Hepatoprotective Effects of Berberis vulgaris L. Extract/β Cyclodextrin on Carbon Tetrachloride–Induced Acute Toxicity in Mice
Abstract
:1. Introduction
2. Results and Discussion
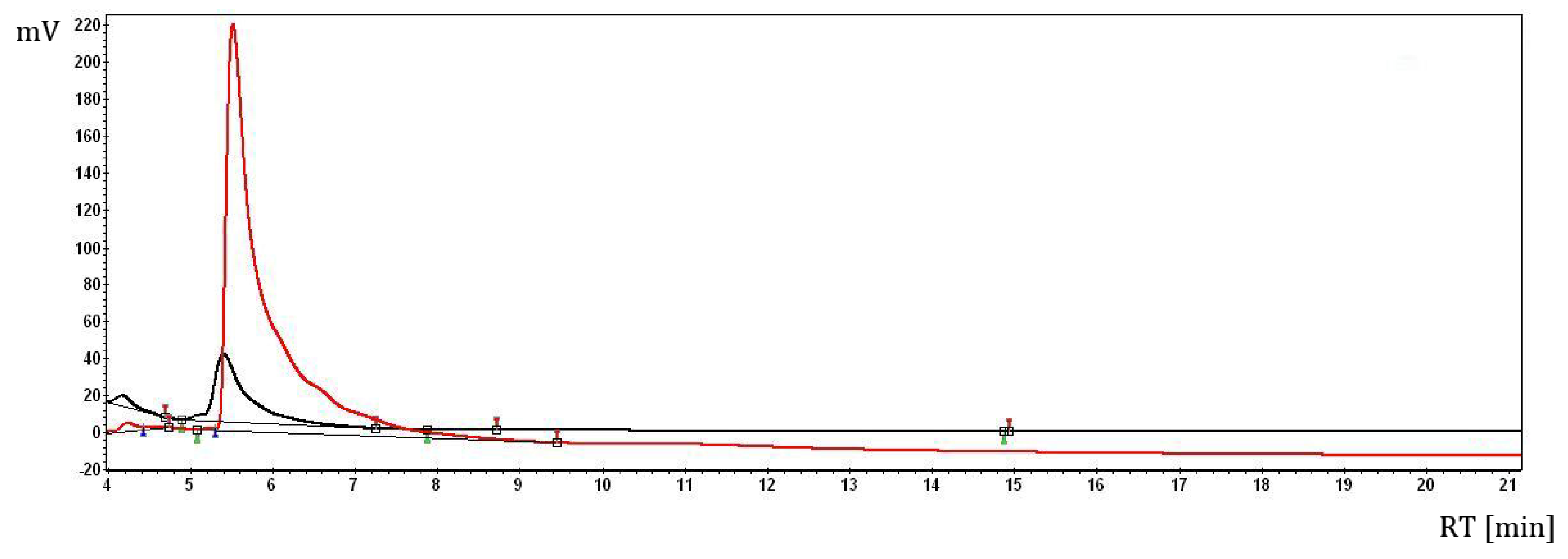
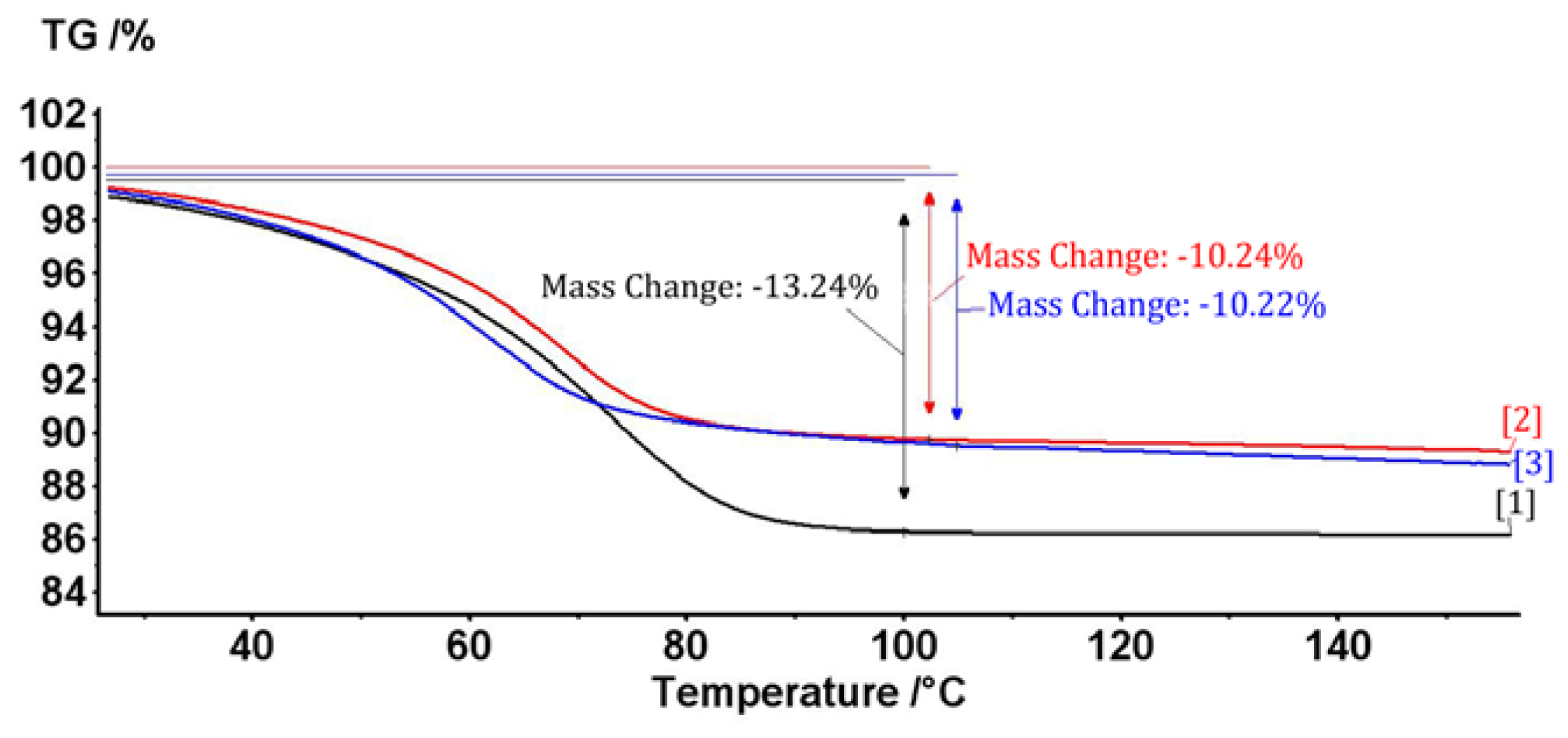
2.1. The Complex of Berberis vulgaris Extract and β Cyclodextrin
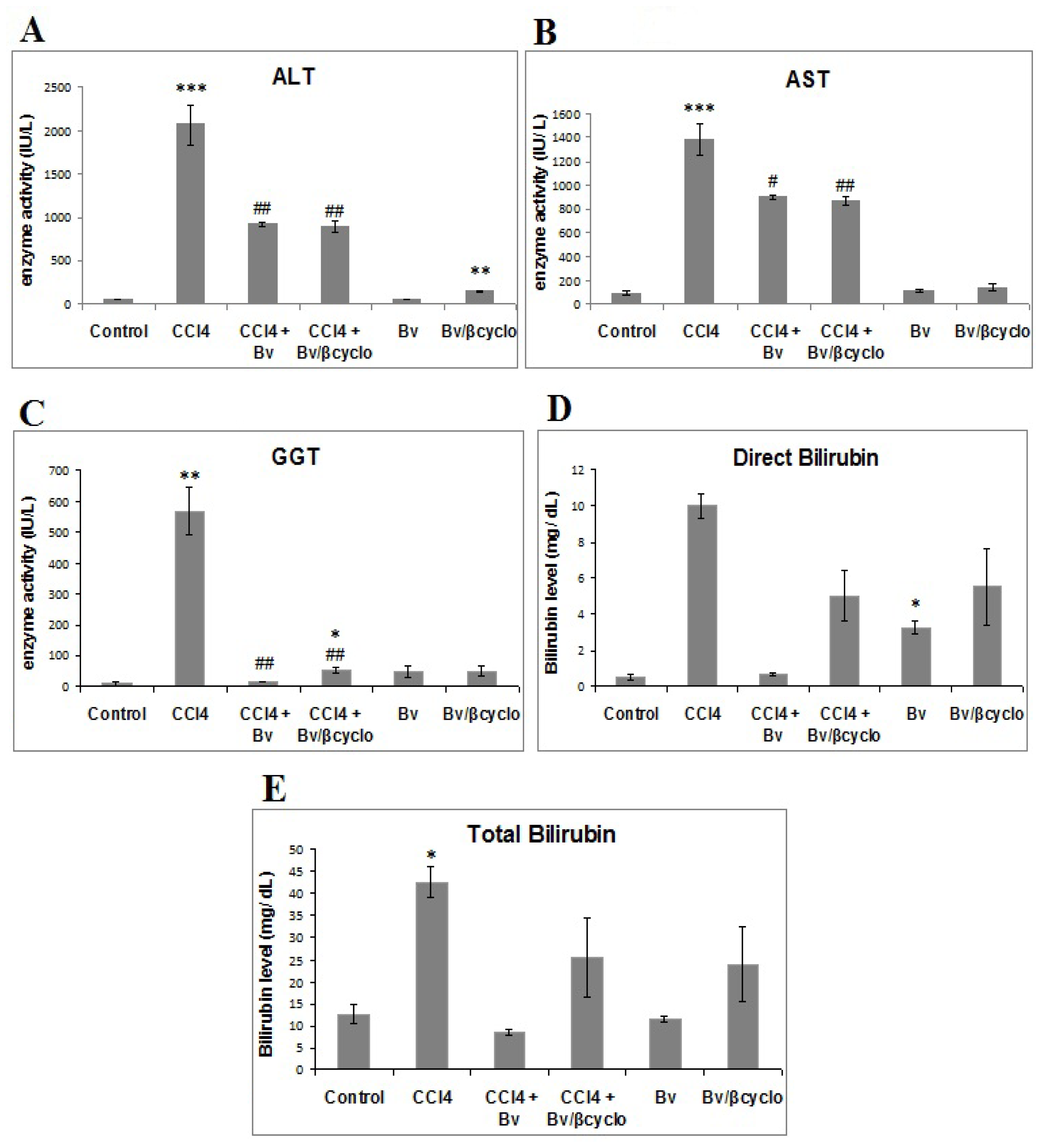
2.2. Effects of Berberis vulgaris Extract/β Cyclodextrin Pre-Treatment on Serum ALT, AST, γ-GT, Total and Direct Bilirubin
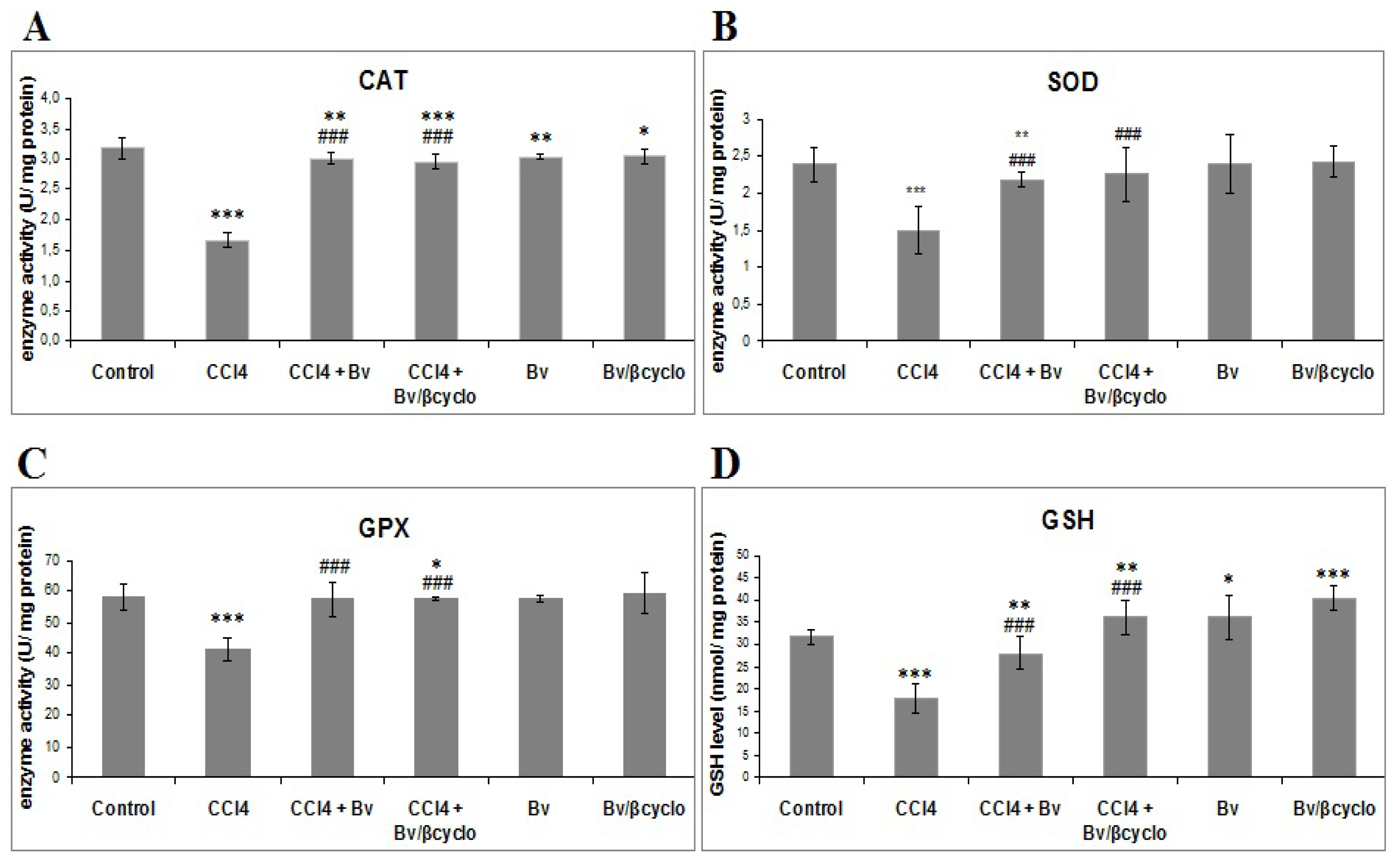
2.3. Effects of Berberis vulgaris Extract/β Cyclodextrin Pre-Treatment on Antioxidant Status and Lipid Peroxidation
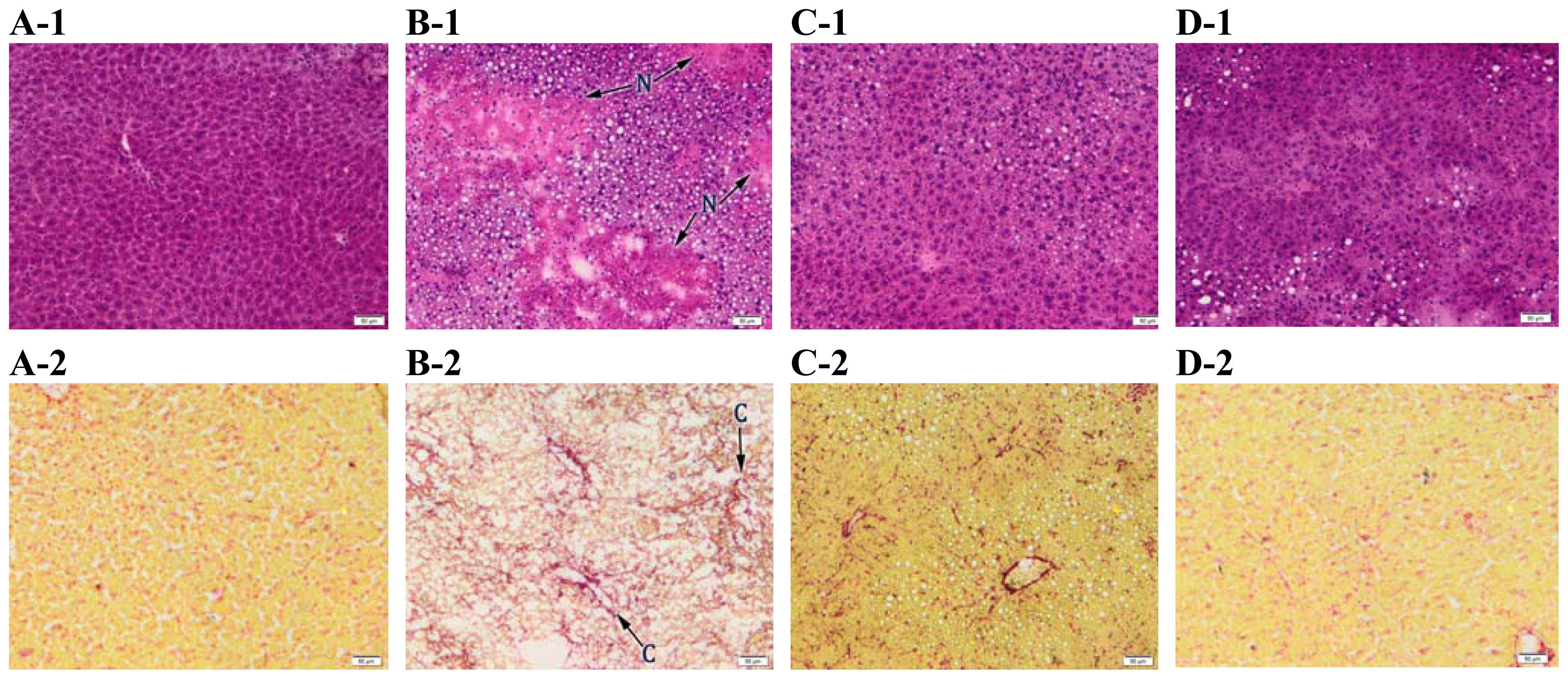
2.4. Histopathology
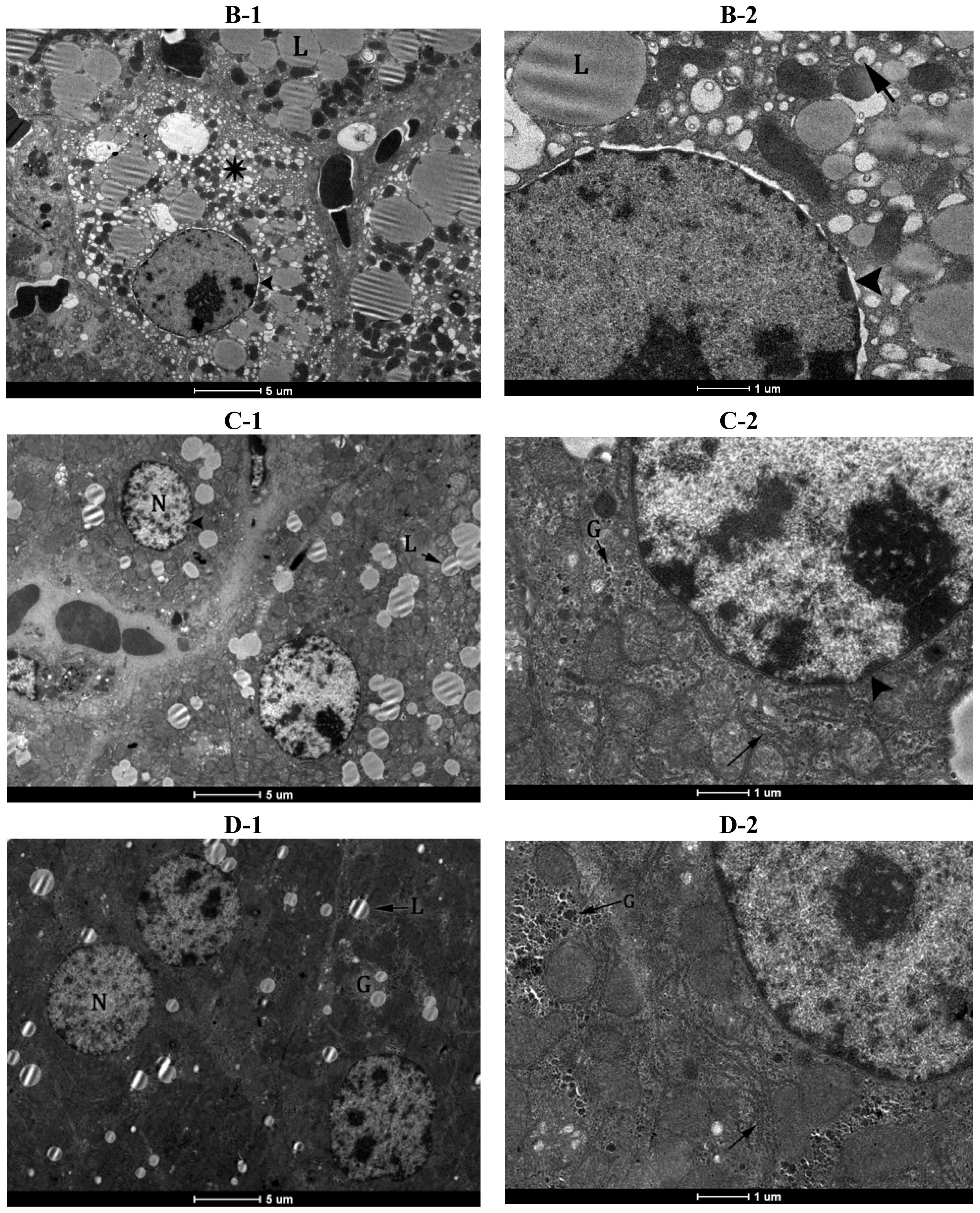
2.5. Electron Microscopy
2.6. Effects of Berberis vulgaris Extract/β-Cyclodextrin Pre-Treatment on DNA Damage
3. Experimental Section
3.1. Berberis vulgaris Extract/β-Cyclodextrin Formulation
3.2. Animals and Experimental Procedure
3.3. Animals and Experimental Procedure
3.4. Histopathology
3.5. Electron Microscopy
3.6. Biochemical Assays
3.6.1. Activities of Serum Hepatic Markers
3.6.2. Assesment of Antioxidant Status
3.6.3. Assay of Lipid Peroxidation
3.6.4. Protein Concentration Measurement
3.7. DNA Fragmentation Assays
3.8. Statistical Analysis
4. Conclusions
Acknowledgment
References
- Jones, A.L. Anatomy of the Normal Liver. In Hepatology: A Textbook of Liver Disease, 3rd ed; Zakin, D., Boyer, T.D., Eds.; WB Saunders: Philadelphia, PA, USA, 1996; pp. 3–32. [Google Scholar]
- Zimmerman, H.J. Hepatotoxicity: The Adverse Effects of Drugs and other Chemicals on the Liver; Appleton-Century Crofts Publisher: New York, NY, USA, 1978. [Google Scholar]
- Kaminski, M.; Wiaderkiewicz, R. The role of the liver in xenobiotic biotransformation. Part I. The role of the liver and its cells and their interactions. Probl. Forensic Sci 2007, 72, 357–378. [Google Scholar]
- Weber, L.W.; Boll, M.; Stampfl, A. Hepatotoxicity and mechanism of action of haloalkanes: Carbon tetrachloride as a toxicological model. Crit. Rev. Toxicol 2003, 33, 105–136. [Google Scholar]
- Merrick, B.A. Toxicoproteomics in liver injury and inflammation. Ann. N. Y. Acad. Sci 2006, 1076, 707–717. [Google Scholar]
- Manibusan, M.K.; Odin, M.; Eastmond, D.A. Postulated carbon tetrachloride mode of action: A review. J. Environ. Sci. Health C 2007, 25, 185–209. [Google Scholar]
- Luper, S. A review of plants used in the treatment of liver disease: Part 1. Altern. Med. Rev 1998, 3, 410–421. [Google Scholar]
- Luper, S. A review of plants used in the treatment of liver disease: Part 2. Altern. Med. Rev 1999, 4, 178–188. [Google Scholar]
- Murriel, P.; Rivera-Espinoza, Y. Beneficial drugs for liver diseases. J. Appl. Toxicol 2008, 28, 93–103. [Google Scholar]
- Ivanovska, N.; Philipov, S. Study on the anti-inflamatory action of Berberis vulgaris root extract, alkaloid fractions and pure alkaloids. Int. J. Immunopharmacol 1996, 18, 553–561. [Google Scholar]
- Saied, S.; Begum, S. Phytochemical studies of Berberis vulgaris. Chem. Nat. Compd 2004, 40, 137–140. [Google Scholar]
- Imanshahidi, M.; Hosseinzadeh, H. Pharmacological and therapeutical effects of Berberis vulgaris and its active constituent, berberine. Phytother. Res 2008, 22, 999–1012. [Google Scholar]
- Freile, M.L.; Giannini, F.; Pucci, G. Antimicrobial activity of aqueolus extracts and of berberine isolates from Berberis heterophylla. Fitoterapia 2003, 74, 702–705. [Google Scholar]
- Mahady, G.B.; Pendland, S.L.; Stoia, A.; Chaadwick, L.R. In vitro susceptibility of Helicobacter pylori to isoquinoline alkaloids from Sanguinaria canadensis and Hydrastis canadensis. Phytother. Res 2003, 17, 217–221. [Google Scholar]
- Iizuka, N.; Miyamoto, K.; Okita, K.; Tangoku, A.; Hayashi, H.; Yosino, S. Inhibitory effect of Coptidis rhizoma and berberine on the proliferation of human esophageal cancer cell lines. Cancer Lett 2000, 148, 19–25. [Google Scholar]
- Thirupurasundari, C.J.; Padmini, R.; Devaraj, S.N. Effect of berberine on the antioxidant status, ultrastructural modifications and protein bound carbohydrates in azoxymethane-induced colon cancer in rats. Chem. Biol. Interact 2009, 177, 190–195. [Google Scholar]
- Wang, N.; Feng, Y.; Zhu, M.; Tsang, C.M.; Man, K.; Tong, Y.; Tsao, S.W. Berberine induces autophagic cell death and mitochondrial apoptosis in liver cancer cells: The cellular mechanism. J. Cell. Biochem 2010, 111, 1426–1436. [Google Scholar] [Green Version]
- Kuo, C.L.; Chi, C.W.; Liu, T.Y. The anti-inflamatory potential of berberine in vitro and in vivo. Cancer Lett 2004, 203, 127–137. [Google Scholar]
- Kupeli, E.; Kosar, M.; Yesilada, E.; Husnu, K.; Baser, C. A comparative study on the anti-inflamatory, antinociceptive and antipyretic effects of isoquinoline alkaloids from the root of Turkish Berberis species. Life Sci 2002, 72, 645–657. [Google Scholar]
- Singh, A.; Duggal, S.; Kaur, N.; Singh, J. Berberine: Alkaloid with wide spectrum of pharmacological activities. J. Nat. Prod 2010, 3, 64–75. [Google Scholar]
- Hobara, N.; Watanabe, A. Berberine-Induced bile bilirubin secretion in the rat. Curr. Ther. Res. Clin. Exp 1984, 35, 663–667. [Google Scholar]
- Rabbani, G.H.; Butler, T.; Knight, J.; Sanyal, S.C.; Alam, K. Randomized controlled trial of berberine sulfate therapy for diarrhea due to enterotoxigenic Escherichia coli and Vibrio cholera. J. Infect. Dis 1978, 155, 979–984. [Google Scholar]
- Zhou, H.; Mineshita, S. The effect of berberine chloride on experimental cholitis in rats in vivo and in vitro. J. Pharmachol. Exp. Ther 2000, 294, 822–829. [Google Scholar]
- Fatehi, M.; Saleh, T.M.; Fatehi-Hassanabad, Z.; Farrokhfal, K.; Jafarzadeh, M.; Davodi, S. A pharmacological study on Berberis vulgaris fruit extract. J. Ethnopharmacol 2005, 102, 46–52. [Google Scholar]
- Wong, K.K. Mechanism of the aortic relaxation induced by low concentrations of berberine. Planta Med 1998, 64, 756–757. [Google Scholar]
- Zeng, X.H.; Zeng, X.J.; Li, Y.Y. Efficacy and safety of berberine for congestive heart failure secondary to ischemic or idiopathic dilated cardiomyophathy. Am. J. Cardiol 2003, 92, 173–176. [Google Scholar]
- Lee, B.; Yang, C.H.; Hahm, D.H.; Choe, E.S.; Lee, H.J.; Pyun, K.H.; Shim, I. Inhibitory Effects of Coptidis rhizoma and Berberine on Cocaine-induced Sensitization. Evid. Complement. Altern. Med 2009, 6, 85–90. [Google Scholar]
- Feng, Y.; Siu, K.Y.; Ye, X.; Wang, N.; Yuen, M.F.; Leung, C.H.; Tong, Y.; Kobayashi, S. Hepatoprotective effects of berberine on carbon tetrachloride-induced acute hepatotoxicity in rats. Chin. Med 2010, 5, 1–6. [Google Scholar]
- Janbaz, K.H.; Gilan, A.H. Studies on preventive and curative effects of berberine on chemical-induced hepatotoxicity in rodents. Fitoterapia 2000, 71, 25–33. [Google Scholar]
- Hwang, J.M.; Wang, C.J.; Chou, F.P. Inhibitory effect of berberine on tert-butyl hydroperoxide-induced oxidative damage in rat liver. Arch. Toxicol 2002, 76, 664–670. [Google Scholar]
- Fallah, H.; Zarrei, M.; Ziai, M.; Mehrazma, M.; Alavian, S.M.; Kianbakht, S.; Mehdizadeh, M. The effects of Taraxacum officinale L. and Berberis vulgaris L. root extracts on carbon tetrachloride induced liver toxicity in rats. J. Med. Plants 2010, 9, 45–52. [Google Scholar]
- Hadaruga, D.I.; Hadaruga, N.G.; Bandur, G.N.; Rivis, A.; Costescu, C.; Ordodi, V.L.; Ardelean, A. Berberis vulgaris extract/β cyclodextrin nanoparticles synthesis and characterization. Rev. Chim. (Bucharest) 2010, 61, 669–675. [Google Scholar]
- Chaung, S.S.; Lin, C.C.; Lin, J.; Yu, K.H.; Hsu, Y.F.; Yen, M.H. The hepatoprotective effects of Limonium sinense against carbon tetrachloride and beta-D-galactosamine intoxication in rats. Phytother. Res 2003, 17, 784–791. [Google Scholar]
- Clawson, G.A. Mechanism of carbon tetrachloride hepatotoxicity. Pathol. Immunopathol. Res 1989, 8, 104–112. [Google Scholar]
- Bhadauria, M. Propolis prevents hepatorenal injury induced by chronic exposure to carbon tetrachloride. Evid. Complement. Altern. Med 2012. [Google Scholar] [CrossRef]
- Shaker, E. Sylimarin, the antioxidant component and Silybum marianum extracts prevent liver damage. Food Chem. Toxicol 2010, 48, 803–806. [Google Scholar]
- Xu, L.; Gao, J.; Wang, Y.; Yu, W.; Zhao, X.; Yang, X.; Zhong, Z.; Qian, Z.M. Myrica rubra extracts protect the liver from CCl4-induced damage. Evid. Complement. Altern. Med 2011. [Google Scholar] [CrossRef]
- Beddowes, E.J.; Faux, S.P.; Chipman, J.K. Chloroform, carbon tetrachloride and glutathione depletion induce secondary genotoxicity in liver cells via oxidative stress. Toxicology 2003, 187, 101–115. [Google Scholar]
- Kohen, R.; Nyska, A. Oxidation of biological systems: Oxidative stress phenomena, antioxidants, redox reactions, and methods for their quantification. Toxicol. Pathol 2010, 30, 620–650. [Google Scholar]
- Motalleb, G.; Hanachi, P.; Kua, S.H.; Fauziah, O.; Asmah, R. Evaluation of phenolic content and total antioxidant activity in Berberis vulgaris fruit extract. J. Biol. Sci 2005, 5, 648–653. [Google Scholar]
- Parichehr, H. Using HPLC to determination the composition and antioxidant activity of Berberis vulgaris. Eur. J. Sci. Res 2009, 29, 47–54. [Google Scholar]
- Zovko, M.K.; Kremer, D.; Karlović, K.; Kosalec, I. Evaluation of antioxidant activities and phenolic content of Berberis vulgaris L. and Berberis croatica Horvat. Food Chem. Toxicol 2010, 48, 2176–2180. [Google Scholar]
- Ozturk, F.; Gul, G.; Ates, B.; Ozturk, I.C.; Cetin, A.; Vardi, N.; Otlu, A.; Yilmaz, I. Protective effect of apricot (Prunus armeniaca L.) on hepatic steatosis and damage induced by carbon tetrachloride in Wistar rats. Br. J. Nutr 2009, 102, 1767–1775. [Google Scholar]
- Junnila, M.; Rahko, T.; Sukura, A. Reduction of carbon tetrachloride-induced hepatotoxic effects by oral administration of betaine in male Han-Wistar rats: A morphometric histological study. Vet. Pathol 2000, 37, 231–238. [Google Scholar]
- Sun, X.; Zhang, X.; Hu, H.; Lu, Y.; Chen, J.; Yasuda, K.; Wang, H. Berberine inhibits hepatic stellate cell proliferation and prevents experimental liver fibrosis. Biol. Pharm. Bull 2009, 32, 1533–1537. [Google Scholar]
- Zhang, Q.; Xiao, X.; Feng, K.; Wang, T.; Li, W.; Yuan, T.; Sun, X.; Sun, Q.; Xiang, H.; Wang, H. Berberine moderates glucose and lipid metabolism through multipathway mechanism. Evid. Complement. Altern. Med 2011. [Google Scholar] [CrossRef]
- Zhou, J.Y.; Zhou, S.W.; Zhang, K.B.; Tang, J.L.; Guang, L.X.; Ying, Y.; Xu, Y.; Zhang, L.; Li, D.D. Chronic effects of berberine on blood, liver glucolipid metabolism and liver PPARs expression in diabetic hyperlipidemic rats. Biol. Pharm. Bull 2008, 31, 1169–1176. [Google Scholar]
- Tasci, I.; Mas, N.; Mas, M.R.; Tuncer, M.; Comert, B. Ultrastructural changes in hepatocytes after taurine treatment in CCl4 induced liver injury. World Gastroenterol 2008, 14, 4897–4902. [Google Scholar]
- Folkmann, J.K.; Risom, L.; Jacobsen, N.R.; Loft, H.W.S.; Moller, P. Oxidatively damaged DNA in rats exposed by oral gavage to C60 fullerenes and single-walled carbon nanotubes. Environ. Health Perspect 2009, 117, 703–708. [Google Scholar]
- Moller, P.; Wallin, H. Adduct formation, mutagenesis and nucleotide excision repair of DNA damage produced by reactive oxygen species and lipid peroxidation product. Mutat. Res 1998, 410, 271–290. [Google Scholar]
- Iseki, S.; Mori, T. Methyl green pyronin stain distinguishes prolifferating from differentiated nonproliferating cell nuclei after acid denaturation of DNA. J. Histochem 1986, 34, 683–687. [Google Scholar]
- Sen, J.Y.; Huang, Q.Y.; Gao, H.Y.; Liu, Y.L.; Cheng, C.F. Modification and application of methyl green-pyronin stain after acid denaturation of DNA. Prog. Anat. Sci 1999, 5, 272–273. [Google Scholar]
- Wang, H.M.; Zheng, N.G.; Wu, J.L.; Gong, C.C.; Wang, Y.L. Dual effects of 8-Br-cAMP on differentiation and apoptosis of human esophageal cancer. World J. Gastroenterol 2005, 7, 6538–6542. [Google Scholar]
- Manna, P.; Bhattacharyya, S.; Das, J.; Ghosh, J.; Parames, C.; Sil, C. Phytomedicinal role of Pithecellobium dulce against CCl4-mediated hepatic oxidative impairments and necrotic cell death. Evid. Complement. Altern. Med 2011. [Google Scholar] [CrossRef]
- Sakr, S.A.; El-Abd, S.F.; Osman, M.; Kandil, A.M.; Helmy, M.S. Ameliorative effect of aqueous leave extract of Ocimum basilicum on CCl4-induced hepatotoxicity and apoptosis in albino rats. J. Am. Sci 2011, 7, 116–127. [Google Scholar]
- Sengupta, M.; Sharma, G.D.; Chakraborty, B. Effect of aqueous extract of Tinospora cordifolia on functions of peritoneal macrophages isolated from CCl4 intoxicated male albino mice. BMC Complement. Altern. Med 2011, 11, 1–9. [Google Scholar]
- Linn, T.T.; Wang, B.M.; Li, X.Y.; Pan, Y.W.; Liu, J.Q.; Shen, J.C.; Luo, G.M. An insight into the protection of rat liver against ischemia/reperfusion injury by 2-selenium-bridged beta-cyclodextrin. Hepatol. Res 2009, 39, 1125–1136. [Google Scholar]
- Liu, Y.; Sakagami, H.; Hasimoto, K.; Kikuchi, H.; Amano, O.; Ishara, M.; Kanda, Y.; Kunii, S.; Kochi, M.; Zhang, W.; Yu, G. Tumor-specific cytotoxicity and type of cell death induced by beta-cyclodextrin benzaldehyde inclusion compound. Anticancer Res 2008, 28, 229–236. [Google Scholar]
- Yadav, V.R.; Prasad, S.; Kannappan, R.; Ravindran, J.; Chatuvedi, M.M.; Vaahtera, L.; Parkkinen, J.; Aggarwal, B.B. Cyclodextrin-complexed cucurmin exhibits anti-inflamatory and antiproliferative activities superior to those of cucurmin through higher cellular uptake. Biochem. Pharmacol 2010, 80, 1021–1032. [Google Scholar]
- Hadaruga, D.I.; Hadaruga, N.G.; Hermenean, A.; Rivis, A.; Paslaru, V.; Codina, G. Biomaterials: Thermal stability of the oleic acid/α and β cyclodextrin complexes. Rev. Chim. (Bucharest) 2008, 59, 994–998. [Google Scholar]
- Aebi, H. Catalase in Methods of Enzymatic Analysis, 2nd ed; Bergmayer, H.U., Ed.; Academic Press: New York, NY, USA, 1974; pp. 673–677. [Google Scholar]
- Beauchamp, C.O.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem 1971, 44, 276–287. [Google Scholar]
- Beutler, E. Red Cell Metabolism: A Manual of Biochemical Methods, 3rd ed; Grune & Stratton Inc: New York, NY, USA, 1974. [Google Scholar]
- Del Rio, D.; Pellegrini, N.; Colombi, B.; Bianchi, M.; Serafini, M.; Torta, F.; Tegoni, F.; Musci, M.; Brighenti, F. Rapid fluorimetric method to detect total plasma malondialdehyde with mild derivatization conditions. Clin. Chem 2003, 49, 690–692. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the folin phenol reagent. J. Biol. Chem 1951, 193, 265–275. [Google Scholar]
- Zhou, B.R.; Gumenscheimer, M.; Freudenberg, M.; Galanos, C. A sriking correaltion between lethal activity and apoptotic DNA fragmentation of liver in response of D-galactosamine-sintetized mice to a non-lethal amount of lipopolysaccharide. Acta Pharmacol. Sin 2003, 24, 193–198. [Google Scholar]






© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hermenean, A.; Popescu, C.; Ardelean, A.; Stan, M.; Hadaruga, N.; Mihali, C.-V.; Costache, M.; Dinischiotu, A. Hepatoprotective Effects of Berberis vulgaris L. Extract/β Cyclodextrin on Carbon Tetrachloride–Induced Acute Toxicity in Mice. Int. J. Mol. Sci. 2012, 13, 9014-9034. https://doi.org/10.3390/ijms13079014
Hermenean A, Popescu C, Ardelean A, Stan M, Hadaruga N, Mihali C-V, Costache M, Dinischiotu A. Hepatoprotective Effects of Berberis vulgaris L. Extract/β Cyclodextrin on Carbon Tetrachloride–Induced Acute Toxicity in Mice. International Journal of Molecular Sciences. 2012; 13(7):9014-9034. https://doi.org/10.3390/ijms13079014
Chicago/Turabian StyleHermenean, Anca, Cristina Popescu, Aurel Ardelean, Miruna Stan, Nicoleta Hadaruga, Ciprian-Valentin Mihali, Marieta Costache, and Anca Dinischiotu. 2012. "Hepatoprotective Effects of Berberis vulgaris L. Extract/β Cyclodextrin on Carbon Tetrachloride–Induced Acute Toxicity in Mice" International Journal of Molecular Sciences 13, no. 7: 9014-9034. https://doi.org/10.3390/ijms13079014




