Detection of Glycomic Alterations Induced by Overexpression of P-Glycoprotein on the Surfaces of L1210 Cells Using Sialic Acid Binding Lectins
Abstract
:1. Introduction
2. Results and Discussion
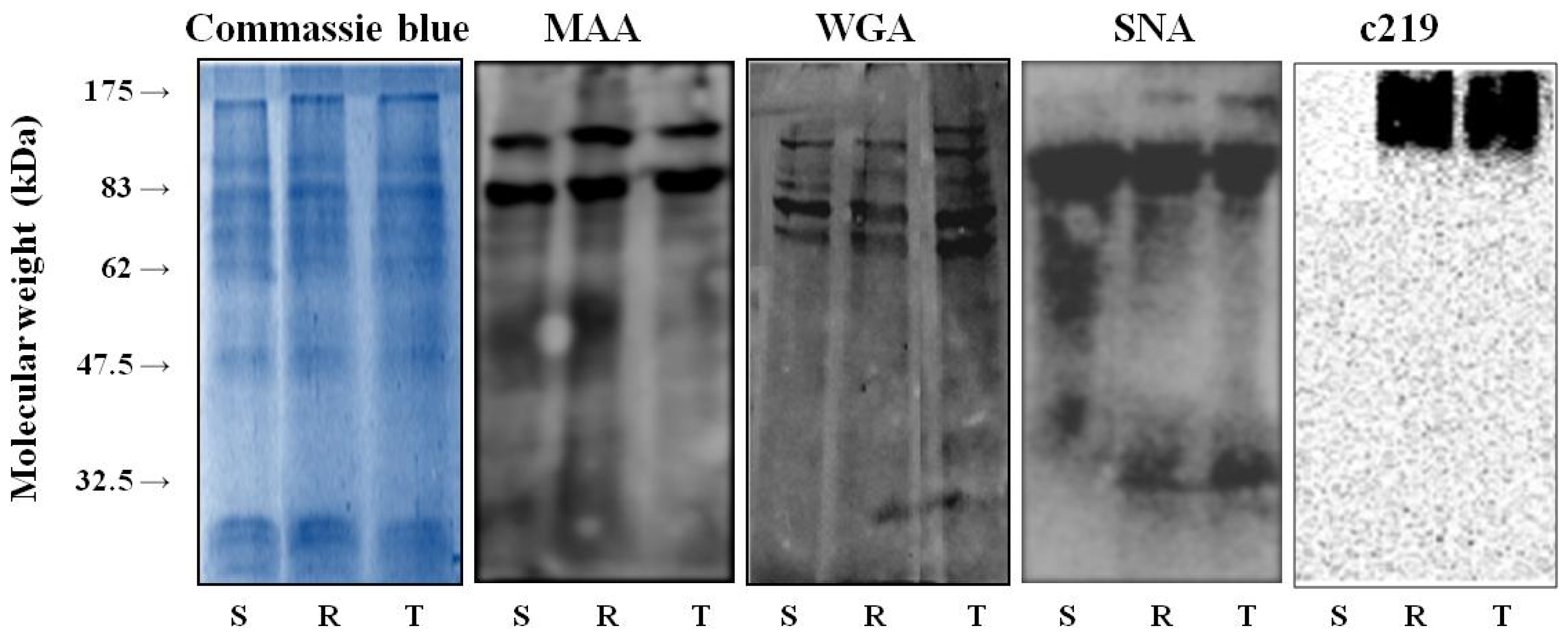
2.1. Binding of WGA, MAA and SNA to Glycoproteins in the Crude Membrane Fraction of S, R and T cells
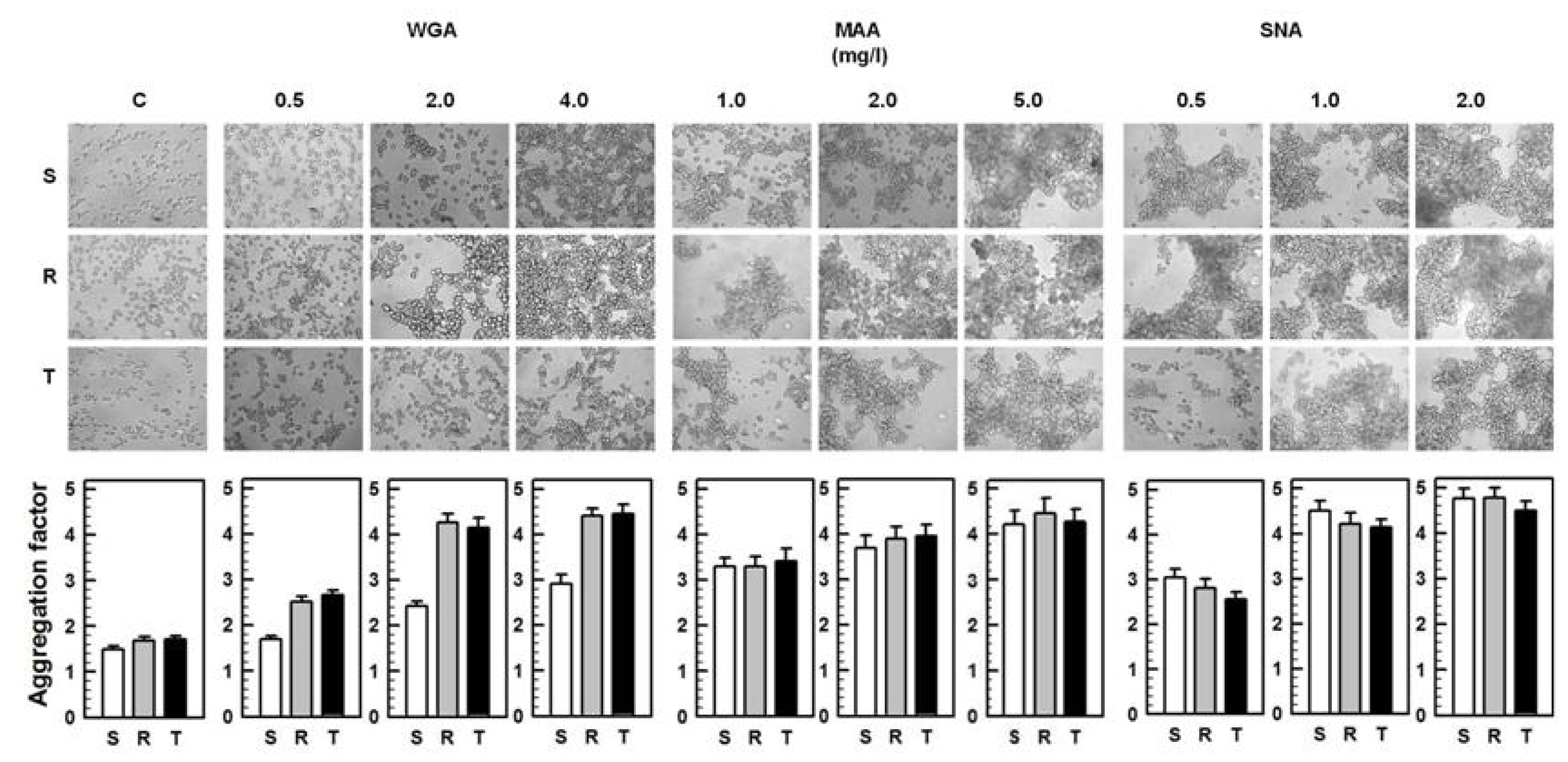
2.2. Agglutinations and Cell death Effects of WGA, MAA and SNA on S, R and T Cells
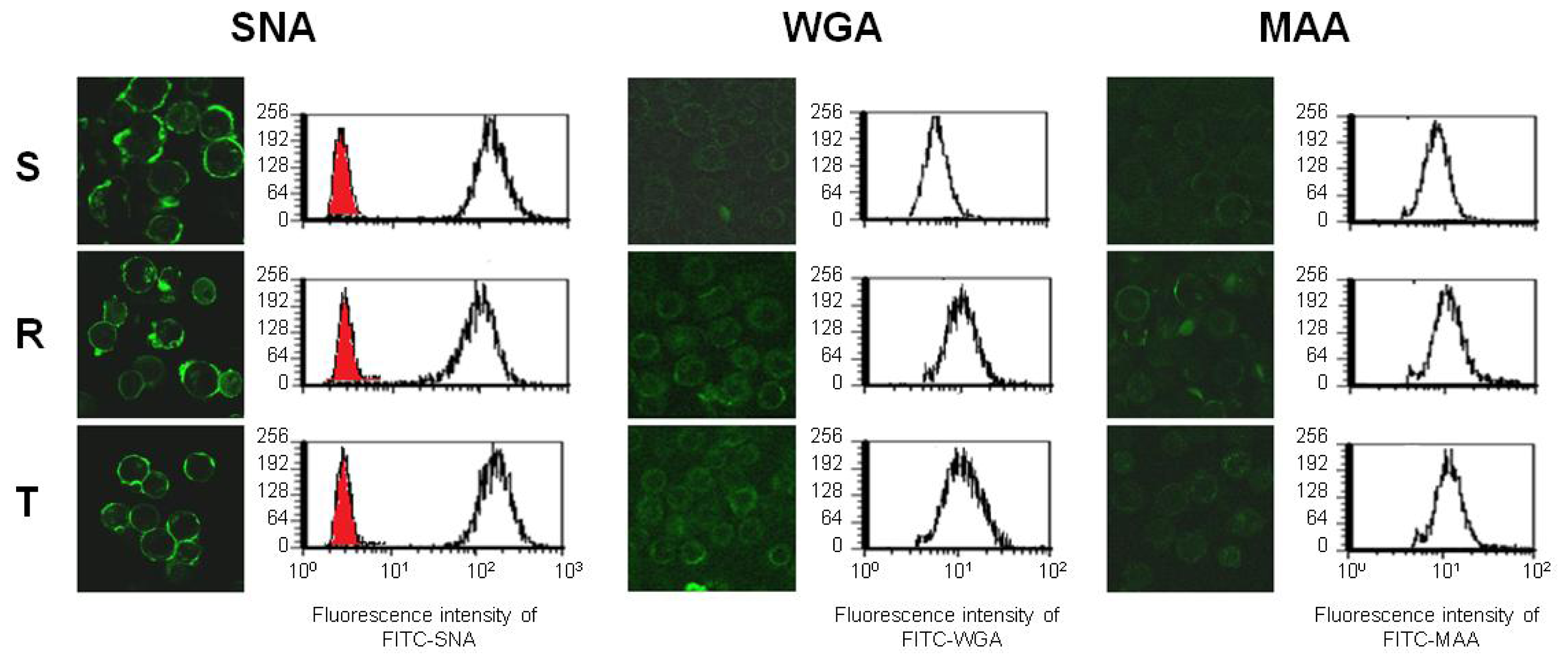
2.3. Binding of Fluorescein Isothiocyanate (FITC) Labeled WGA, MAA and SNA to the Cell Surface of S, R and T Cells
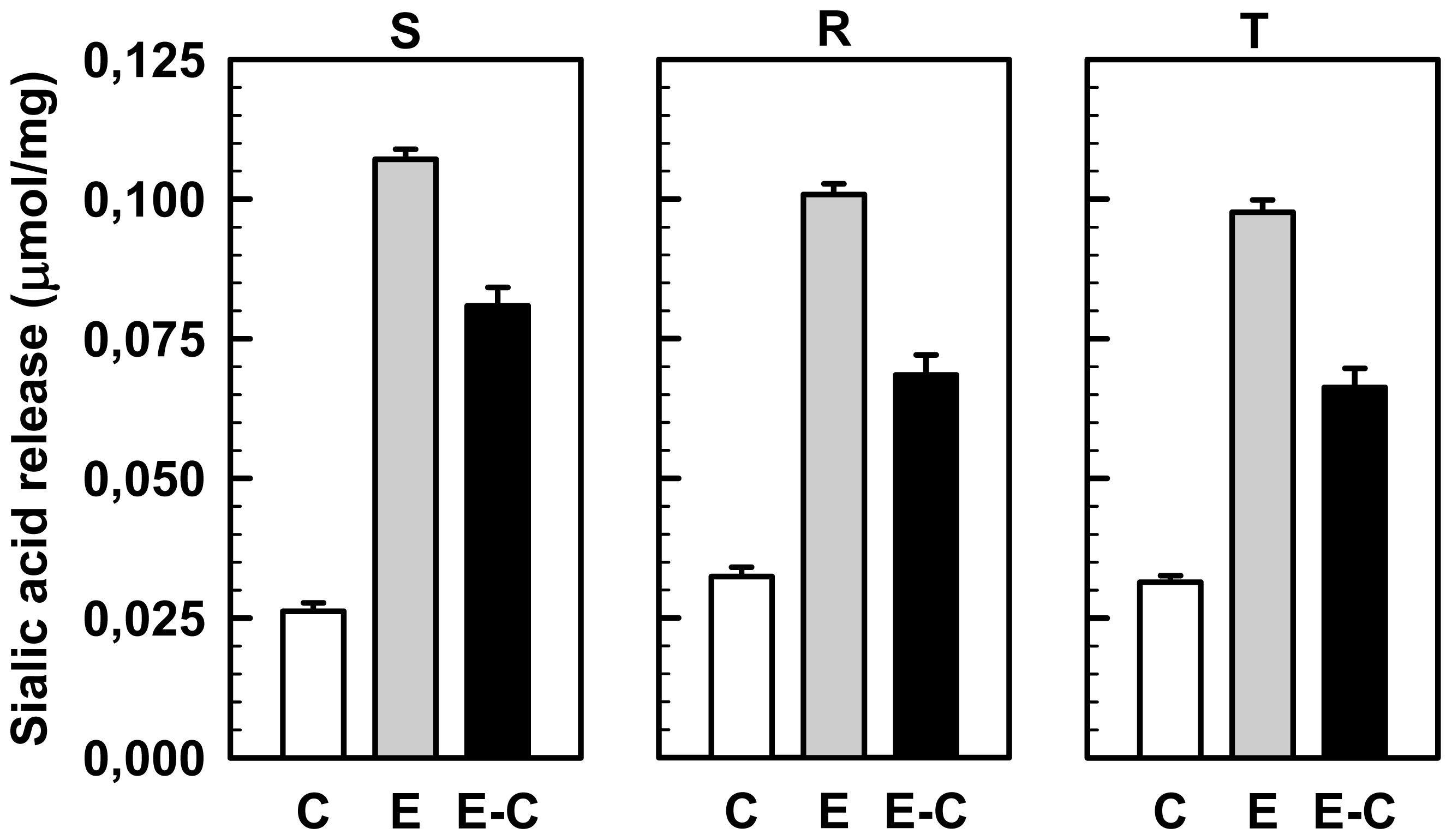
2.4. Effect of Sialidase Treatment on WGA, SNA and MAA Binding to the Cell Surface of S, R and T Cells
3. Experimental Section
3.1. Cell Culture Conditions
3.2. Western Blot and Lectin Blot Procedures
3.3. Detection of S, R and T Cells Agglutination by WGA, MAA and SNA
3.4. Effect of WGA, MAA and SNA on S, R and T Cell Survival
3.5. Detection of FITC Labeled WGA, MAA and SNA Binding to the Surface of S, R and T Cells by Confocal Microscopy and Flow Cytometry
3.6. Treatment of S, R and T cells with Sialidase and Estimation of the Amount of Sialic Acid Released from Cells to the External Medium
3.7. Statistical Analysis and Data Processing
4. Conclusions
Supplementary Information
ijms-13-15177-s001.pdfAcknowledgments
References
- Perez-Tomas, R. Multidrug resistance: Retrospect and prospects in anti-cancer drug treatment. Curr. Med. Chem 2006, 13, 1859–1876. [Google Scholar]
- Breier, A.; Barancik, M.; Sulova, Z.; Uhrik, B. P-glycoprotein--implications of metabolism of neoplastic cells and cancer therapy. Curr. Cancer Drug Targets 2005, 5, 457–468. [Google Scholar]
- Breier, A.; Gibalova, L.; Seres, M.; Barancik, M.; Sulova, Z. New Insight into P-Glycoprotein as a Drug Target. Anticancer Agents Med. Chem. 2012, in press. [Google Scholar]
- Sulova, Z.; Brtko, J.; Macejova, D.; Breier, A. Are Nuclear Receptors for Retinoids Involved in the Control of the Expression and Activity of P-Glycoprotein? In Retinoic Acid: Structure, Mechanisms and Roles in Disease; Cheng, L.-H., Ito, Y., Eds.; NOVA Publisher: New York, NY, USA, 2012; pp. 29–52. [Google Scholar]
- Cerveny, L.; Svecova, L.; Anzenbacherova, E.; Vrzal, R.; Staud, F.; Dvorak, Z.; Ulrichova, J.; Anzenbacher, P.; Pavek, P. Valproic acid induces CYP3A4 and MDR1 gene expression by activation of constitutive androstane receptor and pregnane X receptor pathways. Drug Metab. Dispos 2007, 35, 1032–1041. [Google Scholar]
- Chan, G.N.; Hoque, M.T.; Cummins, C.L.; Bendayan, R. Regulation of P-glycoprotein by orphan nuclear receptors in human brain microvessel endothelial cells. J. Neurochem 2011, 118, 163–175. [Google Scholar]
- Sulova, Z.; Macejova, D.; Seres, M.; Sedlak, J.; Brtko, J.; Breier, A. Combined treatment of P-gp-positive L1210/VCR cells by verapamil and all-trans retinoic acid induces down-regulation of P-glycoprotein expression and transport activity. Toxicol. In Vitro 2008, 22, 96–105. [Google Scholar]
- Greer, D.A.; Ivey, S. Distinct N-glycan glycosylation of P-glycoprotein isolated from the human uterine sarcoma cell line MES-SA/Dx5. Biochim. Biophys. Acta 2007, 1770, 1275–1282. [Google Scholar]
- Loo, T.W.; Bartlett, M.C.; Clarke, D.M. Thapsigargin or curcumin does not promote maturation of processing mutants of the ABC transporters, CFTR, and P-glycoprotein. Biochem. Biophys. Res. Commun 2004, 325, 580–585. [Google Scholar]
- Zhang, Z.; Wu, J.Y.; Hait, W.N.; Yang, J.M. Regulation of the stability of P-glycoprotein by ubiquitination. Mol. Pharmacol 2004, 66, 395–403. [Google Scholar]
- Ledoux, S.; Yang, R.; Friedlander, G.; Laouari, D. Glucose depletion enhances P-glycoprotein expression in hepatoma cells: Role of endoplasmic reticulum stress response. Cancer Res 2003, 63, 7284–7290. [Google Scholar]
- Seres, M.; Cholujova, D.; Bubencikova, T.; Breier, A.; Sulova, Z. Tunicamycin depresses P-glycoprotein glycosylation without an effect on its membrane localization and drug efflux activity in l1210 cells. Int. J. Mol. Sci 2011, 12, 7772–7784. [Google Scholar]
- Zhang, Z.; Zhao, Y.; Jiang, L.; Miao, X.; Zhou, H.; Jia, L. Glycomic alterations are associated with multidrug resistance in human leukemia. Int. J. Biochem. Cell Biol 2012, 44, 1244–1253. [Google Scholar]
- Fiala, R.; Sulova, Z.; El-Saggan, A.H.; Uhrik, B.; Liptaj, T.; Dovinova, I.; Hanusovska, E.; Drobna, Z.; Barancik, M.; Breier, A. P-glycoprotein-mediated multidrug resistance phenotype of L1210/VCR cells is associated with decreases of oligo- and/or polysaccharide contents. Biochim. Biophys. Acta 2003, 1639, 213–224. [Google Scholar]
- Luft, J.H. Ruthenium red and violet. II. Fine structural localization in animal tissues. Anat. Rec 1971, 171, 369–415. [Google Scholar]
- Luft, J.H. Ruthenium red and violet. I. Chemistry, purification, methods of use for electron microscopy and mechanism of action. Anat. Rec 1971, 171, 347–368. [Google Scholar]
- Uhrik, B.; El-Saggan, A.H.; Seres, M.; Gibalova, L.; Breier, A.; Sulova, Z. Structural differences between sensitive and resistant L1210 cells. Gen. Physiol. Biophys 2006, 25, 427–438. [Google Scholar]
- Vitte, J.; Benoliel, A.M.; Pierres, A.; Bongrand, P. Regulation of cell adhesion. Clin. Hemorheol. Microcirc 2005, 33, 167–188. [Google Scholar]
- Csuka, O.; Sugar, J. Cell surface properties of L1210 leukaemia cells. Acta Morphol. Acad. Sci. Hung 1976, 24, 297–305. [Google Scholar]
- Sulova, Z.; Mislovicova, D.; Gibalova, L.; Vajcnerova, Z.; Polakova, E.; Uhrik, B.; Tylkova, L.; Kovarova, A.; Sedlak, J.; Breier, A. Vincristine-induced overexpression of P-glycoprotein in L1210 cells is associated with remodeling of cell surface saccharides. J. Proteome Res 2009, 8, 513–520. [Google Scholar]
- Sulova, Z.; Ditte, P.; Kurucova, T.; Polakova, E.; Rogozanova, K.; Gibalova, L.; Seres, M.; Skvarkova, L.; Sedlak, J.; Pastorek, J.; et al. The presence of P-glycoprotein in L1210 cells directly induces down-regulation of cell surface saccharide targets of concanavalin A. Anticancer Res 2010, 30, 3661–3668. [Google Scholar]
- Lehmann, F.; Tiralongo, E.; Tiralongo, J. Sialic acid-specific lectins: occurrence, specificity and function. Cell Mol. Life Sci 2006, 63, 1331–1354. [Google Scholar]
- Polekova, L.; Barancik, M.; Mrazova, T.; Pirker, R.; Wallner, J.; Sulova, Z.; Breier, A. Adaptation of mouse leukemia cells L1210 to vincristine. Evidence for expression of P-glycoprotein. Neoplasma 1992, 39, 73–77. [Google Scholar]
- Gibalova, L.; Seres, M.; Rusnak, A.; Ditte, P.; Labudova, M.; Uhrik, B.; Pastorek, J.; Sedlak, J.; Breier, A.; Sulova, Z. P-glycoprotein depresses cisplatin sensitivity in L1210 cells by inhibiting cisplatin-induced caspase-3 activation. Toxicol. In Vitro 2012, 26, 435–444. [Google Scholar]
- Seres, M.; Ditte, P.; Breier, A.; Sulova, Z. Effect of thapsigargin on P-glycoprotein-negative and P-glycoprotein-positive L1210 mouse leukaemia cells. Gen. Physiol. Biophys 2010, 29, 396–401. [Google Scholar]
- Fakla, I.; Hever, A.; Molnar, J.; Fischer, J. Tomato lectin labels the 180 kD glycoform of P-glycoprotein in rat brain capillary endothelia and mdr tumor cells. Anticancer Res 1998, 18, 3107–3111. [Google Scholar]
- Molnar, J.; Kars, M.D.; Gunduz, U.; Engi, H.; Schumacher, U.; van Damme, E.J.; Peumans, W.J.; Makovitzky, J.; Gyemant, N.; Molnar, P. Interaction of tomato lectin with ABC transporter in cancer cells: Glycosylation confers functional conformation of P-gp. Acta Histochem 2009, 111, 329–333. [Google Scholar]
- Dalla Pellegrina, C.; Matucci, A.; Zoccatelli, G.; Rizzi, C.; Vincenzi, S.; Veneri, G.; Andrighetto, G.; Peruffo, A.D.; Chignola, R. Studies on the joint cytotoxicity of Wheat Germ Agglutinin and monensin. Toxicol. In Vitro 2004, 18, 821–827. [Google Scholar]
- Carvalho, F.C.; Soares, S.G.; Tamarozzi, M.B.; Rego, E.M.; Roque-Barreira, M.C. The recognition of N-glycans by the lectin ArtinM mediates cell death of a human myeloid leukemia cell line. PLoS One 2011, 6, e27892. [Google Scholar]
- Aubery, M.; Reynier, M.; Lopez, M.; Ogier-Denis, E.; Font, J.; Bardin, F. WGA binding to the surface of two autologous human melanoma cell lines: different expression of sialyl and N-acetylglucosaminyl residues. Cell Biol. Int. Rep 1990, 14, 275–286. [Google Scholar]
- Ree, H.J. Lectin histochemistry of malignant tumors. II. Concanavalin A: A new histochemical marker for macrophage-histiocytes in follicular lymphoma. Cancer 1983, 51, 1639–1646. [Google Scholar]
- Ree, H.J.; Hsu, S.M. Lectin histochemistry of malignant tumors. I. Peanut agglutinin (PNA) receptors in follicular lymphoma and follicular hyperplasia: An immunohistochemical study. Cancer 1983, 51, 1631–1638. [Google Scholar]
- Ree, H.J.; Raine, L.; Crowley, J.P. Lectin binding patterns in diffuse large cell lymphoma. Cancer 1983, 52, 2089–2099. [Google Scholar]
- Vetri, V.; Carrotta, R.; Picone, P.; di Carlo, M.; Militello, V. Concanavalin A aggregation and toxicity on cell cultures. Biochim. Biophys. Acta 2010, 1804, 173–183. [Google Scholar]
- Rutishauser, U. Polysialic acid at the cell surface: Biophysics in service of cell interactions and tissue plasticity. J. Cell Biochem 1998, 70, 304–312. [Google Scholar]
- Soeiro, M.N.; Silva-Filho, F.C.; Meirelles, M.N. The nature of anionic sites and the endocytic pathway in heart muscle cells. J. Submicrosc. Cytol. Pathol 1994, 26, 121–130. [Google Scholar]
- Wieraszko, A. Evidence that ruthenium red disturbs the synaptic transmission in the rat hippocampal slices through interacting with sialic acid residues. Brain Res 1986, 378, 120–126. [Google Scholar]
- Kim, S.; Oh, D.B.; Kang, H.A.; Kwon, O. Features and applications of bacterial sialidases. Appl. Microbiol. Biotechnol 2011, 91, 1–15. [Google Scholar]
- Barancik, M.; Bohacova, V.; Gibalova, L.; Sedlak, J.; Sulova, Z.; Breier, A. Potentiation of anticancer drugs: Effects of pentoxifylline on neoplastic cells. Int. J. Mol. Sci 2012, 13, 369–382. [Google Scholar]
- Thies, A.; Moll, I.; Berger, J.; Schumacher, U. Lectin binding to cutaneous malignant melanoma: HPA is associated with metastasis formation. Br. J. Cancer 2001, 84, 819–823. [Google Scholar]
- Knibbs, R.N.; Goldstein, I.J.; Ratcliffe, R.M.; Shibuya, N. Characterization of the carbohydrate binding specificity of the leukoagglutinating lectin from Maackia amurensis. Comparison with other sialic acid-specific lectins. J. Biol. Chem 1991, 266, 83–88. [Google Scholar]
- Garner, R.E.; Walker, A.N.; Horst, M.N. Morphologic and biochemical studies of chitin expression in Pneumocystis carinii. J. Protozool 1991, 38, 12S–14S. [Google Scholar]
- De Graaf, T.W.; Slot, S.S.; Peters, G.J.; van Dijk, W. Changes in glycosylation of L1210 cells after exposure to various antimetabolites. Eur. J. Cancer 1993, 29A, 1760–1765. [Google Scholar]
- Liang, X.J.; Shen, D.W.; Garfield, S.; Gottesman, M.M. Mislocalization of membrane proteins associated with multidrug resistance in cisplatin-resistant cancer cell lines. Cancer Res 2003, 63, 5909–5916. [Google Scholar]
- Pastan, I.; Gottesman, M.M.; Ueda, K.; Lovelace, E.; Rutherford, A.V.; Willingham, M.C. A retrovirus carrying an MDR1 cDNA confers multidrug resistance and polarized expression of P-glycoprotein in MDCK cells. Proc. Natl. Acad. Sci. USA 1988, 85, 4486–4490. [Google Scholar]
- Cell Counter + Analyser System Model TT, Operator Manual. Available online: http://www.ibgm.med.uva.es/files/upload/manual//casyoperatormanual.pdf accessed on 30 April 2002.
- Gerlier, D.; Thomasset, N. Use of MTT colorimetric assay to measure cell activation. J. Immunol. Methods 1986, 94, 57–63. [Google Scholar]
- Warren, L. The thiobarbituric acid assay of sialic acids. J. Biol. Chem 1959, 234, 1971–1975. [Google Scholar]


| Lectin from | Abbreviation | Specificity |
|---|---|---|
| Triticum vulgaris (Wheat germ) | WGA | Internal β-d-N-acetyl glucosamine> β-d-N-acetyl glucosamine–SA > N-acetyl galactosamine > lactose > galalactose |
| Maackia amurensis | MAA | α-2,3 branched SA |
| Sambucus nigra | SNA | α-2,6 branched SA |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bubencíkova, T.; Cholujová, D.; Messingerová, L.; Mislovicova, D.; Seres, M.; Breier, A.; Sulova, Z. Detection of Glycomic Alterations Induced by Overexpression of P-Glycoprotein on the Surfaces of L1210 Cells Using Sialic Acid Binding Lectins. Int. J. Mol. Sci. 2012, 13, 15177-15192. https://doi.org/10.3390/ijms131115177
Bubencíkova T, Cholujová D, Messingerová L, Mislovicova D, Seres M, Breier A, Sulova Z. Detection of Glycomic Alterations Induced by Overexpression of P-Glycoprotein on the Surfaces of L1210 Cells Using Sialic Acid Binding Lectins. International Journal of Molecular Sciences. 2012; 13(11):15177-15192. https://doi.org/10.3390/ijms131115177
Chicago/Turabian StyleBubencíkova, Tatiana, Dana Cholujová, Lucia Messingerová, Danica Mislovicova, Mario Seres, Albert Breier, and Zdena Sulova. 2012. "Detection of Glycomic Alterations Induced by Overexpression of P-Glycoprotein on the Surfaces of L1210 Cells Using Sialic Acid Binding Lectins" International Journal of Molecular Sciences 13, no. 11: 15177-15192. https://doi.org/10.3390/ijms131115177




