Genetic Diversity of the Endemic and Medicinally Important Plant Rheum officinale as Revealed by Inter-Simpe Sequence Repeat (ISSR) Markers
Abstract
:1. Introduction
2. Results
2.1. Genetic Diversity
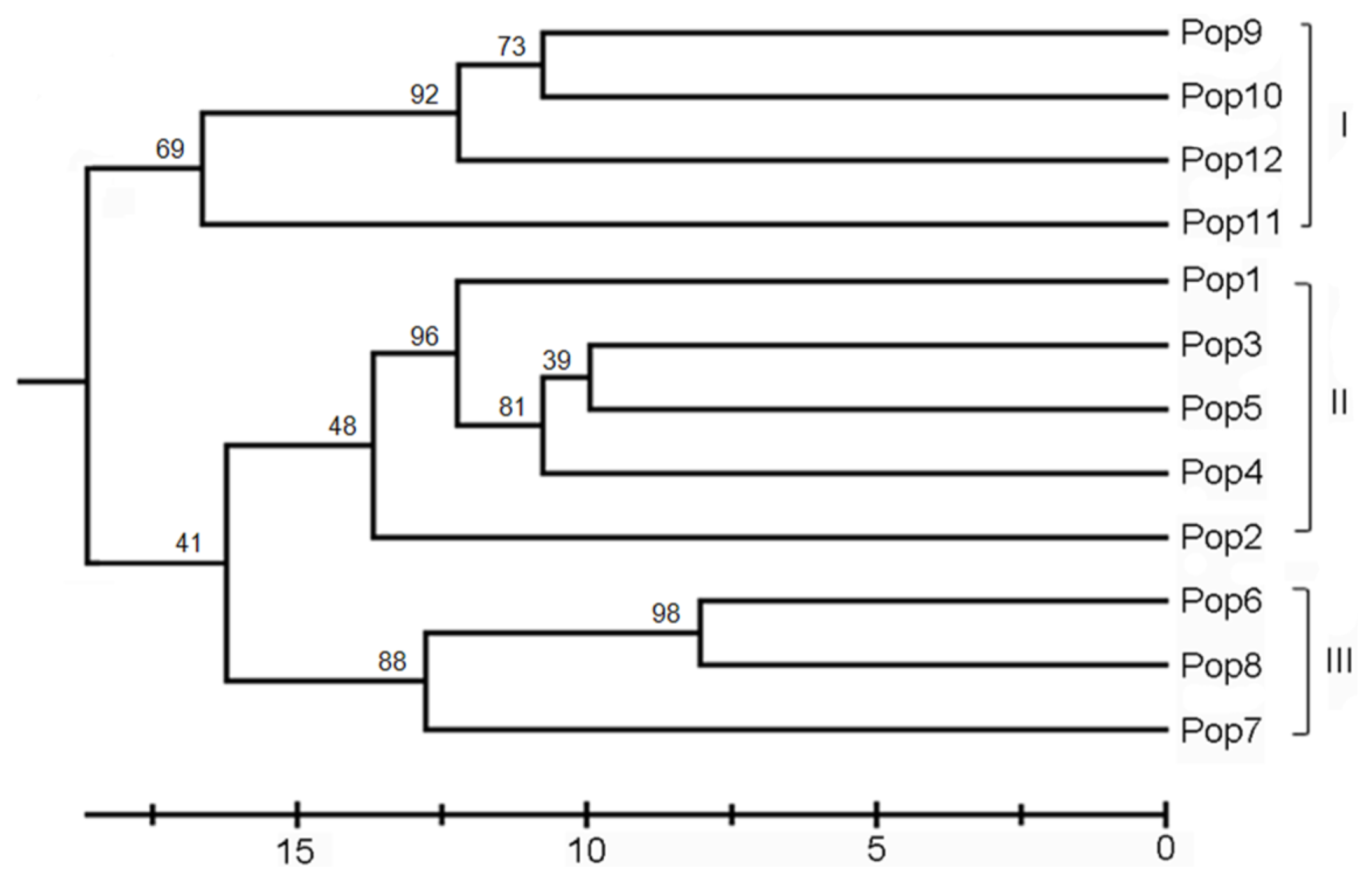
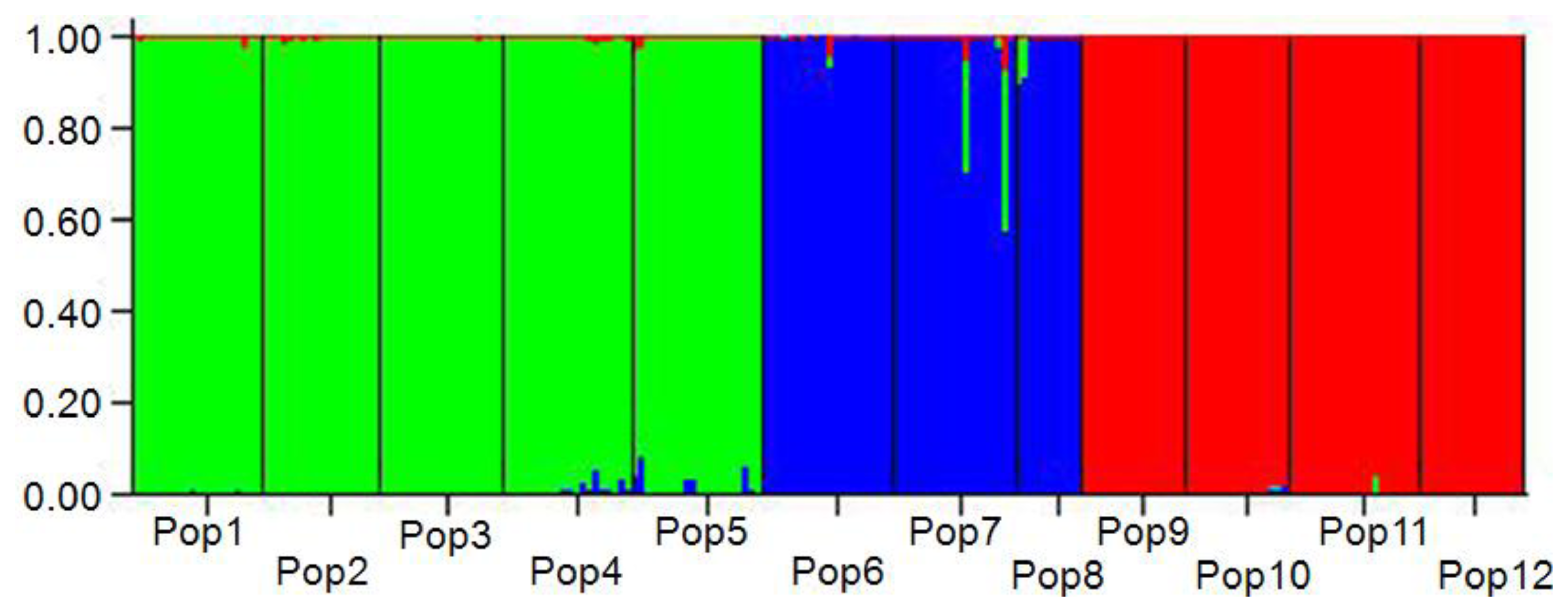
2.2. Genetic Differentiation and Relationships
3. Discussion
3.1. Genetic Diversity
3.2. Genetic Differentiation
3.3. Implications for Conservation and Cultivation
4. Experimental Section
4.1. Plant Sampling
4.2. DNA Extraction and PCR Amplification
4.3. Data Analysis
5. Conclusions
Acknowledgments
References
- Cote, C.T. Genetic variation in rare and common plants. Annu. Rev. Ecol. Syst 2003, 34, 213–237. [Google Scholar]
- Qian, W.; Ge, S.; Hong, D.Y. Genetic variation within and among populations of a wild rice Oryza granulata from China detected by RAPD and ISSR markers. Theor. Appl. Genet 2001, 102, 440–449. [Google Scholar]
- Kim, S.C.; Lee, C.; Santos-Guerra, A. Genetic analysis and conservation of the endangered Canary Island woody sow-thistle, Sonchus gandogeri (Asteraceae). J. Plant Res 2005, 118, 147–153. [Google Scholar]
- Ayala, F.J.; Kiger, J.A. Modern Genetics, 2nd ed; Benjamin/Cumings: Menlo Park, CA, USA, 1984. [Google Scholar]
- Xiao, P.; He, L.; Wang, L. Ethnopharmacologic study of Chinese rhubarb. J. Ethnopharmacol 1984, 10, 275–293. [Google Scholar]
- Tseng, S.H.; Lee, H.H.; Chen, L.G.; Wu, C.H.; Wang, C.C. Effects of three purgative decoctions on inflammatory mediators. J. Ethnopharmacol 2006, 105, 118–124. [Google Scholar]
- Liu, L.; Guo, Z.J.; Lv, Z.G.; Sun, Y.; Cao, W.; Zhang, R.; Liu, Z.G.; Li, C.; Cao, S.S.; Mei, Q.B. The beneficial effect of Rheum tanguticum polysaccharide on protecting against diarrhea, colonic inflammation and ulceration in rats with TNBS-induced colitis: The role of macrophage mannose receptor in inflammation and immune response. Int. J. Immunopharmacol 2008, 8, 1481–1492. [Google Scholar]
- Li, W.Y.; Chan, S.W.; Guo, D.J.; Chung, M.K.; Leung, T.Y.; Yu, P.H. Water extract of Rheum officinale Baill. induces apoptosis in human lung adenocarcinoma A549 and human breast cancer MCF-7 cell lines. J. Ethnopharmacol 2009, 124, 251–256. [Google Scholar]
- Chinese Pharmacopoeia Committee, Pharmacopoeia of the People’s Republic of China; China Medical Science and Technology Press: Beijing, China, 2010; pp. 22–23.
- Wang, A.L.; Yang, M.H.; Liu, J.Q. Molecular phylogeny, recent radiation and evolution of gross morphology of the rhubarb genus Rheum (Polygonaceae) inferred from chloroplast DNA trnL-F sequences. Ann. Bot 2005, 96, 489–498. [Google Scholar]
- Bao, B.J.; Grabovskaya-Borodina, A.E. Rheum. In Flora of China; Li, A.R., Bao, B.J., Eds.; Science Press: Beijing, China; Missouri Botanical Garden: St. Louis, USA, 2003; Volume 5, pp. 341–350. [Google Scholar]
- Wang, X.M.; Hou, X.Q.; Zhang, Y.Q.; Li, Y. Distribution pattern of genuine species of rhubarb as traditional Chinese medicine. J. Med. Plants Res 2010, 4, 1865–1876. [Google Scholar]
- Chen, F.J.; Wang, A.L.; Chen, K.M.; Wan, D.S.; Liu, J.Q. Genetic diversity and population structure of the endangered and medically important Rheum tanguticum (Polygonaceae) revealed by SSR markers. Biochem. Syst. Ecol 2009, 37, 613–621. [Google Scholar]
- Hu, Y.P.; Wang, L.; Xie, X.L.; Yang, J.; Li, Y.; Zhang, H.G. Genetic diversity of wild populations of Rheum tanguticum endemic to China as revealed by ISSR analysis. Bioch. Syst. Ecol 2010, 38, 264–274. [Google Scholar]
- Yang, X.J.; Ma, X.X.; Yang, L.J.; Yu, D.Z.; Qian, Y.X.; Ni, H.W. Efficacy of Rheum officinale liquid formulation on cucumber powdery mildew. Crop Prot 2009, 28, 1031–1035. [Google Scholar]
- Shen, S.L.; Li, Z.G.; Zhang, J.Y. Resources distribution and usage of medicinal plants of Rheum in Gansu. J. Gansu Sci 1996, 8, 20–26. [Google Scholar]
- Yang, F.Q.; Zhang, T.Y.; Tian, G.L.; Cao, H.F.; Liu, Q.H.; Ito, Y. Preparative isolation and purification of hydroxyanthraquinones from Rheum officinale Baill. by high-speed counter-current chromatography using pH-modulated stepwise elution. J. Chromatogr. A 1999, 858, 103–107. [Google Scholar]
- Xu, Z.X.; Sun, Y.; Zhang, H.Q. Study on the anti-aging effect of rhapontinum. J. Anhui Med. Pharmaceu 2002, 6, 9–10. [Google Scholar]
- Tsai, J.C.; Tsai, S.; Chang, W.C. Effect of ethanol extracts of three Chinese medicinal plants with laxative properties on ion transport of the rat intestinal epithelia. Biol. Pharm. Bull 2004, 27, 162–165. [Google Scholar]
- Moon, M.K.; Kang, D.G.; Lee, J.K.; Kim, J.S.; Lee, H.S. Vasodilatory and anti-inflammatory effects of the aqueous extract of rhubarb via a NO-cGMP pathway. Life Sci 2006, 78, 1550–1557. [Google Scholar]
- Zietkiewicz, E.; Rafalski, A.; Labuda, D. Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 1994, 20, 176–183. [Google Scholar]
- Godwin, I.D.; Aitken, E.A.; Smith, L.W. Application of inter simple sequence repeat (ISSR) markers to plant genetics. Electrophoresis 1997, 18, 1524–1528. [Google Scholar]
- Esselman, E.J.; Li, J.Q.; Crawford, D.J.; Winduss, J.L.; Wolfe, A.D. Clonal diversity in the rare Calamagrostis porteri ssp. insperata (Poaceae): Comparative results for allozymes and randomamplified polymorphic DNA (RAPD) and inter simple sequence repeat (ISSR) markers. Mol. Ecol 1999, 8, 443–451. [Google Scholar]
- Reddy, M.P.; Sarla, N.; Siddiq, E.A. Inter simple sequence repeat (ISSR) polymorphism and its application in plant breeding. Euphytica 2002, 128, 9–17. [Google Scholar]
- Song, Z.Q.; Li, X.F.; Wang, H.G.; Wang, J.H. Genetic diversity and population structure of Salvia miltiorrhiza Bge. in China revealed by ISSR and SRAP. Genetica 2010, 138, 241–249. [Google Scholar]
- Camacho, F.J.; Liston, A. Population structure and genetic diversity of Botrychium pumicola (Ophioglossaceae) based on inter-simple sequence repeats (ISSR). Am. J. Bot 2001, 88, 1065–1070. [Google Scholar]
- Lei, Y.D.; Gao, H.; Tsering, T.; Shi, S.H.; Zhong, Y. Determination of genetic variation in Rhodiola crenulata from the Hengduan Mountains Region, China using inter-simple sequence repeats. Genet. Mol. Biol 2006, 29, 339–344. [Google Scholar]
- Xiao, M.; Li, Q.; Wang, L.; Guo, L.; Li, J.; Tang, L.; Chen, F. ISSR Analysis of the genetic diversity of the endangered species Sinopodophyllum hexandrum (Royle) Ying from western Sichuan Province, China. J. Integr. Plant Biol 2006, 48, 1140–1146. [Google Scholar]
- Qiu, Y.Q.; Zong, M.; Yao, H.; Chen, B.L.; Zhou, X.L.; Chen, Z.L.; Fu, C.X. Genetic variation in wild and cultivated Rhizoma Corydalis revealed by ISSRs markers. Planta Med 2008, 74, 1–5. [Google Scholar]
- Nei, M. Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 1978, 89, 583–590. [Google Scholar]
- Tajima, F. Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 1989, 123, 585–595. [Google Scholar]
- Fu, Y.X. Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 1997, 147, 915–925. [Google Scholar]
- Hamrick, J.L.; Godt, M.J.W. Allozyme Diversity in Plant Species. In Plant Population Genetics, Breeding, and Genetic Resources; Brown, A.H.D., Clegg, M.T., Kahler, A.L., Weir, B.S., Eds.; Sinauer Associates: Sunderland, MA, USA, 1990; pp. 43–63. [Google Scholar]
- Xiao, L.Q.; Ge, X.J.; Gong, X.; Hao, G.; Zheng, S.X. ISSR variation in the endemic and endangered plant Cycas guizhouensis (Cycadaceae). Ann. Bot 2004, 94, 133–138. [Google Scholar]
- Li, F.G.; Xia, N.H. Population structure and genetic diversity of an endangered species, Glyptostrobus pensilis (Cupressaceae). Bot. Bull. Acad. Sin 2005, 46, 155–162. [Google Scholar]
- Ge, Y.Q.; Qiu, Y.X.; Ding, B.Y.; Fu, C.X. An ISSR analysis on population genetic diversity of the relict plant Ginkgo biloba. Biodiv. Sci 2003, 11, 276–287. [Google Scholar]
- Luan, S.; Chiang, T.Y.; Gong, X. High genetic diversity vs. low genetic differentiation in Nouelia insignis (Asteraceae), a narrowly distributed and endemic species in China, revealed by ISSR fingerprinting. Ann. Bot 2006, 98, 583–589. [Google Scholar]
- Li, J.M.; Jin, Z.X. Genetic variation and differentiation in Torreya jackii Chun, an endangered plant endemic to China. Plant Sci 2007, 172, 1048–1053. [Google Scholar]
- Knuth, P. Handbook of Flower Pollination Based upon Hermann Müller’s Work ‘The Fertilisation of Flowers by Insects’, 2nd ed; Clarendon Press: Oxford, UK, 1906; Volume 3, p. 340. [Google Scholar]
- Xiao, S.P.; Chen, M.; Huang, L.Q.; Gao, F. Primary study on shapes of fruits and germination characters of seeds of Radix et Rhizoma Rhei. China J. Chin. Mat. Med 2007, 32, 195–199. [Google Scholar]
- Nybom, H. Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Mol. Ecol 2004, 13, 1143–1155. [Google Scholar]
- Zhao, Y.; Chen, X.Y.; Wang, X.R.; Pian, R.Q. ISSR analysis of genetic diversity among Lespedeza bicolor populations. J. Plant Genet. Resour 2007, 8, 195–199. [Google Scholar]
- Chen, L.Z. Present Situation of Biodiversity and Its Protection in China; Science Press: Beijing, China, 1993; pp. 12–43. [Google Scholar]
- Fu, C.J.; Liu, Y.H.; Xiong, W.J. Regeneration characteristics of gaps and their effect on Abies fargesii forests in the Shennongjia National Nature Reserve, China. For. Stud. China 2010, 12, 206–212. [Google Scholar]
- Ge, X.J.; Zhang, L.B.; Yuan, Y.M.; Hao, G.; Chang, T.Y. Strong genetic differentiation of the East-Himalayan Megacodon stylophorus (Gentianaceae) detected by inter-simple sequence repeats (ISSR). Biodiv. Conserv 2005, 14, 849–861. [Google Scholar]
- Xia, T.; Chen, S.L.; Chen, S.Y.; Ge, X.J. Genetic variation within and among populations of Rhodiola alsia (Crassulaceae) native to the Tibetan Plateau as detected by ISSR markers. Biochem. Genet 2005, 43, 87–101. [Google Scholar]
- Pfeifer, M.; Jetschke, G. Influence of geographical isolation on genetic diversity of Himantoglossum hircinum (Orchidaceae). Folia Geobot 2006, 41, 3–20. [Google Scholar]
- Slatkin, M. Gene flow in natural populations. Annu. Rev. Ecol. Syst 1985, 16, 393–430. [Google Scholar]
- Ellstrand, N.C.; Elam, D.R. Population genetic consequences of small population size: Implications for plant conservation. Annu. Rev. Ecol. Syst 1993, 24, 217–242. [Google Scholar]
- Templeton, A.R.; Shaw, K.; Routman, E.; Davis, S.K. The genetic consequences of habitat fragmentation. Ann. Mo. Bot. Gard 1990, 77, 13–27. [Google Scholar]
- Wright, S. Isolation by distance. Genetics 1943, 28, 114–138. [Google Scholar]
- Milligan, B.G.; Leebens-Mack, J.; Strand, A.E. Conservation genetics: Beyond the maintenance of marker diversity. Mol. Ecol 1994, 12, 844–855. [Google Scholar]
- Avise, J.C.; Hamrick, J.L. Conservation Genetics: Case Histories from Nature; Chapman and Hall: New York, NY, USA, 1996. [Google Scholar]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh leaf material. Phytochem. Bull 1987, 19, 11–15. [Google Scholar]
- Wang, X.M. Optimization of DNA isolation, ISSR-PCR system and primers screening of genuine species of rhubarb, an important herbal medicine in China. J. Med. Plants Res 2010, 4, 904–908. [Google Scholar]
- Nei, M. Analysis of gene diversity in subdivided populations. Proc. Natl. Acad. Sci. USA 1973, 70, 3321–3323. [Google Scholar]
- Lewontin, R.C. Apportionment of human diversity. Evol. Biol 1972, 6, 381–398. [Google Scholar]
- Yeh, F.; Yang, R.; Boyle, T. POPGENE Version 1.32; Microsoft Window-based Freeware for Population Genetic Analysis; Molecular Biology and Biotechnology Center, University of Alberta: Edmonton, AB, Canada, 1999. [Google Scholar]
- McDermott, J.; McDonald, B. Gene flow in plant pathosystems. Annu. Rev. Phytopathol 1993, 31, 353–373. [Google Scholar]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA 4: Molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol. Biol. Evol 2007, 24, 1596–1599. [Google Scholar]
- Excoffier, L.; Smouse, P.E.; Quattro, J.M. Analysis of molecular variance inferred from metric distances among DNA haplotypes: Applications to human mitochondrial DNA restriction data. Genetics 1992, 131, 479–491. [Google Scholar]
- Zhang, F.M.; Ge, S. Data analysis in population genetics. I. Analysis of RAPD data with AMOVA. Biodivers. Sci 2002, 10, 438–444. [Google Scholar]
- Miller, M.P. Tools for Population Genetic Analyses (TFPGA) Version 1.3: A Windows program for the Analysis of Allozyme and Molecular Population Genetic Data; Utah State University: Logan, UT, USA, 1997. [Google Scholar]
- Excoffier, L.; Laval, G.; Schneider, S. Arlequin (version 3.0): An integrated software package for population genetics data analysis. Evol. Bioinform 2005, 1, 47–50. [Google Scholar]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol 2005, 14, 2611–2620. [Google Scholar]
| Population | Locality | Longitude (E) | Latitude (N) | Altitude (m) | Sample size | Voucher |
|---|---|---|---|---|---|---|
| Pop1 | Baotianman Reserve, Mt. Funiu, Neixiang county, Henan | 33°29.179′ | 111°54.96′ | 1100 | 20 | Xu-mei Wang 08082417 |
| Pop2 | Muyu, Mt. Shennongjia, Xingshan county, Hubei | 31°27.088′ | 110°16.172′ | 2908 | 18 | Xu-mei Wang and Xiao-qi Hou 09071101 |
| Pop3 | Doumugong, Mt. Taibai, Mei county, Shannxi | 34°02.286′ | 107°42.869′ | 2841 | 19 | Xiao-qi Hou 10072001 |
| Pop4 | Taibaimiao, Ningshan county, Shaanxi | 33°25.406′ | 108°31.833′ | 1878 | 20 | Xiao-qi Hou 10071301 |
| Pop5 | Mt. Hualong, Pingli county, Shaanxi | 32°01.393′ | 109°21.499′ | 2919 | 20 | Xiao-qi Hou 2010072801 |
| Pop6 | Daping, Mt. Jinfo, Nanchuan county, Chongqing | 28°58.414′ | 107°11.023′ | 1412 | 5 | Xu-mei Wang and Xiao-qi Hou 09072711 |
| Pop7 | Nanjiang county, Sichuan | 32°35.668′ | 107°06.78′ | 1809 | 19 | Yu-qu Zhang 10080801 |
| Pop8 | Hailongtun, Gaoping, Zunyi county, Guizhou | 27°48.766′ | 106°49.097′ | 1252 | 10 | Xu-mei Wang and Xiao-qi Hou 20090731 |
| Pop9 | Gelachang, Haba, Sanba, Xianggelila county, Yunan | 27°23.219′ | 100°02.754′ | 3995 | 16 | Xiao-qi Hou 09080718 |
| Pop10 | Parch house, Haba, Sanba, Xianggelila county, Yunan | 27°23.741′ | 100°02.257′ | 3727 | 16 | Xiao-qi Hou 09080720 |
| Pop11 | Xiaozhongdian, Xianggelila county, Yunan | 27°35.118′ | 99°50.835′ | 3441 | 20 | Xiao-qi Hou 1010202 |
| Pop12 | Hongxi, Meigu county, Sichuan | 28°40.243′ | 102°58.341′ | 3623 | 16 | Yu-qu Zhang and Xiao-qi Hou 09091824 |
| Primer code | Sequence (5′→3′) | Annealing temperature (°C) | No. of amplified bands | No. of polymorphic bands |
|---|---|---|---|---|
| UBC807 | (AG)8T | 51 | 12 | 12 |
| UBC811 | (GA)8C | 53 | 13 | 11 |
| UBC816 | (CA)8T | 52 | 16 | 15 |
| UBC825 | (AC)8T | 52 | 12 | 11 |
| UBC834 | (AG)8YT | 52 | 14 | 14 |
| UBC835 | (AG)8YC | 52 | 10 | 9 |
| UBC836 | (AG)8YA | 52 | 16 | 16 |
| UBC841 | (GA)8YC | 52 | 15 | 14 |
| UBC842 | (GA)8YG | 56 | 11 | 11 |
| UBC888 | BDB(CA)7 | 52 | 19 | 18 |
| UBC889 | DBD(AC)7 | 52 | 17 | 17 |
| UBC890 | VHV(GT)7 | 56 | 18 | 17 |
| UBC891 | HVH(TG)7 | 52 | 16 | 15 |
| Total | - | - | 189 | 180 |
| Populations | Na | Ne | H | I | PPB (%) |
|---|---|---|---|---|---|
| Pop1 | 1.4457 | 1.2626 | 0.1555 | 0.2332 | 44.57 |
| Pop2 | 1.4457 | 1.2771 | 0.1614 | 0.2400 | 44.57 |
| Pop3 | 1.3943 | 1.2210 | 0.1305 | 0.1970 | 39.43 |
| Pop4 | 1.3657 | 1.2156 | 0.1251 | 0.1877 | 36.57 |
| Pop5 | 1.4343 | 1.2730 | 0.1563 | 0.2318 | 43.43 |
| Pop6 | 1.0400 | 1.0213 | 0.0139 | 0.0213 | 4.00 |
| Pop7 | 1.3200 | 1.1129 | 0.0699 | 0.1115 | 32.00 |
| Pop8 | 1.1143 | 1.0766 | 0.0435 | 0.0640 | 11.43 |
| Pop9 | 1.2457 | 1.1737 | 0.0990 | 0.1447 | 24.57 |
| Pop10 | 1.2229 | 1.1614 | 0.0914 | 0.1329 | 22.29 |
| Pop11 | 1.1771 | 1.1190 | 0.0688 | 0.1014 | 17.71 |
| Pop12 | 1.2686 | 1.1613 | 0.0940 | 0.1402 | 26.86 |
| Average | 1.2895 | 1.1730 | 0.1008 | 0.1505 | 28.95 |
| Species level | 1.9543 | 1.5696 | 0.3341 | 0.5000 | 95.24 |
| Source of variation | d.f. | SSD | MSD | VC | TVP (%) | P-value * |
|---|---|---|---|---|---|---|
| Among populations | 11 | 2076.62 | 207.66 | 24.37 | 74.38 | <0.001 |
| Within populations | 187 | 663.00 | 8.39 | 8.39 | 25.62 | <0.001 |
| Among geographic regions | 2 | 712.42 | 356.21 | 11.42 | 32.89% | <0.001 |
| Within geographic regions | 196 | 2027.20 | 23.30 | 23.30 | 67.11% | <0.001 |
| Population | Pop1 | Pop2 | Pop3 | Pop4 | Pop5 | Pop6 | Pop7 | Pop8 | Pop9 | Pop10 | Pop11 | Pop12 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pop1 | **** | 0.7674 | 0.8084 | 0.7491 | 0.7928 | 0.6960 | 0.7165 | 0.7238 | 0.6944 | 0.7279 | 0.7072 | 0.7026 |
| Pop2 | 0.2648 | **** | 0.7455 | 0.7783 | 0.7513 | 0.6690 | 0.6904 | 0.6920 | 0.6890 | 0.7054 | 0.6778 | 0.6903 |
| Pop3 | 0.2127 | 0.2937 | **** | 0.8022 | 0.8196 | 0.6954 | 0.7258 | 0.7520 | 0.7027 | 0.6922 | 0.6784 | 0.7258 |
| Pop4 | 0.2889 | 0.2507 | 0.2205 | **** | 0.8108 | 0.7420 | 0.7408 | 0.7770 | 0.6770 | 0.7140 | 0.7289 | 0.7044 |
| Pop5 | 0.2322 | 0.2859 | 0.1990 | 0.2098 | **** | 0.7347 | 0.7313 | 0.7647 | 0.7084 | 0.7173 | 0.7350 | 0.6925 |
| Pop6 | 0.3625 | 0.4019 | 0.3633 | 0.2984 | 0.3083 | **** | 0.7499 | 0.8513 | 0.6349 | 0.6627 | 0.6383 | 0.6522 |
| Pop7 | 0.3334 | 0.3704 | 0.3204 | 0.3000 | 0.3129 | 0.2878 | **** | 0.7997 | 0.6230 | 0.6961 | 0.6543 | 0.6810 |
| Pop8 | 0.3232 | 0.3682 | 0.2850 | 0.2523 | 0.2683 | 0.1610 | 0.2235 | **** | 0.6851 | 0.7119 | 0.6642 | 0.6888 |
| Pop9 | 0.3647 | 0.3725 | 0.3528 | 0.3900 | 0.3447 | 0.4542 | 0.4732 | 0.3783 | **** | 0.8064 | 0.7108 | 0.7865 |
| Pop10 | 0.3175 | 0.3490 | 0.3678 | 0.3369 | 0.3323 | 0.4114 | 0.3623 | 0.3398 | 0.2152 | **** | 0.7364 | 0.7803 |
| Pop11 | 0.3464 | 0.3890 | 0.3880 | 0.3162 | 0.3079 | 0.4490 | 0.4241 | 0.4092 | 0.3413 | 0.3059 | **** | 0.7040 |
| Pop12 | 0.3529 | 0.3707 | 0.3204 | 0.3504 | 0.3674 | 0.4275 | 0.3842 | 0.3728 | 0.2401 | 0.2481 | 0.3510 | **** |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, X.-M.; Hou, X.-Q.; Zhang, Y.-Q.; Yang, R.; Feng, S.-F.; Li, Y.; Ren, Y. Genetic Diversity of the Endemic and Medicinally Important Plant Rheum officinale as Revealed by Inter-Simpe Sequence Repeat (ISSR) Markers. Int. J. Mol. Sci. 2012, 13, 3900-3915. https://doi.org/10.3390/ijms13033900
Wang X-M, Hou X-Q, Zhang Y-Q, Yang R, Feng S-F, Li Y, Ren Y. Genetic Diversity of the Endemic and Medicinally Important Plant Rheum officinale as Revealed by Inter-Simpe Sequence Repeat (ISSR) Markers. International Journal of Molecular Sciences. 2012; 13(3):3900-3915. https://doi.org/10.3390/ijms13033900
Chicago/Turabian StyleWang, Xu-Mei, Xiao-Qi Hou, Yu-Qu Zhang, Rui Yang, Shi-Fang Feng, Yan Li, and Yi Ren. 2012. "Genetic Diversity of the Endemic and Medicinally Important Plant Rheum officinale as Revealed by Inter-Simpe Sequence Repeat (ISSR) Markers" International Journal of Molecular Sciences 13, no. 3: 3900-3915. https://doi.org/10.3390/ijms13033900




