Combined Phosphatase and Tensin Homolog (PTEN) Loss and Fatty Acid Synthase (FAS) Overexpression Worsens the Prognosis of Chinese Patients with Hepatocellular Carcinoma
Abstract
:1. Introduction
2. Results
2.1. Clinicopathological Characteristics of Patients with HCC
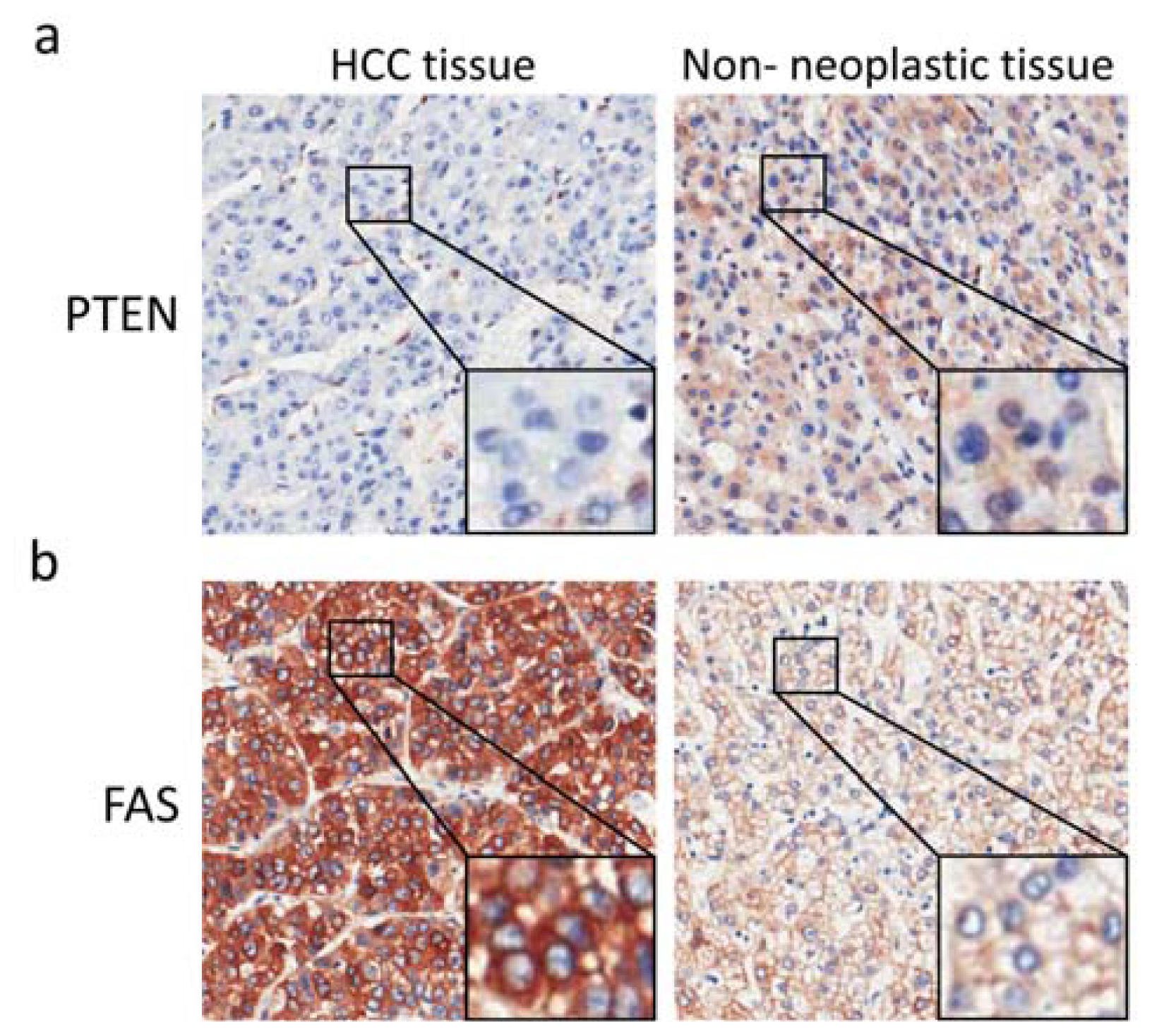
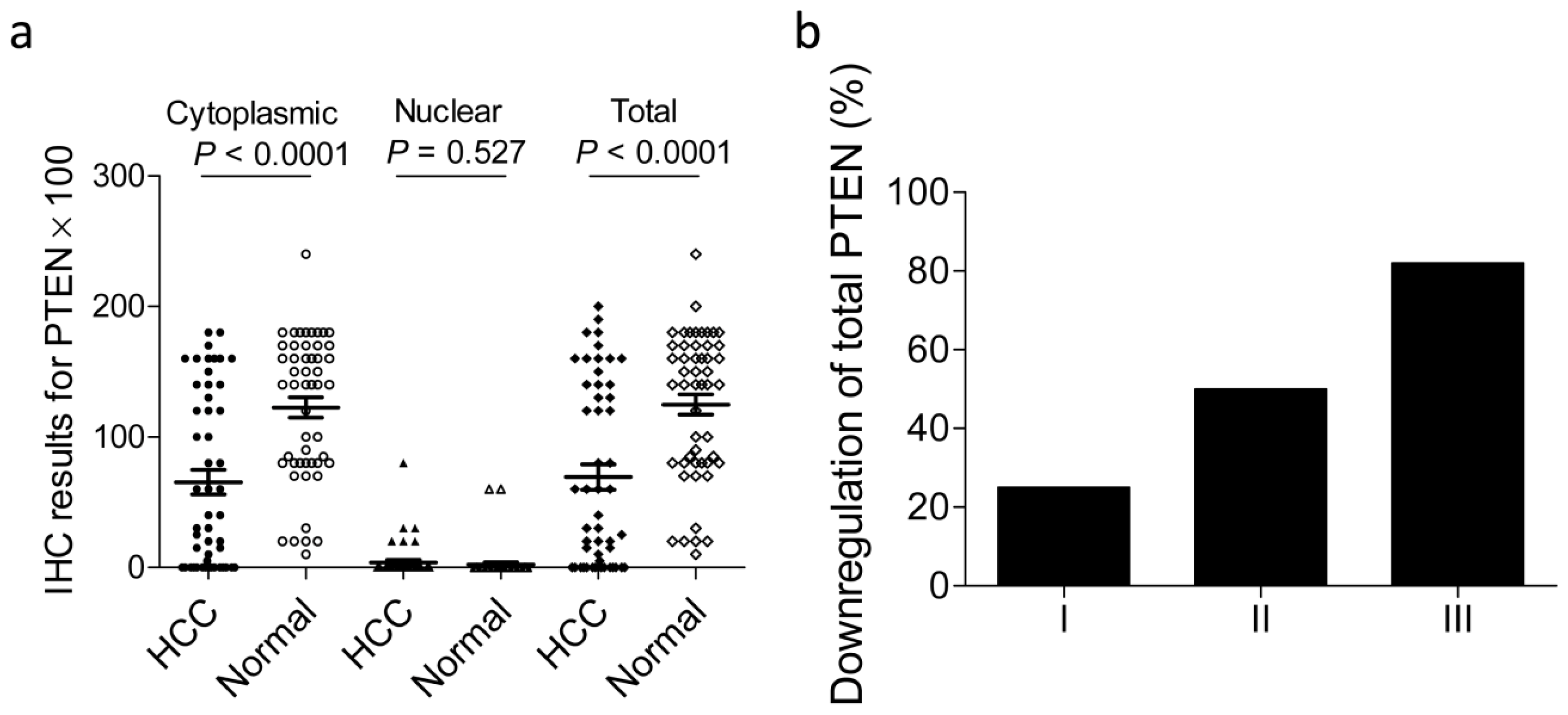
2.2. Loss of PTEN Expression in Patients with HCC
2.3. Correlations between Downregulated PTEN Expression in HCC and Clinicopathological Characteristics
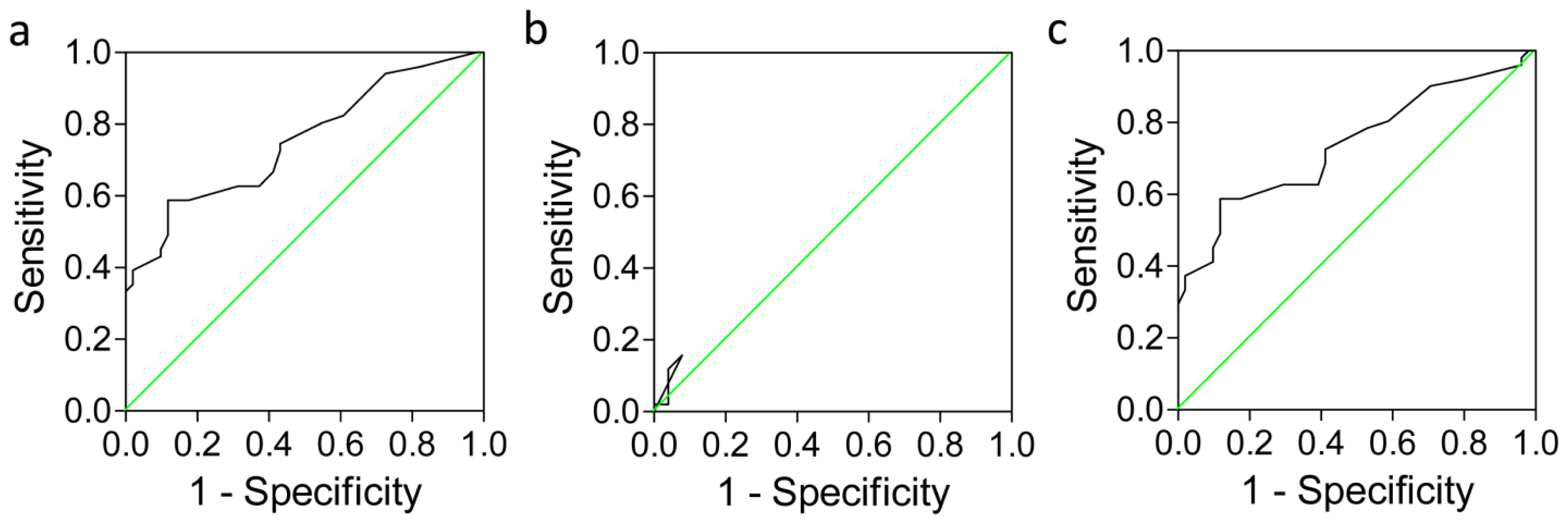
2.4. Clinical Sensitivity and Specificity of PTEN Expression in HCC
2.5. Correlation between PTEN Loss and FAS Overexpression in HCC
2.6. Effects of PTEN Loss and FAS Overexpression on Survival of Patients with HCC
3. Experimental Section
3.1. Tissue Microarray
3.2. Immunohistochemistry and Scoring
3.3. Statistical Analysis
4. Discussion
5. Conclusions
Acknowledgments
- Competing InterestsGlaxoSmithKline Research and Development Center is a division of GlaxoSmithKline. Xia Qin, Joel Greshock, Kurtis E. Bachman, Richard Wooster and Crystal Ying Qin are employees of GlaxoSmithKline. This does not alter our adherence to all the International Journal of Molecular Sciences policies on sharing data and materials.
References
- Okuda, K. Hepatocellular carcinoma. J. Hepatol 2000, 32, 225–237. [Google Scholar]
- Chen, C.J.; Yu, M.W.; Liaw, Y.F. Epidemiological characteristics and risk factors of hepatocellular carcinoma. J. Gastroenterol. Hepatol 1997, 12, S294–S308. [Google Scholar]
- Sherman, M. Hepatocellular carcinoma: Epidemiology, risk factors, and screening. Semin. Liver Dis 2005, 25, 143–154. [Google Scholar]
- Li, J.; Yen, C.; Liaw, D.; Podsypanina, K.; Bose, S.; Wang, S.I.; Puc, J.; Miliaresis, C.; Rodgers, L.; McCombie, R.; et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 1997, 275, 1943–1947. [Google Scholar]
- Tamura, M.; Gu, J.; Tran, H.; Yamada, K.M. PTEN gene and integrin signaling in cancer. J. Natl. Cancer Inst 1999, 91, 1820–1828. [Google Scholar]
- Eng, C. PTEN: One gene, many syndromes. Hum. Mutat 2003, 22, 183–198. [Google Scholar]
- Di Cristofano, A.; Pandolfi, P.P. The multiple roles of PTEN in tumor suppression. Cell 2000, 100, 387–390. [Google Scholar]
- Hollander, M.C.; Blumenthal, G.M.; Dennis, P.A. PTEN loss in the continuum of common cancers, rare syndromes and mouse models. Nat. Rev. Cancer 2011, 11, 289–301. [Google Scholar]
- Menendez, J.A.; Lupu, R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat. Rev. Cancer 2007, 7, 763–777. [Google Scholar]
- Van de Sande, T.; de Schrijver, E.; Heyns, W.; Verhoeven, G.; Swinnen, J.V. Role of the phosphatidylinositol 3′-kinase/PTEN/Akt kinase pathway in the overexpression of fatty acid synthase in LNCaP prostate cancer cells. Cancer Res 2002, 62, 642–646. [Google Scholar]
- Stiles, B.; Wang, Y.; Stahl, A.; Bassilian, S.; Lee, W.P.; Kim, Y.J.; Sherwin, R.; Devaskar, S.; Lesche, R.; Magnuson, M.A.; Wu, H. Liver-specific deletion of negative regulator Pten results in fatty liver and insulin hypersensitivity [corrected]. Proc. Natl. Acad. Sci. USA 2004, 101, 2082–2087. [Google Scholar]
- Remmele, W.; Stegner, H.E. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 1987, 8, 138–140. [Google Scholar]
- Salmena, L.; Carracedo, A.; Pandolfi, P.P. Tenets of PTEN tumor suppression. Cell 2008, 133, 403–414. [Google Scholar]
- Manning, B.D.; Cantley, L.C. AKT/PKB signaling: Navigating downstream. Cell 2007, 129, 1261–1274. [Google Scholar]
- Kim, D.; Kim, S.; Koh, H.; Yoon, S.O.; Chung, A.S.; Cho, K.S.; Chung, J. Akt/PKB promotes cancer cell invasion via increased motility and metalloproteinase production. FASEB J 2001, 15, 1953–1962. [Google Scholar]
- Song, M.S.; Carracedo, A.; Salmena, L.; Song, S.J.; Egia, A.; Malumbres, M.; Pandolfi, P.P. Nuclear PTEN regulates the APC-CDH1 tumor-suppressive complex in a phosphatase-independent manner. Cell 2011, 144, 187–199. [Google Scholar]
- Gimm, O.; Perren, A.; Weng, L.P.; Marsh, D.J.; Yeh, J.J.; Ziebold, U.; Gil, E.; Hinze, R.; Delbridge, L.; Lees, J.A.; et al. Differential nuclear and cytoplasmic expression of PTEN in normal thyroid tissue, and benign and malignant epithelial thyroid tumors. Am. J. Pathol 2000, 156, 1693–1700. [Google Scholar]
- McMenamin, M.E.; Soung, P.; Perera, S.; Kaplan, I.; Loda, M.; Sellers, W.R. Loss of PTEN expression in paraffin-embedded primary prostate cancer correlates with high Gleason score and advanced stage. Cancer Res 1999, 59, 4291–4296. [Google Scholar]
- Halvorsen, O.J.; Haukaas, S.A.; Akslen, L.A. Combined loss of PTEN and p27 expression is associated with tumor cell proliferation by Ki-67 and increased risk of recurrent disease in localized prostate cancer. Clin. Cancer Res 2003, 9, 1474–1479. [Google Scholar]
- Sze, K.M.; Wong, K.L.; Chu, G.K.; Lee, J.M.; Yau, T.O.; Ng, I.O. Loss of phosphatase and tensin homolog enhances cell invasion and migration through AKT/Sp-1 transcription factor/matrix metalloproteinase 2 activation in hepatocellular carcinoma and has clinicopathologic significance. Hepatology 2011, 53, 1558–1569. [Google Scholar]
- Chen, J.S.; Wang, Q.; Fu, X.H.; Huang, X.H.; Chen, X.L.; Cao, L.Q.; Chen, L.Z.; Tan, H.X.; Li, W.; Bi, J.; et al. Involvement of PI3K/PTEN/AKT/mTOR pathway in invasion and metastasis in hepatocellular carcinoma: Association with MMP-9. Hepatol. Res 2009, 39, 177–186. [Google Scholar]
- Hu, T.H.; Huang, C.C.; Lin, P.R.; Chang, H.W.; Ger, L.P.; Lin, Y.W.; Changchien, C.S.; Lee, C.M.; Tai, M.H. Expression and prognostic role of tumor suppressor gene PTEN/MMAC1/TEP1 in hepatocellular carcinoma. Cancer 2003, 97, 1929–1940. [Google Scholar]
- Wu, S.K.; Wang, B.J.; Yang, Y.; Feng, X.H.; Zhao, X.P.; Yang, D.L. Expression of PTEN, PPM1A and P-Smad2 in hepatocellular carcinomas and adjacent liver tissues. World J. Gastroenterol 2007, 13, 4554–4559. [Google Scholar]
- Bandyopadhyay, S.; Pai, S.K.; Watabe, M.; Gross, S.C.; Hirota, S.; Hosobe, S.; Tsukada, T.; Miura, K.; Saito, K.; Markwell, S.J.; et al. FAS expression inversely correlates with PTEN level in prostate cancer and a PI 3-kinase inhibitor synergizes with FAS siRNA to induce apoptosis. Oncogene 2005, 24, 5389–5395. [Google Scholar]
- Hu, T.H.; Wang, C.C.; Huang, C.C.; Chen, C.L.; Hung, C.H.; Chen, C.H.; Wang, J.H.; Lu, S.N.; Lee, C.M.; Changchien, C.S.; et al. Down-regulation of tumor suppressor gene PTEN, overexpression of p53, plus high proliferating cell nuclear antigen index predict poor patient outcome of hepatocellular carcinoma after resection. Oncol. Rep 2007, 18, 1417–1426. [Google Scholar]

| Characteristic | Total Number | Cytoplasmic PTEN Negative | p Value | Nuclear PTEN Negative | p Value | Total PTEN Negative | p Value | Ca/CN Total ≤ 0.5 | p Value |
|---|---|---|---|---|---|---|---|---|---|
| 15 (29%) | 43 (84%) | 13 (25%) | 29 (57%) | ||||||
| Sex | |||||||||
| M | 45 | 13 (29%) | 1 | 38 (84%) | 1 | 11 (24%) | 0.638 | 26 (58%) | 1 |
| F | 6 | 2 (33%) | 5 (83%) | 2 (33%) | 3 (50%) | ||||
| Age | |||||||||
| Median | 52 | 55 | 52 | 54 | |||||
| Range | 43–72 | 38–72 | 43–72 | 39–72 | |||||
| Histological grade | |||||||||
| I | 4 | 1 (25%) | 0.478 | 4 (100%) | 0.404 | 1 (25%) | 0.106 | 1 (25%) | 0.043 a |
| II | 34 | 9 (26%) | 29 (85%) | 7 (21%) | 17 (50%) | ||||
| III | 11 | 5 (45%) | 8 (73%) | 5 (45%) | 9 (82%) | ||||
| TNM stage | |||||||||
| I | 38 | 13 (34%) | 0.336 | 33 (87%) | 0.564 | 11 (29%) | 0.419 | 22 (58%) | 0.406 |
| II | 5 | 0 (0%) | 4 (80%) | 0 (0%) | 3 (60%) | ||||
| III | 6 | 2 (33%) | 4 (67%) | 2 (33%) | 4 (67%) | ||||
| IV | 2 | 0 (0%) | 2 (100%) | 0 (0%) | 0 (0%) | ||||
| Depth of invasion | |||||||||
| T1 | 40 | 13 (33%) | 0.315 | 35 (88%) | 0.409 | 11 (28%) | 0.37 | 22 (55%) | 0.856 |
| T2 | 5 | 0 (0%) | 4 (80%) | 0 (0%) | 3 (60%) | ||||
| T3 | 6 | 2 (33%) | 4 (67%) | 2 (33%) | 4 (67%) | ||||
| Lymph node metastasis | |||||||||
| N0 | 50 | 15 (30%) | 0.515 | 42 (84%) | 0.663 | 13 (26%) | 0.555 | 29 (58%) | 0.246 |
| N1 | 1 | 0 (0%) | 1 (100%) | 0 (0%) | 0 (0%) | ||||
| Distant metastasis | |||||||||
| M0 | 50 | 15 (30%) | 0.515 | 42 (84%) | 0.663 | 13 (26%) | 0.555 | 29 (58%) | 0.246 |
| M1 | 1 | 0 (0%) | 1 (100%) | 0 (0%) | 0 (0%) |
| PTEN Expression Pattern | AUC (95% CI) | p Value | Optimal Cut-Off Point | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Sensitivity | Specificity | +PV | −PV | +LR | −LR | DISEASE Accuracy | |||
| Cytoplasmic | 0.752 (0.656–0.832) | <0.0001 | 58.8% | 88.2% | 83.3% | 68.2% | 5.00 | 0.47 | 83.3% |
| Nuclear | 0.539 (0.438–0.638) | 0.494 | 11.8% | 96.1% | 75.0% | 52.1% | 3.00 | 0.92 | 75.2% |
| Total | 0.739 (0.642–0.821) | <0.0001 | 58.8% | 88.2% | 83.3% | 68.2% | 5.00 | 0.47 | 83.3% |
| PTEN Expression Pattern | Total Number | FAS Overexpression | p Value |
|---|---|---|---|
| Cytoplasmic PTEN | |||
| Negative | 15 | 4 (26.7%) | 0.054 |
| Positive | 36 | 2 (5.6%) | |
| Nuclear PTEN | |||
| Negative | 43 | 6 (14.0%) | 0.572 |
| Positive | 8 | 0 (0.0%) | |
| Total PTEN | |||
| Negative | 13 | 4 (30.8%) | 0.014 |
| Positive | 38 | 2 (5.3%) | |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhu, X.; Qin, X.; Fei, M.; Hou, W.; Greshock, J.; Bachman, K.E.; Wooster, R.; Kang, J.; Qin, C.Y. Combined Phosphatase and Tensin Homolog (PTEN) Loss and Fatty Acid Synthase (FAS) Overexpression Worsens the Prognosis of Chinese Patients with Hepatocellular Carcinoma. Int. J. Mol. Sci. 2012, 13, 9980-9991. https://doi.org/10.3390/ijms13089980
Zhu X, Qin X, Fei M, Hou W, Greshock J, Bachman KE, Wooster R, Kang J, Qin CY. Combined Phosphatase and Tensin Homolog (PTEN) Loss and Fatty Acid Synthase (FAS) Overexpression Worsens the Prognosis of Chinese Patients with Hepatocellular Carcinoma. International Journal of Molecular Sciences. 2012; 13(8):9980-9991. https://doi.org/10.3390/ijms13089980
Chicago/Turabian StyleZhu, Xuehua, Xia Qin, Maogui Fei, Wenmin Hou, Joel Greshock, Kurtis E. Bachman, Richard Wooster, Jiuhong Kang, and Crystal Ying Qin. 2012. "Combined Phosphatase and Tensin Homolog (PTEN) Loss and Fatty Acid Synthase (FAS) Overexpression Worsens the Prognosis of Chinese Patients with Hepatocellular Carcinoma" International Journal of Molecular Sciences 13, no. 8: 9980-9991. https://doi.org/10.3390/ijms13089980




