Conventional Study on Novel Dicationic Ionic Liquid Inclusion with β-Cyclodextrin
Abstract
:1. Introduction
2. Experimental
2.1. Reagent and Solution
2.2. Instrumentation
2.3. Synthesis
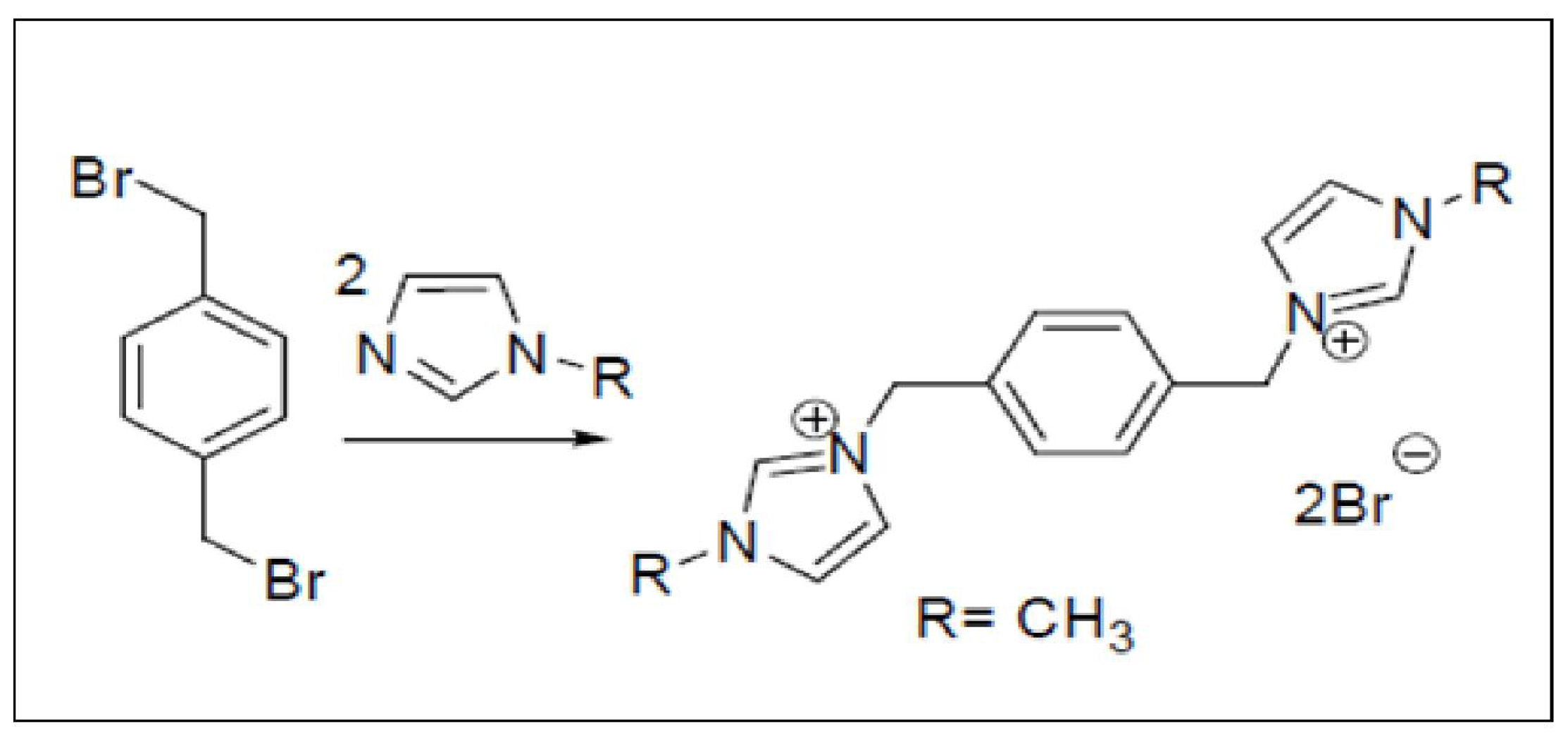
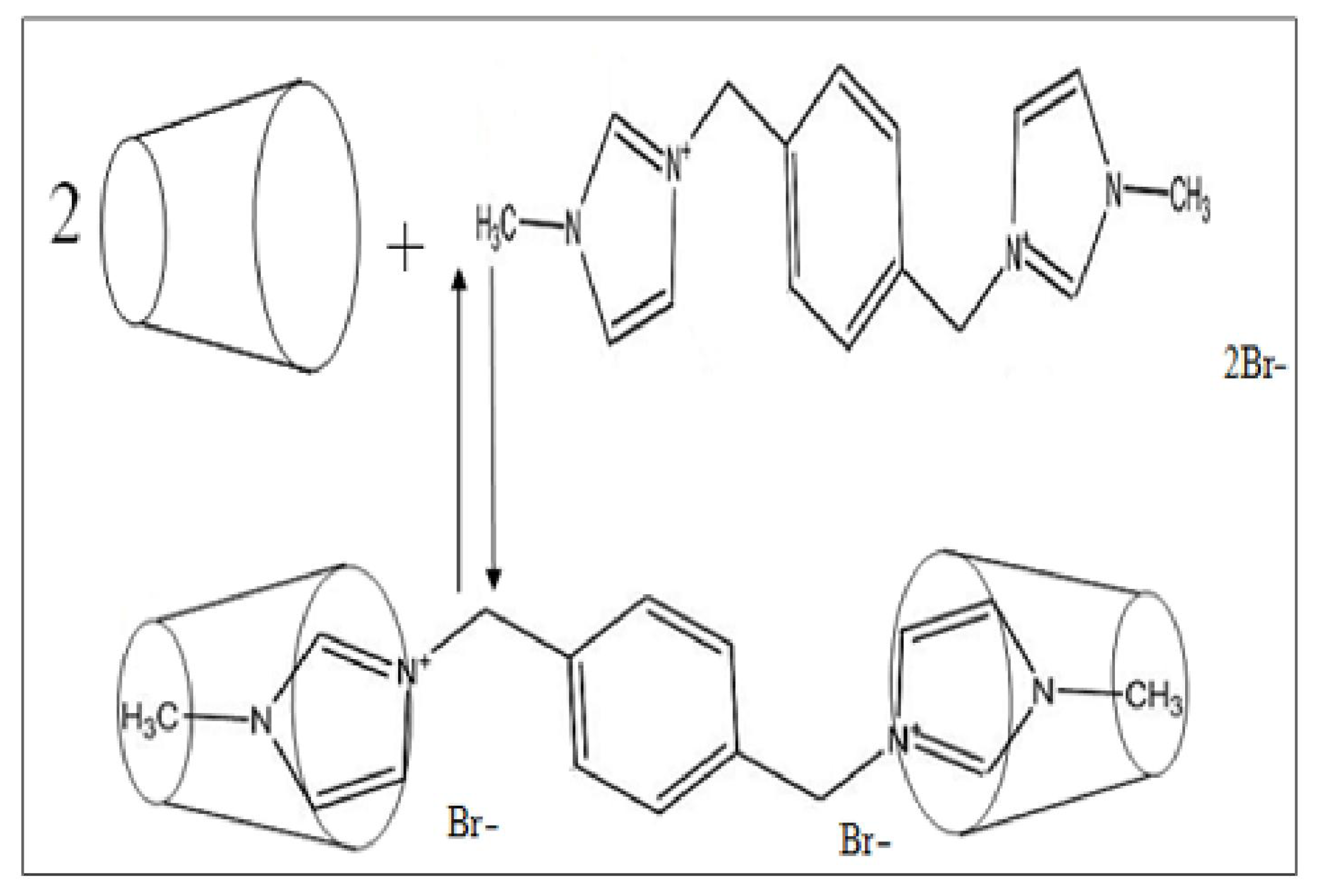
2.3.1. Preparation of 3,3′-(1,4-Phenylenebis [methylene]) bis(1-methyl-1H-imidazol-3-ium) di(bromide) (PhenmimBr)
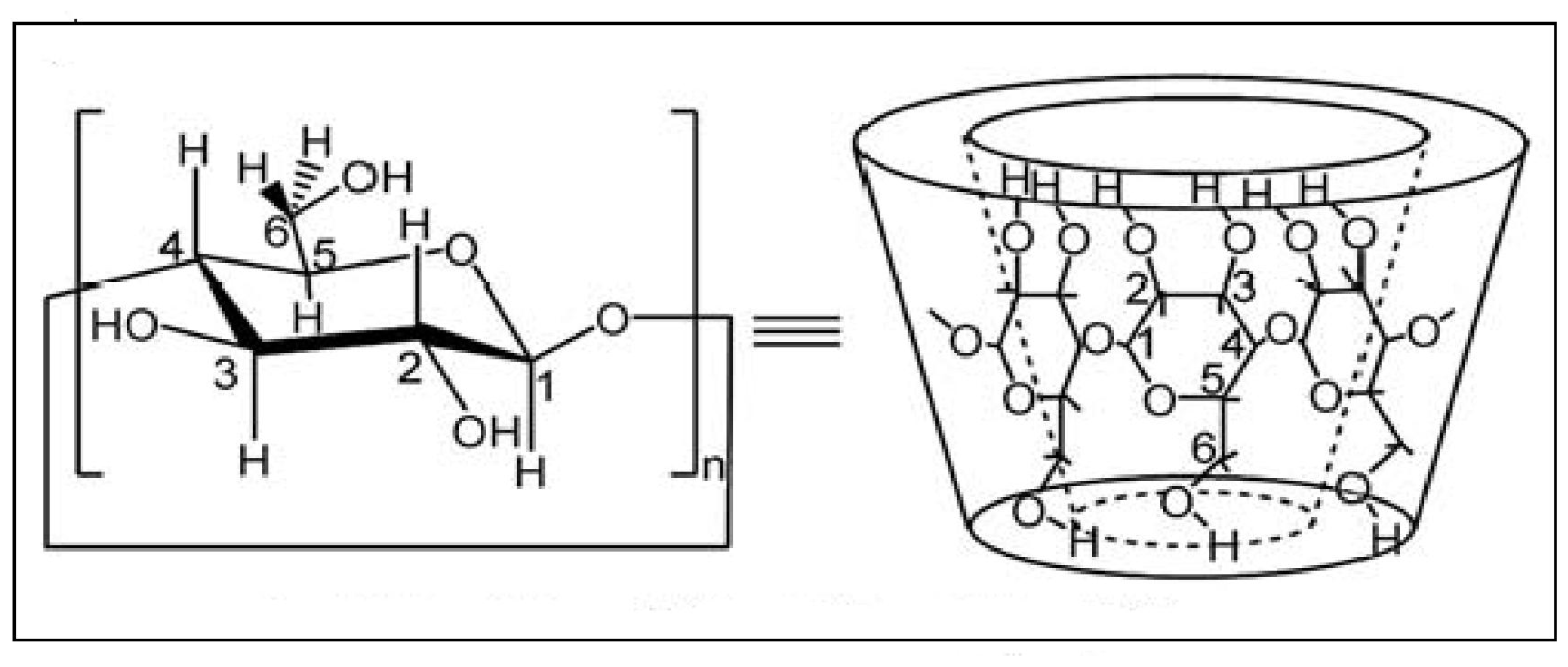
2.3.2. Synthesis of β-Cyclodextrin-PhenmimBr (β-CD-PhenmimBr)
2.4. Procedure
2.4.1. Preparation of β-CD-PhenmimBr for Spectroscopic Studies
3. Results and Discussion
3.1. 1H Nuclear Magnetic Resonance (NMR) Spectra
3.2. Correlation Spectroscopy (COSY)
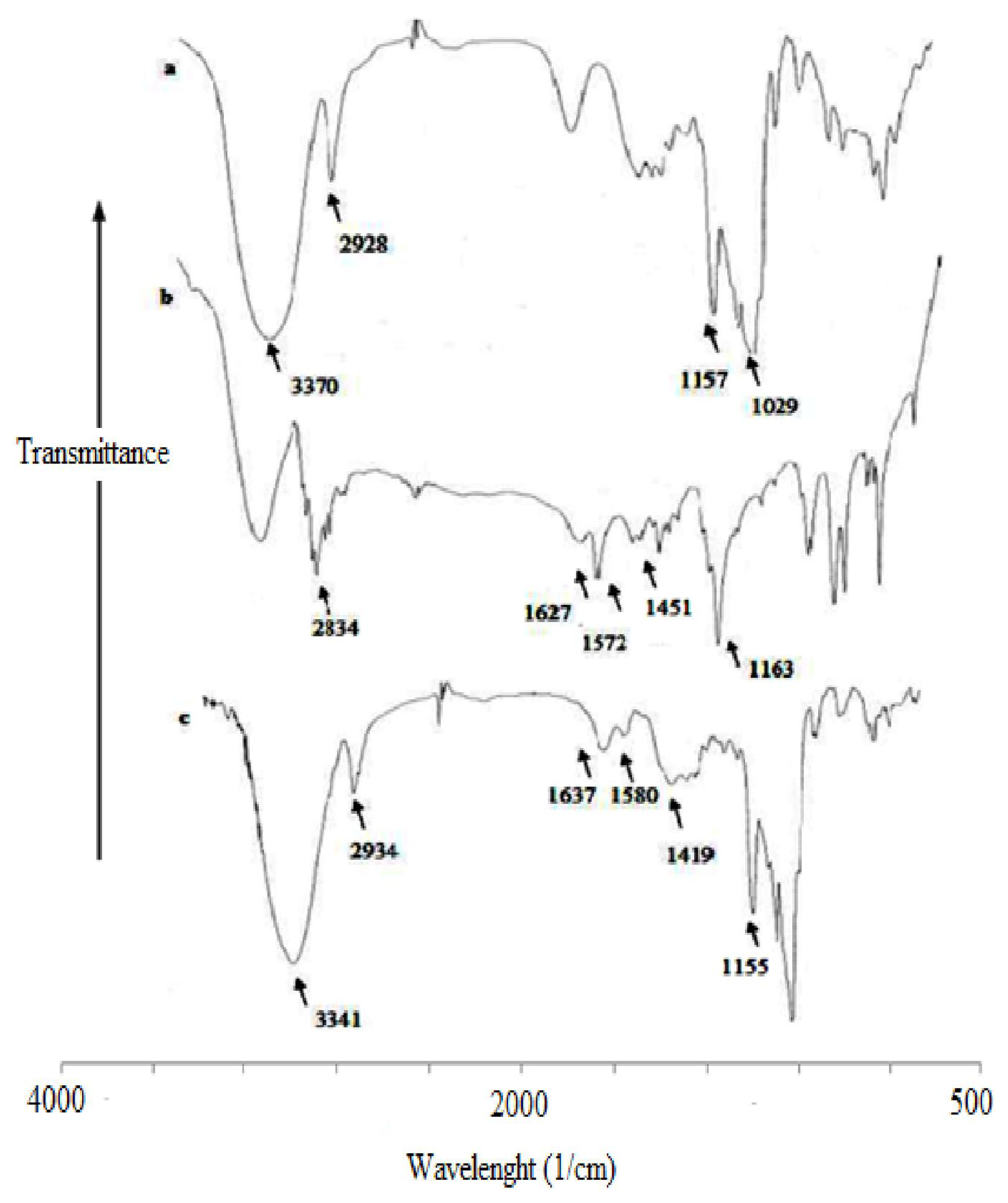
3.3. Fourier Transform Infrared (FT-IR) Spectroscopy
3.4. X-ray Diffraction (XRD) Analysis
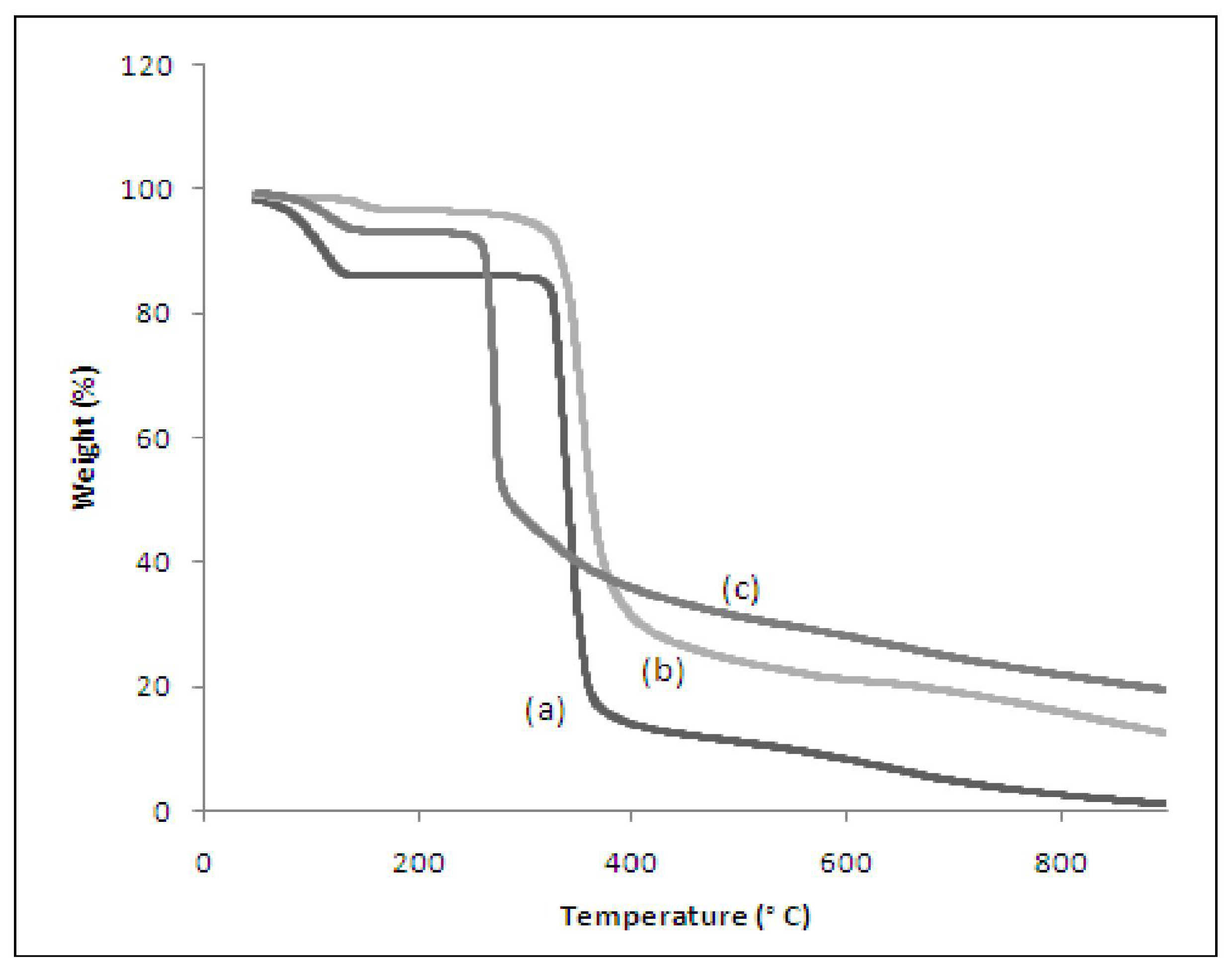
3.5. Thermogravimetric Analysis (TGA)
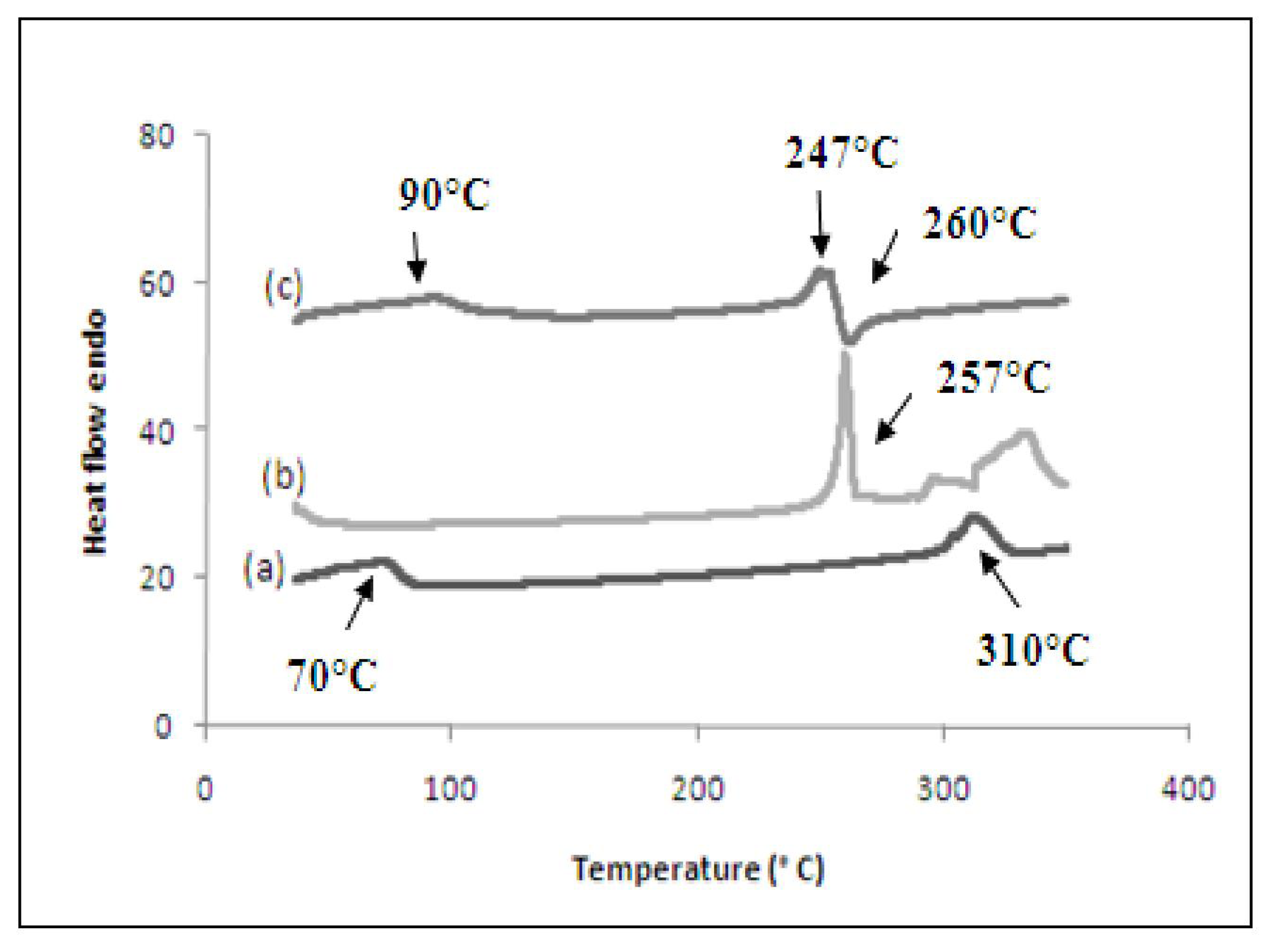
3.6. Differential Scanning Calorimetry (DSC)
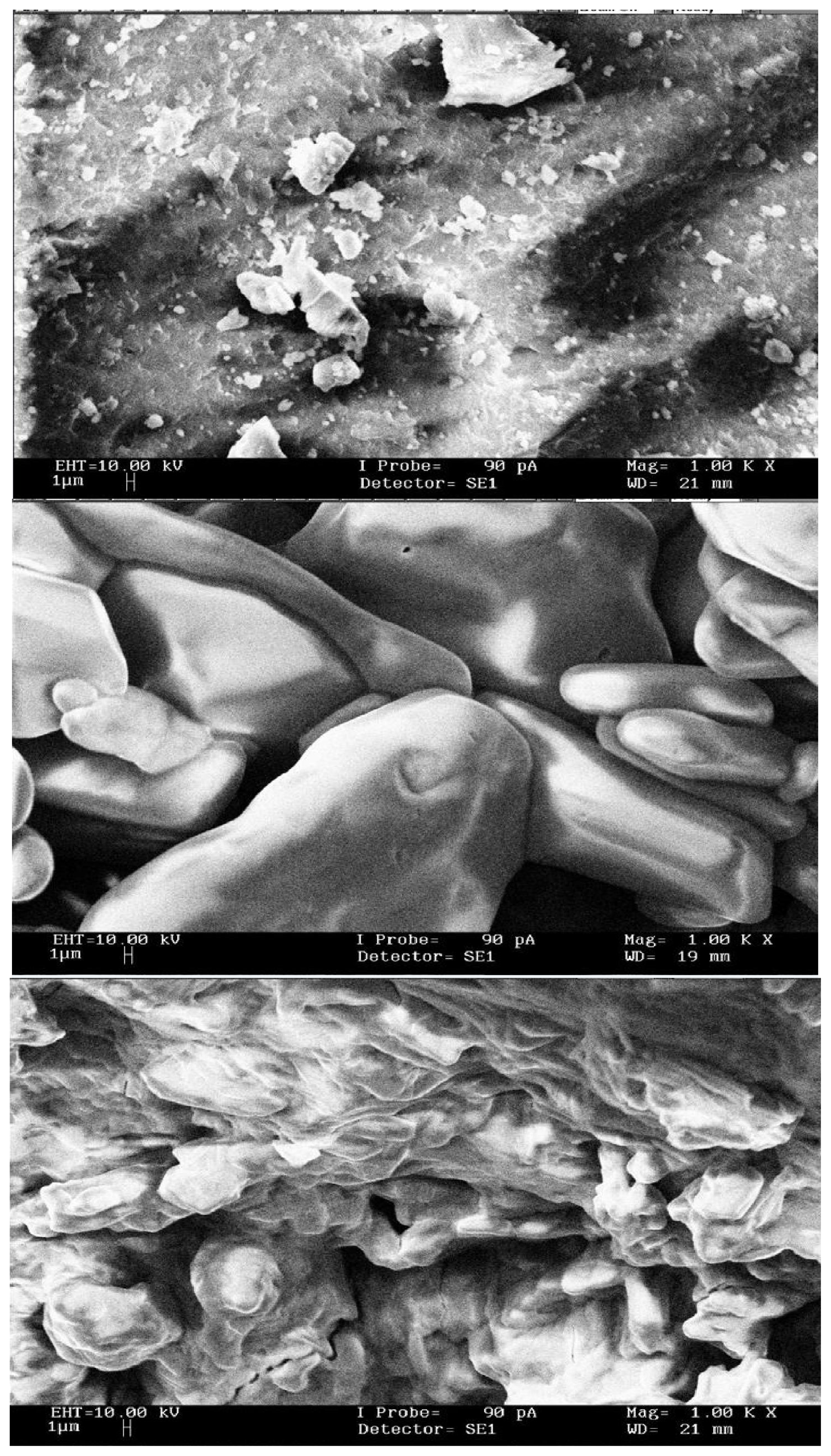
3.7. Scanning Electron Microscopy (SEM)
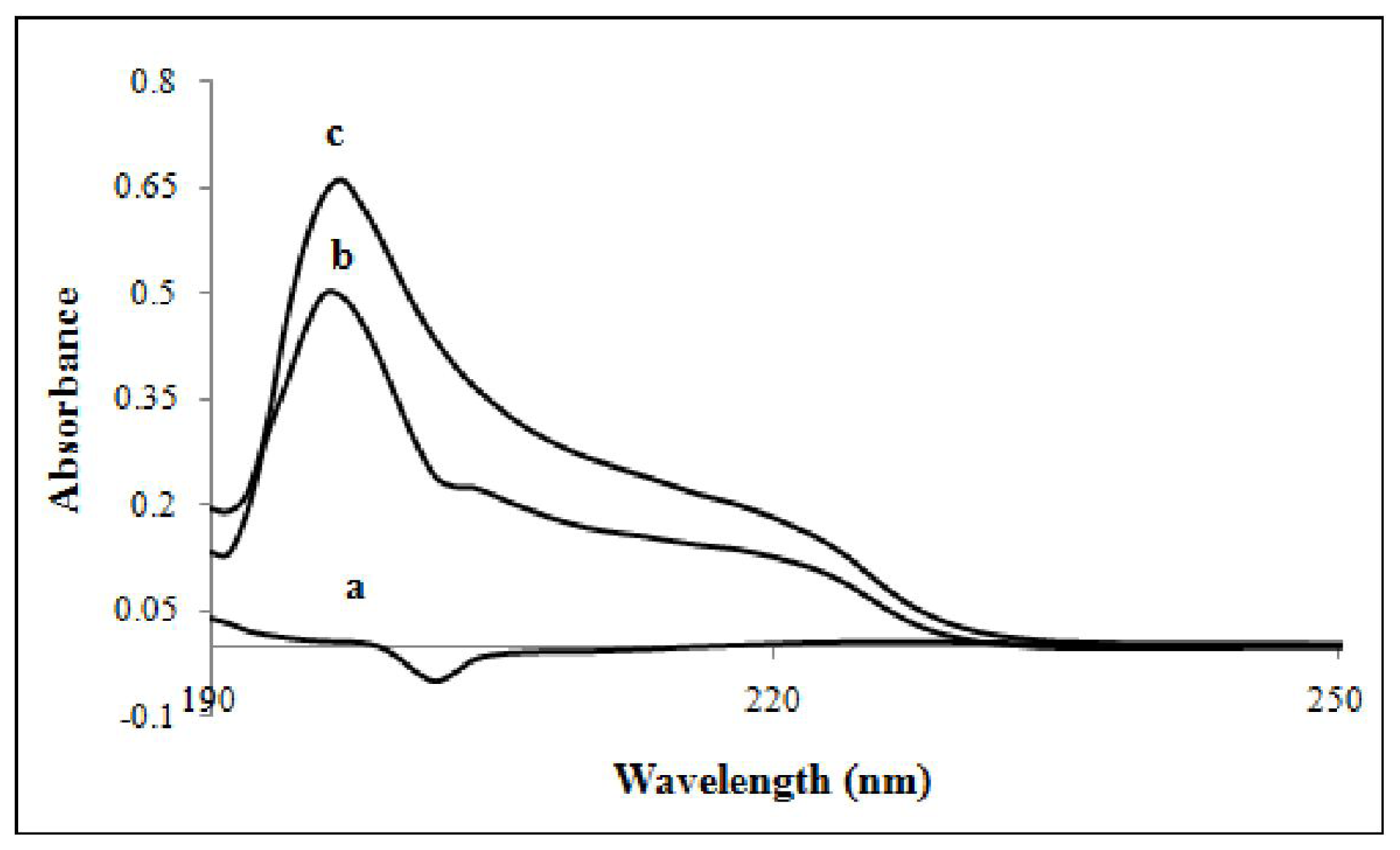
3.8. UV-Visible Spectroscopy
3.8.1. Stoichiometry of the Complex and Formation Constant
3.8.2. Inclusion Structure
4. Conclusion
Acknowledgement
Reference
- Szejtli, J. Introduction and general overview of cyclodextrin chemistry. Chem Rev 1998, 98, 1743–1753. [Google Scholar]
- Ashnagar, A; Naseri, NG; Khanaki, B. Study of the β-cyclodextrin imipramine hydrochloride inclusion complex and determination of its stability constant (K) by UV-Visible spectroscopy. E-J Chem 2007, 4, 550–558. [Google Scholar]
- Letellier, S; Maupas, B; Gramond, JP; Guyon, F; Gareil, P. Determination of the formation constant for the inclusion complex between rutin and methyl-β-cyclodextrin. Anal Chim Acta 1995, 315, 357–363. [Google Scholar]
- Schneiderman, E; Stalcup, AM. Cyclodextrins: A versatile tool in separation science. J Chromatogr B 2000, 745, 83–102. [Google Scholar]
- Bernini, A; Spiga, O; Ciutti, A; Scarselli, M; Bottoni, G; Mascagni, P; Niccolai, N. NMR studies of the inclusion complex between beta-cyclodextrin and paroxetine. Eur J Pharm Sci 2004, 22, 445–450. [Google Scholar]
- Bonenfant, D; Niquette, P; Mimeault, M; Furtos-Matei, A; Hauslera, R. UV-VIS and FTIR spectroscopic analyses of inclusion complexes of nonylphenol and nonylphenol ethoxylate with β-cyclodextrin. Water Res 2009, 43, 3575–3581. [Google Scholar]
- Waleczek, KJ; Marques, C; Hempel, B; Schmidt, PC. Phase solubility study of pure (−)-α-bisabolol and camomile essential oil with β-cyclodextrin. Eur J Pharm Biopharm 2003, 55, 247–251. [Google Scholar]
- He, Y; Shen, X. Interaction between β-cyclodextrin and ionic liquids in aqueous solutions investigated by a competitive method using a substituted 3H-indole probe. J Photochem Photobiol Chem 2008, 197, 253–259. [Google Scholar]
- Yáñez, C; Salazar, R; Núñez-Vergara, LJ; Squella, JA. Spectrophotometric and electrochemical study of the inclusion complex between β-cyclodextrin and furnidipine. J Pharm Biomed Anal 2004, 35, 51–56. [Google Scholar]
- Li, N; Zhao, X; Gao, Y; Zheng, L; Zhang, J; Yu, L. Complex formation of ionic liquid surfactant and β-cyclodextrin. Colloids Surf A 2007, 292, 196–201. [Google Scholar]
- Tavornvipas, S; Hirayama, F; Arima, H; Uekama, K; Ishiguro, T; Oka, M; Hamayasu, K; Hashimoto, H. 6-O-a-(4-O-a-d-glucuronyl)-d-glucosyl-β-cyclodextrin solubilizing ability and some cellular effects. Int J Pharm 2002, 249, 199–209. [Google Scholar]
- Padukka, I; Bhandari, B; Darcy, B. Evaluation of various extraction methods of encapsulated oil from β-cyclodextrin-lemon oil complex powder. J Food Compos Anal 2000, 13, 59–70. [Google Scholar]
- Szente, L; Szejtli, J. Cyclodextrins as food ingredients. Trends Food Sci Technol 2004, 15, 137–142. [Google Scholar]
- Perrin, C; Fabre, H; Maftouh, M; Massart, DL; Heyden, YV. Robustness testing of chiral separations by capillary electrophoresis using highly-sulfated cyclodextrins. J Chromatogr A 2003, 1007, 165–177. [Google Scholar]
- Morillo, E; Perez-Martinez, JI; Gines, JM. Leaching of 2,4-d from a soil in the presence of β-cyclodextrin: Laboratory columns experiments. Chemosphere 2001, 44, 1065–1069. [Google Scholar]
- Jozefaciuk, G; Muranyi, A; Fenyvesi, E. Effect of randomly methylated β-cyclodextrin on physical properties of soils. Environ Sci Technol 2003, 37, 3012–3017. [Google Scholar]
- McEwen, AB; Ngo, HL; LeCompte, K; Goldman, JL. Electrochemical properties of imidazolium salt electrolytes for electrochemical capacitor applications. J Electrochem Soc 1999, 146, 1687–1695. [Google Scholar]
- Xue, H; Shreeve, MJ. Ionic Liquids with Fluorine-Containing Cations. Eur J Inorg Chem 2005, 13, 2573–2580. [Google Scholar]
- Wilkes, JS. Ionic Liquid in Synthesis; Wasserscheid, P, Welton, T, Eds.; Wiley-VCH: Hoboken, NJ, USA, 2002. [Google Scholar]
- Anderson, JL; Ding, R; Ellern, A; Armstrong, DW. Structure and properties of high stability geminal dicationic ionic liquids. J Am Chem Soc 2005, 127, 593–604. [Google Scholar]
- Qi, S; Cui, S; Chen, X; Hu, Z. Rapid and sensitive determination of anthraquinones in Chinese herb using 1-butyl-3-methylimiazolium-based ionic liquid with β-cyclodextrin as modifier in capillary zone electrophoresis. J Chromatogr A 2004, 1059, 191–198. [Google Scholar]
- Yu, L; Qin, W; Li, SFY. Ionic liquids as additives for separation of benzoic acid and chlorophenoxy acid herbicides by capillary electrophoresis. Anal Chim Acta 2006, 547, 165–171. [Google Scholar]
- Jing, B; Chen, X; Hao, JC; Qiu, HY; Chai, YC; Zhang, GD. Supramolecular self-assembly of polypseudorotaxanes in ionic liquid. Colloids Surf A 2007, 292, 51–55. [Google Scholar]
- Tian, K; Wang, YS; Chen, YL; Chen, YG; Hu, ZD. Application of 1-alkyl-3-methylimidazolium-based ionic liquids as background electrolyte in capillary zone electrophoresis for the simultaneous determination of five anthraquinones in Rhubarb. Talanta 2007, 72, 587–593. [Google Scholar]
- Gao, YA; Li, ZH; Du, JM; Han, BX; Li, GZ; Hou, WG; Shen, D; Zheng, LQ; Zhang, GY. Preparation and characterization of inclusion complexes of β-cyclodextrin with ionic liquid. Chem Eur J 2005, 11, 5875–5880. [Google Scholar]
- Gao, Y; Zhao, X; Dong, B; Zheng, L; Li, N; Zhang, S. Inclusion complexes of β-cyclodextrin with ionic liquid surfactants. J Phys Chem B 2006, 110, 8576–8581. [Google Scholar]
- Amajjahe, S; Ritter, H. Anion complexation of vinylimidazolium salts and its influence on polymerization. Macromolecules 2008, 41, 716–718. [Google Scholar]
- He, Y; Chen, Q; Xu, C; Zhang, J; Shen, X. Interaction between ionic liquid and β-Cyclodextrin: A discussion of association pattern. J Phys Chem B 2009, 113, 231–238. [Google Scholar]
- Li, HG; Zhang, Q; Zhao, Q; Liu, M; Liu, J; Sun, DZ. Studies on interaction of ionic liquids with cyclodextrin aqueous solution. Indian J Chem 2010, 49A, 752–756. [Google Scholar]
- Puvaneswary, S; Sharifah, M; Alias, Y. Synthesis and characterization of the inclusion complex of dicationic ionic liquid and β-cyclodextrin. Int J Mol Sci 2010, 11, 3675–3685. [Google Scholar]
- Kilivelu, G; Yatimah, A. Synthesis and characterization of novel dimeric ionic liquids by conventional approaches. Int J Mol Sci 2008, 9, 1207–1213. [Google Scholar]
- Cwiertnia, B; Hladon, T; Stobiecki, M. Stability of diclofenac sodium in the inclusion complex with β-cyclodextrin in the solid state. J Pharm Pharmacol 1999, 51, 1213–1218. [Google Scholar]
- Li, J; Ni, X; Zhou, Z; Leong, KW. Preparation and characterization of polypseudorotaxanes based on block-selected inclusion complexation between poly(propylene oxide)-poly(ethylene oxide)-poly(propylene oxide) triblock copolymers and α-cyclodextrin. J Am Chem Soc 2003, 125, 1788–1795. [Google Scholar]
- Qian, L; Guan, Y; Xiao, H. Preparation and characterization of inclusion complexes of a cationic β-cyclodextrin polymer with butylparaben or triclosan. Int J Pharm 2008, 357, 244–251. [Google Scholar]
- Nostro, PL; Santoni, I; Bonini, M; Baglioni, P. Inclusion compound from a semifluorinated alkane and β-cyclodextrin. Langmuir 2003, 19, 2313–2317. [Google Scholar]
- Dupont, J. On the solid, liquid and solution structural organization of imidazolium ionic liquids. J Braz Chem Soc 2004, 15, 341–350. [Google Scholar]
- Okumura, H; Kawaguchi, Y; Harada, A. Preparation and characterization of the inclusion complexes of poly(dimethylsilane)s with cyclodextrins. Macromolecules 2003, 36, 6422–6429. [Google Scholar]
- Liu, L; Zhu, S. A study on the supramolecular structure of inclusion complex of β-cyclodextrin with prazosin hydrochloride. Carbohydr Polym 2007, 68, 472–476. [Google Scholar]
- Pralhad, T; Rajendrakumar, K. Study of freeze-dried quercetin–cyclodextrin binary systems by DSC, FT-IR, X-ray diffraction and SEM analysis. J Pharmaceut Biomed Anal 2004, 34, 333–339. [Google Scholar]
- Wang, HY; Han, J; Feng, XG. Spectroscopic study of orange G–β-cyclodextrin complex and its analytical application. Spectrochim Acta Part A 2007, 66, 578–585. [Google Scholar]
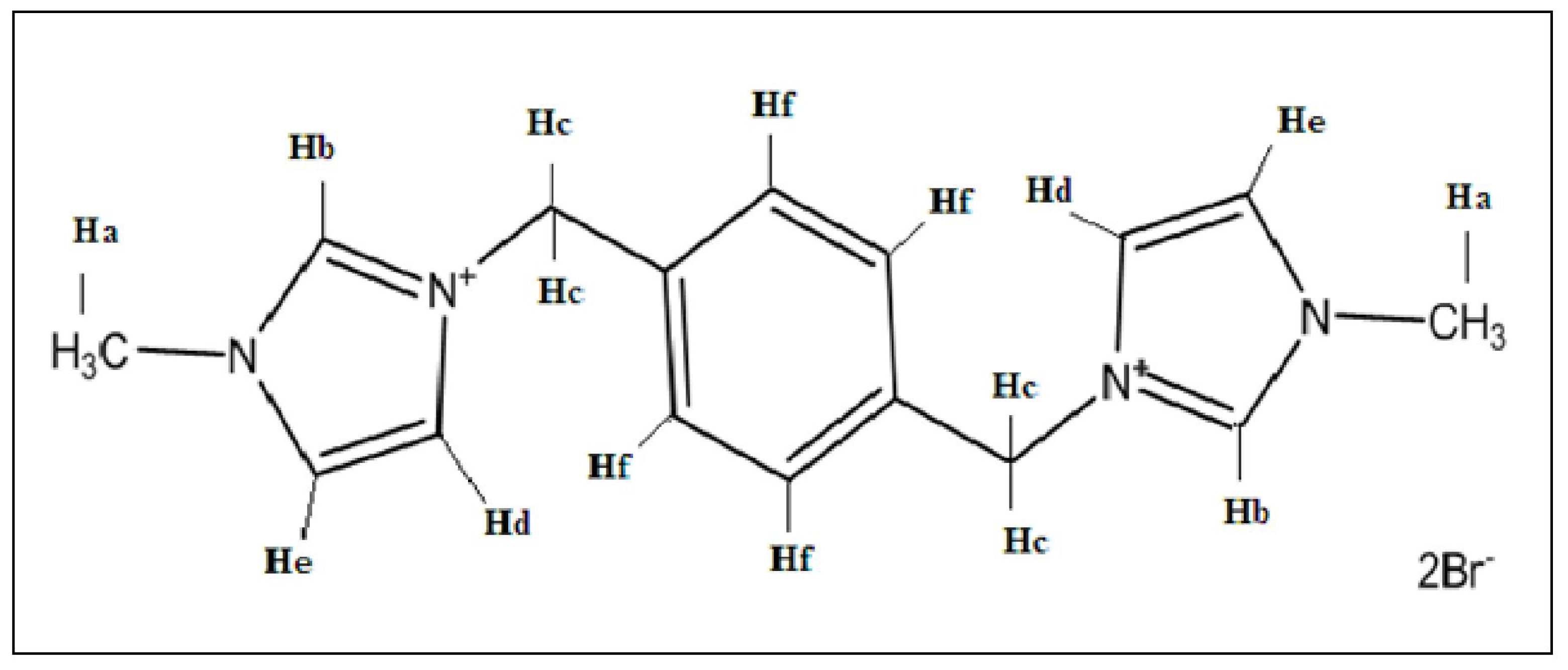




| β-CD | PhenmimBr | β-CD-PhenmimBr | ||
|---|---|---|---|---|
| δ | δ | δ | Δδ | |
| H1 | 4.8191 | 4.8094 | −0.0097 | |
| H2 | 3.3144 | 3.2955 | −0.0189 | |
| H3 | 3.6402 | 3.5987 | −0.0415 | |
| H4 | 3.3607 | 3.3504 | −0.0103 | |
| H5 | 3.6006 | 3.5591 | −0.0415 | |
| H6 | 3.6469 | 3.6189 | −0.0280 | |
| Ha | 3.8532 | 3.8410 | −0.0122 | |
| Hb | 9.3449 | 9.2430 | −0.1019 | |
| Hc | 5.4598 | 5.4226 | −0.0372 | |
| Hd | 7.8243 | 7.7633 | −0.0610 | |
| He | 7.7291 | 7.6974 | −0.0317 | |
| Hf | 7.4948 | 7.4637 | −0.0311 | |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mohamad, S.; Surikumaran, H.; Raoov, M.; Marimuthu, T.; Chandrasekaram, K.; Subramaniam, P. Conventional Study on Novel Dicationic Ionic Liquid Inclusion with β-Cyclodextrin. Int. J. Mol. Sci. 2011, 12, 6329-6345. https://doi.org/10.3390/ijms12096329
Mohamad S, Surikumaran H, Raoov M, Marimuthu T, Chandrasekaram K, Subramaniam P. Conventional Study on Novel Dicationic Ionic Liquid Inclusion with β-Cyclodextrin. International Journal of Molecular Sciences. 2011; 12(9):6329-6345. https://doi.org/10.3390/ijms12096329
Chicago/Turabian StyleMohamad, Sharifah, Hemavathy Surikumaran, Muggundha Raoov, Tilagam Marimuthu, Kumuthini Chandrasekaram, and Puvaneswary Subramaniam. 2011. "Conventional Study on Novel Dicationic Ionic Liquid Inclusion with β-Cyclodextrin" International Journal of Molecular Sciences 12, no. 9: 6329-6345. https://doi.org/10.3390/ijms12096329
APA StyleMohamad, S., Surikumaran, H., Raoov, M., Marimuthu, T., Chandrasekaram, K., & Subramaniam, P. (2011). Conventional Study on Novel Dicationic Ionic Liquid Inclusion with β-Cyclodextrin. International Journal of Molecular Sciences, 12(9), 6329-6345. https://doi.org/10.3390/ijms12096329






