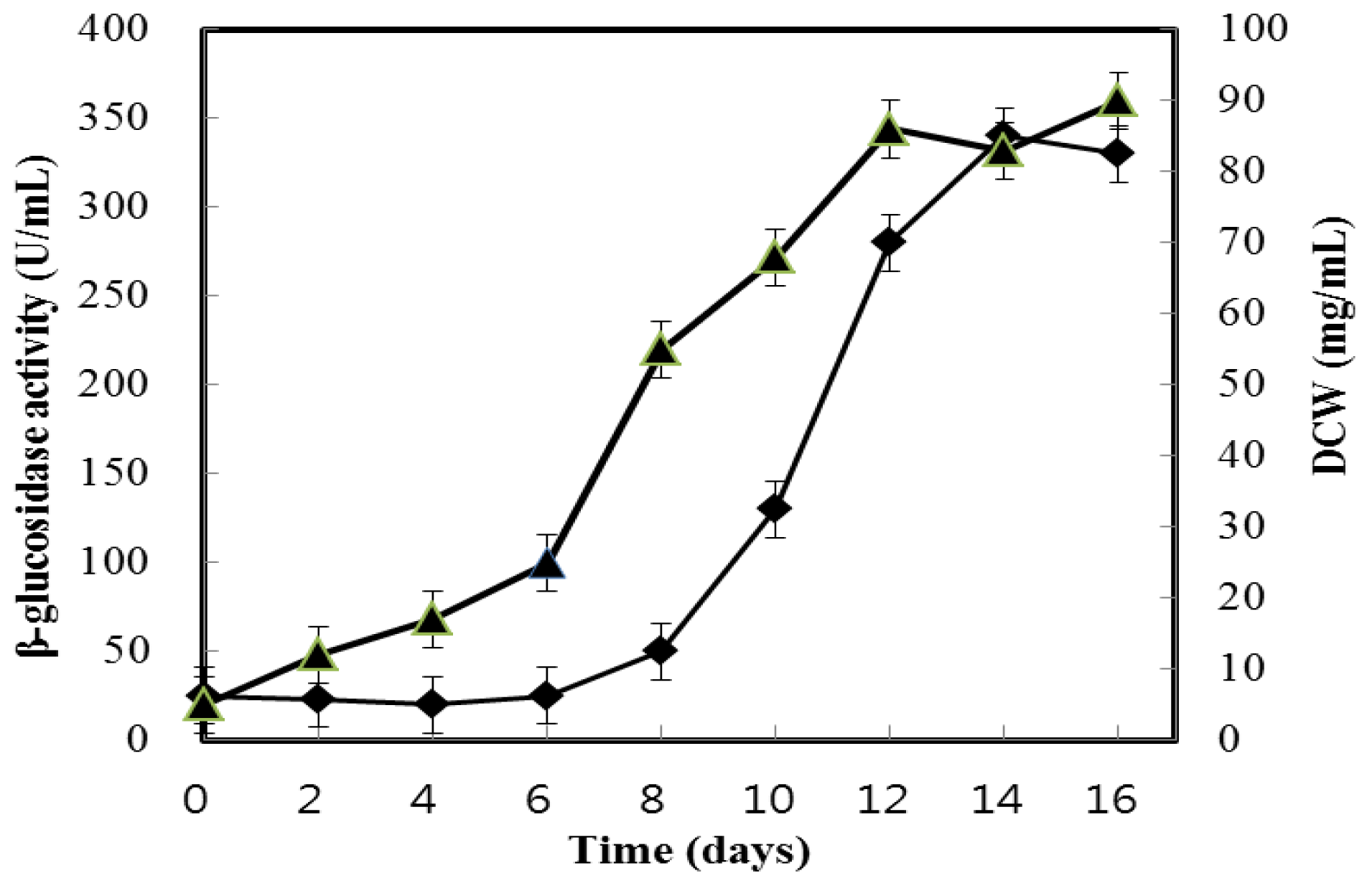
2.1. Growth and β-Glucosidase Production
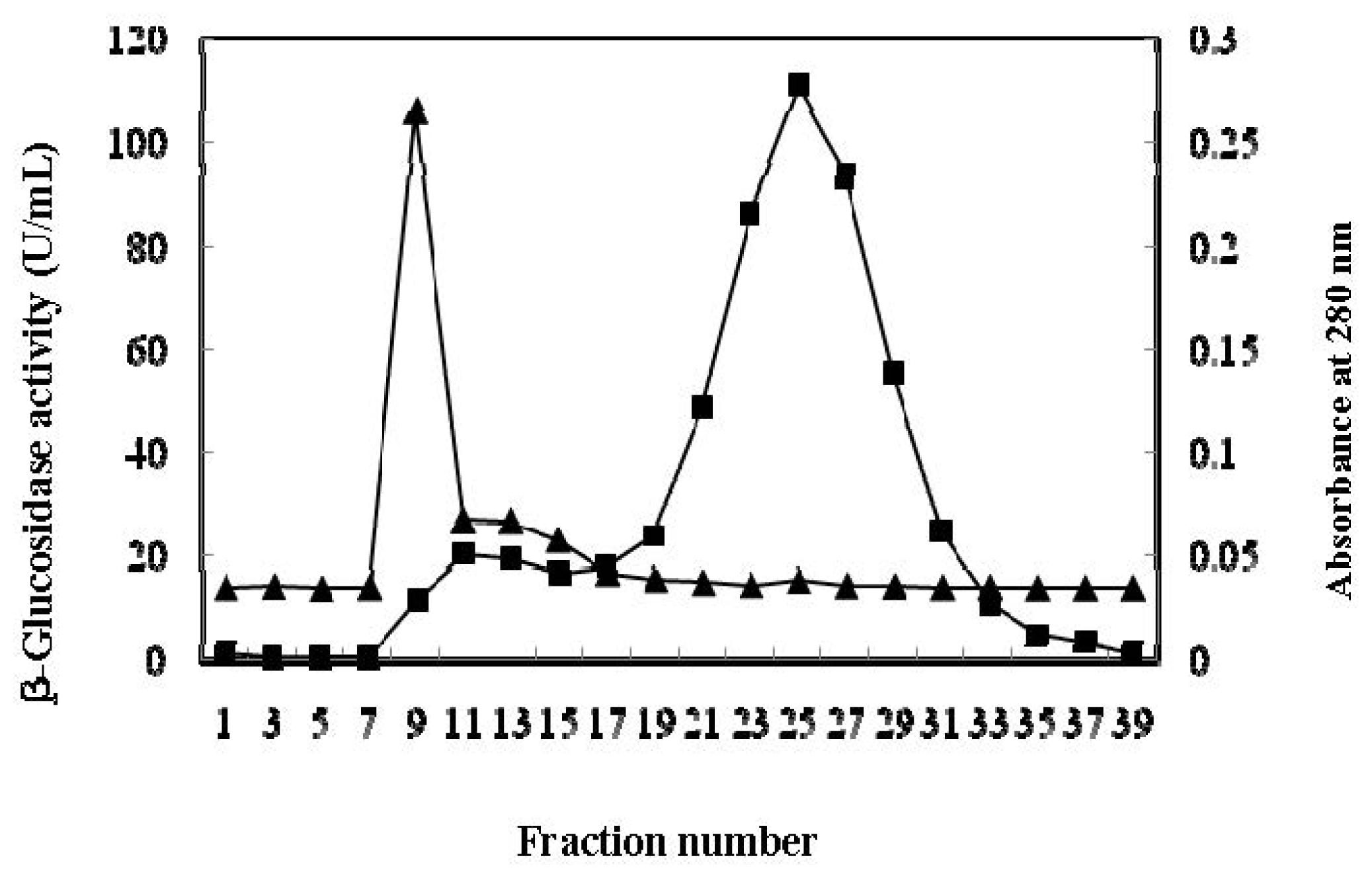
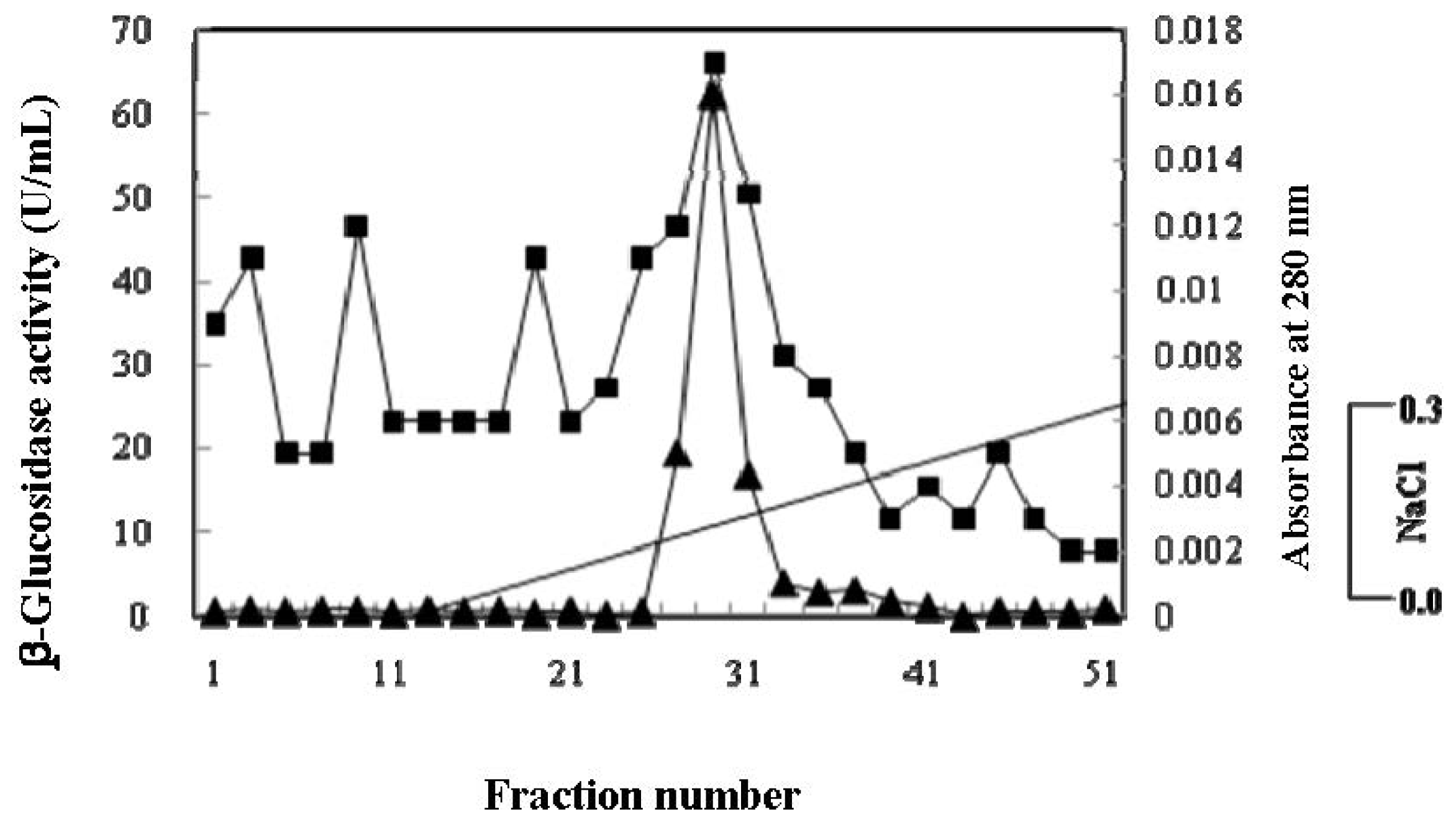
2.2. Purification of β-Glucosidase
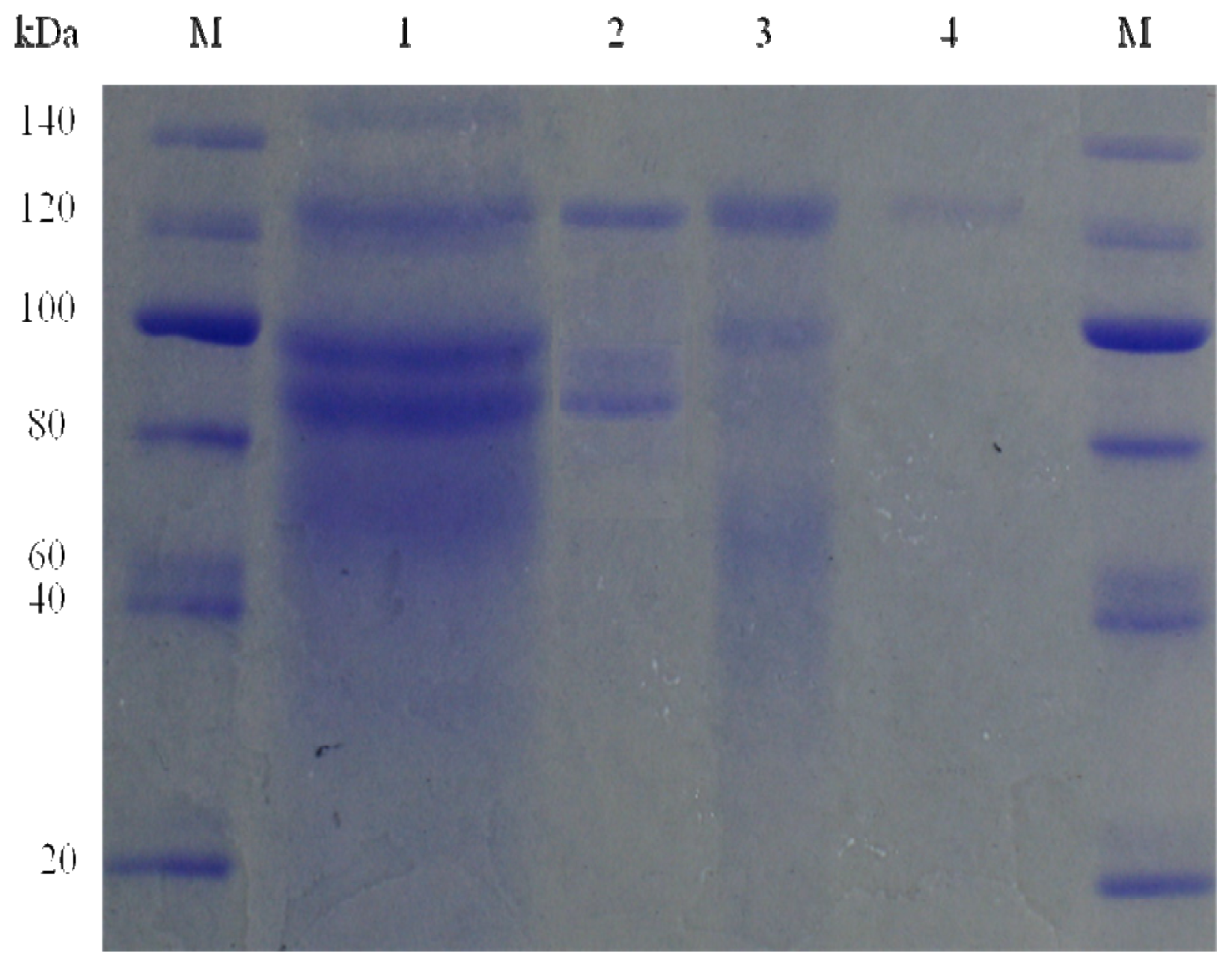
2.3. Molecular Mass of the Purified Enzyme
2.4. Effects of pH and Temperature
2.5. Effects of Metal Ions and Reagents
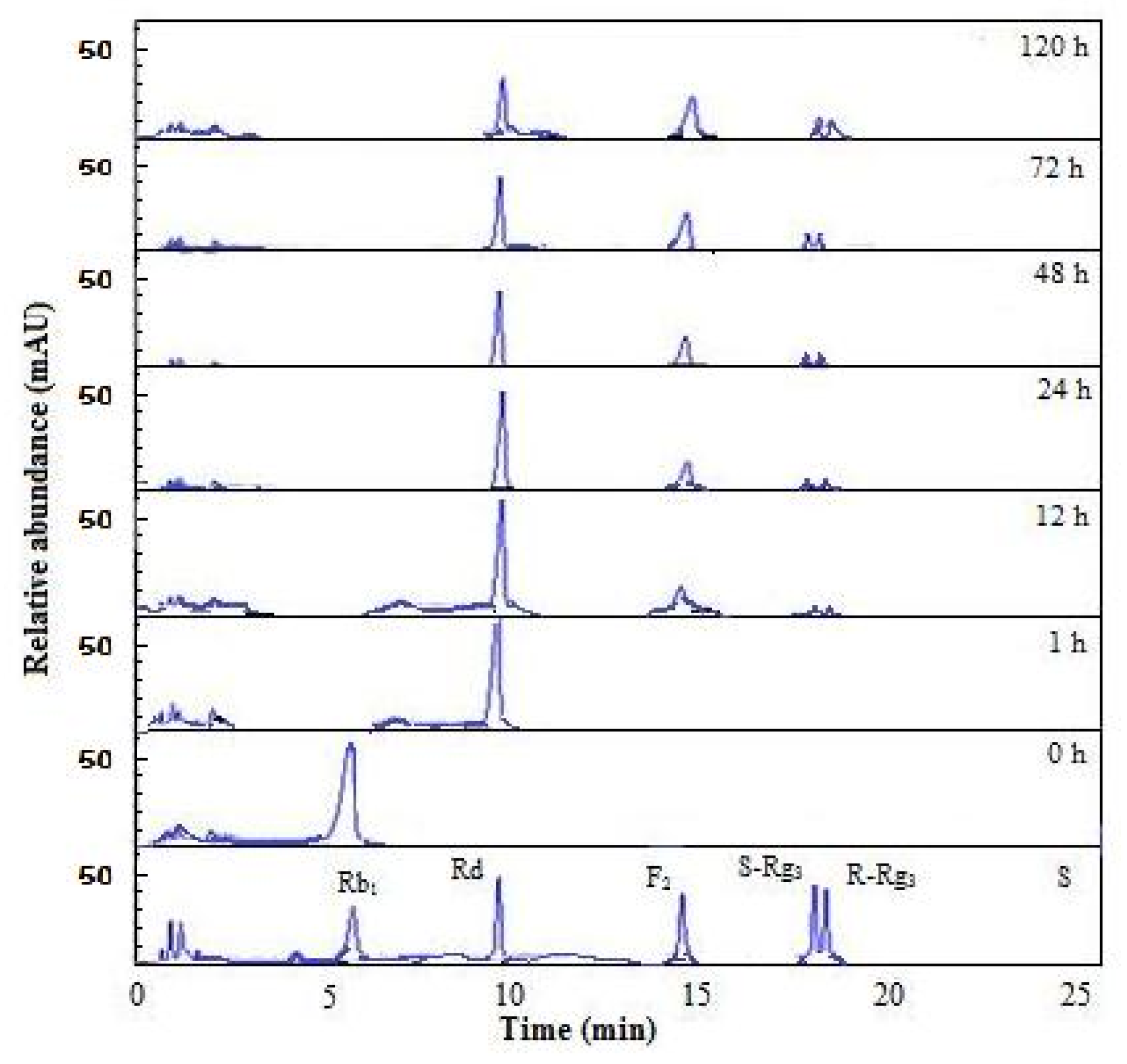
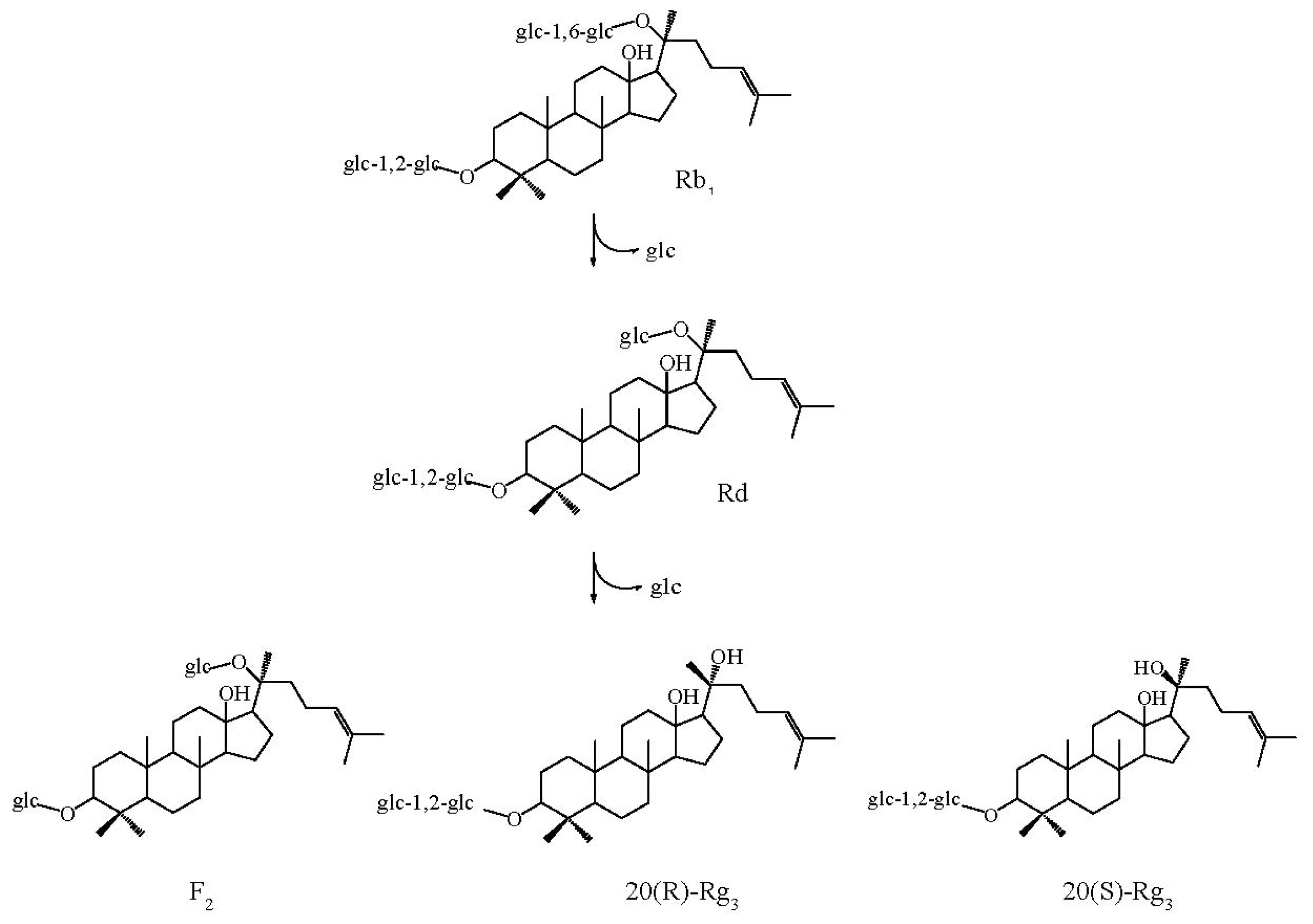
2.6. Substrate Specificity and Time Course of Hydrolysis of Ginsenoside Rb1
3. Experimental Section
3.1. Materials
3.2. Growth and Enzyme Production
3.3. Assay of Enzyme Activity
3.4. Protein Measurement
3.5. Purification of β-Glucosidase
3.6. SDS-PAGE
3.7. Assay of Optimal pH and pH Stability
3.8. Assay of Optimal Temperature and Thermal Stability
3.9. Effects of Metal Ions and Reagents
3.10. Substrate (Ginsenosides) Specificity
3.11. Time Course of Hydrolysis of Ginsenoside Rb1
4. Conclusions
Acknowledgments
References
- Pitson, S.M.; Seviour, R.J.; McDougall, B.M. Purification and characterization of an extracellular β-glucosidase from the filamentous fungus Acremonium persicinum and its probable role in β-glucan degradation. Enzyme Microb. Technol 1997, 21, 182–190. [Google Scholar]
- Hsieh, M.C.; Graham, T.L. Partial purification and characterization of a soybean β-glucosidase with high specific activity towards isoflavone conjugates. Phytochemistry 2001, 58, 995–1005. [Google Scholar]
- Esen, A. β-Glucosidases: Overview. In β-Glucosidases-Biochemistry and Molecular Biology; American Chemical Society: Washington DC, USA, 1993; pp. 1–14. [Google Scholar]
- Su, J.H.; Xu, J.H.; Yu, H.L.; He, Y.C.; Lu, W.Y.; Lin, G.Q. Properties of a novel β-glucosidase from Fusarium proliferatum ECU2042 that converts ginsenoside Rg3 into Rh2. J. Mol. Catal. B 2009, 57, 278–283. [Google Scholar]
- Terra, W.R.; Ferreira, C. Insect digestive enzymes: Properties, compartmentalization and function. Comp. Biochem. Physiol. B 1994, 109, 1–62. [Google Scholar]
- Dan, S.; Marton, I.; Dekel, M.; Bravdo, B.; He, S.; Withers, S.G.; Shoseyov, O. Cloning, expression, characterization, and nucleophile identification of family 3, Aspergillus niger β-glucosidase. J. Biol. Chem 2000, 275, 4973–4980. [Google Scholar]
- Pontoh, J.; Low, N.H. Purification and characterization of beta-glucosidase from honey bees (Apis mellifera). Insect Biochem. Mol. Biol 2002, 32, 679–690. [Google Scholar]
- Park, J.D.; Rhee, D.K.; Lee, Y.H. Biological activities and chemistry of saponins from Panax ginseng C.A. Meyer. Phytochem. Rev 2005, 4, 159–175. [Google Scholar]
- Cheng, L.Q.; Na, J.R.; Bang, M.H.; Kim, M.K.; Yang, D.C. Conversion of major ginsenoside Rb1 to 20(S)-ginsenoside Rg3 by Microbacterium sp. GS514. Phytochemistry 2008, 69, 218–224. [Google Scholar]
- Zhao, X.; Gao, L.; Wang, J.; Bi, H.; Gao, J.; Du, X.; Zhou, Y.; Tai, G. A novel ginsenoside Rb1-hydrolyzing β-d-glucosidase from Cladosporium fulvum. Process Biochem 2009, 44, 612–618. [Google Scholar]
- Han, B.H.; Park, M.H.; Han, Y.N.; Woo, L.K.; Sankawa, U.; Yahara, S.; Tanaka, O. Degradation of ginseng under mild acidic condition. Planta Med 1982, 44, 146–149. [Google Scholar]
- Yang, L.; He, K.J.; Yang, Y. A preparation method of less polar individual ginsenosides and saponins by base hydrolysis. China Patent CN03134090.3, 30 March 2005. [Google Scholar]
- Park, J.H. Sun ginseng—A new processed ginseng with fortified activity. Food Ind. Nutr 2004, 9, 23–27. [Google Scholar]
- Cheng, L.Q.; Na, J.R.; Kim, M.K.; Bang, M.H.; Yang, D.C. Microbial conversion of ginsenoside Rb1 to minor ginsenoside F2 and gypenoside XVII by Intrasporangium sp. GS603 isolated from soil. J. Micobiol. Biotechnol 2007, 17, 1937–1943. [Google Scholar]
- Schuster, E.; Dunn-Coleman, N.; Frisvad, J.C.; van Dijck, P.W.M. On the safety of Aspergillus niger—A review. Appl. Microbiol. Biotechnol 2009, 59, 426–435. [Google Scholar]
- Sung, C.K.; Lee, S.W.; Park, S.K.; Park, J.R.; Moon, I.L. Purification and characterization of β-glucosidase from Aspergillus niger SFN-416. Korean J. Appl. Microbiol. Biotechnol 1997, 25, 44–50. [Google Scholar]
- Rashid, M.H.; Siddiqui, K.S. Purification and characterization of a beta-glucosidase from Apsergillus niger. Folia Microbiol 1997, 42, 544–550. [Google Scholar]
- Watanabe, T.; Sato, T.; Yoshioka, S.; Koshijima, T.; Kuwahara, M. Purification and properties of Aspergillus niger β-glucosidase. Eur. J. Biochem 1992, 209, 651–659. [Google Scholar]
- Kaur, J.; Chadha, B.S.; Kumar, B.A.; Kaur, G.S.; Saini, H.S. Purification and characterization of β-glucosidase from Melanocarpus sp. MTCC 3922. Electron. J. Biotechnol 2007, 10, 260–270. [Google Scholar]
- Yan, T.R.; Lin, Y.H.; Lin, C.L. Purification and characterization of an extracellular β-glucosidase II with high hydrolysis and transglucosylation activities from Aspergillus niger. J. Agric. Food Chem 1998, 46, 431–437. [Google Scholar]
- Lee, J.K.; Choi, S.S.; Lee, H.K.; Han, K.J.; Han, E.J.; Suh, H.W. Effects of ginsenoside Rd and decursinol on the neurotoxic responses induced by kainic acid in mice. Planta Med 2003, 69, 230–234. [Google Scholar]
- Zeng, S.; Guan, Y.Y.; Liu, D.Y.; He, H.; Wang, W.; Qiu, Q.Y.; Wang, X.R.; Wang, Y.D. Synthesis of 12-epi-ginsenoside Rd and its effects on contractions of rat aortic rings. Chin. Pharmacol. Bull 2003, 19, 282–286. [Google Scholar]
- Tachikawa, E.; Kudo, K.; Hasegawa, H.; Kashimoto, T.; Sasaki, K.; Miyazaki, M.; Taira, H.; Lindstorm, J.M. In vitro inhibition of adrenal catecholamine secretion by steroidal metabolites of ginseng saponins. Biochem. Pharmacol 2003, 66, 2213–2221. [Google Scholar]
- Shinkai, K.; Akedo, H.; Mukai, M.; Imamura, F.; Isoai, A.; Kobayashi, M.; Kitagawa, I. Inhibition of in vitro tumor cell invasion by ginsenoside Rg3. Jpn. J. Cancer Res 1996, 87, 357–362. [Google Scholar]
- Lee, H.U.; Bae, E.A.; Han, M.J.; Kim, D.H. Hepatoprotective effect of 20(S)-ginsenoside Rg3 and its metabolite 20(S)-ginsenoside Rh2 on tert-butyl hydroperoxide-induced liver injury. Biol. Pharm. Bull 2005, 28, 1992–1994. [Google Scholar]
- Choi, I.Y.; Lee, J.H.; Kang, T.H.; An, H.J.; Kim, S.J.; Moon, P.D.; Kim, N.H.; Myung, N.Y.; Yand, D.C.; Kang, I.C.; et al. Synergistic effects of KH-red ginseng/chlorella on the endurance capacity and immune enhancing in forced swimming tested mice. Food Sci. Biotech 2009, 18, 1351–1357. [Google Scholar]
- Kohchi, C.; Toh-E, A. Cloning of Candida pelliculosa beta-glucosidase gene and its expression in Saccharomyces cerevisiae. Mol. Gen. Genet 1986, 203, 89–94. [Google Scholar]
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of protein using bicinchoninic acid. Anal. Biochem 1985, 150, 76–85. [Google Scholar]
- Laemmli, U.K. Cleavage of the structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar]
- Luan, H.; Liu, X.; Qi, X.; Hu, Y.; Hao, D.; Cui, Y.; Yang, L. Purification and characterization of a novel stable ginsenoside Rb1-hydrolyzing β-d-glucosidase from China white jade snail. Process Biochem 2006, 41, 1974–1980. [Google Scholar]
- Chang, K.H.; Jee, H.S.; Lee, N.K.; Park, S.H.; Lee, N.W.; Paik, H.D. Optimization of the enzymatic production of 20(S)-ginsenoside Rg3 from white ginseng extract using response surface methodology. New Biotechnol 2009, 26, 181–186. [Google Scholar]
| Purification step | Total activity (U) | Total protein (mg) | Specific activity (U/mg) | Yield (%) | Purification (Fold) |
|---|---|---|---|---|---|
| Crude extract (supernatant), 400 mL | 86,971.0 | 8,797.0 | 9.9 | 100 | 1.0 |
| (NH4)2SO4 Precipitation, 86 mL | 22,655.0 | 248.8 | 91.0 | 21.5 | 9.2 |
| Sephadex G-100 Chromatography, 44 mL | 3,073.8 | 13.5 | 227.1 | 11.0 | 22.9 |
| DEAE Sephadex, Chromatography, 6 mL | 712.3 | 1.5 | 460.5 | 1.5 | 46.5 |
| Metal ions or reagents a | Relative activity (%) b |
|---|---|
| None c | 100.00 ± 0.00 d |
| FeCl2 | 92.00 ± 1.79 |
| CuSO4 | 79.00 ± 0.27 |
| CoCl2 | 88.00 ± 1.50 |
| KCl | 97.00 ± 1.45 |
| MnCl2 | 97.00 ± 4.11 |
| ZnSO4 | 94.00 ± 2.76 |
| CaCl2 | 101.00 ± 2.95 |
| MgCl2 | 87.00 ± 8.39 |
| NaCl | 101.00 ± 1.29 |
| Ethanol | 97.00 ± 3.98 |
| Glycerol | 95.00 ± 3.60 |
| Acetic acid | 86.00 ± 4.25 |
| Triton X-100 | 110.00 ± 5.25 |
| Methanol | 94.00 ± 2.77 |
| SDS | 95.00 ± 1.54 |
| EDTA | 97.00 ± 5.00 |
| Ginsenoside | Configuration of glycoside linkage | Relative activity (%) a |
|---|---|---|
| Rb1 | −glc(2→1)glc, −glc(6→1)glc | 100 ± 0.00 b |
| Rb2 | −glc(2→1)glc, −glc(6→1)arap | ND c |
| Rc | −glc(2→1)glc, −glc(6→1)araf | 2.00 ± 0.26 |
| Rd | −glc(2→1)glc | ND |
| Re | −glc(2→1)rha | ND |
| Rg1 | −glc | 21.00 ± 0.71 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chang, K.H.; Jo, M.N.; Kim, K.-T.; Paik, H.-D. Purification and Characterization of a Ginsenoside Rb1-Hydrolyzing β-Glucosidase from Aspergillus niger KCCM 11239. Int. J. Mol. Sci. 2012, 13, 12140-12152. https://doi.org/10.3390/ijms130912140
Chang KH, Jo MN, Kim K-T, Paik H-D. Purification and Characterization of a Ginsenoside Rb1-Hydrolyzing β-Glucosidase from Aspergillus niger KCCM 11239. International Journal of Molecular Sciences. 2012; 13(9):12140-12152. https://doi.org/10.3390/ijms130912140
Chicago/Turabian StyleChang, Kyung Hoon, Mi Na Jo, Kee-Tae Kim, and Hyun-Dong Paik. 2012. "Purification and Characterization of a Ginsenoside Rb1-Hydrolyzing β-Glucosidase from Aspergillus niger KCCM 11239" International Journal of Molecular Sciences 13, no. 9: 12140-12152. https://doi.org/10.3390/ijms130912140




