Protective Effect of Anthocyanin from Lonicera Caerulea var. Edulis on Radiation-Induced Damage in Mice
Abstract
:1. Introduction
2. Results and Discussion
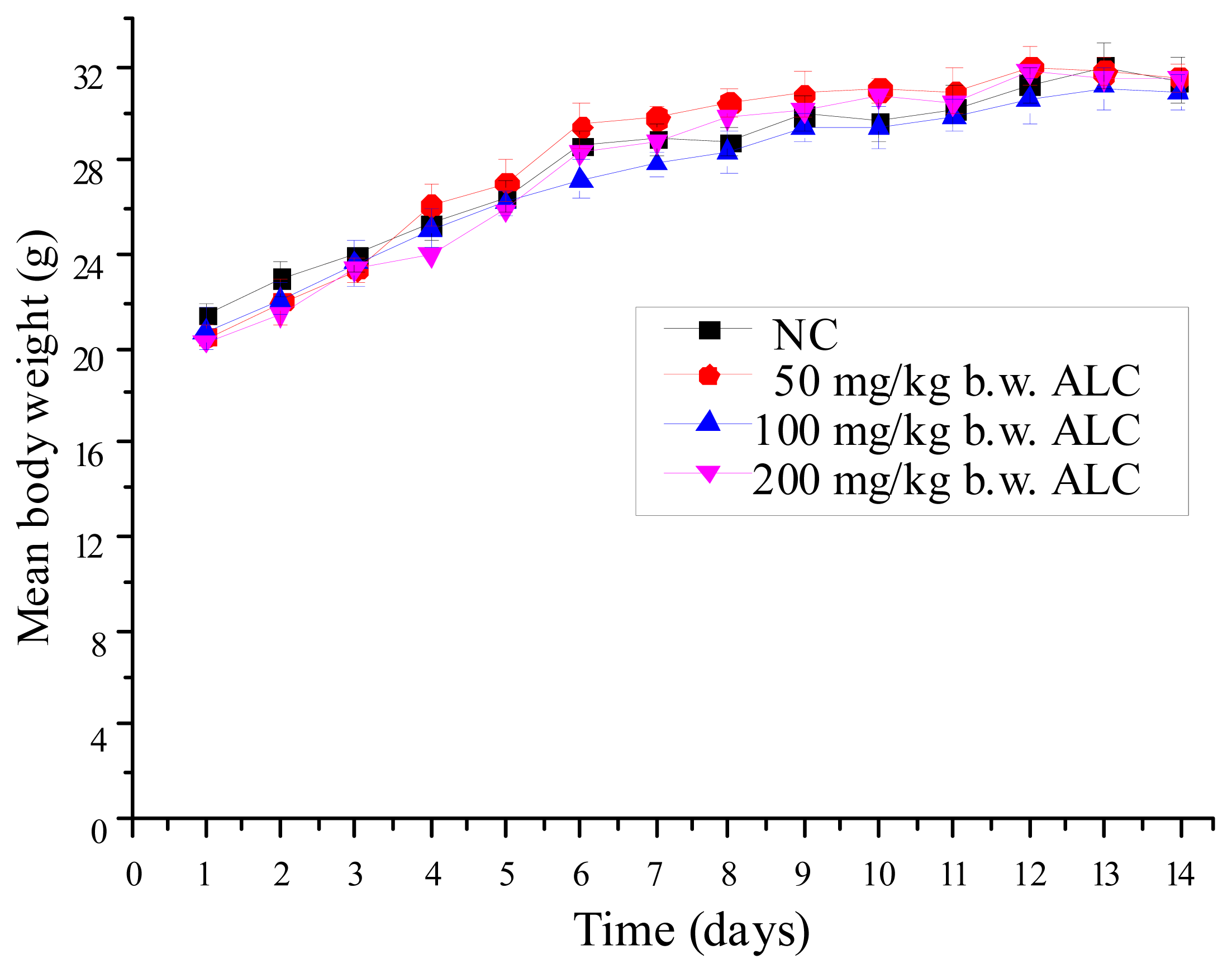
2.1. Effect of ALC Treatment on Body Weight Change of Mice
2.2. Effect of ALC on the Immune Organ Indices of Mice
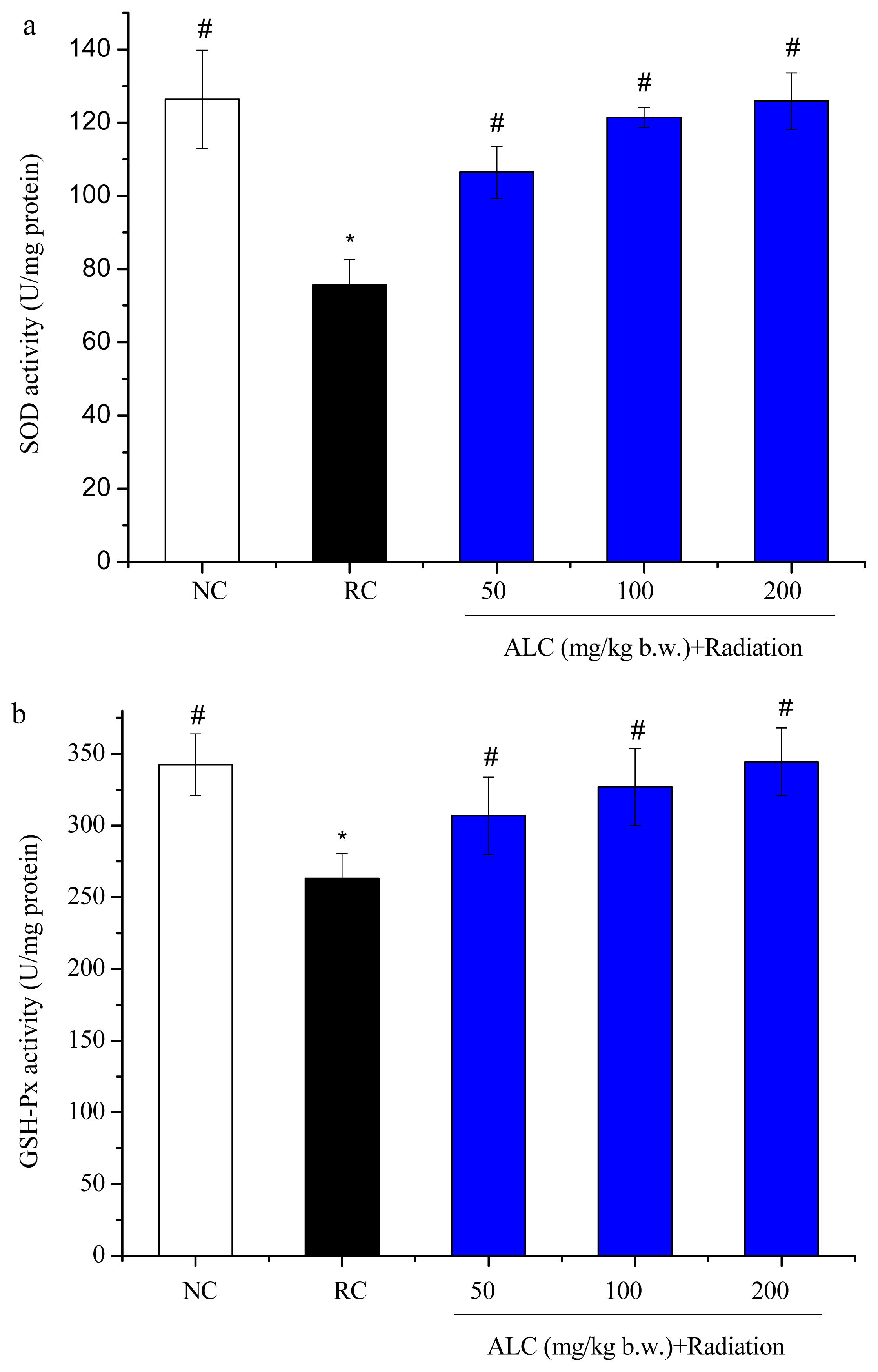
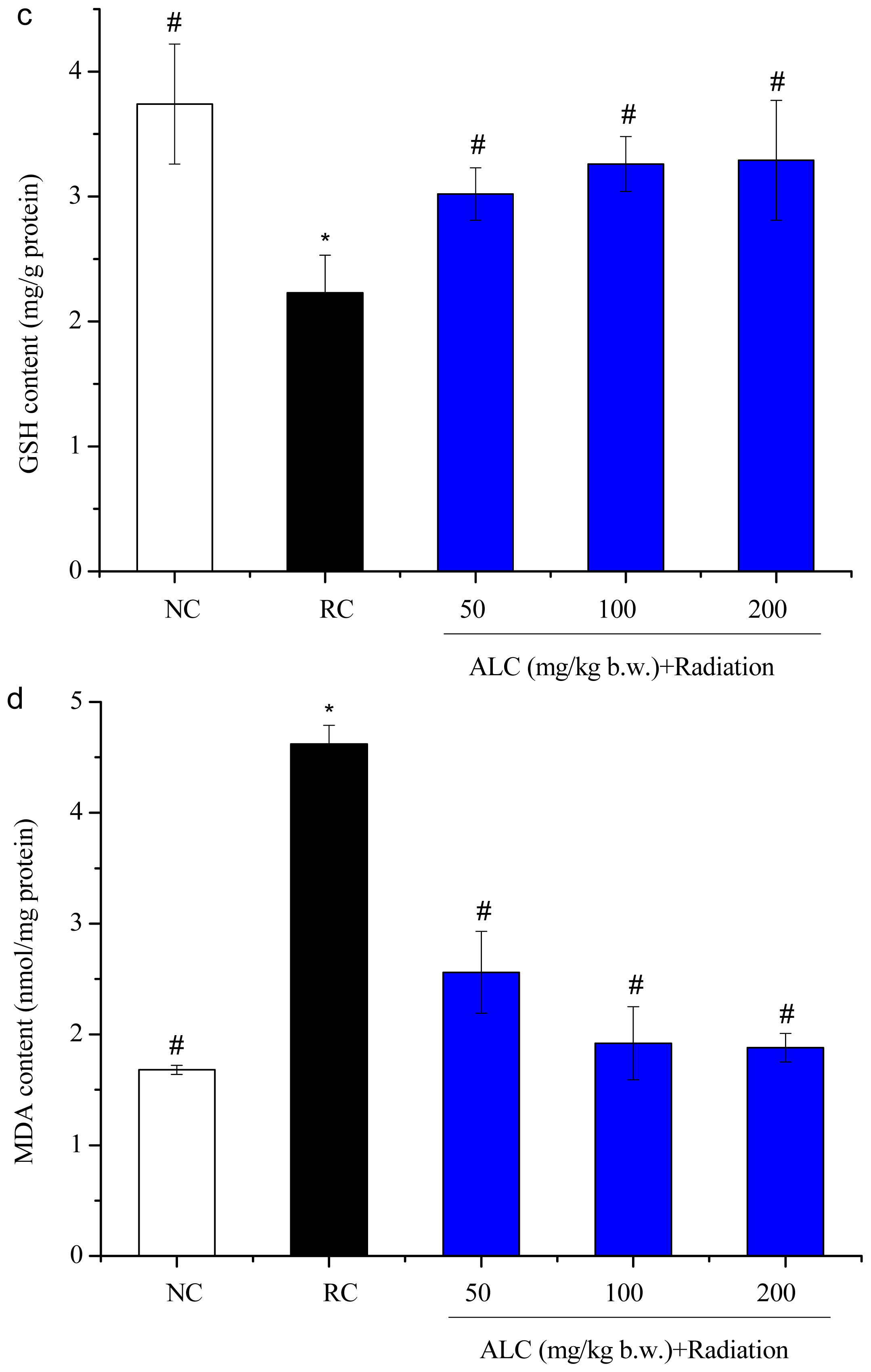
2.3. Effect of ALC on Oxidant/Antioxidant Status in Liver Tissue
3. Experimental Section
3.1. Chemicals and Reagents
3.2. Animals
3.3. Irradiation
3.4. Experimental Design
3.5. Thymus and Spleen Indices
3.6. Activities of SOD and GSH-Px
3.7. Determination of Lipid Peroxidation and Reduced GSH Content in Liver Tissue
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Kalpana, K.B.; Devipriya, N.; Thayalan, K.; Menon, V.P. Protection against X-ray radiation-induced cellular damage of human peripheral blood lymphocytes by an aminothiazole derivative of dendrodoine. Chem. Biol. Interact 2010, 186, 267–274. [Google Scholar]
- Tominaga, H.; Kodama, S.; Matsuda, N.; Suzuki, K.; Watanabe, M. Involvement of reactive oxygen species (ROS) in the induction of genetic instability by radiation. J. Radit. Res 2004, 45, 181–188. [Google Scholar]
- Weiss, J.F.; Landauer, M.R. Radioprotection by antioxidants. Ann. N. Y. Acad. Sci 2000, 899, 44–60. [Google Scholar]
- Singh, V.K.; Yadav, V.S. Role of cytokines and growth factors in radioprotection. Exp. Mol. Pathol 2005, 78, 156–169. [Google Scholar]
- Nair, C.K.; Parida, D.K.; Nomura, T. Radioprotectors in radiotherapy. J. Radit. Res 2001, 42, 21–37. [Google Scholar]
- Svobodová, A.; Rambousková, J.; Walterová, D.; Vostálová, J. Protective effects of phenolic fraction of blue honeysuckle fruits against UVA-induced damage to human keratinocytes. Arch. Dermatol. Res 2008, 300, 225–233. [Google Scholar]
- Chaovanalikit, A.; Thompson, M.M.; Wrolstad, R.E. Characterization and quantification of anthocyanins and polyphenolics in blue honeysuckle (Lonicera caerulea L.). J. Agric. Food. Chem 2004, 52, 848–852. [Google Scholar]
- Zdařilová, A.; Svobodová, A.R.; Chytilová, K.; Šimánek, V.; Ulrichová, J. Polyphenolic fraction of Lonicera caerulea L. fruits reduces oxidative stress and inflammatory markers induced by lipopolysaccharide in gingival fibroblasts. Food Chem. Toxicol 2010, 48, 1555–1561. [Google Scholar]
- Svobodová, A.; Zdarilová, A.; Vostálová, J. Lonicera caerulea and Vaccinium myrtillus fruit polyphenols protect HaCaT keratinocytes against UVB-induced phototoxic stress and DNA damage. J. Dermatol. Sci 2009, 56, 196–204. [Google Scholar]
- Fan, Z.-L.; Wang, Z.-Y.; Liu, J.-R. Cold-field fruit extracts exert different antioxidant and antiproliferative activities in vitro. Food Chem 2011, 129, 402–407. [Google Scholar]
- Jin, X.-H.; Ohgami, K.; Shiratori, K.; Suzuki, Y.; Koyama, Y.; Yoshida, K.; Ilieva, I.; Tanaka, T.; Onoe, K.; Ohno, S. Effects of blue honeysuckle (Lonicera caerulea L.) extract on lipopolysaccharide-induced inflammation in vitro and in vivo. Exp. Eye Res 2006, 82, 860–867. [Google Scholar]
- Zhao, H.; Wang, Z.; Cheng, C.; Yao, L.; Wang, L.; Lu, W.; Yang, X.; Ma, F. In vitro free radical scavenging activities of anthocyanins from three berries. J. Med. Plants Res 2011, 5, 7036–7042. [Google Scholar]
- Yoshino, F.; Yoshida, A.; Okada, E.; Okada, Y.; Maehata, Y.; Miyamoto, C.; Kishimoto, S.; Otsuka, T.; Nishimura, T.; Lee, M.C.-I. Dental resin curing blue light induced oxidative stress with reactive oxygen species production. J. Photochem. Photobiol. B 2012, 114, 73–78. [Google Scholar]
- Riley, P.A. Free radicals in biology: Oxidative stress and the effects of ionizing radiation. Int. J. Radiat. Biol 1994, 65, 27–33. [Google Scholar]
- Pillai, T.G.; Krishnan, C.; Nair, K.; Janardhanan, K.K. Polysaccharides isolated from Ganoderma lucidum occurring in Southern parts of India, protects radiation induced damages both in vitro and in vivo. Environ. Toxicol. Pharmacol 2008, 26, 80–85. [Google Scholar]
- Yao, L.; Wang, Z.; Zhao, H.; Cheng, C.; Fu, X.; Liu, J.; Yang, X. Protective Effects of polysaccharides from soybean meal against X-ray radiation induced damage in mouse spleen lymphocytes. Int. J. Mol. Sci 2011, 12, 8096–8104. [Google Scholar]
- Qi, L.; Liu, C.; Wu, W.; Gu, Z.; Guo, C. Fitoterapia Protective effect of flavonoids from Astragalus complanatus on radiation induced damages in mice. Fitoterapia 2011, 82, 383–392. [Google Scholar]
- Paul, P.; Bansal, P.; Nayak, P.G.; Pannakal, S.T.; Priyadarsini, K.I.; Unnikrishnan, M.K. Polyphenolic fraction of Pilea microphylla (L.) protects Chinese hamster lung fibroblasts against γ-radiation-induced cytotoxicity and genotoxicity. Environ. Toxicol. Pharmacol 2011, 33, 107–119. [Google Scholar]
- Abdel-Aal, E.-S.M.; Abou-Arab, A.A.; Gamel, T.H.; Hucl, P.; Young, J.C.; Rabalski, I. Fractionation of blue wheat anthocyanin compounds and their contribution to antioxidant properties. J. Agric. Food. Chem 2008, 56, 11171–11177. [Google Scholar]
- De Pascual-Teresa, S.; Moreno, D.A.; García-Viguera, C. Flavanols and anthocyanins in cardiovascular health: A review of current evidence. Int. J. Mol. Sci 2010, 11, 1679–1703. [Google Scholar]
- Janero, D.R. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med 1990, 9, 515–540. [Google Scholar]
- Taysi, S.; Uslu, C.; Akcay, F.; Sutbeyaz, M.Y. Malondialdehyde and nitric oxide levels in the plasma of patients with advanced laryngeal cancer. Surg. Today 2003, 33, 651–654. [Google Scholar]
- Krishnan, C.; Nair, K. Radiation protection by 6-palmitoyl ascorbic acid-2-glucoside: Studies on DNA damage in vitro, ex vivo, in vivo and oxidative stress in vivo. Cancer Res 2009, 50, 203–212. [Google Scholar]
- Ahaskar, M.; Sharma, K.V.; Singh, S.; Sisodia, R. Radioprotective effect of fruit extract of Grewia asiatica in Swiss albino mice against lethal dose of γ-irradiation. Asian J. Exp. Sci 2007, 21, 297–310. [Google Scholar]
- Zelko, I.N.; Mariani, T.J.; Folz, R.J. Superoxide dismutase multigene family: A comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free. Radic. Biol. Med 2002, 33, 337–349. [Google Scholar]
- Taysi, S.; Polat, F.; Gul, M.; Sari, R.; Bakan, E. Lipid peroxidation, some extracellular antioxidants, and antioxidant enzymes in serum of patients with rheumatoid arthritis. Rheumatol. Int 2002, 21, 200–204. [Google Scholar]
- Su, X.-Y.; Wang, Z.-Y.; Liu, J.-R. In vitro and in vivo antioxidant activity of Pinus koraiensis seed extract containing phenolic compounds. Food Chem 2009, 117, 681–686. [Google Scholar]
- Eroglu, C.; Yildiz, O.G.; Saraymen, R.; Soyuer, S.; Kilic, E.; Ozcan, S. Aminoguanidine ameliorates radiation-induced oxidative lung damage in rats. Clin. Invest. Med 2008, 31, E182–E188. [Google Scholar]
- Mansour, H.H. Protective role of carnitine ester against radiation-induced oxidative stress in rats. Pharmacol. Res 2006, 54, 165–171. [Google Scholar]
- Bhatia, A.L.; Sharma, A.; Patni, S.; Sharma, A.L. Prophylactic Effect of flaxseed oil against radiation-induced hepatotoxicity in mice. Phytother. Res 2007, 21, 852–859. [Google Scholar]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem 1979, 95, 351–358. [Google Scholar]
| Group | Body weight (g) | Sleep weight (mg) | Sleep index (mg/g) | Thymus weight (mg) | Thymus index (mg/g) | |
|---|---|---|---|---|---|---|
| NC | 31.54 ± 0.97 | 153.62 ± 9.12 # | 4.90 ± 0.70 # | 76.66 ± 6.13 # | 2.44 ± 0.33 # | |
| RC | 30.07 ± 0.88 | 44.19 ± 6.91 * | 1.41 ± 0.28 * | 21.65 ± 2.64 * | 0.69 ± 0.27 * | |
| ALC | 50 | 31.43 ± 0.57 | 57.92 ± 7.98 *,# | 1.84 ± 0.34 *,# | 25.16 ± 3.07 * | 0.79 ± 0.26 * |
| (mg/kg b.w.) | 100 | 30.85 ± 0.75 | 64.35 ± 6.26 *,# | 2.09 ± 0.33 *,# | 60.31 ± 5.38 *,# | 1.95 ± 0.31 *,# |
| + Radiation | 200 | 31.51 ± 0.6 | 73.30 ± 7.15 *,# | 2.33 ± 0.43 *,# | 61.45 ± 3.56 *,# | 1.95 ± 0.31 *,# |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, H.; Wang, Z.; Ma, F.; Yang, X.; Cheng, C.; Yao, L. Protective Effect of Anthocyanin from Lonicera Caerulea var. Edulis on Radiation-Induced Damage in Mice. Int. J. Mol. Sci. 2012, 13, 11773-11782. https://doi.org/10.3390/ijms130911773
Zhao H, Wang Z, Ma F, Yang X, Cheng C, Yao L. Protective Effect of Anthocyanin from Lonicera Caerulea var. Edulis on Radiation-Induced Damage in Mice. International Journal of Molecular Sciences. 2012; 13(9):11773-11782. https://doi.org/10.3390/ijms130911773
Chicago/Turabian StyleZhao, Haitian, Zhenyu Wang, Fengming Ma, Xin Yang, Cuilin Cheng, and Lei Yao. 2012. "Protective Effect of Anthocyanin from Lonicera Caerulea var. Edulis on Radiation-Induced Damage in Mice" International Journal of Molecular Sciences 13, no. 9: 11773-11782. https://doi.org/10.3390/ijms130911773




