Proteomic Analysis of the Organ of Corti Using Nanoscale Liquid Chromatography Coupled with Tandem Mass Spectrometry
Abstract
:1. Introduction
2. Results
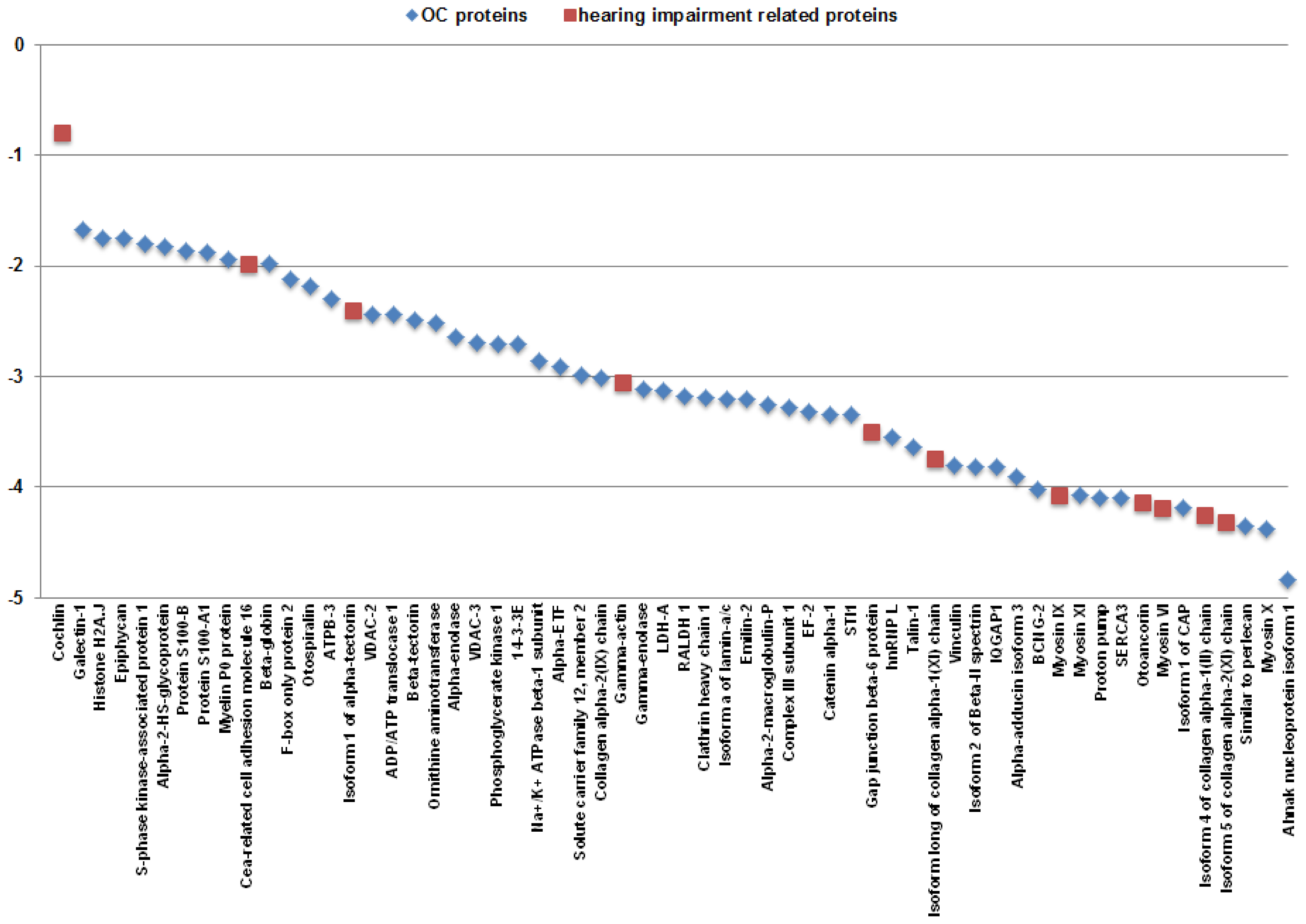
2.1. Identification of Proteins from the OC
2.2. Identification of PTMs from the OC
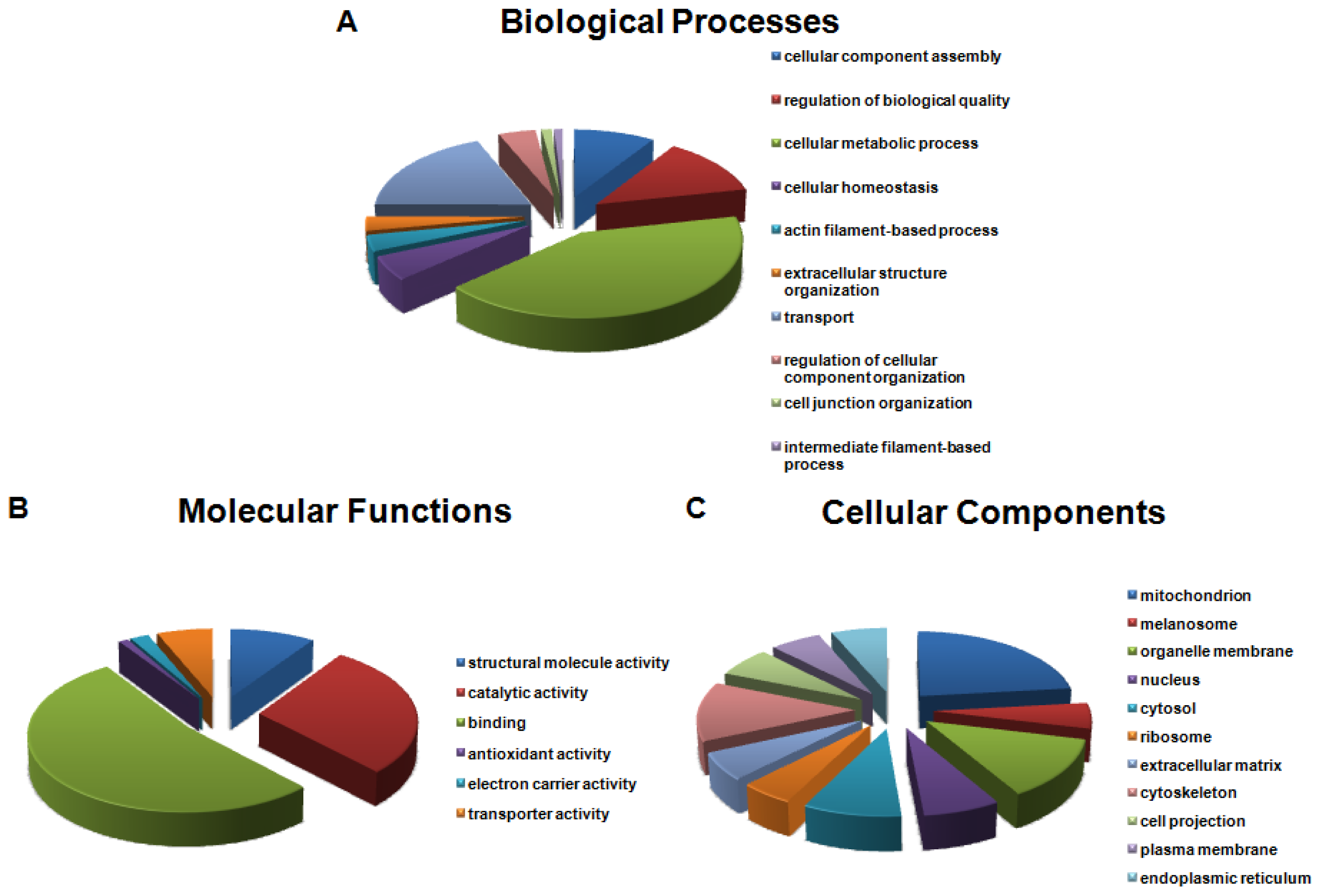
2.3. Gene Ontology (GO) Analysis of Identified OC Proteins
3. Discussion
4. Experimental Section
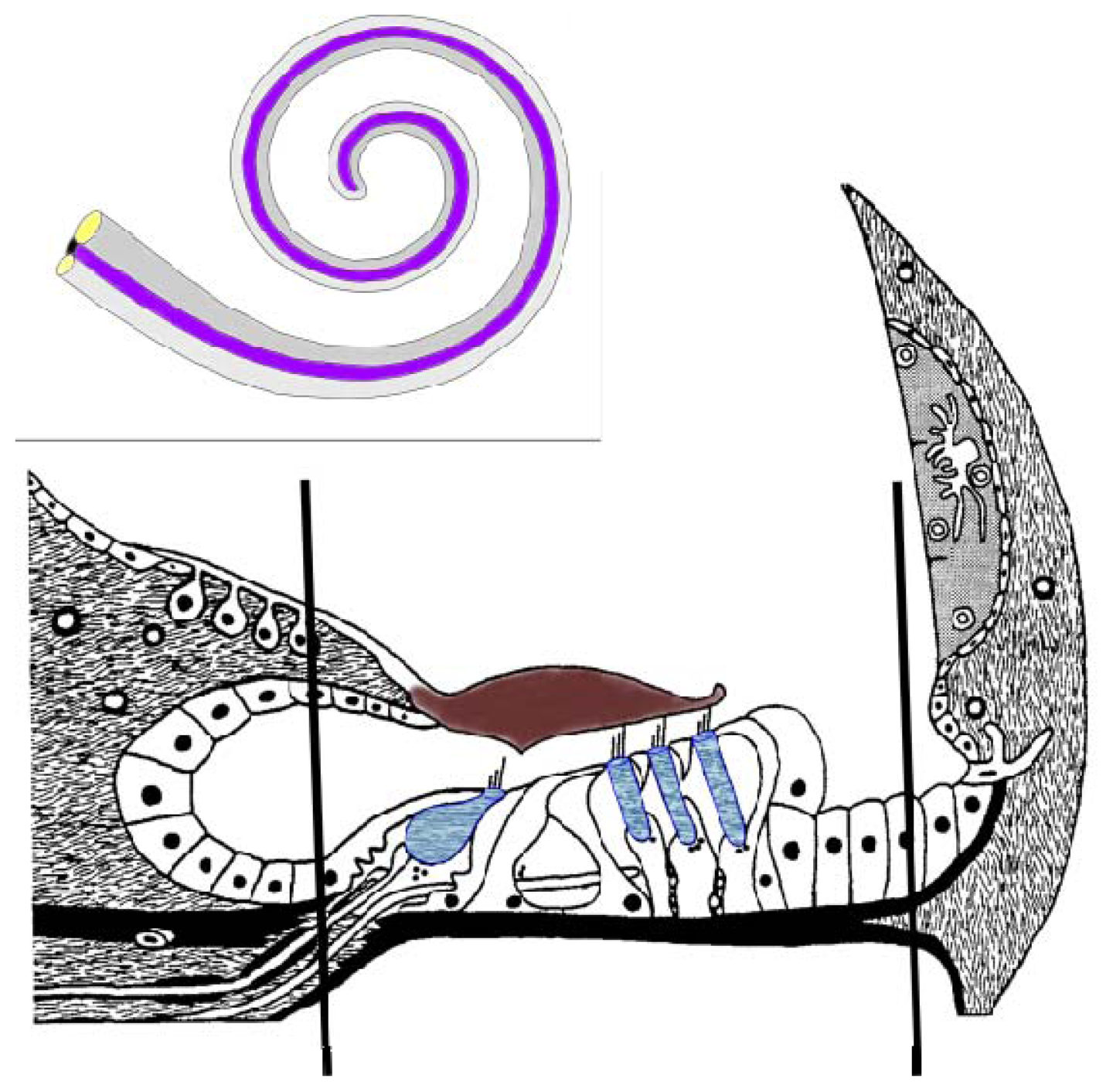
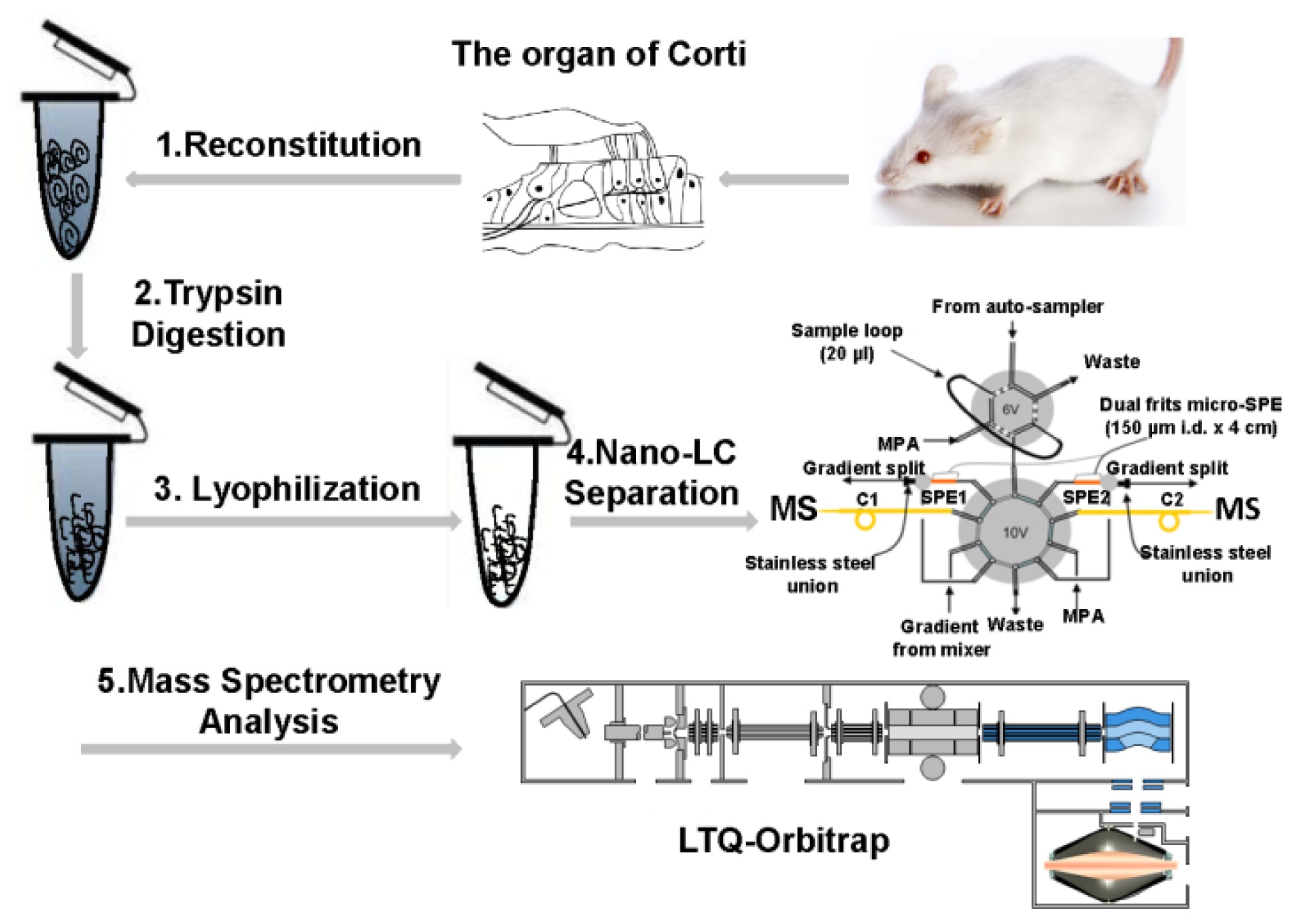
4.1. OC Isolation
4.2. In-Solution Trypsin Digestion
4.3. LC-MS/MS Analysis
4.4. Protein Identification
4.5. Semi-Quantitative and GO Analyses
5. Conclusions
Acknowledgments
References
- Hilgert, N.; Smith, R.J.; van Camp, G. Function and expression pattern of nonsyndromic deafness genes. Curr. Mol. Med 2009, 9, 546–564. [Google Scholar]
- Martini, A.; Calzolari, F.; Sensi, A. Genetic syndromes involving hearing. Int. J. Pediatr. Otorhinolaryngol 2009, 73, S2–S12. [Google Scholar]
- Zybailov, B.; Mosley, A.L.; Sardiu, M.E.; Coleman, M.K.; Florens, L.; Washburn, M.P. Statistical analysis of membrane proteome expression changes in Saccharomyces cerevisiae. J. Proteome Res 2006, 5, 2339–2347. [Google Scholar]
- Leibovici, M.; Safieddine, S.; Petit, C. Mouse models for human hereditary deafness. Curr. Top. Dev. Biol 2008, 84, 385–429. [Google Scholar]
- Chatterjee, S.; Kraus, P.; Lufkin, T. A symphony of inner ear developmental control genes. BMC Genet 2010, 11, 68. [Google Scholar]
- Dror, A.A.; Avraham, K.B. Hearing impairment: A panoply of genes and functions. Neuron 2010, 68, 293–308. [Google Scholar]
- Raviv, D.; Dror, A.A.; Avraham, K.B. Hearing loss: A common disorder caused by many rare alleles. Ann. N. Y. Acad. Sci 2010, 1214, 168–179. [Google Scholar]
- Frolenkov, G.I.; Belyantseva, I.A.; Friedman, T.B.; Griffith, A.J. Genetic insights into the morphogenesis of inner ear hair cells. Nat. Rev. Genet 2004, 5, 489–498. [Google Scholar]
- Deafness and hearing impairment. Available online: http://www.who.int/mediacentre/factsheets/fs300/en/ accessed on 1 February 2012.
- Edge, A.S.; Chen, Z.Y. Hair cell regeneration. Curr. Opin. Neurobiol 2008, 18, 377–382. [Google Scholar]
- Thalmann, I. Inner ear proteomics: A fad or hear to stay. Brain Res 2006, 1091, 103–112. [Google Scholar]
- Gygi, S.P.; Rochon, Y.; Franza, B.R.; Aebersold, R. Correlation between protein and mRNA abundance in yeast. Mol. Cell. Biol 1999, 19, 1720–1730. [Google Scholar]
- Sanborn, K.B.; Mace, E.M.; Rak, G.D.; Difeo, A.; Martignetti, J.A.; Pecci, A.; Bussel, J.B.; Favier, R.; Orange, J.S. Phosphorylation of the myosin IIA tailpiece regulates single myosin IIA molecule association with lytic granules to promote NK-cell cytotoxicity. Blood 2011, 118, 5862–5871. [Google Scholar]
- Friedman, L.M.; Dror, A.A.; Avraham, K.B. Mouse models to study inner ear development and hereditary hearing loss. Int. J. Dev. Biol 2007, 51, 609–631. [Google Scholar]
- Zheng, Q.Y.; Rozanas, C.R.; Thalmann, I.; Chance, M.R.; Alagramam, K.N. Inner ear proteomics of mouse models for deafness, a discovery strategy. Brain Res 2006, 1091, 113–121. [Google Scholar]
- Thalmann, I.; Hughes, I.; Tong, B.D.; Ornitz, D.M.; Thalmann, R. Microscale analysis of proteins in inner ear tissues and fluids with emphasis on endolymphatic sac, otoconia, and organ of Corti. Electrophoresis 2006, 27, 1598–1608. [Google Scholar]
- Gagnon, L.H.; Longo-Guess, C.M.; Berryman, M.; Shin, J.B.; Saylor, K.W.; Yu, H.; Gillespie, P.G.; Johnson, K.R. The chloride intracellular channel protein CLIC5 is expressed at high levels in hair cell stereocilia and is essential for normal inner ear function. J. Neurosci 2006, 26, 10188–10198. [Google Scholar]
- Shin, J.B.; Streijger, F.; Beynon, A.; Peters, T.; Gadzala, L.; McMillen, D.; Bystrom, C.; van der Zee, C.E.; Wallimann, T.; Gillespie, P.G. Hair bundles are specialized for ATP delivery via creatine kinase. Neuron 2007, 53, 371–386. [Google Scholar]
- Coling, D.E.; Ding, D.; Young, R.; Lis, M.; Stofko, E.; Blumenthal, K.M.; Salvi, R.J. Proteomic analysis of cisplatin-induced cochlear damage: Methods and early changes in protein expression. Hear. Res 2007, 226, 140–156. [Google Scholar]
- Jamesdaniel, S.; Ding, D.; Kermany, M.H.; Jiang, H.; Salvi, R.; Coling, D. Analysis of cochlear protein profiles of Wistar, Sprague-Dawley, and Fischer 344 rats with normal hearing function. J. Proteome Res 2009, 8, 3520–3528. [Google Scholar]
- Yang, Y.; Dai, M.; Wilson, T.M.; Omelchenko, I.; Klimek, J.E.; Wilmarth, P.A.; David, L.L.; Nuttall, A.L.; Gillespie, P.G.; Shi, X. Na+/K+-ATPase alpha1 identified as an abundant protein in the blood-labyrinth barrier that plays an essential role in the barrier integrity. PLoS One 2011, 6. [Google Scholar] [CrossRef]
- Petit, C.; Richardson, G.P. Linking genes underlying deafness to hair-bundle development and function. Nat. Neurosci 2009, 12, 703–710. [Google Scholar]
- Ding, S.J.; Qian, W.J.; Smith, R.D. Quantitative proteomic approaches for studying phosphotyrosine signaling. Expert Rev. Proteomics 2007, 4, 13–23. [Google Scholar]
- Hereditary hearing loss homepage. Available online: http://hereditaryhearingloss.org/ accessed on 9 March 2012.
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc 2009, 4, 44–57. [Google Scholar]
- Zheng, Q.Y.; Rozanas, C.R.; Thalmann, I.; Chance, M.R.; Alagramam, K.N. Inner ear proteomics of mouse models for deafness, a discovery strategy. Brain Res 2006, 1091, 113–121. [Google Scholar]
- Kathiresan, T.; Harvey, M.; Orchard, S.; Sakai, Y.; Sokolowski, B. A protein interaction network for the large conductance Ca(2+)-activated K(+) channel in the mouse cochlea. Mol. Cell. Proteomics 2009, 8, 1972–1987. [Google Scholar]
- Gillespie, P.G.; Muller, U. Mechanotransduction by hair cells: Models, molecules, and mechanisms. Cell 2009, 139, 33–44. [Google Scholar]
- Richardson, G.P.; de Monvel, J.B.; Petit, C. How the genetics of deafness illuminates auditory physiology. Annu. Rev. Physiol 2011, 73, 311–334. [Google Scholar]
- Walker, R.G.; Hudspeth, A.J.; Gillespie, P.G. Calmodulin and calmodulin-binding proteins in hair bundles. Proc. Natl. Acad. Sci. USA 1993, 90, 2807–2811. [Google Scholar]
- Manor, U.; Disanza, A.; Grati, M.; Andrade, L.; Lin, H.; di Fiore, P.P.; Scita, G.; Kachar, B. Regulation of stereocilia length by myosin XVa and whirlin depends on the actin-regulatory protein Eps8. Curr. Biol 2011, 21, 167–172. [Google Scholar]
- Uthaiah, R.C.; Hudspeth, A.J. Molecular anatomy of the hair cell’s ribbon synapse. J. Neurosci 2010, 30, 12387–12399. [Google Scholar]
- Zhao, R.; Ding, S.J.; Shen, Y.; Camp, D.G., II; Livesay, E.A.; Udseth, H.; Smith, R.D. Automated metal-free multiple-column nanoLC for improved phosphopeptide analysis sensitivity and throughput. J. Chromatogr. B 2009, 877, 663–670. [Google Scholar]
- Yates, J.R.; Ruse, C.I.; Nakorchevsky, A. Proteomics by mass spectrometry: Approaches, advances, and applications. Annu. Rev. Biomed. Eng 2009, 11, 49–79. [Google Scholar]
- Robertson, N.G.; Cremers, C.W.; Huygen, P.L.; Ikezono, T.; Krastins, B.; Kremer, H.; Kuo, S.F.; Liberman, M.C.; Merchant, S.N.; Miller, C.E.; et al. Cochlin immunostaining of inner ear pathologic deposits and proteomic analysis in DFNA9 deafness and vestibular dysfunction. Hum. Mol. Genet 2006, 15, 1071–1085. [Google Scholar]
- Robertson, N.G.; Khetarpal, U.; Gutierrez-Espeleta, G.A.; Bieber, F.R.; Morton, C.C. Isolation of novel and known genes from a human fetal cochlear cDNA library using subtractive hybridization and differential screening. Genomics 1994, 23, 42–50. [Google Scholar]
- Robertson, N.G.; Resendes, B.L.; Lin, J.S.; Lee, C.; Aster, J.C.; Adams, J.C.; Morton, C.C. Inner ear localization of mRNA and protein products of COCH, mutated in the sensorineural deafness and vestibular disorder, DFNA9. Hum. Mol. Genet 2001, 10, 2493–2500. [Google Scholar]
- Ikezono, T.; Omori, A.; Ichinose, S.; Pawankar, R.; Watanabe, A.; Yagi, T. Identification of the protein product of the Coch gene (hereditary deafness gene) as the major component of bovine inner ear protein. Biochim. Biophys. Acta 2001, 1535, 258–265. [Google Scholar]
- Goodyear, R.J.; Richardson, G.P. Extracellular matrices associated with the apical surfaces of sensory epithelia in the inner ear: Molecular and structural diversity. J. Neurobiol 2002, 53, 212–227. [Google Scholar]
- Richardson, G.P.; Russell, I.J.; Duance, V.C.; Bailey, A.J. Polypeptide composition of the mammalian tectorial membrane. Hear. Res 1987, 25, 45–60. [Google Scholar]
- Thalmann, I.; Thallinger, G.; Crouch, E.C.; Comegys, T.H.; Barrett, N.; Thalmann, R. Composition and supramolecular organization of the tectorial membrane. Laryngoscope 1987, 97, 357–367. [Google Scholar]
- Legan, P.K.; Rau, A.; Keen, J.N.; Richardson, G.P. The mouse tectorins. Modular matrix proteins of the inner ear homologous to components of the sperm-egg adhesion system. J. Biol. Chem 1997, 272, 8791–8801. [Google Scholar]
- Rau, A.; Legan, P.K.; Richardson, G.P. Tectorin mRNA expression is spatially and temporally restricted during mouse inner ear development. J. Comp. Neurol 1999, 405, 271–280. [Google Scholar]
- Zwaenepoel, I.; Mustapha, M.; Leibovici, M.; Verpy, E.; Goodyear, R.; Liu, X.Z.; Nouaille, S.; Nance, W.E.; Kanaan, M.; Avraham, K.B.; et al. Otoancorin, an inner ear protein restricted to the interface between the apical surface of sensory epithelia and their overlying acellular gels, is defective in autosomal recessive deafness DFNB22. Proc. Natl. Acad. Sci. USA 2002, 99, 6240–6245. [Google Scholar]
- Avraham, K.B.; Hasson, T.; Sobe, T.; Balsara, B.; Testa, J.R.; Skvorak, A.B.; Morton, C.C.; Copeland, N.G.; Jenkins, N.A. Characterization of unconventional MYO6, the human homologue of the gene responsible for deafness in Snell’s waltzer mice. Hum. Mol. Genet 1997, 6, 1225–1231. [Google Scholar]
- Self, T.; Sobe, T.; Copeland, N.G.; Jenkins, N.A.; Avraham, K.B.; Steel, K.P. Role of myosin VI in the differentiation of cochlear hair cells. Dev. Biol 1999, 214, 331–341. [Google Scholar]
- Roux, I.; Hosie, S.; Johnson, S.L.; Bahloul, A.; Cayet, N.; Nouaille, S.; Kros, C.J.; Petit, C.; Safieddine, S. Myosin VI is required for the proper maturation and function of inner hair cell ribbon synapses. Hum. Mol. Genet 2009, 18, 4615–4628. [Google Scholar]
- Lalwani, A.K.; Atkin, G.; Li, Y.; Lee, J.Y.; Hillman, D.E.; Mhatre, A.N. Localization in stereocilia, plasma membrane, and mitochondria suggests diverse roles for NMHC-IIa within cochlear hair cells. Brain Res 2008, 1197, 13–22. [Google Scholar]
- Mhatre, A.N.; Li, J.; Kim, Y.; Coling, D.E.; Lalwani, A.K. Cloning and developmental expression of nonmuscle myosin IIA (Myh9) in the mammalian inner ear. J. Neurosci. Res 2004, 76, 296–305. [Google Scholar]
- Mhatre, A.N.; Li, Y.; Atkin, G.; Maghnouj, A.; Lalwani, A.K. Expression of Myh9 in the mammalian cochlea: Localization within the stereocilia. J. Neurosci. Res 2006, 84, 809–818. [Google Scholar]
- Slepecky, N.; Chamberlain, S.C. Distribution and polarity of actin in the sensory hair cells of the chinchilla cochlea. Cell Tissue Res 1982, 224, 15–24. [Google Scholar]
- Flock, A.; Cheung, H.C.; Flock, B.; Utter, G. Three sets of actin filaments in sensory cells of the inner ear. Identification and functional orientation determined by gel electrophoresis, immunofluorescence and electron microscopy. J. Neurocytol 1981, 10, 133–147. [Google Scholar]
- Belyantseva, I.A.; Perrin, B.J.; Sonnemann, K.J.; Zhu, M.; Stepanyan, R.; McGee, J.; Frolenkov, G.I.; Walsh, E.J.; Friderici, K.H.; Friedman, T.B.; et al. Gamma-actin is required for cytoskeletal maintenance but not development. Proc. Natl. Acad. Sci. USA 2009, 106, 9703–9708. [Google Scholar]
- Delpire, E.; Lu, J.; England, R.; Dull, C.; Thorne, T. Deafness and imbalance associated with inactivation of the secretory Na-K-2Cl co-transporter. Nat. Genet 1999, 22, 192–195. [Google Scholar]
- Henzl, M.T.; O’Neal, J.; Killick, R.; Thalmann, I.; Thalmann, R. OCP1, an F-box protein, co-localizes with OCP2/SKP1 in the cochlear epithelial gap junction region. Hear. Res 2001, 157, 100–111. [Google Scholar]
- Henzl, M.T.; Thalmann, I.; Larson, J.D.; Ignatova, E.G.; Thalmann, R. The cochlear F-box protein OCP1 associates with OCP2 and connexin 26. Hear. Res 2004, 191, 101–109. [Google Scholar]
- Thalmann, R.; Henzl, M.T.; Thalmann, I. Specific proteins of the organ of Corti. Acta Otolaryngol 1997, 117, 265–268. [Google Scholar]
- Morris, J.K.; Maklad, A.; Hansen, L.A.; Feng, F.; Sorensen, C.; Lee, K.F.; Macklin, W.B.; Fritzsch, B. A disorganized innervation of the inner ear persists in the absence of ErbB2. Brain Res 2006, 1091, 186–199. [Google Scholar]
- Lee, M.J.; Calle, E.; Brennan, A.; Ahmed, S.; Sviderskaya, E.; Jessen, K.R.; Mirsky, R. In early development of the rat mRNA for the major myelin protein P(0) is expressed in nonsensory areas of the embryonic inner ear, notochord, enteric nervous system, and olfactory ensheathing cells. Dev. Dyn 2001, 222, 40–51. [Google Scholar]
- Aarnisalo, A.A.; Green, K.M.; O’Malley, J.; Makary, C.; Adams, J.; Merchant, S.N.; Evans, J.E. A method for MS(E) differential proteomic analysis of archival formalin-fixed celloidin-embedded human inner ear tissue. Hear. Res 2010, 270, 15–20. [Google Scholar]
- Basappa, J.; Turcan, S.; Vetter, D.E. Corticotropin-releasing factor-2 activation prevents gentamicin-induced oxidative stress in cells derived from the inner ear. J. Neurosci. Res 2010, 88, 2976–2990. [Google Scholar]
- Elkan-Miller, T.; Ulitsky, I.; Hertzano, R.; Rudnicki, A.; Dror, A.A.; Lenz, D.R.; Elkon, R.; Irmler, M.; Beckers, J.; Shamir, R.; et al. Integration of transcriptomics, proteomics, and microRNA analyses reveals novel microRNA regulation of targets in the mammalian inner ear. PLoS One 2011, 6, e18195. [Google Scholar]
- Morris, K.A.; Snir, E.; Pompeia, C.; Koroleva, I.V.; Kachar, B.; Hayashizaki, Y.; Carninci, P.; Soares, M.B.; Beisel, K.W. Differential expression of genes within the cochlea as defined by a custom mouse inner ear microarray. J. Assoc. Res. Otolaryngol 2005, 6, 75–89. [Google Scholar]
- Zheng, J.; Miller, K.K.; Yang, T.; Hildebrand, M.S.; Shearer, A.E.; DeLuca, A.P.; Scheetz, T.E.; Drummond, J.; Scherer, S.E.; Legan, P.K.; et al. Carcinoembryonic antigen-related cell adhesion molecule 16 interacts with alpha-tectorin and is mutated in autosomal dominant hearing loss (DFNA4). Proc. Natl. Acad. Sci. USA 2011, 108, 4218–4223. [Google Scholar]
- Voges, D.; Zwickl, P.; Baumeister, W. The 26S proteasome: A molecular machine designed for controlled proteolysis. Annu. Rev. Biochem 1999, 68, 1015–1068. [Google Scholar]
- Nelson, R.F.; Glenn, K.A.; Zhang, Y.; Wen, H.; Knutson, T.; Gouvion, C.M.; Robinson, B.K.; Zhou, Z.; Yang, B.; Smith, R.J.; et al. Selective cochlear degeneration in mice lacking the F-box protein, Fbx2, a glycoprotein-specific ubiquitin ligase subunit. J. Neurosci 2007, 27, 5163–5171. [Google Scholar]
- Nakano, Y.; Kim, S.H.; Kim, H.M.; Sanneman, J.D.; Zhang, Y.; Smith, R.J.; Marcus, D.C.; Wangemann, P.; Nessler, R.A.; Banfi, B. A claudin-9-based ion permeability barrier is essential for hearing. PLoS Genet 2009, 5. [Google Scholar] [CrossRef]
- Kikuchi, T.; Kimura, R.S.; Paul, D.L.; Adams, J.C. Gap junctions in the rat cochlea: Immunohistochemical and ultrastructural analysis. Anat. Embryol. (Berl.) 1995, 191, 101–118. [Google Scholar]
- Shen, Y.; Moore, R.J.; Zhao, R.; Blonder, J.; Auberry, D.L.; Masselon, C.; Pasa-Tolic, L.; Hixson, K.K.; Auberry, K.J.; Smith, R.D. High-efficiency on-line solid-phase extraction coupling to 15–150-microm-i.d. column liquid chromatography for proteomic analysis. Anal. Chem 2003, 75, 3596–3605. [Google Scholar]
- Geer, L.Y.; Markey, S.P.; Kowalak, J.A.; Wagner, L.; Xu, M.; Maynard, D.M.; Yang, X.; Shi, W.; Bryant, S.H. Open mass spectrometry search algorithm. J. Proteome Res 2004, 3, 958–964. [Google Scholar]
- Kersey, P.J.; Duarte, J.; Williams, A.; Karavidopoulou, Y.; Birney, E.; Apweiler, R. The International Protein Index: An integrated database for proteomics experiments. Proteomics 2004, 4, 1985–1988. [Google Scholar]
- Elias, J.E.; Gygi, S.P. Target-decoy search strategy for increased confidence in large-scale protein identifications by mass spectrometry. Nat. Methods 2007, 4, 207–214. [Google Scholar]
- Huang, X.; Tolmachev, A.V.; Shen, Y.; Liu, M.; Huang, L.; Zhang, Z.; Anderson, G.A.; Smith, R.D.; Chan, W.C.; Hinrichs, S.H.; et al. UNiquant, a program for quantitative proteomics analysis using stable isotope labeling. J. Proteome Res 2011, 10, 1228–1237. [Google Scholar]
- Paoletti, A.C.; Parmely, T.J.; Tomomori-Sato, C.; Sato, S.; Zhu, D.; Conaway, R.C.; Conaway, J.W.; Florens, L.; Washburn, M.P. Quantitative proteomic analysis of distinct mammalian Mediator complexes using normalized spectral abundance factors. Proc. Natl. Acad. Sci. USA 2006, 103, 18928–18933. [Google Scholar]
- Ashburner, M.; Ball, C.A.; Blake, J.A.; Botstein, D.; Butler, H.; Cherry, J.M.; Davis, A.P.; Dolinski, K.; Dwight, S.S.; Eppig, J.T.; et al. Gene ontology: Tool for the unification of biology. The Gene Ontology Consortium. Nat. Genet 2000, 25, 25–29. [Google Scholar]
| Protein Name | IPI IDa | Gene Name | Locusb |
|---|---|---|---|
| γ-actin | IPI00648420 | Actg1 | DFNA20 |
| Cochlin | IPI00127100 | Coch | DFNA9 |
| Collagen α-1(XI) chain | IPI00230065 IPI00400048 | Col11a1 | STL2 |
| Collagen α-2(XI) chain | IPI00283629 | Col11a2 | DFNA13 DFNB53 STL3 |
| Collagen α-1(II) chain | IPI00828467 | Col2a1 | STL1 |
| Gap junction β-6 protein | IPI00111902 | Gjb6 | DFNA3B DFNB1 |
| Myosin IX | IPI00123181 | Myh9 | DFNA17 |
| Myosin VI | IPI00462752 IPI00776187 | Myo6 | DFNA22 DFNB37 |
| Otoancorin | IPI00265452 | Otoa | DFNB22 |
| α-tectorin | IPI00114338 IPI00415299 | Tecta | DFNA12 |
| Carcinoembryonic antigen-related cell adhesion molecule 16 | IPI00356708 | Ceacam16 | DFNA4 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Peng, H.; Liu, M.; Pecka, J.; Beisel, K.W.; Ding, S.-J. Proteomic Analysis of the Organ of Corti Using Nanoscale Liquid Chromatography Coupled with Tandem Mass Spectrometry. Int. J. Mol. Sci. 2012, 13, 8171-8188. https://doi.org/10.3390/ijms13078171
Peng H, Liu M, Pecka J, Beisel KW, Ding S-J. Proteomic Analysis of the Organ of Corti Using Nanoscale Liquid Chromatography Coupled with Tandem Mass Spectrometry. International Journal of Molecular Sciences. 2012; 13(7):8171-8188. https://doi.org/10.3390/ijms13078171
Chicago/Turabian StylePeng, Hong, Miao Liu, Jason Pecka, Kirk W. Beisel, and Shi-Jian Ding. 2012. "Proteomic Analysis of the Organ of Corti Using Nanoscale Liquid Chromatography Coupled with Tandem Mass Spectrometry" International Journal of Molecular Sciences 13, no. 7: 8171-8188. https://doi.org/10.3390/ijms13078171




