Indoleamine 2,3-Dioxygenase (IDO) Downregulates the Cell Surface Expression of the CD4 Molecule
Abstract
:1. Introduction
2. Results and Discussion
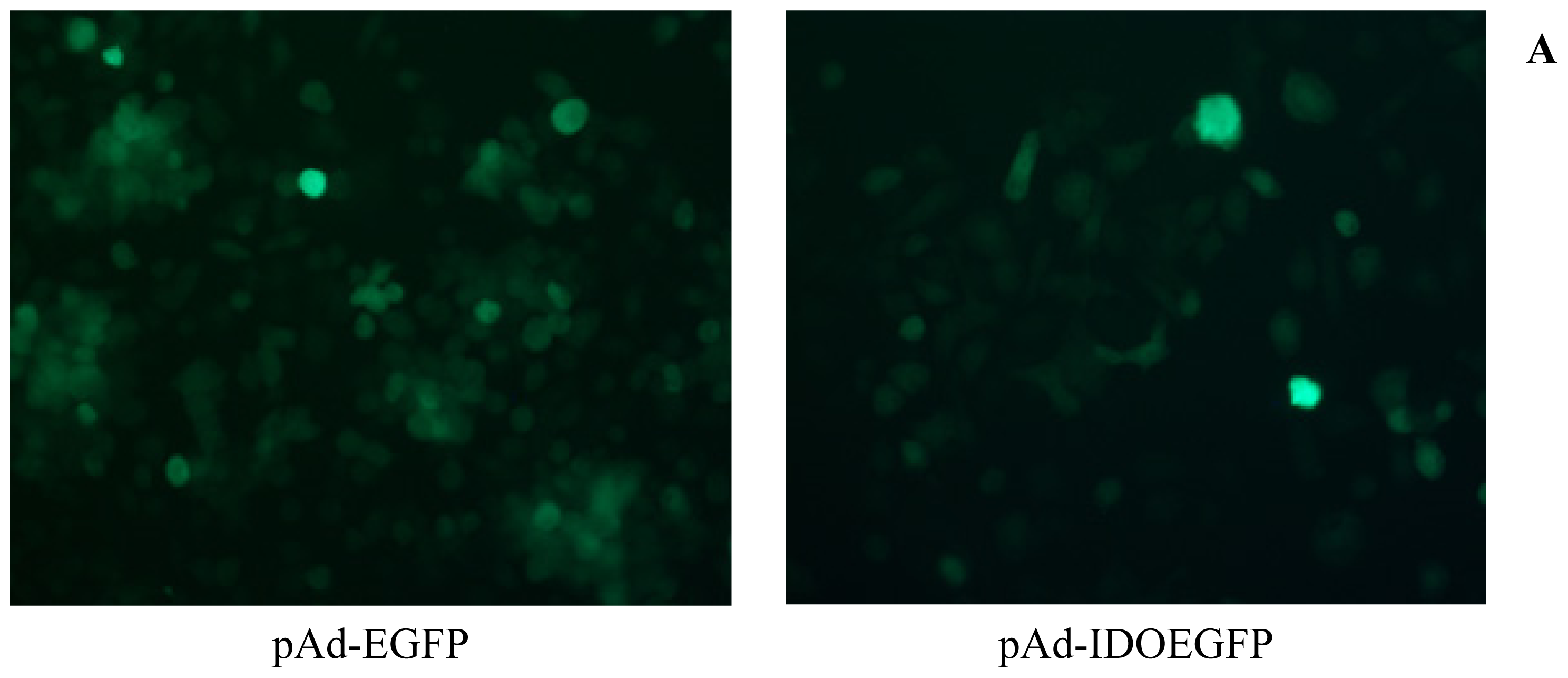
2.1. Infection of MT-2 Cells with Either pAd-EGFP or pAd-IDOEGFP
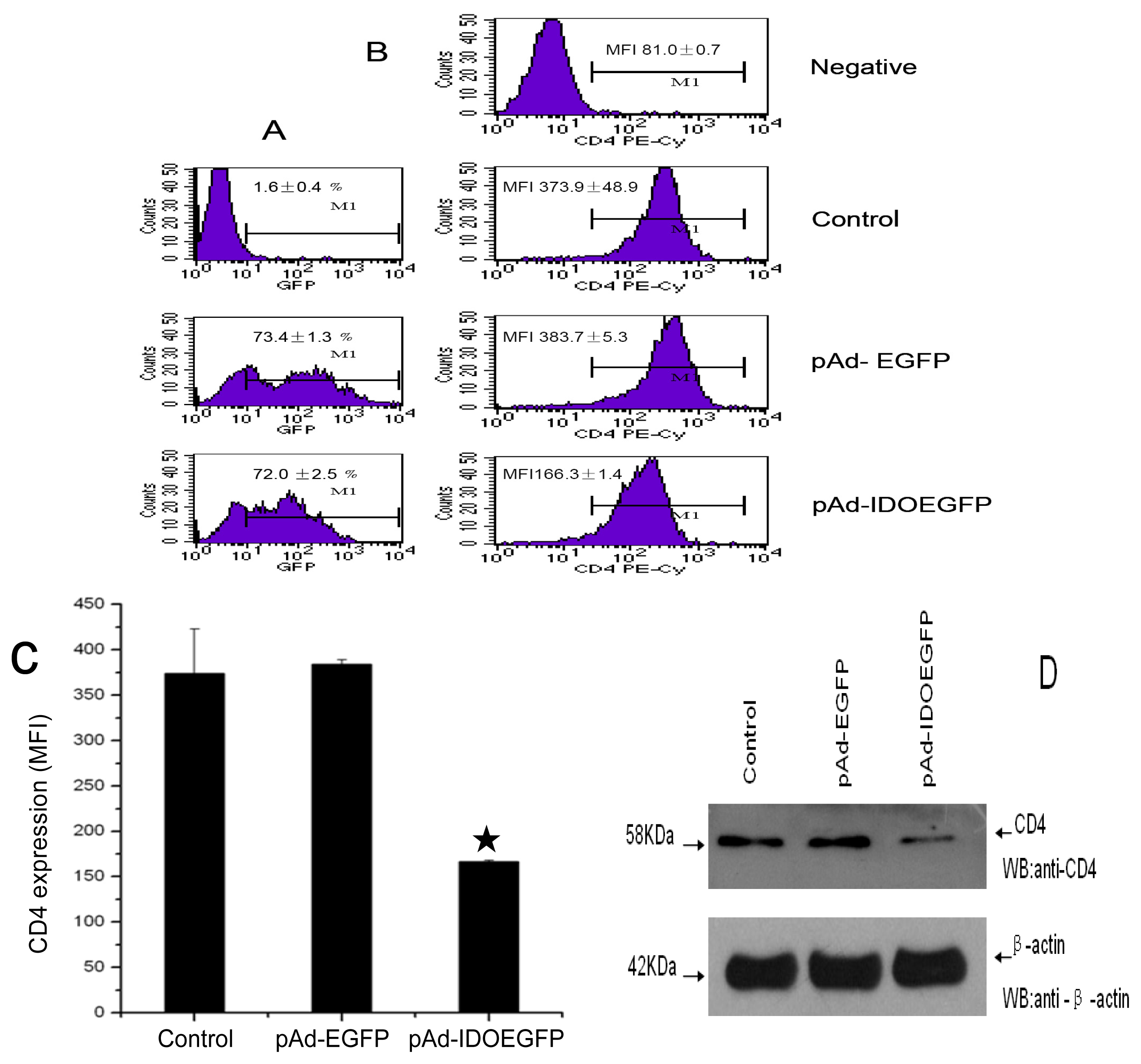
2.2. IDO Downregulates Expression of CD4
2.3. Downregulation of the CD4 Molecule by IDO Is Partially Tryptophan Dependent
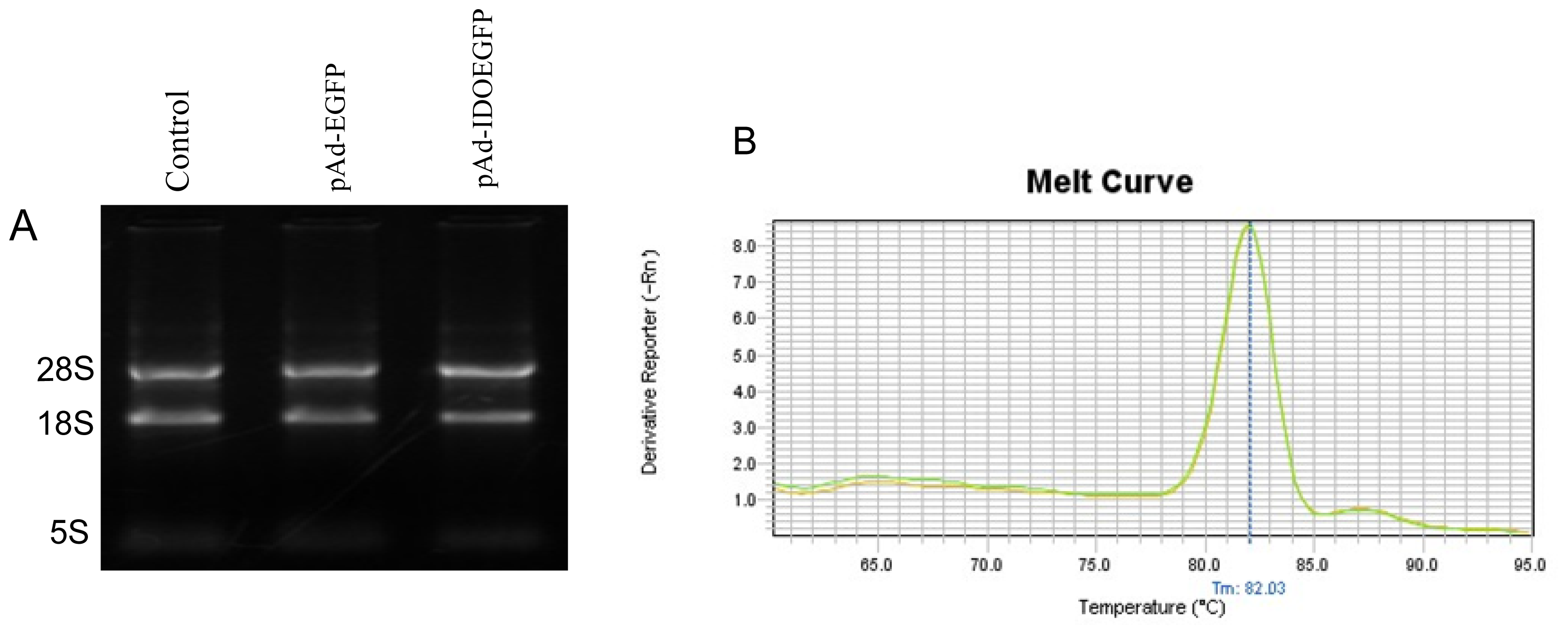
2.4. Effect of IDO on the Level of CD4 mRNA in C8166 Cells
3. Experimental Section
3.1. Cell Culture
3.2. Adenovirus Vector Construction
3.3. Infection and GFP Detection
3.4. Flow Cytometric Analysis
3.5. Western Blot Analysis
3.6. Real Time PCR
3.7. Statistics
4. Conclusions
Acknowledgments
Abbreviations
| EGFP | enhanced green fluorescent protein |
| IDO | indoleamine 2,3-dioxygenase |
| MOI | multiplicity of infection |
| 1-MT | 1-methyl-dl-tryptophan |
| pAd-EGFP | adenoviral vector carry EGFP, or recombinant adenovirus containing EGFP gene |
| pAd-IDOEGFP | adenoviral vector carrying IDOEGFP, or recombinant adenovirus containing IDOEGFP gene |
References
- Taylor, M.W.; Feng, G.S. Relationship between interferon-gamma, indoleamine 2,3-dioxygenase, and tryptophan catabolism. FASEB J 1991, 5, 2516–2522. [Google Scholar]
- Hönig, A.; Rieger, L.; Kapp, M.; Sütterlin, M.; Dietl, J.; Kämmerer, U. Indoleamine 2,3-dioxygenase (IDO) expression in invasive extravillous trophoblast supports role of the enzyme for materno-fetal tolerance. J. Reprod. Immunol 2004, 61, 79–86. [Google Scholar]
- Mellor, A.L.; Munn, D.H. IDO expression in dendritic cells: Tolerance and tryptophan catabolism. Nat. Rev. Immunol 2004, 4, 762–774. [Google Scholar]
- Takikawa, O.; Yoshida, R.; Kido, R.; Hayaishi, O. Tryptophan degradation in mice initiated by indoleamine 2,3-dioxygenase. J. Biol. Chem 1986, 261, 3648–3653. [Google Scholar]
- Munn, D.H.; Shafizadeh, E.; Attwood, J.T.; Bondarev, I.; Pashine, A.; Mellor, A.L. Inhibition of T cell proliferation by macrophage tryptophan catabolism. J. Exp. Med 1999, 9, 1363–1372. [Google Scholar]
- Frumento, G.; Rotondo, R.; Tonetti, M.; Damonte, G.; Benatti, U.; Ferrara, G.B. Tryptophan-derived catabolites are responsible for inhibition of T and natural killer cell proliferation induced by indoleamine 2,3-dioxygenase. J. Exp. Med 2002, 196, 459–468. [Google Scholar]
- Munn, D.H.; Zhou, M.; Attwood, J.T.; Bondarev, I.; Conway, S.J.; Marshall, B.; Brown, C.; Mellor, A.L. Prevention of allogeneic fetal rejection by tryptophan catabolism. Science 1998, 281, 1191–1193. [Google Scholar]
- Mellor, A.L.; Sivakumar, J.; Chandler, P.; Smith, K.; Molina, H.; Mao, D.; Munn, D.H. Prevention of T cell-driven complement activation and inflammation by tryptophan catabolism during pregnancy. Nat. Immunol 2001, 2, 64–68. [Google Scholar]
- Frumento, G.; Rotondo, R.; Tonetti, M.; Ferrara, G.B. T cell proliferation is blocked by indoleamine 2,3-dioxygenase. Transplant. Proc 2001, 33, 428–430. [Google Scholar]
- Boasso, A.; Herbeuval, J.P.; Hardy, A.W.; Anderson, S.A.; Dolan, M.J.; Fuchs, D.; Shearer, G.M. HIV inhibits CD4+ T-cell proliferation by inducing indoleamine 2,3-dioxygenase in plasmacytoid dendritic cells. Blood 2007, 109, 3351–3359. [Google Scholar]
- Maddon, P.J.; Littman, D.R.; Godfrey, M.; Maddon, D.E.; Chess, L.; Axel, R. The isolation and nucleotide sequence of a cDNA encoding the T cell surface protein T4: A new member of the immunoglobulin gene family. Cell 1985, 42, 93–104. [Google Scholar]
- Bowers, K.; Pitcher, C.; Marsh, M. CD4: A co-receptor in the immune response and HIV infection. Int. J. Biochem. Cell Biol 1997, 29, 871–875. [Google Scholar]
- Brady, R.L.; Barclay, A.N. The structure of CD4. Curr. Top. Microbiol 1996, 205, 1–18. [Google Scholar]
- Forouzandeh, F.; Jalili, R.B.; Germain, M.; Duronio, V.; Ghahary, A. Differential immunosuppressive effect of indoleamine 2,3-dioxygenase (IDO) on primary human CD4+ and CD8+ T cells. Mol. Cell Biochem 2008, 309, 1–7. [Google Scholar]
- Li, Y.; Tredget, E.E.; Ghahary, A. Cell surface expression of MHC class I antigen is suppressed in indoleamine 2,3-dioxygenase genetically modified keratinocytes: Implications in allogeneic skin substitute engraftment. Hum. Immunol 2004, 65, 114–123. [Google Scholar]
- Potula, R.; Poluektova, L.; Knipe, B.; Chrastil, J.; Heilman, D.; Dou, H.; Takikawa, O.; Munn, D.H.; Gendelman, H.E.; Persidsky, Y. Inhibition of indoleamine 2,3-dioxygenase (IDO) enhances elimination of virus-infected macrophages in an animal model of HIV-1 encephalitis. Blood 2005, 106, 2382–2390. [Google Scholar]
- He, X.; Xu, L.; Liu, Y.; Cai, X.; Jiang, X.; Zeng, Y. Construction of mammalian expression vector for IDO and EGFP fusion protein and its expression in human chondrocytes (in Chinese). Immunol. J 2004, 20, 172–176. [Google Scholar]
- Entrican, G.; Wattegedera, S.; Rocchi, M.; Wheelhouse, N. Pregnancy indoleamine 2,3-dioxygenase (IDO) and chlamydial abortion: An unresolved paradox. Vet. Microbiol 2009, 135, 98–102. [Google Scholar]
- Grohmann, U.; Orabona, C.; Fallarino, F.; Vacca, C.; Calcinaro, F.; Falorni, A.; Candeloro, P.; Belladonna, M.L.; Bianchi, R.; Fioretti, M.C.; et al. CTLA-4-Ig regulates tryptophan catabolism in vivo. Nat. Immunol 2002, 3, 1097–1101. [Google Scholar]
- Liu, H.; Liu, L.; Visner, G.A. Nonviral gene delivery with indoleamine 2,3-dioxygenase targeting pulmonary endothelium protects against ischemia-reperfusion injury. Am. J. Transplant 2007, 7, 2291–2300. [Google Scholar]
- Yu, G.; Dai, H.; Chen, J.; Duan, L.; Gong, M.; Liu, L.; Xiong, P.; Wang, C.Y.; Fang, M.; Gong, F. Gene delivery of indoleamine 2,3-dioxygenase prolongs cardiac allograft survival by shaping the types of T-cell responses. J. Gene Med 2008, 10, 754–761. [Google Scholar]
- Palafox, D.; Llorente, L.; Alberú, J.; Torres-Machorro, A.; Camorlinga, N.; Rodríguez, C.; Granados, J. The role of indoleamine 2,3 dioxygenase in the induction of immunetolerance in organ transplantation. Transplant. Rev 2010, 24, 160–165. [Google Scholar]
- Kwidzinski, E.; Bunse, J.; Aktas, O.; Richter, D.; Mutlu, L.; Zipp, F.; Nitsch, R.; Bechmann, I. Indolamine 2,3-dioxygenase is expressed in the CNS and down-regulates autoimmune inflammation. FASEB J 2005, 19, 1347–1349. [Google Scholar]
- Platten, M.; Ho, P.P.; Youssef, S.; Fontoura, P.; Garren, H.; Hur, E.M.; Gupta, R.; Lee, L.Y.; Kidd, B.A.; Robinson, W.H.; et al. Treatment of autoimmune neuroinflammation with a synthetic tryptophan metabolite. Science 2005, 310, 850–855. [Google Scholar]
- Fallarino, F.; Volpi, C.; Zelante, T.; Vacca, C.; Calvitti, M.; Fioretti, M.C.; Fioretti, M.C.; Puccetti, P.; Romani, L.; Grohmann, U. IDO mediates TLR9-driven protection from experimental autoimmune diabetes. J. Immunol 2009, 183, 6303–6312. [Google Scholar]
- Larrea, E.; Riezu-Boj, J.I.; Gil-Guerrero, L.; Casares, N.; Aldabe, R.; Sarobe, P.; Civeira, M.P.; Heeney, J.L.; Rollier, C.; Verstrepen, B.; et al. Upregulation of indoleamine 2,3-dioxygenase in hepatitis C virus infection. J. Virol 2007, 81, 3662–3666. [Google Scholar]
- Prendergast, G.C.; Chang, M.Y.; Mandik-Nayak, L.; Metz, R.; Muller, A.J. Indoleamine 2,3-dioxygenase as a modifier of pathogenic inflammation in cancer and other inflammation-associated diseases. Curr. Med. Chem 2011, 18, 2257–2262. [Google Scholar]
- Uyttenhove, C.; Pilotte, L.; Théate, I.; Stroobant, V.; Colau, D.; Parmentier, N.; Boon, T.; van den Eynde, B.J. Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat. Med 2003, 9, 1269–1274. [Google Scholar]
- Muller, A.J.; DuHadaway, J.B.; Donover, P.S.; Sutanto-Ward, E.; Prendergast, G.C. Inhibition of indoleamine 2,3-dioxygenase, an immunoregulatory target of the cancer suppression gene Bin1, potentiates cancer chemotherapy. Nat. Med 2005, 11, 312–319. [Google Scholar]
- Nonaka, H.; Saga, Y.; Fujiwara, H.; Akimoto, H.; Yamada, A.; Kagawa, S.; Takei, Y.; Machida, S.; Takikawa, O.; Suzuki, M. Indoleamine 2,3-dioxygenase promotes peritoneal dissemination of ovarian cancer through inhibition of natural killer cell function and angiogenesis promotion. Int. J. Oncol 2011, 38, 113–1120. [Google Scholar]
- Fallarino, F.; Grohmann, U.; Vacca, C.; Bianchi, R.; Orabona, C.; Spreca, A.; Fioretti, M.C.; Puccetti, P. T cell apoptosis by tryptophan catabolism. Cell Death Differ 2002, 9, 1069–1077. [Google Scholar]
- Fallarino, F.; Grohmann, U.; You, S.; McGrath, B.C.; Cavener, D.R.; Vacca, C.; Orabona, C.; Bianchi, R.; Belladonna, M.L.; Volpi, C.; et al. The combined effects of tryptophan starvation and tryptophan catabolites down-regulate T cell receptor zeta-chain and induce a regulatory phenotype in naive T cells. J. Immunol 2006, 176, 6752–6761. [Google Scholar]
- Curti, A.; Pandolfi, S.; Valzasina, B.; Aluigi, M.; Isidori, A.; Ferri, E.; Salvestrini, V.; Bonanno, G.; Rutella, S.; Durelli, I.; et al. Modulation of tryptophan catabolism by human leukemic cells results in the conversion of CD25− into CD25+ T regulatory cells. Blood 2007, 109, 2871–2877. [Google Scholar]
- Wahlfors, J.; Loimas, S.; Pasanen, T.; Hakkarainen, T. Green fluorescent protein (GFP) fusion constructs in gene therapy research. Histochem. Cell Biol 2001, 115, 59–65. [Google Scholar]
- Ao, Z.; Huang, G.; Yao, H.; Xu, Z.; Labine, M.; Cochrane, A.W.; Yao, X. Interaction of human immunodeficiency virus type 1 integrase with cellular nuclear import receptor importin7 and its impact on viral replication. J. Biol. Chem 2007, 282, 13456–13467. [Google Scholar]
- Zamoyska, R. CD4 and CD8: Modulators of T-cell receptor recognition of antigen and of immune responses? Curr. Opin. Immunol 1998, 10, 82–87. [Google Scholar]
- Marodon, G. CD4 down modulation on T-cells: An ‘immune’ checkpoint for HIV. Immunol. Lett 2001, 79, 165–168. [Google Scholar]
- Wildum, S.; Schindler, M.; Münch, J.; Kirchhoff, F. Contribution of Vpu, Env, and Nef to CD4 down-modulation and resistance of human immunodeficiency virus type 1-infected T cells to superinfection. J. Virol 2006, 80, 8047–8059. [Google Scholar]
- Chaudhuri, R.; Lindwasser, O.W.; Smith, W.J.; Hurley, J.H.; Bonifacino, J.S. Downregulation of CD4 by human immunodeficiency virus type 1 Nef is dependent on clathrin and involves direct interaction of Nef with the AP2 clathrin adaptor. J. Virol 2007, 81, 3877–3890. [Google Scholar]
- Coleman, S.H.; Madrid, R.; van Damme, N.; Mitchell, R.S.; Bouchet, J.; Servant, C.; Pillai, S.; Benichou, S.; Guatelli, J.C. Modulation of cellular protein trafficking by human immunodeficiency virus type 1 Nef: Role of the acidic residue in the ExxxLL motif. J. Virol 2006, 80, 1837–1849. [Google Scholar]



| β-actin Ct (Mean ± SD) | CD4 Ct (Mean ± SD) | 2−ΔΔCt | GAPDH Ct (Mean ± SD) | CD4 Ct (Mean ± SD) | 2−ΔΔCt | |
|---|---|---|---|---|---|---|
| Control | 30.35 ± 0.10 | 32.44 ± 2.63 | 1 | 31.34 ± 0.06 | 32.33 ± 2.51 | 1 |
| pAd-EGFP | 31.03 ± 0.46 | 33.58 ± 2.40 | 1.04 ± 0.05 | 32.34 ± 0.21 | 33.42 ± 2.55 | 1.02 ± 0.07 |
| pAd-IDOEGFP | 30.89 ± 0.27 | 33.02 ± 2.25 | 1.08 ± 0.09 | 31.86 ± 0.42 | 32.78 ± 2.10 | 1.05 ± 0.02 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, G.; Zeng, Y.; Liang, P.; Zhou, C.; Zhao, S.; Huang, X.; Wu, L.; He, X. Indoleamine 2,3-Dioxygenase (IDO) Downregulates the Cell Surface Expression of the CD4 Molecule. Int. J. Mol. Sci. 2012, 13, 10863-10879. https://doi.org/10.3390/ijms130910863
Huang G, Zeng Y, Liang P, Zhou C, Zhao S, Huang X, Wu L, He X. Indoleamine 2,3-Dioxygenase (IDO) Downregulates the Cell Surface Expression of the CD4 Molecule. International Journal of Molecular Sciences. 2012; 13(9):10863-10879. https://doi.org/10.3390/ijms130910863
Chicago/Turabian StyleHuang, Guanyou, Yaoying Zeng, Peiyan Liang, Congrong Zhou, Shuyun Zhao, Xiuyan Huang, Lingfei Wu, and Xianhui He. 2012. "Indoleamine 2,3-Dioxygenase (IDO) Downregulates the Cell Surface Expression of the CD4 Molecule" International Journal of Molecular Sciences 13, no. 9: 10863-10879. https://doi.org/10.3390/ijms130910863




