Micro Electromechanical Systems (MEMS) Based Microfluidic Devices for Biomedical Applications
Abstract
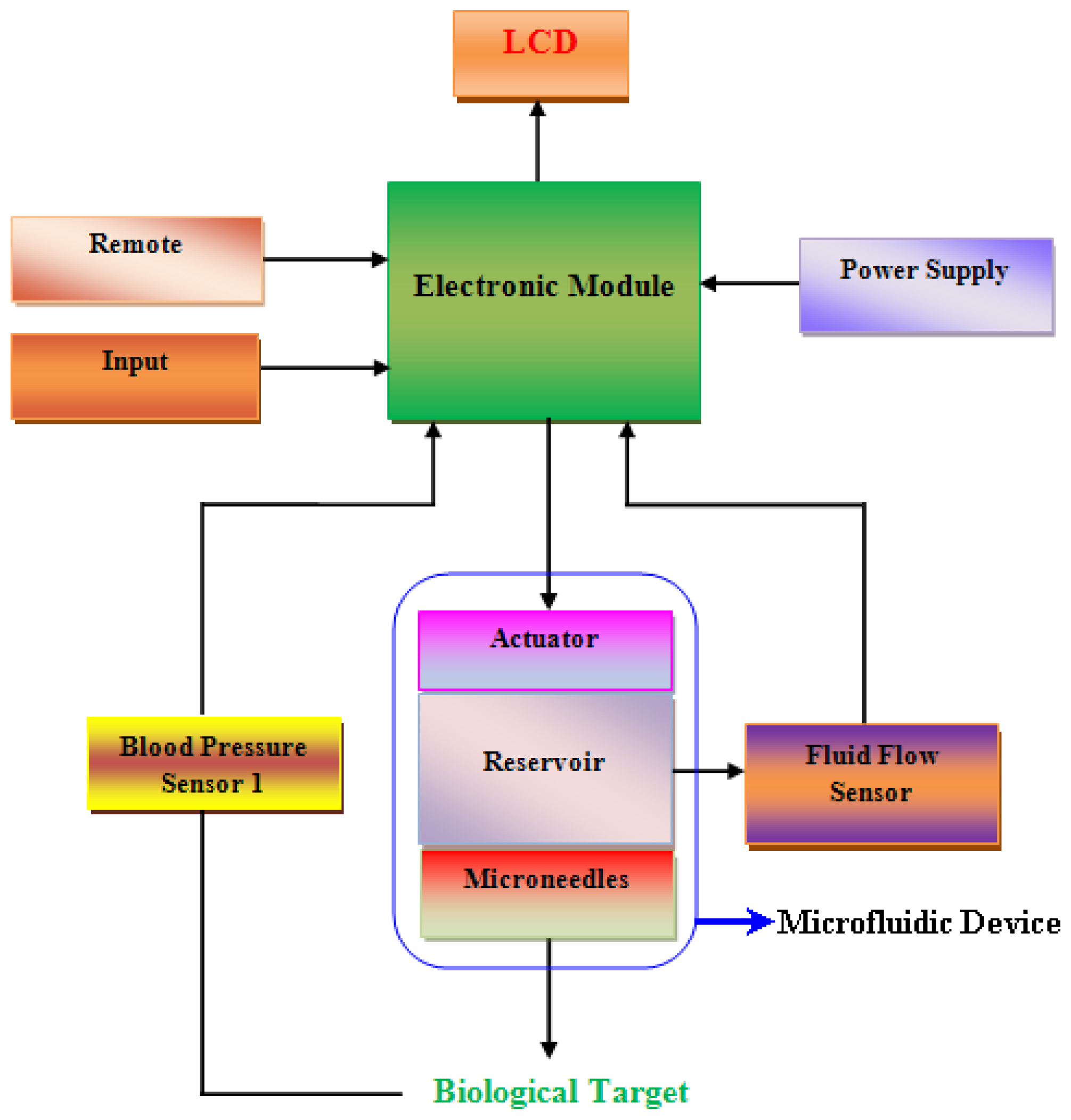
:1. Introduction
2. Micropumps
2.1. Design Specifications and Parameters of Micropumps
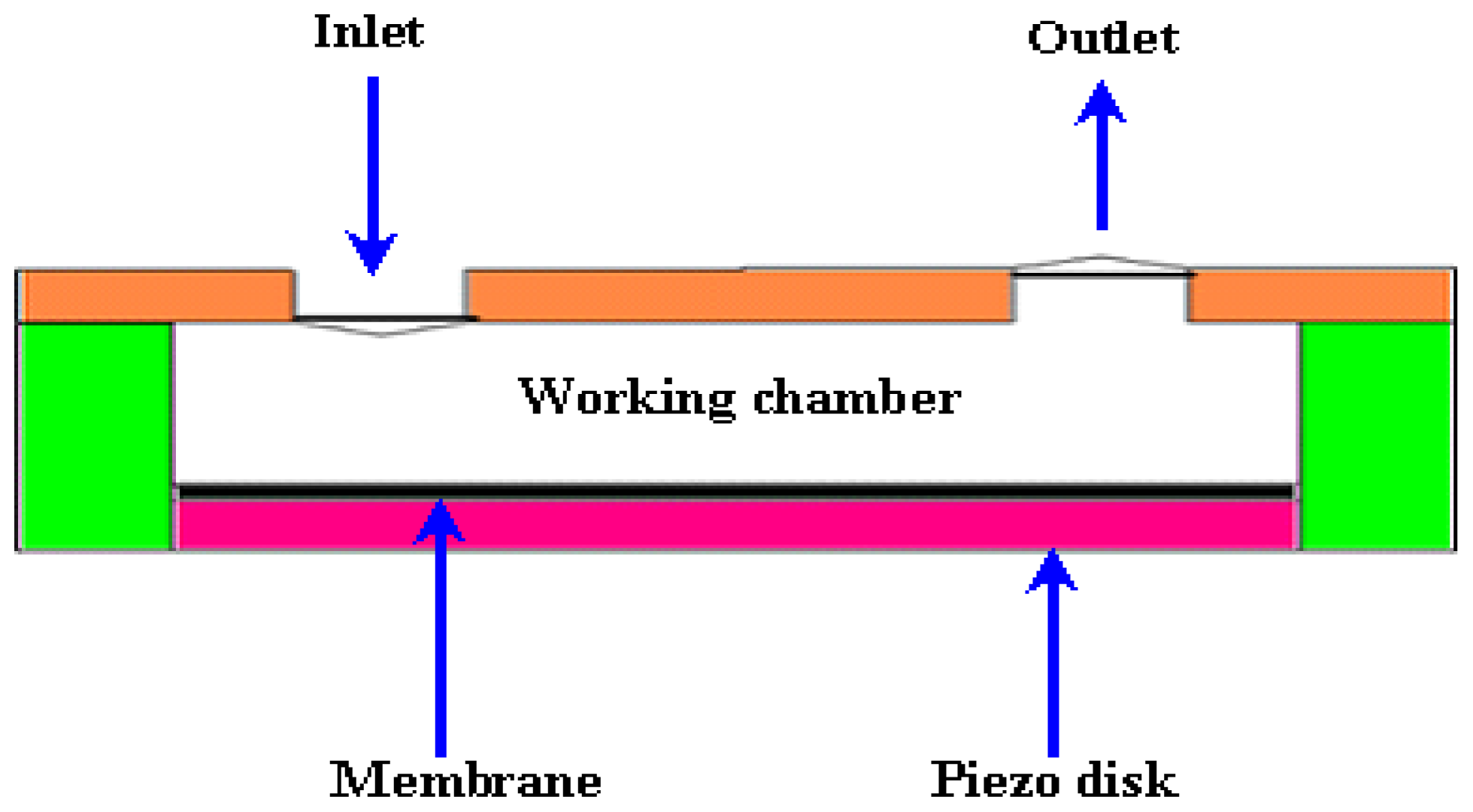
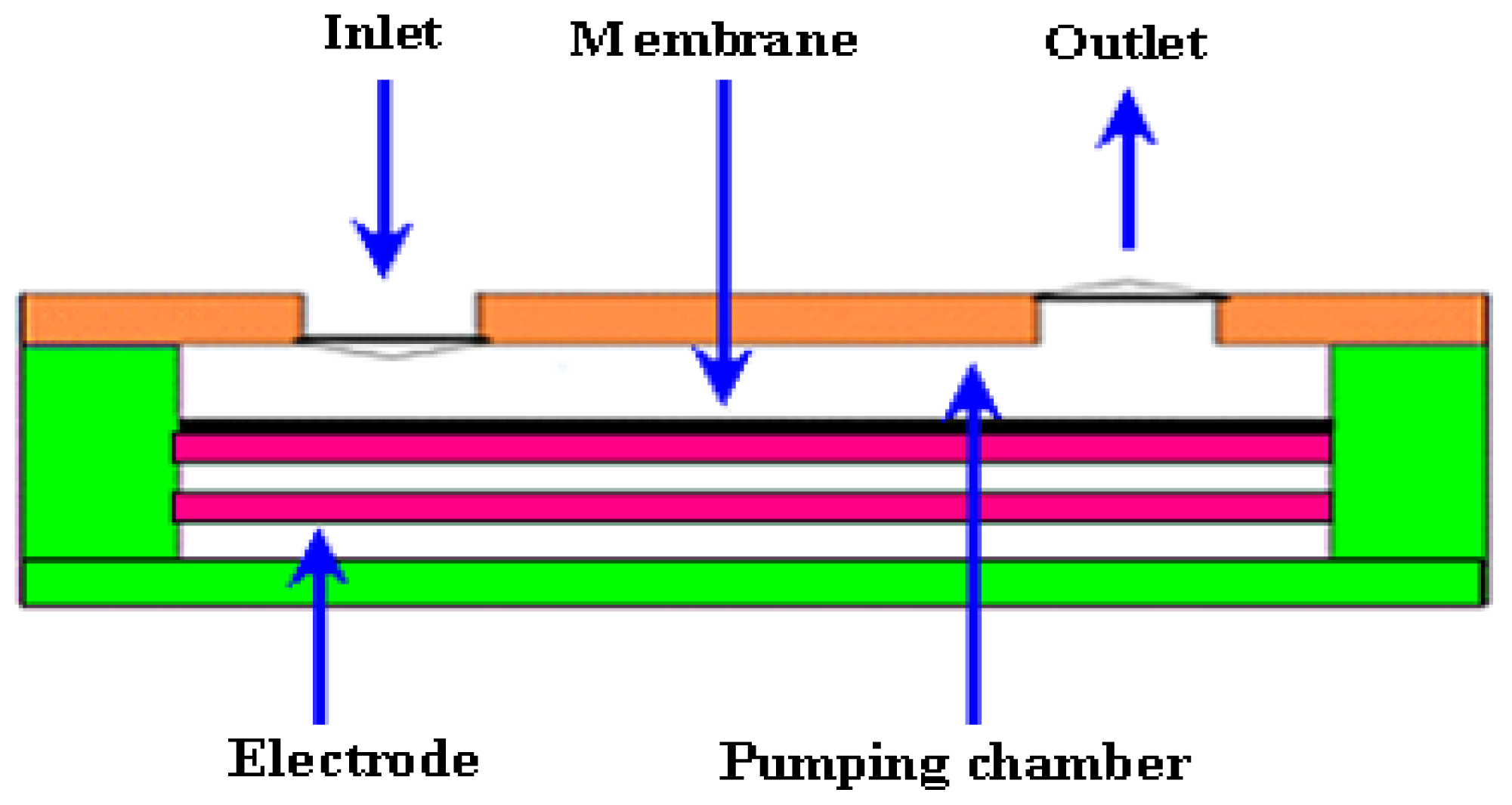
2.1.1. Actuator
2.1.2. Valves
2.1.3. Chamber or Reservoir
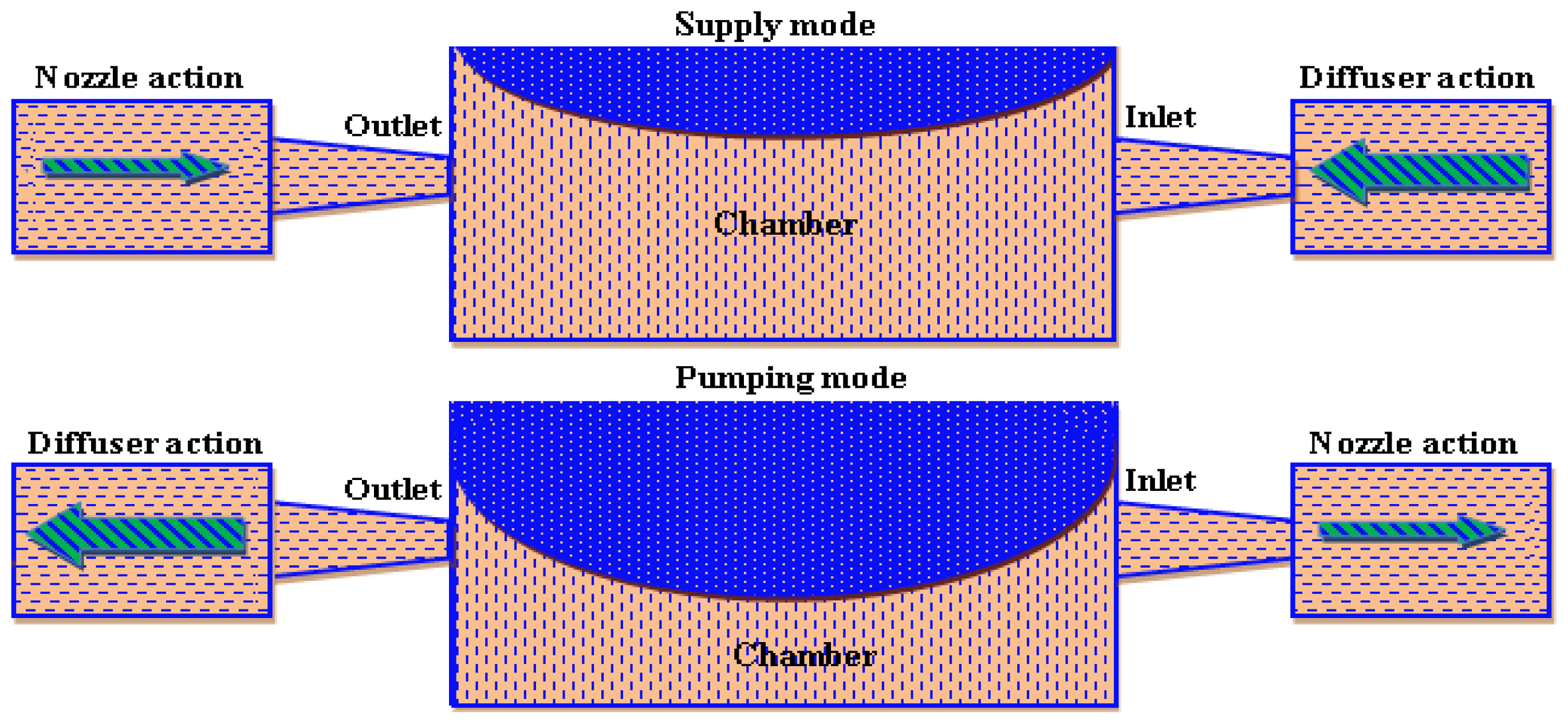
2.1.4. Nozzle/Diffuser Element
2.1.5. Pumping Parameters
2.2. Mechanical Micropumps
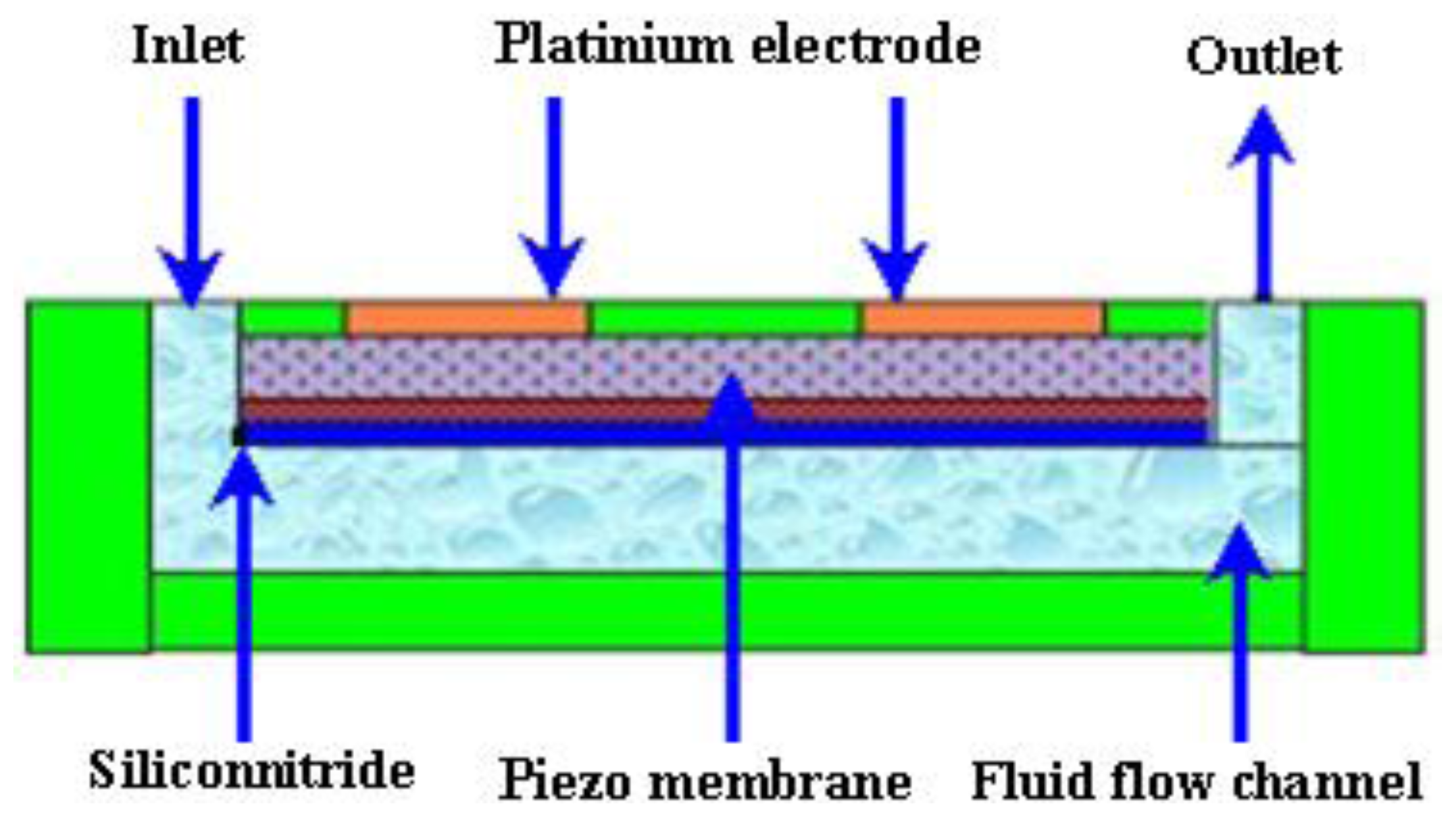
2.2.1. Piezoelectric Micropumps
2.2.2. Electrostatic Micropumps
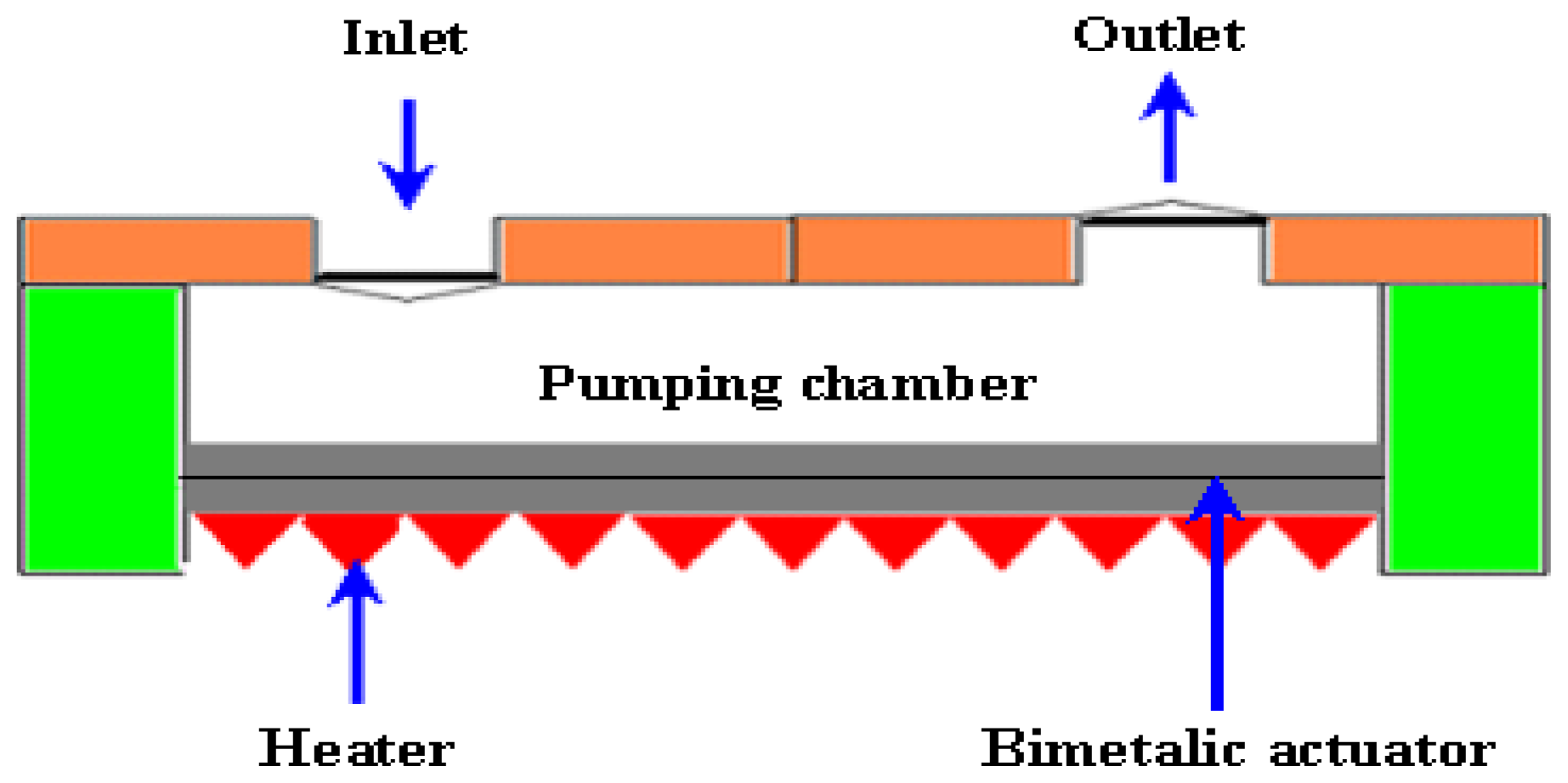
2.2.3. Thermopneuamtic Micropumps
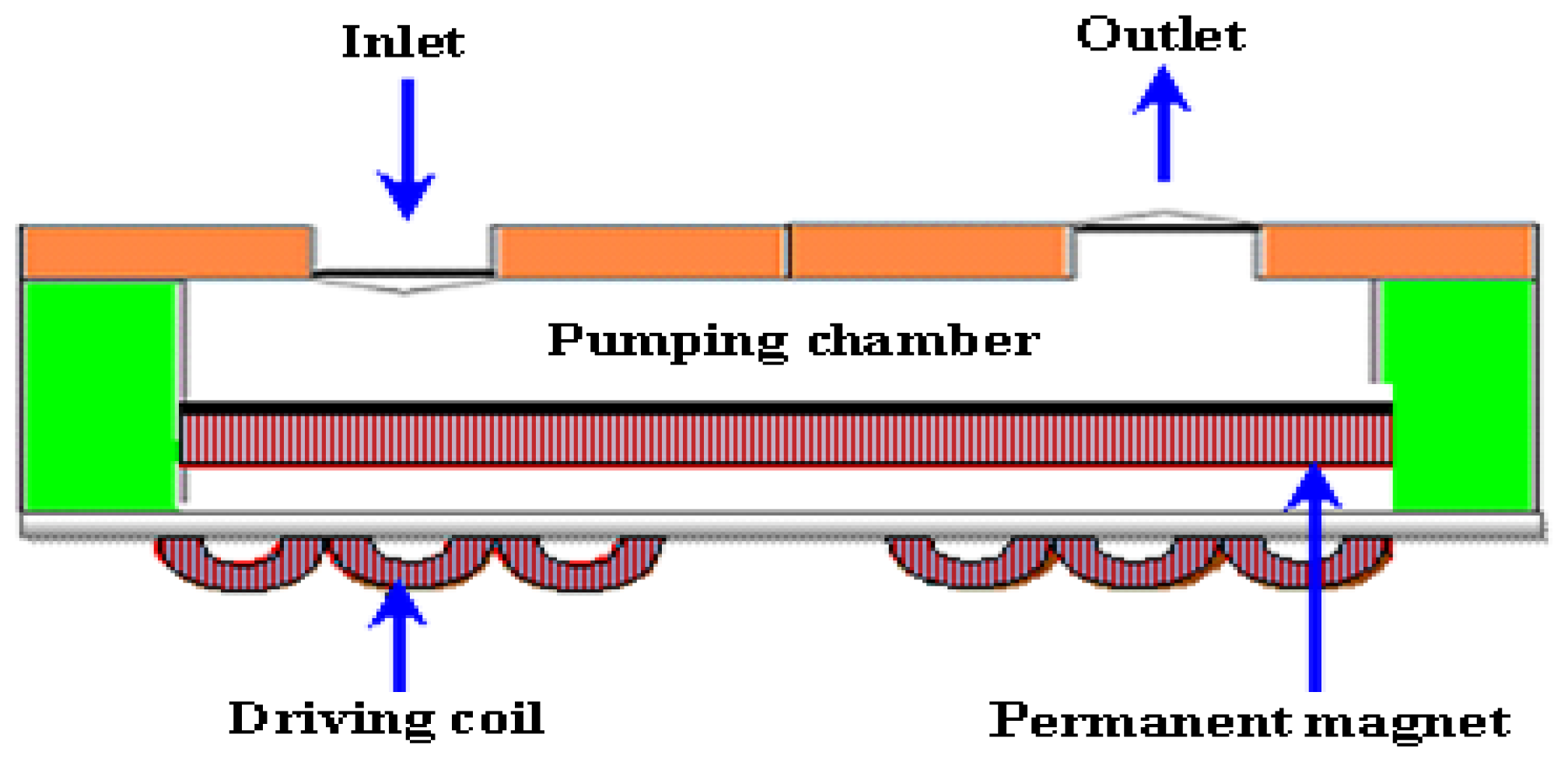
2.2.4. Electromagnetic Micropumps
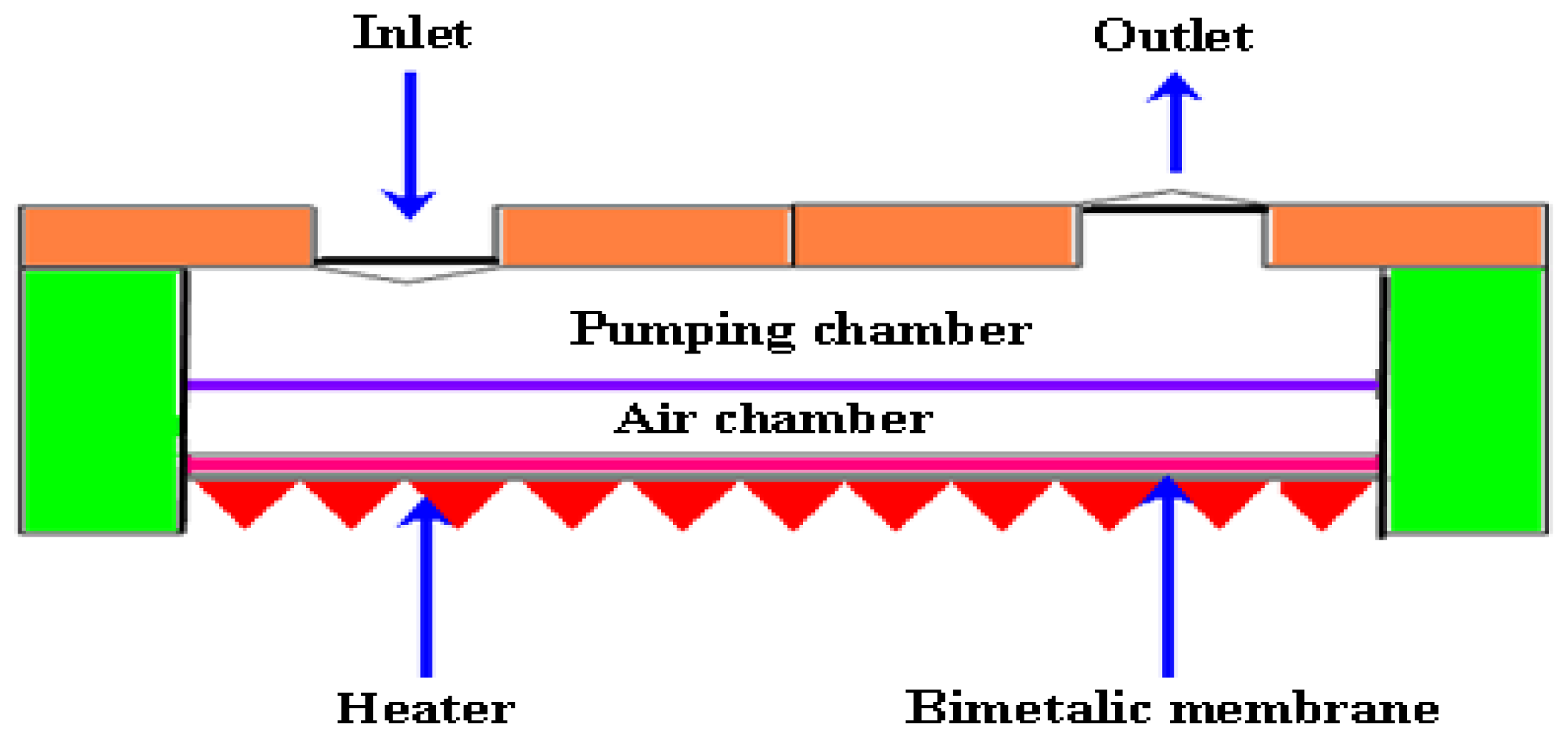
2.2.5. Bimetallic Micropumps
2.2.6. Ion Conductive Polymer Film (ICPF) Micropumps
2.2.7. Phase Change Micropumps
2.2.8. Shape Memory Alloy (SMA) Micropumps
2.3. Non-Mechanical Micropumps
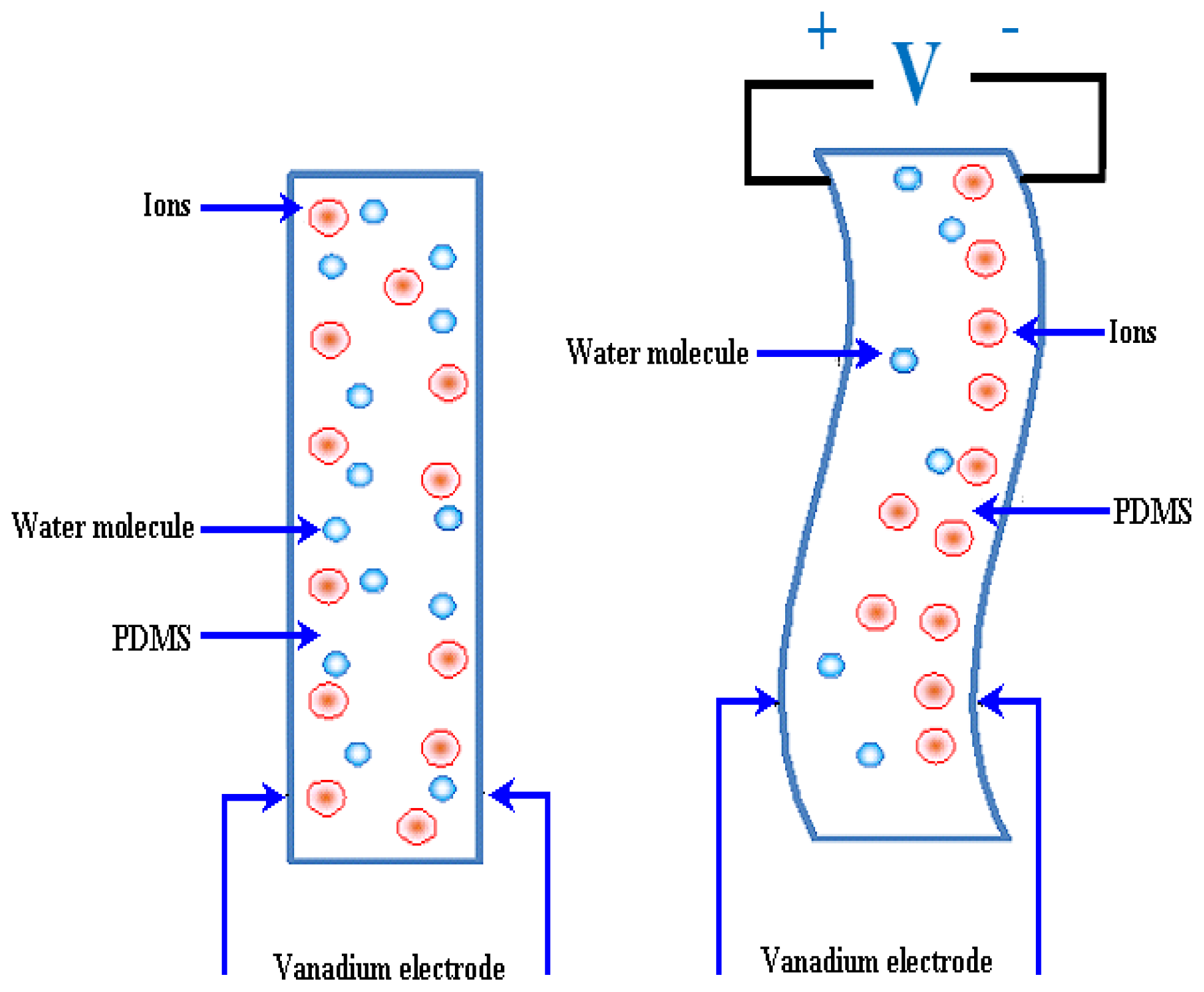
2.3.1. Electroosmotic (EO) Micropumps
2.3.2. Electrowetting (EW) Micropumps
2.3.3. Electrochemical Micropumps
2.3.4. Evaporation Micropumps
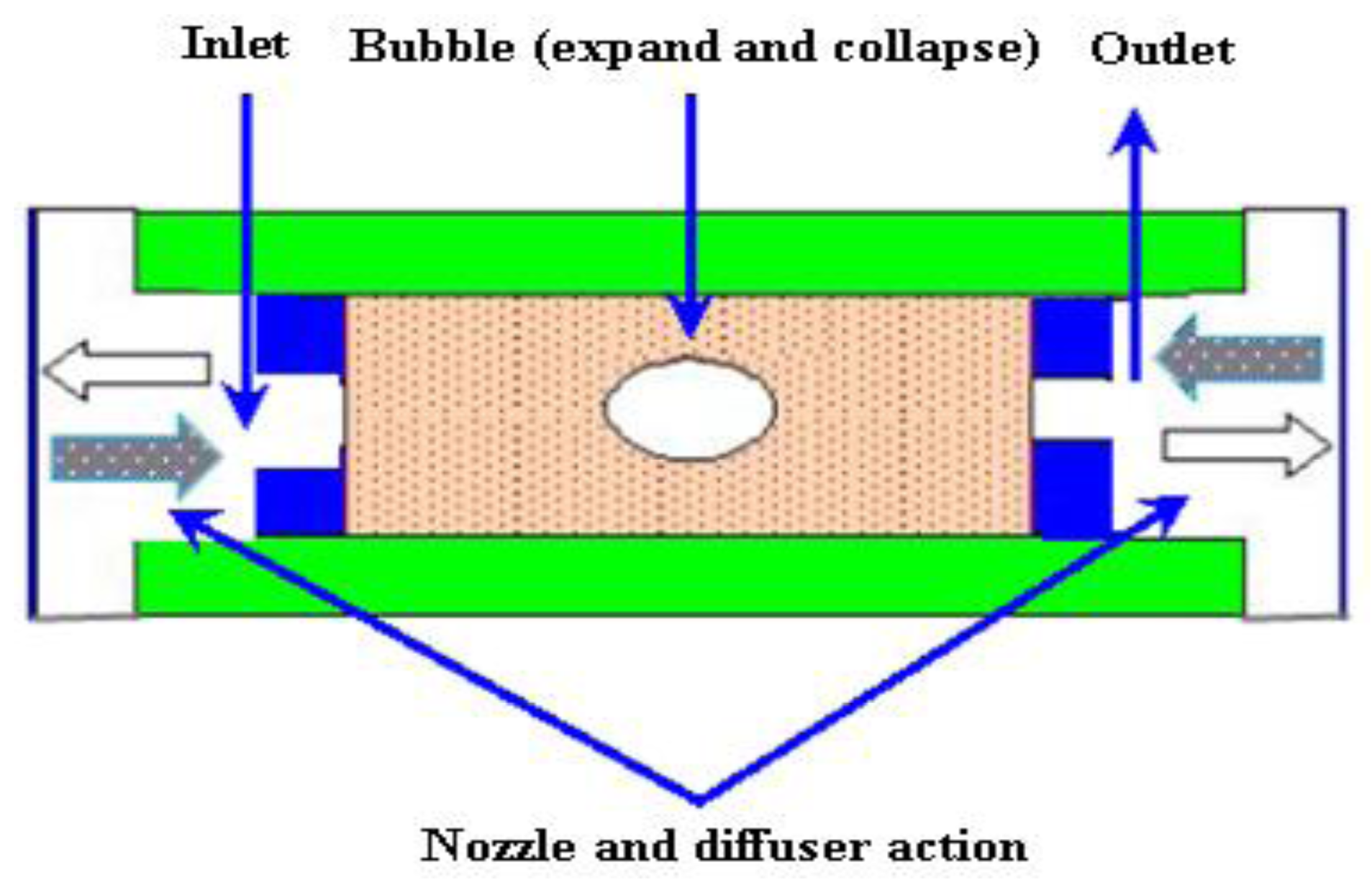
2.3.5. Bubble Micropumps
2.3.6. Magnetohydrodynamic (MHD) Micropumps
2.3.7. Flexural Planer Wave (FPW) Micropumps
2.3.8. Electrohydrodynamic (EHD) Micropumps
3. Microneedles
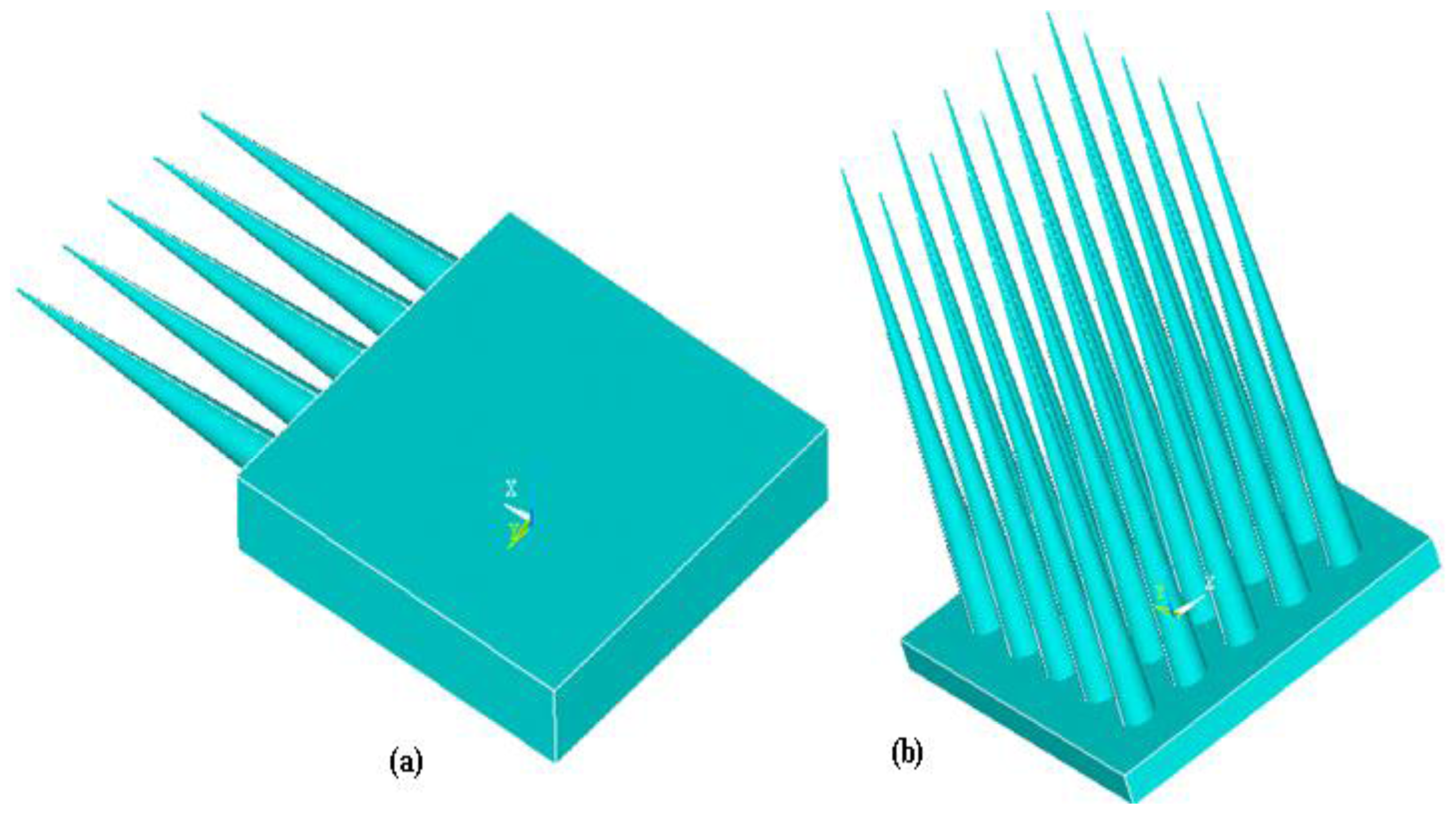
3.1. Categories of Microneedles
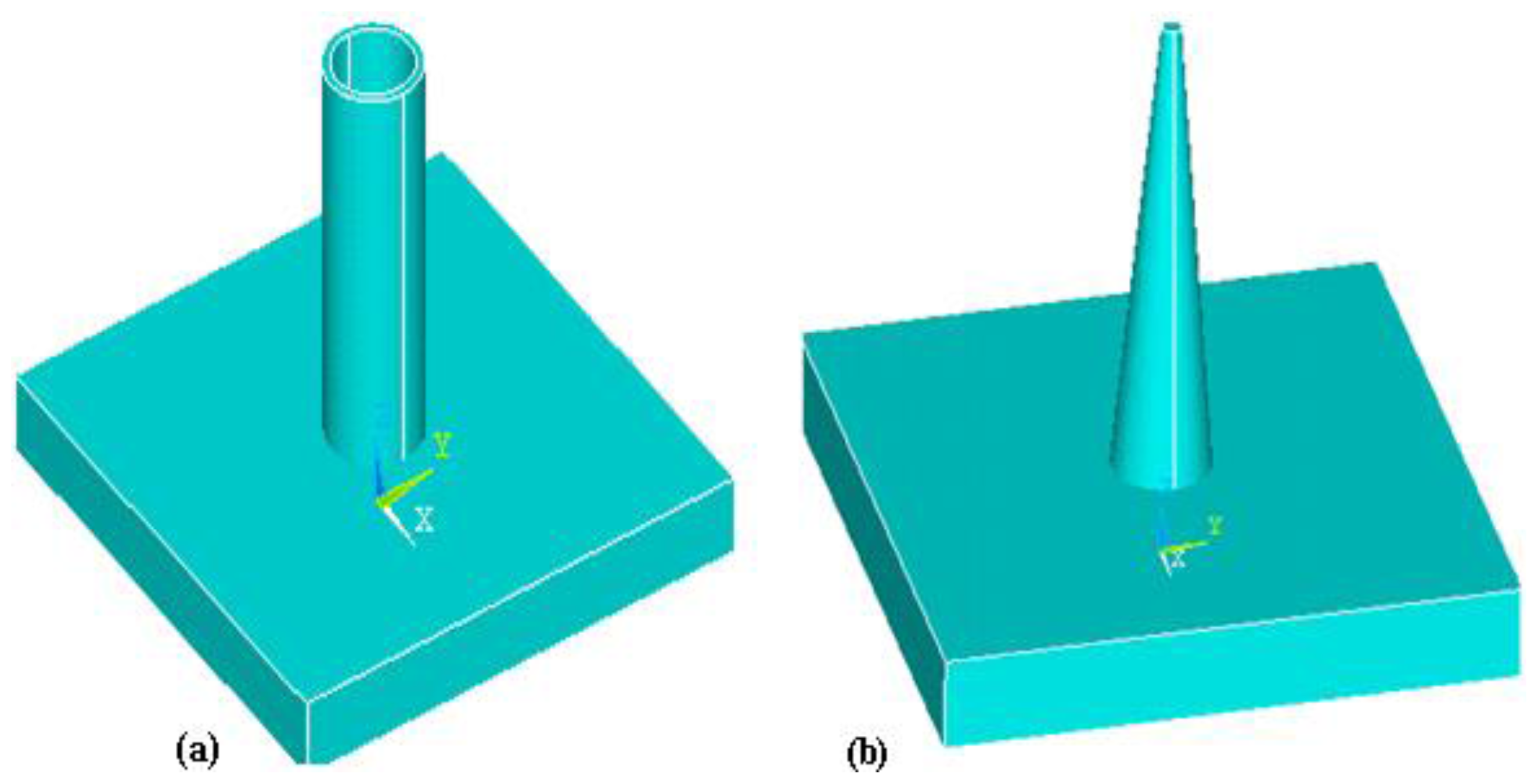
3.1.1. Structure of Microneedles
- In-plane microneedles
- Out-of-plane microneedles
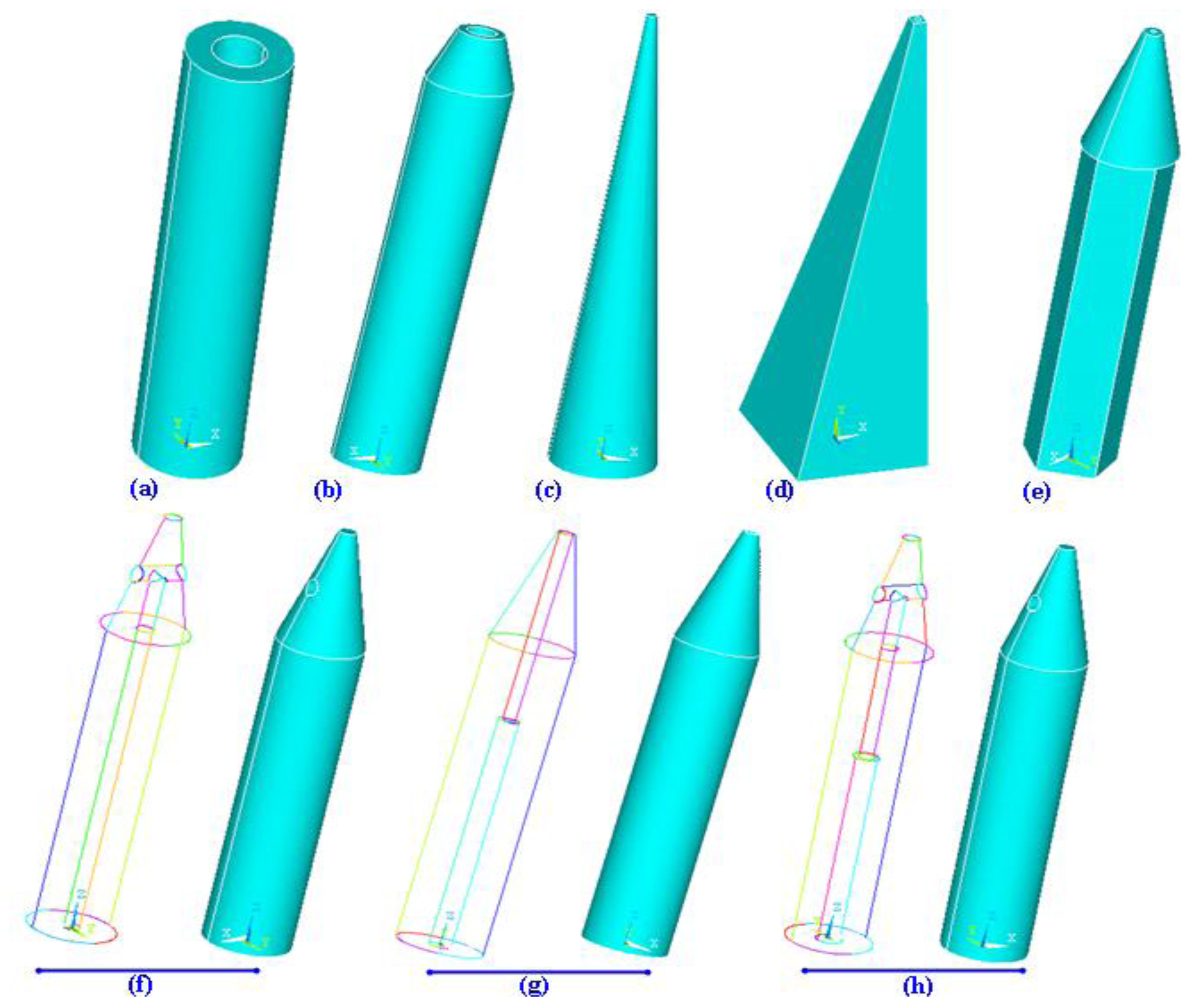
3.1.2. Shape of Microneedles
3.1.3. Materials Used for Microneedles
3.1.4. Microneedles Applications
3.2. Forces Experienced by Microneedles during Penetration
3.3. Fabrication of Microneedles
3.4. Microneedles Testing
4. Discussion
5. Challenges and Future Aspects
6. Conclusions
References
- Hsu, T. MEMS & Microsystem Design and Manufacturing, 1st ed; McGraw-Hill: New York, NY, USA, 2002. [Google Scholar]
- Hardt, S; Schonfeild, F. Microfluidic Technologies for Maniaturized Analysis System, 1st ed; Springer: Berlin, Germany, 2010. [Google Scholar]
- Ashraf, MW; Tayyaba, S; Afzulpurkar, N; Nisar, A. Fabrication and analysis of tapered tip silicon microneedles for mems based drug delivery system. Sens. Transducer 2010, 122, 158–173. [Google Scholar]
- Ashraf, MW; Tayyaba, S; Nisar, A; Afzulpurkar, N; Bodhale, DW; Lomas, T; Poyai, A; Tuantranont, A. Design, fabrication and analysis of silicon hollow microneedles for transdermal drug delivery system for Treatment of Hemodynamic Dysfunctions. Cardiovasc. Eng. J 2010, 3, 91–108. [Google Scholar]
- Prausnitz, MR; Langer, R. Transdermal drug delivery. Nat. Biotechnol 2008, 26, 1261–1268. [Google Scholar]
- Polla, DL. BioMEMS Applications in Medicine. Proceedings of International Symposium on Micromechatronics and Human Science, Nagoya, Japan, 9–12 September 2001; pp. 13–15.
- Shawgo, SR; Amy, C; Grayson, R; Yawen, L; Cima, JM. BioMEMS for drug delivery. Curr. Opin. Solid-State Mater. Sci 2002, 6, 329–334. [Google Scholar]
- Amy, C; Grayson, R; Shawgo, RS; Li, Y; Cima, MJ. Electronic MEMS for triggered delivery. Adv. Drug Deliv. Rev 2004, 56, 173–184. [Google Scholar]
- Staples, M; Daniel, K; Cima, MJ; Langer, R. Application of micro and nano electromechanical devices to drug delivery. Pharm. Res 2006, 23, 847–863. [Google Scholar]
- Laser, DJ; Santiago, JG. Areview of micropumps. J. Micromech. Microeng 2004, 14, 35–64. [Google Scholar]
- Woias, P. Micropumps-past, progress and future prospects. Sens. Actuators B 2005, 105, 28–38. [Google Scholar]
- Tsai, NC; Sue, CY. Review of MEMS based drug delivery and dosing systems. Sens. Actuators A 2007, 134, 555–564. [Google Scholar]
- Nisar, A; Afzulpurkar, N; Mahaisavariya, B; Tuantranont, A. MEMS-based micropumps in drug delivery and biomedical applications. Sens. Actuators B 2008, 130, 917–942. [Google Scholar]
- Amirouche, F; Zhou, Y; Johnson, T. Current micropump technologies their biomedical applications. Microsyst. Technol 2009, 15, 647–666. [Google Scholar]
- Grayson, AR; Shawgo, RS; Johnson, AM; Flynn, NT; Yawen, L; Cima, MJ; Langer, R. A BioMEMS review: MEMS technology for physiologically integrated devices. Proc. IEEE 2004, 92, 6–21. [Google Scholar]
- Karman, S; Ibrahim, F; Soin, N. A review of MEMS drug delivery in medical application, Biomed 06. Proc. IFMBE 2007, 15, 312–315. [Google Scholar]
- Bao-jian, X; Qing-hui, J; Jian-long, Z. An review of MEMS-Based microneedles technology developments. Micronanoelectron Technol 2005, 4. 1671-4776.0.2005-04-002.. [Google Scholar]
- Khanna, P; Storm, JA; Malone, JI; Bhansali, S. Microneedle-Based Automated Therapy for Diabetes Mellitus. J. Diabetes Sci. Technol 2008, 2, 1122–1129. [Google Scholar]
- Sachdeva, V; Banga, AK. Microneedles and their applications. Recent Pat. Drug Deliv. Formul 2011, 5, 95–132. [Google Scholar]
- Stemme, E; Stemme, G. A valveless diffuser/nozzle based fluid pump. Sens. Actuators A 1993, 39, 159–167. [Google Scholar]
- White, FM. Fluid Mechanics, International Student Edition; McGraw Hill Inc: Columbus, OH, USA, 1979; pp. 161–162. [Google Scholar]
- Van Lintel, HTG; Van de Pol, FCM; Bouwstra, S. A piezoelectric micropump based on micromachining of silicon. Sens. Actuators A 1988, 15, 153–168. [Google Scholar]
- Esashi, M; Shoji, S; Nakano, A. Normally close microvalve and micropump fabricated on a silicon wafer. Proceedings of IEEE Micro Electro Mechanical Systems, An Investigation of Micro Structures, Sensors, Actuators Machines and Robots, Salt Lake City, UT, USA, 20–22 February 1989; pp. 29–34.
- Olsson, A; Stemme, G; Stemme, E. A valve-less planar fluid pump with two pump chambers, Sens. Actuators A 1995, 47, 549–556. [Google Scholar]
- Koch, M; Harris, N; Evans, AGR; White, NM; Brunnschweiler, A. A novel micromachined pump based on thick film piezoelectric actuation. Sens. Actuators A 1998, 70, 98–103. [Google Scholar]
- Schabmueller, CGJ; Koch, M; Mokhtari, ME; Evans, AGR; Brunnschweiler, A; Sehr, H. Self-aligning gas/liquid micropump. J. Micromech. Microeng 2002, 12, 420–424. [Google Scholar]
- Feng, GH; Kim, ES. Piezoelectrically actuated dome-shaped diaphragm micropump. J. Microelectromech. Syst 2005, 14, 92–199. [Google Scholar]
- Geipel, A; Doll, A; Goldschmidtboing, F; Jantscheff, P; Esser, N; Massing, U; Woias, P. Istanbul, Turkey, 22–26 January 2006; 2006; pp. 786–789.
- Trenkle, F; Haeberle, S; Zengerle, R. Normally-closed peristaltic micropump with re-usable actuator and disposable fluidic chip. Procedia Chem 2009, 1, 1515–1518. [Google Scholar]
- Johari, J; Yunas, J; Yeopmajlis, B. Piezoeletric Micropump for Drug Delivery System Fabricated Using Two Optical Masks. Adv. Mat. Res 2009, 74, 279–282. [Google Scholar]
- Wang, C; Leu, T; Sun, J. Asymmetrical flow effect applied to pumping performance of simple duct channel. Sens. Actuators A 2009, 155, 203–209. [Google Scholar]
- Ali, MY; Kuang, C; Khan, J; Wang, G. A dynamic piezoelectric micropumping phenomenon. Microfluid. Nanofluid 2010, 9, 385–396. [Google Scholar]
- Liu, G; Shen, C; Yang, Z; Cai, X; Zhang, H. A disposable piezoelectric micropump with high performance for closed-loop insulin therapy system. Sens. Actuators A 2010, 163, 291–296. [Google Scholar]
- Judy, JW; Tamagawa, T; Polla, DL. Surface-machined Micromechanical Membrane Pump. Proceedings of the IEEE Micro Electro Mechanical systems, Nara, Japan, 30 January–2 February 1991; pp. 182–186.
- Zengerle, R; Richter, A; Sandmaier, H. A Micro Membrane Pump with Electrostatic Actuation. Proceedings of Micro Electro Mechanical Systems 1992, An Investigation of Micro Structures, Sensors, Actuators, Machines and Robot IEEE, Travemunde, Germany, 4–7 February 1992; pp. 19–24.
- Cabuz, C; Herb, WR; Cabuz, EI; Lu, ST. The dual diaphragm pump. Proceedings of the 14th IEEE International Conference on Micro Electro Mechanical Systems, Interlaken, Switzerland, 21–25 January 2001; pp. 519–522.
- Machauf, A; Nemirovsky, Y; Dinnar, U. A membrane micropump electrostatically actuated across the working fluid. J. Micromech. Microeng 2005, 15, 2309–2316. [Google Scholar]
- Astle, AA; Kim, HS; Bernal, LP; Najafi, K; Washabaugh, PD. Theoretical and experimental performance of a high frequency gas micropump. Sens. Actuators A 2006, 134, 245–256. [Google Scholar]
- Lee, S; Yee, SY; Besharatian, A; Kim, H; Bernal, LP; Najafi, K. Adaptive gas pumping by colntrolled timing of active microvalves in peristaltic micropumps. Proceedings of Transducers, Denver, CO, USA, 21–25 June 2009.
- Liu, W. Changsha, China, 11–12 May 2010.
- Lil, L; Zhu, R; Zhou, Z; Ren, J. Modeling of a Micropump Membrane with Electrostatic Actuator. Proceedings of 2nd International Conference on Advanced Computer Control (ICACC), Shenyang, China, 27–29 March 2010.
- Van de Pol, FCM; van Lintel, HTG; Elwenspoek, M; Fluitman, JHJ. A thermopneumatic micropump based on microengineering techniques. Sens. Actuators A 1990, 21, 198–202. [Google Scholar]
- Jeong, OC; Yang, SS. Fabrication and test of a thermopneumatic micropump with a corrugated p diaphragm, Sens. Actuators 2000, 83, 249–255. [Google Scholar]
- Cooney, CG; Towe, BC. A thermopneumatic dispensing micropump. Sens. Actuators A 2004, 116, 519–524. [Google Scholar]
- Kim, JH; Na, KH; Kang, CJ; Kim, YS. A disposable thermopneumatic actuated micropump stacked with PDMS layers and ITO coated glass, Sens. Actuators A 2005, 120, 365–369. [Google Scholar]
- Jeong, OC; Konishi, S. Fabrication of a peristaltic micro pump with novel cascaded actuators. J. Micromech. Microeng 2008, 18, 025022. [Google Scholar]
- Chia, BT; Liao, H; Yang, Y. A novel thermo-pneumatic peristaltic micropump with low temperature elevation on working fluid. Sens. Actuators A 2010, 165, 86–93. [Google Scholar]
- Tan, HY; Loke, WK; Nguyen, N. A reliable method for bonding polydimethylsiloxane (PDMS) to polymethylmethacrylate (PMMA) and its application in micropumps. Sens. Actuators B 2010, 151, 133–139. [Google Scholar]
- Zheng, W; Ahn, CH. A Bi-directional Magnetic Micropump on a Silicon Wafer. Proceedings of Technical Digest Solid-State Sensor and Actuator Workshop 1996, Hilton Head Island, SC, USA, 3–6 June 1996.
- Bohm, S; Olthuis, W; Bergveld, P. A plastic micropump constructed with conventional techniques and materials. Sens. Actuators A 1999, 77, 223–228. [Google Scholar]
- Gong, Q; Zhou, Z; Yang, Y; Wang, X. Design, optimization and simulation on microelectromagnetic pump. Sens. Actuators A 2000, 83, 200–207. [Google Scholar]
- Yamahata, C; Lotto, C; Al Assaf, E; Gijs, MAM. A PMMA valveless micropump using electromagnetic actuation. Microfluid. Nanofluid 2005, 1, 197–207. [Google Scholar]
- Su, Y; Chen, W; Cui, F; Zhang, W. Analysis and fabrication process of an electromagnetically actuated valveless micropump with two parallel flexible diaphragms. Proc. IMechE 2005, 219, 1007–1014. [Google Scholar]
- Balaji, G; Singh, S; Ananthasuresh, GK. Electro-magnetically Actuated Minute Polymer Pump Fabricated using Packaging Technology. J. Phys 2006, 34, 258–263. [Google Scholar]
- Yu-feng, SU; Wen-yuan, C; Feng, C; Wei-ping, Z. Design and fabrication process of electromagnetically actuated valveless micropump with two parallel flexible diaphragms. J. Shanghai Univ 2007, 11, 79–83. [Google Scholar]
- Shen, M; Yamahata, C; Gijs, MAM. Miniaturized PMMA ball-valve micropump with cylindrical electromagnetic actuator. Microelectr. Eng 2008, 85, 1104–1107. [Google Scholar]
- Al-Halhouli, AT; Kilani, MI; Büttgenbacha, S. Development of a novel electromagnetic pump for biomedical applications. Sens. Actuators A 2010, 162, 172–176. [Google Scholar]
- Zhan, C; Lo, T; Liu, LP. A silicon membrane micropump with integrated bimetallic actuator. Chin. J. Electron 1996, 5, 29–35. [Google Scholar]
- Zou, JX; Ye, YZ; Zhou, Y; Yang, Y. A Novel Thermally Actuated Silicon Micropump. Proceedings of International Symposium on Micromechatronics and Human Science, Nagoya, Japan, 5–8 October 1997.
- Pang, J; Zou, Q; Tan, Z; Qian, X; Liu, L; Li, Z. The Study of Single Chip Integrated Microfluidic System. Proceedings of 5th International Conference on Solid State and Integrated Circuit Technology, Beijing, China, 21–23 October 1998; pp. 895–898.
- Yang, Y; Zhaoying, Z; Xiongying, Y; Xiaoning, J. Bimetallic Thermally Actuated Micropump. Proceedings of International Mechnical Engineering Congross and Exposition (ASME 1996), Atlanta, GA, USA, 17–22 November 1996.
- Guo, S; Nakamura, T; Fukuda, T; Oguro, K. Design and Experiments of Micropump Using ICPF Actuator. Proceedings of 7th International IEEE Symposium on Micro Machine and Human Science, Nagoya, Japan, 2–4 October 1996; pp. 235–240.
- Tadokoro, S; Yamagami, S; Ozawa, M. Soft Micromanipulation Device with Multiple Degrees of Freedom Consisting of High Polymer Gel Actuators. Proceedings of IEEE International Conference on Microelectromechanical Systems, Orlando, FL, USA, 17–21 January 1999; pp. 37–42.
- Guo, S; Asaka, K. Polymer Based New Type of Micropump for Biomedical Applications. Proceedings of IEEE Conference on Robotics & Automation, Taipei, Taiwan, 14–19 September 2003; pp. 1830–1835.
- Nguyen, TT; Goo, NS; Nguyen, VK; Yoo, Y; Park, S. Design, fabrication, and experimental characterization of a flap valve IPMC micropump with a flexibly supported diaphragm. Sens. Actuators A 2008, 141, 640–648. [Google Scholar]
- Chen, Z; Kwon, K; Tan, X. Integrated IMPC/PVDF sensory actuator and its validation in feedback control. Sens. Actuators A 2008, 144, 231–241. [Google Scholar]
- Fang, T; Tan, X. A novel diaphragm micropump actuated by conjugated polymer petals: Fabrication, modeling, and experimental results. Sens. Actuators A 2010, 158, 121–131. [Google Scholar]
- Sim, WY; Yoon, HJ; Jeong, OC; Yang, SS. A phase change type of micropump with aluminum flap valves. J. Micromech. Microeng 2003, 13, 286–294. [Google Scholar]
- Boden, R; Lehto, M; Simu, U; Thornell, G; Hjort, K; Schweitz, JA. A Polymeric Paraffin Micropump with Active Valves for High Pressure Microfluidics. Proceedings of 13th International Conference on Solid State Sensors, Actuators and Microsystems, Seoul, Korea, 5–9 June 2005; pp. 201–204.
- Sim, WY; Lee, SW; Yang, SS. The Fabrication and Test of a Phase-change Type Micropump. 2008. Available online: http://click.ndsl.kr/servlet/LinkingFullTextView?service_code=04&dbt=JAKO&cn=JAKO200011919587765 accessed on 20 February 2011.
- Benard, WL; Kahn, H; Heuer, AH; Huff, MA. Thin-film shapememory alloy actuated micropumps. J. Microelectromech. Syst 1998, 7, 245–251. [Google Scholar]
- Makino, E; Mitsuya, T; Shibata, T. Fabrication of TiNi shape memory micropump. Sens. Actuators A 2001, 88, 256–262. [Google Scholar]
- Xu, D; Wang, L; Ding, G; Zhou, Y; Yu, A; Cai, B. Characteristics and fabrication of NiTi/Si diaphragm micropump. Sens. Actuators A 2001, 93, 87–92. [Google Scholar]
- Shuxiang, G; Fukuda, T. SMA Actuator Based Novel Type of Micropump for Biomedical Application. Proceedings of IEEE International Conference Robotics and Automation, Takamatsu, Japan, 26 April–1 May 2004; 2, pp. 1616–1621.
- Zhang, HJ; Qiu, CJ. A TiNiCu thin film micropump made by magnetron Co-sputtered method. Mat. Trans 2006, 47, 532–535. [Google Scholar]
- Setiawan, MA. The performance evaluation of SMA Spring as actuator for gripping manipulation. J. Teknik Elektro 2007, 7, 110–120. [Google Scholar]
- Zhu, M; Kirby, P; Wacklerle, M; Herz, M; Richter, M. Optimization design of multi-material micropump using finite element method. Sens. Actuators A 2009, 149, 130–135. [Google Scholar] [Green Version]
- Kang, J; Auner, GW. Simulation and verification of a piezoelectrically actuated diaphragm for check valve micropump design. Sens Actuators A 2011, in press. [Google Scholar]
- Shen, M; Dovat, L; Gijs, MAM. Magnetic active-valve micropump actuated by a rotating magnetic assembly. Sens. Actuators B 2009, 154, 52–58. [Google Scholar]
- Teymoori, MM; Abbaspour-Sani, E. Design and simulation of a novel electrostatic peristaltic micromachined pump for drug delivery applications. Sens. Actuators A 2005, 117, 222–229. [Google Scholar]
- Zeng, S; Chen, CH; Mikkelsen, JC; Santiago, JG. Fabrication and characterization of electroosmotic micropumps. Sens. Actuators B 2001, 79, 107–114. [Google Scholar]
- Takemori, Y; Horiike, S; Nishimoto, T; Nakanishi, H; Yoshida, T. High Pressure Electroosmotic Pump Packed with Uniform Silica Nanosphers. Proceedings of 13th International Conference on Solid State Sensors, Actuators and Microsystems, Seoul, Korea, 5–9 June 2005.
- Hu, JS; Chao, CYH. Numerical study of electroosmotic (EO) flow in microfabricated EO pump with overlapped electrical double layer (EDL). Int. J. Refrig 2007, 30, 290–298. [Google Scholar]
- Good, BT; Bowman, CN; Davis, RT. A water-activated pump for portable microfluidic applications. J. Colloid Interface Sci 2007, 305, 239–249. [Google Scholar]
- Ryu, WH; Huang, Z; Prinz, FB; Goodman, SB; Fasching, R. Biodegradable microosmotic pump for long term and controlled release of basic fibroblast growth factor. J. Control. Release 2007, 124, 98–105. [Google Scholar]
- Yairi, M; Richter, C. Massively parallel microfluidic pump. Sens. Actuators A 2007, 137, 350–356. [Google Scholar]
- Borowsky, J; Lu, Q; Collins, GE. High pressure electroosmotic pump based on a packed bed planar microchip. Sens. Actuators B 2008, 131, 333–339. [Google Scholar]
- Wang, X; Wang, S; Gendhar, B; Cheng, C; Byun, CK; Li, G; Zhao, M; Liu, S. Electroosmotic pumps for microflow analysis. Trends Anal. Chem 2009, 28, 64–74. [Google Scholar]
- Yun, KS; Cho, IJ; Bu, JU; Kim, CJ; Yoon, E. A surface tension driven micropump for low voltage and low power operations. J. MEMS 2002, 11, 454–461. [Google Scholar]
- Hoshino, K; Triteyaprasert, S; Matsumoto, K; Shimoyama, I. Electrowetting-based pico-liter liquid actuation in a glass-tube microinjector. Sens. Actuators A 2004, 114, 473–477. [Google Scholar]
- Colgate, ED; Matsumoto, H. An investigation of electrowetting based microactuation. J. Vac. Sci. Technol. A 1990, 4, 3625–3633. [Google Scholar]
- Chang, J; Choi, DY; Han, S; Pak, JJ. Driving characteristics of the electrowetting-on-dielectric device using atomic-layer-deposited aluminum oxide as the dielectric. Microfluid. Nanofluid 2010, 8, 269–273. [Google Scholar]
- Suzuki, H; Yoneyama, R. Areversible electrochemical nanosyringe pump and some considerations to realize low power consumption. Sens. Actuators B 2002, 86, 242–250. [Google Scholar]
- Suzuki, H; Yoneyama, R. Integrated microfluidic system with electrochemically actuated on-chip pumps and valves. Sens. Actuators B 2003, 96, 38–45. [Google Scholar]
- Yoshimi, Y; Shinoda, K; Mishima, M; Nakao, K; Munekane, K. Development of an artificial synapse using an electrochemical micropump. J. Artif. Organs 2004, 7, 210–215. [Google Scholar]
- Kim, JH; Laua, KT; Shepherd, R; Wu, Y; Wallace, G; Diamond, D. Performance characteristics of a polypyrrole modified polydimethylsiloxane (PDMS) membrane based microfluidic pump. Sens. Actuators A 2008, 148, 239–244. [Google Scholar]
- Effenhauser, CS; Harttig, H; Kramer, P. An evaporation-based disposable micropump concept for continuous monitoring applications. Biomed. Microdev 2002, 4, 27–33. [Google Scholar]
- Namasivayam, V; Larson, RG; Burke, DT; Burns, MA. Transpirationbased micropump for delivering continuous ultra low flow rates. J. Micromech. Microeng 2003, 13, 261–271. [Google Scholar]
- Guan, Y; Xua, Z; Dai, J; Fang, Z. The use of a micropump based on capillary and evaporation effects in a microfluidic flow injection chemiluminescence system. Talanta 2006, 68, 1384–1389. [Google Scholar]
- Heuck, F; Hug, T; Akiyama, T; Frederix, PLTM; Engel, A; Meister, A; Heinzelmann, H; de Rooij, NF; Staufer, U. Evaporation based micro pump integrated into a scanning force microscope probe. Microelectr. Eng 2008, 85, 1302–1305. [Google Scholar]
- Tsai, JH; Lin, L. A Thermal Bubble Actuated Micro Nozzle-diffuser Pump. Proceedings of 14th IEEE International Conference Microelectromechanical Systems, Interlaken, Switzerland, 21–25 January 2001; pp. 409–412.
- Tsai, JH; Lin, L. Active microfluidic mixture and gas bubble filter driven by thermal bubble micropump. Sens Actuators A 2002, 97-98, 665–671. [Google Scholar]
- Lew, KSF; Klaseboer, E; Khoo, BC. A collapsing bubble-induced micropump: An experimental study. Sens. Actuators A 2007, 133, 161–172. [Google Scholar]
- Jung, JY; Kwak, HY. Fabrication and testing of bubble powered micropumps using embedded microheater. Microfluid. Nanofluid 2007, 3, 161–169. [Google Scholar]
- Cheng, CM; Liu, CH. An Electrolysis-Bubble-Actuated micropump based on the roughness gradient design of hydrophobic surface. J. Microelectromech. Syst 2007, 16, 1095–1105. [Google Scholar]
- Chan, SC; Chen, CR; Liu, CH. A bubble-activated micropump with high-frequency flow reversal. Sens. Actuators A 2010, 163, 501–509. [Google Scholar]
- Jang, J; Lee, SS. Theoretical and experimental study of MHD (magnetohydrodynamic) micropump. Sens. Actuators A 2000, 80, 84–89. [Google Scholar]
- Zhong, J; Yi, M; Bau, HH. Magneto Hydrodynamic (MHD) pump fabricated with ceramic tapes. Sens. Actuators A 2002, 96, 59–66. [Google Scholar]
- Eijkel, JCT; Dalton, C; Hayden, CJ; Burt, JPH; Manz, A. A circular ac magnetohydrodynamic micropump for chromatographic applications. Sens. Actuators B 2003, 92, 215–221. [Google Scholar]
- Patel, V; Kassegne, SK. Electroosmosis and thermal effects in Magnetohydrodynamic (MHD) micropumps using 3D MHD equations. Sens. Actuators B 2007, 122, 42–52. [Google Scholar]
- Duwairi, HM; Abdullah, M. Numerical Computation of Fluid Flow in a Magnetohydrodynamic Micropump. Turkish J. Eng. Env. Sci 2008, 32, 1–5. [Google Scholar]
- Kang, H; Choi, B. Development of the MHD micropump with mixing function. Sens. Actuators A 2010, 165, 439–445. [Google Scholar]
- Moroney, RM; White, RM; Howe, RT. Microtransport induced by ultrasonic lamb waves. Appl. Phys. Lett 1991, 59, 774–776. [Google Scholar]
- Nguyen, NT; White, RM. Design and optimization of an ultrasonic flexural plate wave micropump using numerical simulation. Sens. Actuators A 1999, 77, 229–236. [Google Scholar]
- Nguyen, NT; Meng, AH; Black, J; White, RM. Integrated flow sensor for insitu measurement and control of acoustic streaming in flexural plate wave micropumps. Sens. Actuators A 2000, 79, 115–121. [Google Scholar]
- Meng, AH; Nguyen, NT; White, RM. Focused flow micropump using ultrasonic flexural plate waves. Biomed Microdevices 2000, 2–3, 169–174. [Google Scholar]
- Jang, LS; Chao, SH; Holl, MR; Meldrum, DR. Microfluidic circulatory flows induced by resonant vibration of diaphragms. Sens. Actuators A 2005, 122, 141–148. [Google Scholar]
- Singh, R; Bhethanabotla, VR. Enhancement in Ultrasonic Micro-Transport Using Focused Inter-Digital Transducers in a Surface Acoustic Wave Device: Fluid-Structure Interaction Study. Proceedings of IEEE Sensors, Christchurch, New Zealand, 25–28 October 2009.
- Richter, A; Sandmaier, H. An Electrohydrodynamic Micropump. Proceedings of IEEE Microelectromechanical Systems, Napa Valley, CA, USA, 11–14 February 1990; pp. 99–104.
- Fuhr, G; Hagedorn, R; Muller, T; Benecke, W; Wagner, B. Pumping of Water Solutions in Microfabricated Electrohydrodynamic Systems. Proceedings of IEEE Micro Electro Mechanical Systems. An Investigation of Micro Structures, Sensors, Actuators, Machines and Robots MEMS, Travemunde, Germany, 4–7 February 1992; pp. 25–30.
- Darabi, J; Ohadi, MM; DeVoe, D. An Electrohydrodynamic polarization micropump for electronic cooling. J. Microelectromech. Syst 2001, 10, 98–106. [Google Scholar]
- Darabi, J; Rada, M; Ohadi, M; Lawler, J. Design, fabrication and testing of an electrohydrodynamic ion drag micropump. J. Microelectromech. Syst 2002, 11, 684–690. [Google Scholar]
- Yang, LJ; Wang, JM; Huang, YL. The micro ion drag pump using indium-tin-oxide (ITO) electrodes to resist aging. Sens. Actuators A 2004, 111, 118–122. [Google Scholar]
- Lin, CW; Jang, JY. 3D numerical micro-cooling analysis for an electrohydrodynamic micro-pump. Sens. Actuators A 2005, 122, 167–176. [Google Scholar]
- Darabi, J; Rhodes, C. CFD modeling of an ion-drag micropump. Sens. Actuators A 2006, 127, 94–103. [Google Scholar]
- Singhal, V; Garimella, SV. Induction electrohydrodynamics micropump for high heat flux cooling. Sens. Actuators A 2007, 134, 650–659. [Google Scholar]
- Wakui, D; Imai, N; Nagaura, Y; Sato, H; Sekiguchi, T; Konishi, S; Shoji, S; Homma, T. EHD Micropump using Pyrolyzed Polymer 3-D Carbon Mesh Electrodes. Proceedings of 22nd IEEE International Conference on Micro Electro Mechanical Systems, Sorrento, Italy, 25–29 January 2009; pp. 499–502.
- Litster, S; Suss, ME; Santiago, JG. A two-liquid electroosmotic pump using low applied voltage and power. Sens. Actuators A 2010, 163, 311–314. [Google Scholar]
- Xu, Z; Yang, C; Liua, C; Zhou, Z; Fang, J; Wang, J. An osmotic micro-pump integrated on a microfluidic chip for perfusion cell culture. Talanta 2010, 80, 1088–1093. [Google Scholar]
- Lim, S; Choi, B. A study on the MHD (magnetohydrodynamic) micropump with side-walled electrodes. J. Mech. Sci. Technol 2009, 23, 739–749. [Google Scholar]
- Luginbuhl, P. Microfabricated lamb wave device based on PZT sol-gel thin film for mechanical transport of solid particles and liquids. J. Microelectromech. Syst 1997, 6, 337–345. [Google Scholar]
- Campbell, PK; Jones, KE; Huber, RJ; Horch, KW; Normann, RA. A silicon-based three dimensional neural interface: Manufacturing processes for an intracortical electrode array. IEEE Trans. Biomed. Eng 1991, 38, 758–768. [Google Scholar]
- Bodhale, DW; Nisar, A; Afzulpurkar, N. Structural and microfluidic analysis of hollow side-open polymeric microneedles for transdermal drug delivery applications. Microfluid. Nanofluid 2010, 8, 373–392. [Google Scholar]
- Najafi, K; Wise, K. An implantable multielectrode array with on-chip signal processing. IEEE J. Solid-State Circ 1986, 21, 1035–1044. [Google Scholar]
- Chen, J; Wise, K; Hetke, J; Bledsoe, S. A multichannel neural probe for selective chemical delivery at the cellular level. IEEE Trans. Biomed. Eng 1997, 44, 760–769. [Google Scholar]
- Bai, Q; Wise, K; Anderson, D. A high-yield microassembly structure for three dimensional microelectrode arrays. IEEE Trans. Biomed. Eng 2000, 47, 281–289. [Google Scholar]
- The World Book Encyclopedia, Hypodermic injection. In The world book encyclopedia; World Book Chic, 1999; Volume 9, pp. 480–481.
- McAllister, DV; Allen, MG; Prausnitz, MR. Microfabricated microneedles for gene drug delivery. Annu. Rev. Biomed. Eng 2000, 2, 289–313. [Google Scholar]
- Lin, L; Pisano, A. Silicon processed microneedles. J. Microelectromech. Syst 1999, 8, 78–84. [Google Scholar]
- Zosano Pharma. Available online: http://www.zosanopharma.com accessed on 27 April 2011.
- Ameri, M; Daddona, P; Maa, Y. Demonstrated solid-state stability of parathyroid hormone PTH(1-34) coated on a novel transdermal microprojection delivery system. Pharm. Res 2009, 26, 2454–2463. [Google Scholar]
- Ameri, M; Daddona, P; Maa, Y. Parathyroid hormone PTH(1–34) formulation that enables uniform coating on a novel transdermal microprojection delivery system. Pharm. Res 2009, 27, 303–313. [Google Scholar]
- Dizon, R; Han, H; Russell, A; Reed, M. An ion milling pattern transfer technique for fabrication of three-dimensional micromechanical structures. J. Microelectromech. Syst 1993, 2, 151–159. [Google Scholar]
- Henry, S; McAllister, D; Allen, M; Prausnitz, M. Microfabricated microneedles: A novel approach to transdermal drug delivery. J. Pharm. Sci 1998, 87, 922–925. [Google Scholar]
- Chun, K; Hashiguchi, G; Toshioyoshi, H; Pioufle, B; Ishikawa, J; Murakami, Y; Tamiya, E; Kikuchi, Y; Fujita, H. DNA injection into cell conglomerates by micromachined hollow microcapillary arrays. Proceedings of Digital Transducers, 10th International Conference Solid-State Sensors and Actuators, Sendai, Japan, 7–10 June 1999; pp. 41–47.
- Oka, K; Aoyagi, S; Arai, Y; Isono, Y; Hashiguchi, G; Fujita, H. Fabrication of a microneedle for a trace blood test. Sens Actuators A 2002, 97-98, 478–485. [Google Scholar]
- Brazzle, J; Papautsky, I; Frazier, B. Hollow metallic micromachined needle arrays. J. Biomed. Devices 2000, 2, 197–205. [Google Scholar]
- Chandrasekaran, S; Brazzle, JD; Frazier, AB. Surface micromachined metallic microneedles. J. Microelectromech. Syst 2003, 12, 289–295. [Google Scholar]
- Chandrasekaran, S; Frazier, AB. Characterization of surface micromachined metallic microneedles. J. Microelectromech. Syst 2003, 12, 281–288. [Google Scholar]
- Stoeber, B; Liepmann, D. Design, Fabrication and Testing of a MEMS Syringe. Proceedings of Solid-State Sensors, Actuator and Microsystems Workshop, Hilton Head Island, SC, USA, 2–6 June 2002; pp. 77–80.
- Hashmi, S; Ling, P; Hashmi, G; Reed, M; Gaugler, R; Trimmer, W. Genetic transformation of nematodes using arrays of micromechanical piercing structures. Biotechniques 1995, 19, 766–770. [Google Scholar]
- Narayan, JR; Doraiswamy, A; Chrisey, DB; Chichkov, BN. Medical prototyping using two photon polymerization. Materialstoday 2010, 12, 43–48. [Google Scholar]
- Wu, Y; Qiu, Y; Zhang, S; Qin, G; Gao, Y. Microneedle-based drug delivery: Studies on delivery parameters and biocompatibility. Biomed. Microdevices 2008, 10, 601–610. [Google Scholar]
- Coulman, SA; Ansteyb, A; Gateleyb, C; Morrisseyc, A; McLoughlind, P; Allendera, C; Birchalla, JC. Microneedle mediated delivery of nanoparticles into human skin. Int. J. Pharm 2009, 366, 190–200. [Google Scholar]
- Haq, MI; Smith, E; John, DN; Kalavala, M; Edwards, C; Anstey, A; Morrissey, A; Birchall, JC. Clinical administration of microneedles: skin puncture, pain and sensation. Biomed. Microdevices 2009, 11, 35–47. [Google Scholar]
- Oh, JH; Park, HH; Do, KY; Han, M; Hyun, DH; Kim, CG; Kim, CH; Lee, SS; Hwang, SJ; Shin, SC; Cho, CW. Influence of the delivery systems using a microneedle array on the permeation of a hydrophilic molecule, calcein. Eur. J. Pharm. Biopharm 2008, 69, 1040–1045. [Google Scholar]
- Stoeber, B; Liepmann, D. Fluid Injection Through Out-of-plane Microneedles. Proceedings of 1st Annual International Conference on Microtechnologies in Medicine and Biology, Berkeley, CA, USA, 12–14 October 2000.
- Griss, P; Stemme, G. Side-opened out-of-plane microneedles for microfluidics transdermal liquid transfer. J Microelectromech. Syst 2003, 12, 296–301. [Google Scholar]
- Paik, SJ; Byuna, S; Lima, JM; Park, Y; Leea, A; Chungb, S; Changa, J; Chuna, K; Cho, D. In-plane single-crystal-silicon microneedles for minimally invasive microfluidic systems. Sens. Actuators A 2004, 114, 276–284. [Google Scholar]
- Wilke, N; Hibert, C; O’Brien, J; Morrissey, A. Silicon microneedle electrode array with temperature monitoring for electroporation. Sens Actuators A 2005, 123–124, 319–325. [Google Scholar]
- Choi, JW; Park, IB; Ha, YM; Jung, MG; Lee, SD; Lee, SH. Insertion Force Estimation of Various Microneedle Array-type Structures Fabricated by a Microstereolithography Apparatus. Proceedings of International Joint Conference SICE-ICASE, Busan, Korea, 18–21 October 2006; pp. 3678–3681.
- Shibata, T; Nakanishi, A; Sakai, T; Kato, N; Kawashima, T; Mineta, T; Makino, E. Fabrication and mechanical characterization of microneedle array for cell surgery. Proceedings of Solid-State Sensors, Actuators and Microsystems Conference, Transducers, Lyon, France, 10–14 June 2007; pp. 719–722.
- Runyam, WR; Bean, KE. Semiconductor Integrated Circuit Processing Technology; Addison-Wisley: New York, NY, USA, 1990. [Google Scholar]
- Ambrose, CG; Clanto, TO. Bioabsorbable implants, review of clinical experience in orthopedic surgery. Ann. Biomed. Eng 2004, 32, 171–177. [Google Scholar]
- Park, JH; Allen, MG; Prausnitz, MR. Biodegradable polymer microneedles: fabrication, mechanics and transdermal drug delivery. J. Control. Release 2005, 104, 51–66. [Google Scholar]
- Aoyagi, S; Izumi, H; Fukuda, M. Biodegradable polymer needle with various tip angles and consideration on insertion mechanism of mosquito’s proboscis. Sens. Actuators A 2008, 143, 20–28. [Google Scholar]
- Jiang, J; Moore, JS; Edelhauser, HF; Prausnitz, MR. Intrascleral drug delivery to the eye using hollow microneedles. Pharm. Res 2009, 26, 395–403. [Google Scholar]
- Parker, ER; Rao, MP; Turner, KL; Meinhart, CD; MacDonald, NC. Bulk micromachined Titanium microneedles. J Microelectromech Syst 2007, 16(2), 289–295. [Google Scholar]
- Yoshida, K; Lewinsky, I; Nielsen, M; Hylleberg, M. Implantation mechanics of tungsten microneedles into peripheral nerve truks. Med. Bio. Eng. Comput 2007, 45, 413–420. [Google Scholar]
- Ashraf, MW; Tayyaba, S; Afzulpurkar, N; Nisar, A; Punyasai, C; Saejok, K; Supadech, J; Atthi, N; Hruanun, C; Poyai, A. Optimization of Fabrication Process for MEMS based Microneedles Using ICP Etching Technology. Proceedings of 6th international Conference on MEMS, Nano and Smart System (ICMENS), Changsha, China, 14–15 December 2010; pp. 356–359.
- Tsuchiya, K; Nakanishi, N; Uetsuji, Y; Nakamachi, E. Development of Blood Extraction System for Health Monitoring System. Biomed. Devices 2005, 7, 347–353. [Google Scholar]
- Ding, Z; Verbaan, FJ; Bivas-Benita, M; Bungener, L; Huckriede, A; van den Berg, DG; Kersten, G; Bouwstra, JA. Microneedles arrays for the transcutaneous immunization of diphtheria and influenza in BALB/c mice. J. Control. Release 2009, 136, 71–78. [Google Scholar]
- Chen, B; Wei, J; Iliescu, C. Sonophoretic enhanced microneedles array (SEMA)-improving the efficiency of transdermal drug delivery. Sens. Actuators B 2010, 145, 54–60. [Google Scholar]
- Zhang, W; Gao, J; Zhu, Q; Zhang, M; Ding, X; Wang, X; Hou, X; Fan, W; Ding, B; Wu, X; Wang, X; Gao, S. Penetration and distribution of PLGA nanoparticles in the human skin treated with microneedles. Int. J. Pharm 2010, 402, 205–212. [Google Scholar]
- Zhang, P; Dalton, C; Jullien, GA. Design and fabrication of MEMS based microneedle arrays for medical applications. Microsyst. Technol 2009, 15, 1073–1082. [Google Scholar]
- Yu, LM; Tay, FEH; Guo, DG; Xu, L; Yap, KL. A microfabricated electrode with Hollow microneedles for ECG measurement. Sens. Actuators A 2009, 151, 17–22. [Google Scholar]
- Chen, B; Wei, J; Tay, FEH. Silicon microneedle array with biodegradable tip for transdermal drug delivery. Microsyst. Technol 2008, 14, 1015–1019. [Google Scholar]
- Roxhed, N; Griss, P; Stemme, G. Membrane-seald hollow microneedles and related administration schemes for transdermal drug delivery. Biomed. Microdevices 2008, 10, 271–279. [Google Scholar]
- Bhandari, R; Negi, S; Solzbacher, F. A novel mask-less method of fabricating high aspect ratio microneedles for blood sampling. Proceedings of IEEE 58th Electronic Components and Technology Conference, Lake Buena Vista, FL, USA, 27–30 May 2008; pp. 1306–1309.
- Donnelly, RF; Morrow, DIJ; McCarron, PA; Woolfson, AD; Morrissey, A; Juzenas, P; Juzeniene, A; Lani, V; McCarthy, HO; Moan, J. Microneedle-mediated Intradermal delivery of 5-aminolevulinic acid: Potential for enhanced topical photodynamic therapy. J. Control. Release 2008, 129, 154–162. [Google Scholar]
- Lee, JW; Park, JH; Prausnitz, MR. Dissolving microneedles for transdermal drug delivery. Biomaterials 2008, 29, 2113–2124. [Google Scholar]
- Park, JH; Choi, S; Seo, S; Choy, YB; Prausnitz, MR. A microneedle roller for transdermal drug delivery. Eur. J. Pharm. Biopharm 2010, 76, 282–289. [Google Scholar]
- Gomaa, YA; Morrow, DIJ; Garland, MJ; Donnelly, RF; El-Khordagui, LK. Effects of microneedle length, densty, insertion time and multiple applications on human skin barrier function: Assessments by transdermal water loss. Toxicol. in Vitro 2010, 24, 1971–1978. [Google Scholar]
- Donnelly, RF; Morrow, DIJ; Fay, F; Scott, CJ; Abdelghany, S; Sing, RRT; Garland, MJ; Woolfson, AD. Microneedle-mediated Intradermal nanopartile delivery: Potential for ehnaced local administration of hydrophobic pre-formed photosensitisers. Photodiagn. Photodynam 2010, 7, 222–231. [Google Scholar]
- Matteucci, M; Fanetti, M; Casella, M; Gramatica, F; Gavioli, L; Tormen, M; Grenci, G; Angelis, FD; Fabrizio, ED. Poly vinyl alcohol re-usable masters for microneedles replication. Microelectron. Eng 2009, 86, 752–756. [Google Scholar]
- Han, MH; Kim, DK; Kang, SH; Yoon, HR; Kim, BY; Lee, SS; Kim, KD; Lee, HG. Improvement in antigen-delivery using fabrication of a grooves-embedded microneedle array. Sens. Actuators B 2009, 137, 274–280. [Google Scholar]
- Jin, CY; Han, MH; Lee, SS. Mass producible and biocompatible microneedle patch and functional verification of its usefulness for transdermal drug delivery. Biomed. Microdevices 2009, 11, 1195–1203. [Google Scholar]
- Emam, M; Abashiya, Y; Chareunsack, B; Skordos, J; Oh, JH; Choi, Y; Kralick, F; Noh, H. A novel microdevice for the treatment of hydrocephalus: design and fabrication of an array of microvales and microneedles. Microsyst. Technol 2008, 14, 371–378. [Google Scholar]
- Hsu, CC; Chen, YT; Tsai, CH; Kangl, SW. Fabrication of Microneedles. Proceedings of 2nd IEEE International Conference on Nano/Micro Engineered and Molicular System, Bangkok, Thailand, 16–19 January 2007; pp. 639–642.
- Kim, YC; Quan, FS; Compans, RW; Kang, SM; Prausnitz, MR. Formulation and coating of microneedles with inactivated influenza virus to improve vaccine stability and immunogenicity. J. Control. Release 2010, 142, 187–195. [Google Scholar]
- Kato, N; Kawashima, T; Shibata, T; Mineta, T; Makino, E. Micromachning of a newly designed AFM probe integrated with hollow microneedle for cellular function analysis. Microelectr. Eng 2010, 87, 1185–1189. [Google Scholar]
- Hou, W; Das, B; Jiang, Y; Qian, S; Zheng, X; Yang, J; Pi, X; Liu, H; Zheng, J; Zhang, Y. On A Microfabricated Ti-alloy-based Microneedle Array for Transdermal Drug Delivery. Proceedings of 3rd IEEE International Conference on Nano/Micro Engineered and Molecular System, Sanya, China, 6–9 January 2008; pp. 453–456.
- Kolli, CS; Banga, AK. Characterization of Solid Maltose Microneedles and their Use for Transdermal Delivery. Pharm. Res 2008, 25, 104–113. [Google Scholar]
- Verbaan, FJ; Bal, SM; van den Berg, dj; Dijksman, JA; van Hecke, M; Verpoorten, H; van den Berg, A; Luttge, R; Bouwstra, JA. Improved piercing of microneedle arrays in dermatomed human skin by an impact insertion method. In J Control Release; 2008; Volume 128, pp. 80–88. [Google Scholar]
- Kim, K; Lee, J. High aspect ratio tapered hollow metallic microneedle arrays with microfluidic interconnector. Microsyst. Technol 2007, 13, 231–235. [Google Scholar]
- Gere, J; Timoshenko, S. Mechanics of Materials, 4th ed; Prentice Hall: Upper Saddle River, NJ, USA, 1999. [Google Scholar]
- Zahn, JD; Talbot, NH; Liepmann, D; Pisano, AP. Micro fabricated polysilicon microneedles for minimally invasive biomedical devices. Biomed. Microdevices 2000, 2, 295–303. [Google Scholar]
- Aggarwal, P; Johnston, CR. Geomertrical effects in mechanical characterizing of microneedle for biomedical applications. Sens. Actuators B 2004, 102, 226–234. [Google Scholar]
- Frick, TB; Maruccia, DD; Cartmill, JA; Martin, CJ; Walsh, WR. Resistance forces acting on suture needles. J. Biomech 2001, 34, 1335–1340. [Google Scholar]
- Khumpuang, S; Horade, M; Fujioka, K; Sugiyama, S. Geometrical Strengthening and tip-sharping of a microneedle array fabricated by X-ray lithography. Microsyst. Technol 2007, 13, 209–214. [Google Scholar]
- Davis, SP; Martanto, W; Allen, MG; Prausnitz, MR. Hollow metal microneedles for insulin delivery to diabetic rats. IEEE Trans. Biomed. Eng 2005, 52, 909–915. [Google Scholar]
- Ashraf, MW; Tayyaba, S; Afzulpurkar, N. MEMS based Polymeric Drug Delivery System. Proceedings of 6th IEEE Conference on Automation Science and Engineering (CASE), Toronto, Canada, 21–24 August 2010; pp. 192–197.
- Rajaraman, S; Henderson, HT. A unique fabrication approach for microneedles using coherent porous silicon technology. Sens. Actuators B 2005, 105, 443–448. [Google Scholar]
- Sammoura, F; Kang, J; Heo, Y; Jung, T; Lin, L. Polymeric microneedle fabrication using a microinjection molding technique. Microsyst. Technol 2007, 13, 517–522. [Google Scholar]
- Chen, X; Kask, AS; Crichton, ML; McNeilly, C; Yukiko, S; Dong, L; Marshak, JO; Jarrahian, C; Fernando, GJP; Chen, D; Koelle, DM; Kendall, MAF. Improved DNA vaccination by skin-targeted delivery using dry-coated densely-packed microprojection arrays. J. Control. Release 2010, 148, 327–333. [Google Scholar]
- Milewski, M; Yerramreddy, TR; Ghosh, P; Crooks, PA; Stinchcomb, AL. In vitro permeation of a pegylated naltrexone prodrug across microneedle-treated skin. J. Control. Release 2010, 146, 37–44. [Google Scholar]
- Khann, P; Silv, H; Bhansali, S. Variation in microneedle geometry to increase the shear strength. Proc. Eng 2010, 5, 977–980. [Google Scholar]
- Bal, SM; Kruithof, AC; Zwier, R; Dietz, E; Bouwstra, JA; Lademann, J; Meinke, MC. Influence of microneedle shape on the transport of a fluorescent dye into human skin in vivo. J. Control. Release 2010, 147, 218–224. [Google Scholar]
- Donnelly, RF; Garland, MJ; Morrow, DIJ; Migalska, K; Singh, TRR; Majithiya, R; Woolfson, AD. Optical coherence tomography is a valuable tool in the study of the effects of microneedle geometry on skin penetration characteristics and in-skin dissolution. J. Control. Release 2010, 147, 333–341. [Google Scholar]
- Crichton, ML; Ansaldo, A; Chen, X; Prowa, TW; Fernando, GJP; Kendall, MAF. The effect of strain rate on the precision of penetration of short densely-packed microprojection array patches coated with vaccine. Biomaterials 2010, 31, 4562–4572. [Google Scholar]
- Pearton, M; Kang, SM; Song, JM; Kim, YC; Quan, FS; Anstey, A; Ivory, M; Prausnitz, MR; Compans, RW; Birchall, JC. Influenza virus-like particles coated onto microneedles can elicit stimulatory effects on Langerhans cells in human skin. Vaccine 2010, 28, 6104–6113. [Google Scholar]
- Li, W-Z; Hou, M-R; Zhou, J-P; Zhou, Y-Q; Hao, B-H; Liu, T; Zhang, Y. Super-short solid silicon microneedles for transdermal drug delivery applications international. J. Pharm 2010, 389, 122–129. [Google Scholar]
- Chu, LY; Prausnitz, MR. Separable arrowhead microneedles. J. Control. Release 2010, 149, 242–249. [Google Scholar]
- Yan, K; Todo, H; Sugibayashi, K. Transdermal drug delivery by in-skin electroporation using a microneedle array. Int. J. Pharm 2010, 397, 77–83. [Google Scholar]
- Yan, G; Warner, KS; Zhang, J; Sharma, S; Gale, BK. Evaluation needle length and density of microneedle arrays in the pretreatment of skin for transdermal drug delivery. Int. J. Pharm 2010, 391, 7–12. [Google Scholar]
- Song, JM; Kim, YC; Barlow, PG; Hossain, MJ; Park, KM; Donis, RO; Prausnitz, MR; Compans, RW; Kang, SM. Improve protection against avian influenza H5N1 virus by a single vaccination with virus-like particles in skin using microneedles. Antivir. Res 2010, 88, 244–247. [Google Scholar]
- Zhou, CP; Liu, YL; Wang, HL; Zhang, PX; Zhang, JL. Transdermal delivery of insulin using microneedle rollers in vivo. Int. J. Pharm 2010, 392, 127–133. [Google Scholar]
- Quan, FS; Kim, YC; Compans, RW; Prausnitz, MR; Kang, SM. Dose sparing enabled by skin immunization with influenza virus-like particle vaccine using microneedles. J. Control. Release 2010, 147, 326–332. [Google Scholar]
- Bussemer, T; Otto, I; Bodmeier, R. Pulsatile drug delivery systems. Crit. Rev. Ther. Drug Carrier Syst 2001, 18, 433–458. [Google Scholar]
- Ma, HK; Chen, BR; Gao, JJ; Lin, CY. Development of an OAPCP-micropump liquid cooling system in a laptop. Int. Commun. Heat Mass Transf 2009, 36, 225–232. [Google Scholar]
- Xia, F; Tadigadapa, S; Zhang, QM. Electroactive polymer based microfluidic pump. Sens. Actuators A 2006, 125, 346–352. [Google Scholar]
- Jeong, OC; Konishi, S. Fabrication and drive test of pneumatic PDMS micro pump Sens. Actuators A 2007, 135, 849–856. [Google Scholar]
- Makino, E; Shibata, T. Micropump with TiNi shape memory diaphragm actuator. J. Surface Finish. Soc. Jpn 2005, 56, 213–218. [Google Scholar]
- Ashraf, MW; Tayyaba, S; Nisar, A; Afzulpurkar, N. MEMS based System for Drug Delivery. Proceedings of 6th International Conference on Emerging Technologies (ICET), Islamabad, Pakistan, 18–19 October, 2010; pp. 82–87.
- Kabata, A; Suzuki, H. Microsystem for Injection of Insulin and Monitoring of Glucose Concentration. Proceedings of 5th International Conference on Sensors, Irvine, CA, USA, 30 October–3 November 2005; pp. 171–174.
- Yang, SC; Liu, CH. An Electrolysis-bubble-actuated micropump using electrowetting on dielectric (EWOD) for 1XN micro-sample switches. Proceedings of Transducers, Denver, CO, USA, 21–25 June 2009; pp. 2018–2021.
- Ma, B; Liu, S; Gan, Z; Liu, G; Cai, X; Zhang, H; Yang, Z. A PZT Insulin Pump Integrated with a Silicon Micro Needle Array for Transdermal Drug Delivery. Proceedings of 56th Electronic Components and Technology Conference, San Diego, CA, USA, 30 May–2 June 2006; pp. 667–681.
- Tayyaba, S; Ashraf, MW; Afzulpurkar, N. Design and Simulation of Double Lumen Polymeric Microneedles for Blood Transport. Proceedings of International Conference on Mechanical and Electrical Technology (ICMET), Singapore, 10–12 September 2010; pp. 615–618.
- Najafi, K. Micropackaging technologies for integrated microsystems: Applications to MEMS and MOEMS. Proc SPIE 2003, 4979. [Google Scholar] [CrossRef]
- 3M. Available online: http://solutions.3m.com/wps/portal/3M/en_WW/DrugDeliverySystems/DDSD accessed on 27 April 2011.
- Birchall, JC; Clemo, R; Anstey, A; John, DN. Microneedles in clinical practice-an exploratory study into the opinions of healthcare professionals and the public. Pharm. Res 2011, 28, 95–106. [Google Scholar]
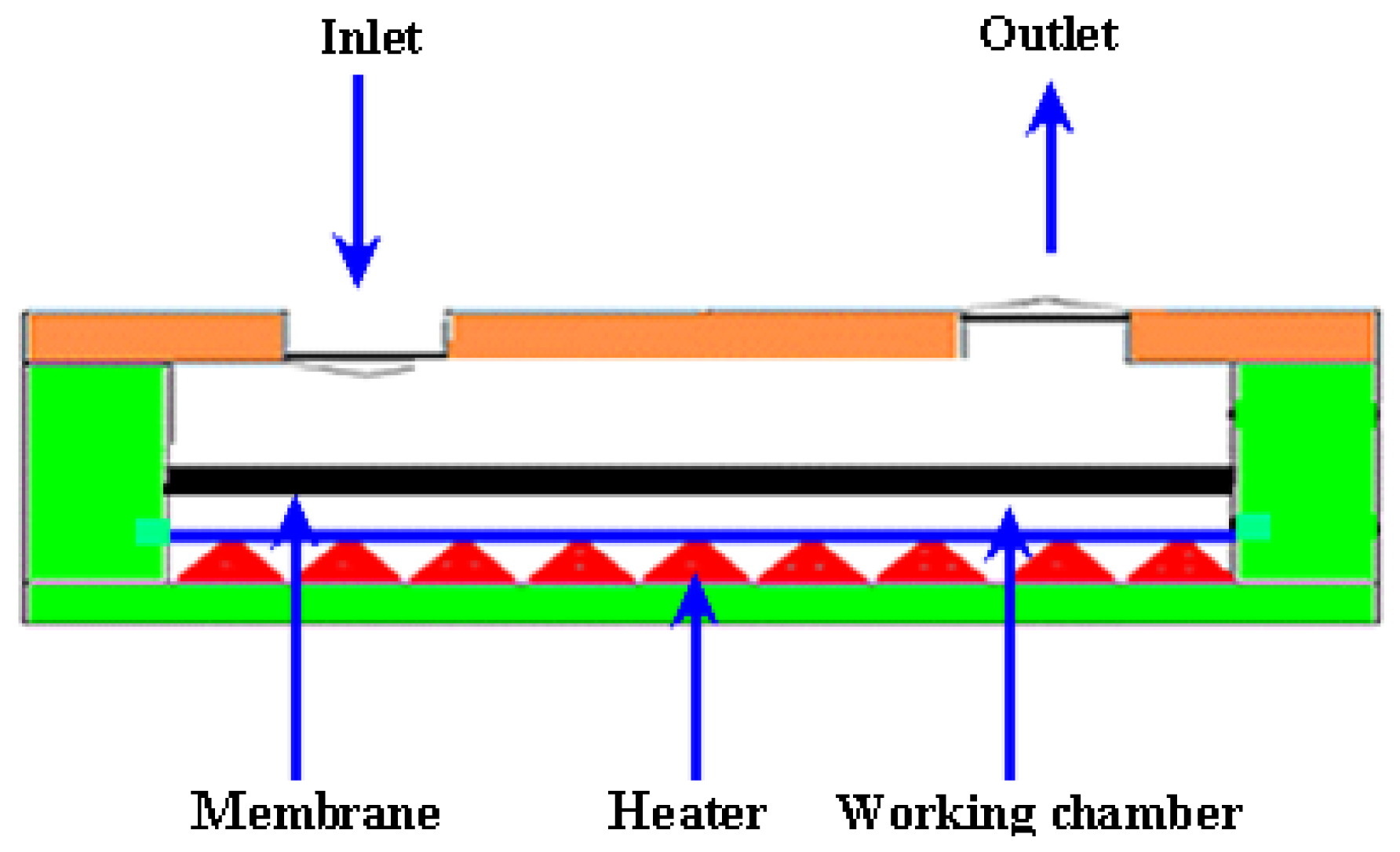

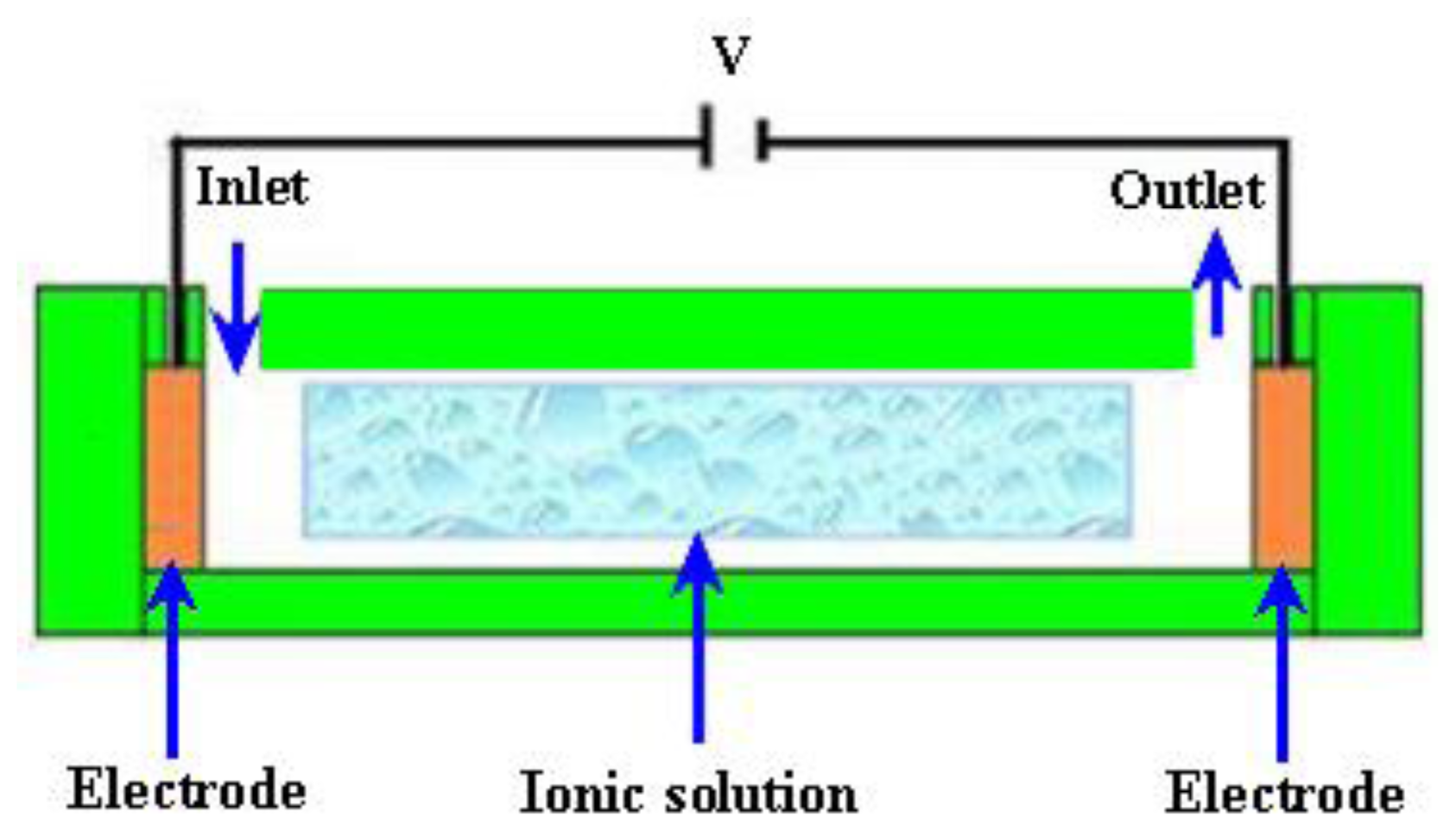
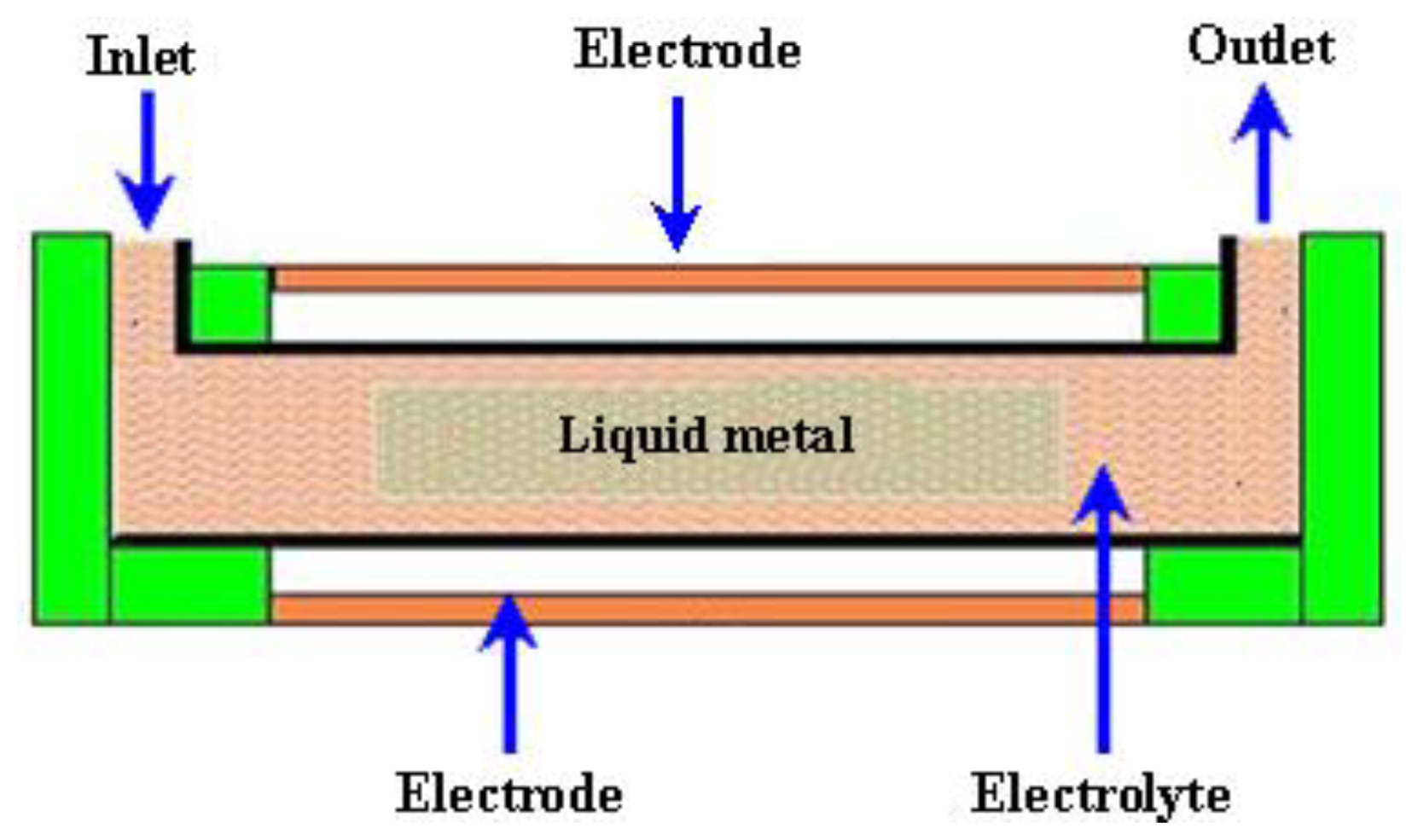

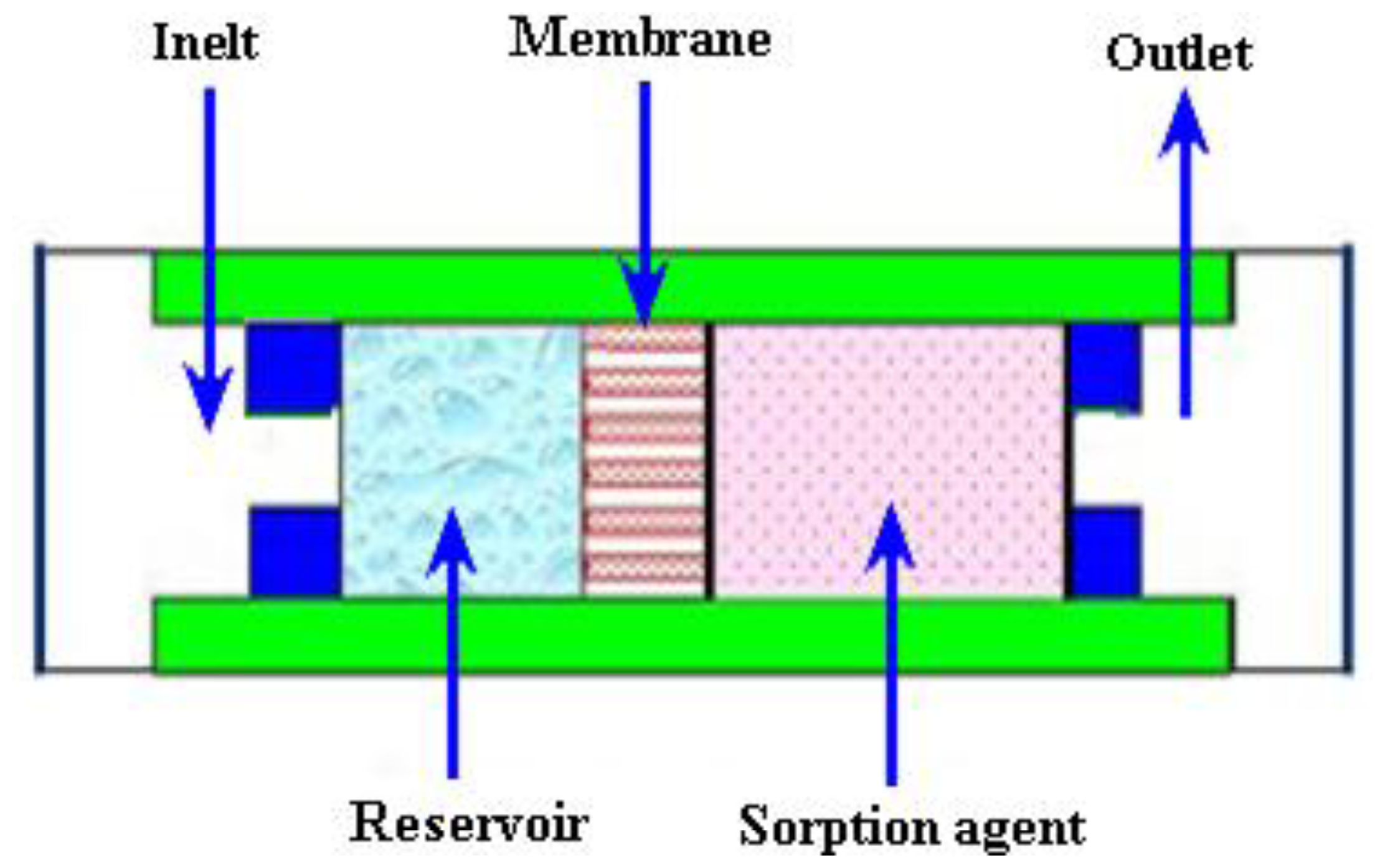


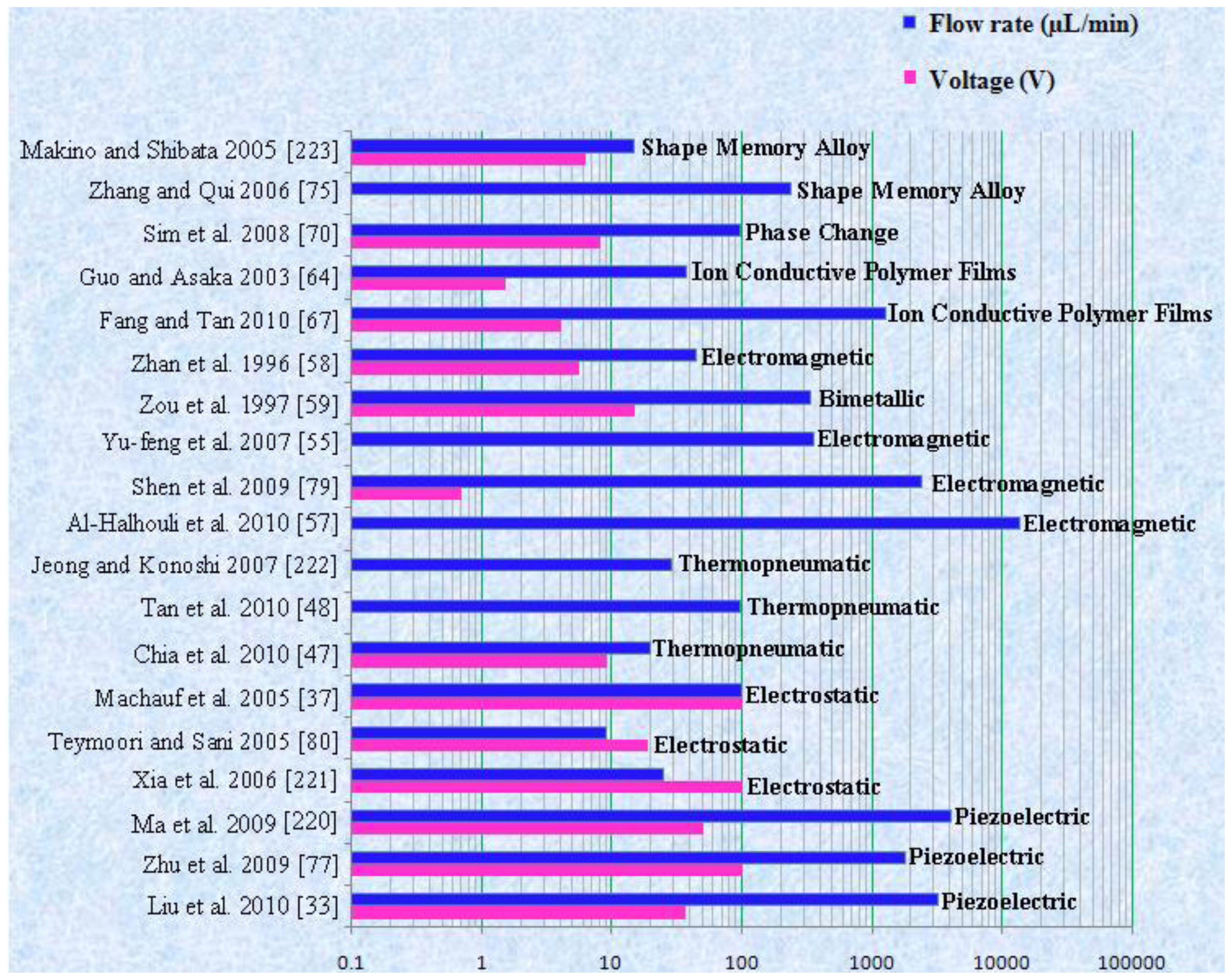

| Reference | Actuation Method | Materials used for fabrication | Size | Pumping Chamber | Pumping Medium | Valve | Voltage (V) | Frequency | Back Pressure/Applied Pressure | Flow Rate (μL/min) | Applications |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Liu et al. 2010 [33] | Piezoelectric | Polycarbonate (PC), PMMA, PDMS, PZT, (Titanium) Ti | 15 × 8 mm | 4 | Insulin | 2 | 36 | 200 Hz | 22 kPa | 6.23 × 10−5 mL/min | Insulin therapy system |
| Zhu et al. 2009 [77] | Piezoelectric | Polyetheretherketone/PDMS/Metal/Ceramics | Not reported | 1 | Air/Water | 2 | 100 | 225 Hz for air, 17 Hz for water | Not reported | 39 mL/min for air, 1.8 mL/min for water | Drug delivery applications |
| Kang and Auner 2011 [78] | Piezoelectric | Si/Epoxy H31/PZT-5A | 14.5 × 9 × 1.1 mm | 1 | Not reported | 2 | 240 | 20–100 Hz | 0–10 psi | 0.52 mL/min | Microfluidic applications |
| Halhouli et al. 2010 [57] | Electromagnetic | PC, Plexiglass | 16 × 18 mm | 1 | Water | Not report ed | Not reported | Not reported | 785 Pa | 13.7 mL/min | Biomedical applications |
| Shen et al. 2009 [79] | Electromagnetic | PDMS, Glass | 24 × 40 × 0.4 mm | 3 | Water | 2 | 0.7 | 12 Hz | 70 mbar | 2.4 mL/min | Portable LOC applications |
| Lee et al. 2009 [39] | Electrostatic | Si | Not reported | 2 | Gas | 19 | Not reported | 2.2–2.8 KHz | 7.3–3.3 kPa | 0.29–0.07 SCCM | Not reported |
| Teymoori and Sani 2005 [80] | Electrostatic | Si, Glass | 7 × 4 × 1 mm | 3 | Not reported | 3 | 18.5 | 50 Hz | Not reported | 9.1 μL/min | Drug delivery applications |
| Chia et al. 2010 [47] | Thermopneumatic | PDMS, Glass | 16 × 18 × 5 mm | 3 | Not reported | Not reported | 9 | 1.2 Hz | 490 Pa | 20.01 μL/min | Biomedical applications |
| Tan et al. 2010 [48] | Thermopneumatic | PDMS, PMMA | Not reported | 3 | Compressed air | 3 | Not reported | 10 Hz | 138 kPa | 96 μL/min | Microfluidic devics |
| Zou et al. 1997 [59] | Bimetallic | Al, Si, Glass | 13 × 7 × 2 mm | 2 | Gas/Water | 2 | 15 | Not reported | 0.5 kPa | 5.6 μL/s | Not reported |
| Fang and Tan 2010 [67] | ICPF | PDMS, Polypyrrole, Stainless steel, Polyvinylidene fluoride | 25 × 25 × 10 mm | 1 | Water | 4 | 4 | 0.5 Hz | 1.3 kPa | 1260 μL/min | Biomedical devices |
| Sim et al. 2008 [70] | Phase Change | Al, Silicon, Silicone rubber, Glass | Not reported | 1 | Water | 2 | 8 | 2 Hz | 0 mm H2O | 97 μL/min | Not reported |
| Zhang and Qiu 2006 [75] | SMA | Ti, Nickel (Ni), Copper (Cu) | 8 × 8 × 1.8 mm | 1 | DI water | 2 | Not reported | 80 Hz | Not reported | 235 μL/min | Not reported |
| Reference | Actuation Method | Material used for Fabrication | Size | Pumping Chamber | Pumping Medium | Valve | Voltage (V) | Frequency | Back Pressure/Applied Pressure | Flow Rate (μL/min) | Applications |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Chan et al. 2010[106] | Bubble type | PDMS, Glass, Si | Not reported | 2 | DI water, Phosphate- buffered solution | Not reported | 5 | 300 Hz | Not reported | 37.8 μL/min | Miniature electronic devices |
| Jung and Kwak 2007 [104] | Bubble type | Si, Pyrex glass | Not reported | 1 | DI water | Not reported | 30 | 0.5–2.0 Hz | Not reported | 6–8 μL/min | Microfluidic applications |
| Wakui et al. 2009 [127] | EHD | Polymer, Carbon, Glass | Not reported | Not reported | Fluorinert | Not reported | 500 | Not reported | 23 Pa | 400 μL/min | Microfluidic devices |
| Singhal and Garimella 2007 [126] | EHD | Al | 1500 × 200 × 50 μm | 1 | Water | Not reported | 3.3 | 373 kHz | Not reported | 10.5 μL/min | Microchannel cooling system |
| Lister et al. 2010 [128] | EO | Glass, Platinum | Not reported | Not reported | Borate buffer, DI water | Not reported | 2.9 | Not reported | 1.6 kPa | 13 μL/min | Drug delivery |
| Xu et al. 2010 [129] | EO | Glass, PDMS | Not reported | 2 | Water | 1 | Not reported | Not reported | Not reported | 0.33 μL/min | Perfusion cell culture |
| Kang and Choi 2010 [112] | MHD | Au (gold), PDMS | Not reported | Not reported | PBS solution | Not reported | 3.6 | Not reported | Not reported | 2.83 μL/min | LOC applications |
| Lim and Choi 2009 [130] | MHD | Si, Pyrex glass, Al | 40 × 25 × 1 mm | Not reported | PBS solution | Not reported | Not reported | Not reported | 100000N/m2 | 0.3 μL/min | Drug delivery |
| Yun et al. 2002 [89] | EW | Glass, Si, Platinum | Not reported | 2 | Mercury | 2 | 2.3 | 25 Hz | 800 Pa | 70 μL/min | Biomedical Devices |
| Kim et al. 2008 [96] | Electrochemical | Ppy, PDMS, PMMA | 5.6 × 16 × 26 mm | 1 | Water | 2 | ±1.5 | Not reported | 11 mbar | 52 μL/min | Microfluidic applications |
| Heuck et al. 2008 [100] | Evaporation | Si | Not reported | Not reported | DI water | Not reported | Not reported | Not reported | Not reported | 11 pL/s | Biological sampling |
| Guan et al. 2006 [99] | Evaporation | Pdms, PMMA, Stainless steel | 25 × 15 × 3 mm | Not reported | Water | Not reported | Not reported | Not reported | 23.5 kPa | 3.02 μL/min | Microfluidics system |
| Luginbuhl 1997 [131] | FPW | Si, Platinum, Ceramic | Not reported | Not reported | Water | Not reported | 6 | 2–3 MHz | Not reported | 0.255 μL/min | Fluid delivery system |
| Structure | Overall Shape | Tip Shape | Material Used | Application |
|---|---|---|---|---|
| Solid | Cylindrical | Volcano | Single crystal silicon | Drug delivery |
| Hollow | Canonical | Snake fang | Polysilicon | Gene delivery |
| In-plane | Pyramid | Cylindrical | Silicon dioxide | Blood extraction |
| Out-of-plane | Candle | Canonical | Silicon nitride | Fluid sampling |
| Spike | Microhypodermis | PGA | Vaccination | |
| Spear | Tapered | PDMS | Micro-dialysis | |
| Square | PMMA | Cancer therapy | ||
| Pentagonal | Glass | Dentistry | ||
| Hexagonal | GaAs | Skin treatment | ||
| Octagonal | Titanium | Cell surgery | ||
| Rocket | Ti- alloy | Allergies diagnosis | ||
| Star | Tungsten | Animal identification | ||
| Tungsten-alloy | Ink-jet printing | |||
| Stainless steel | Sensing electrodes | |||
| Reference | Material | Structure of microneedles | Shapes of microneedles | Dimensions | Array size/Needles | Analysis type | Testing | Fabrication techniques | Application |
|---|---|---|---|---|---|---|---|---|---|
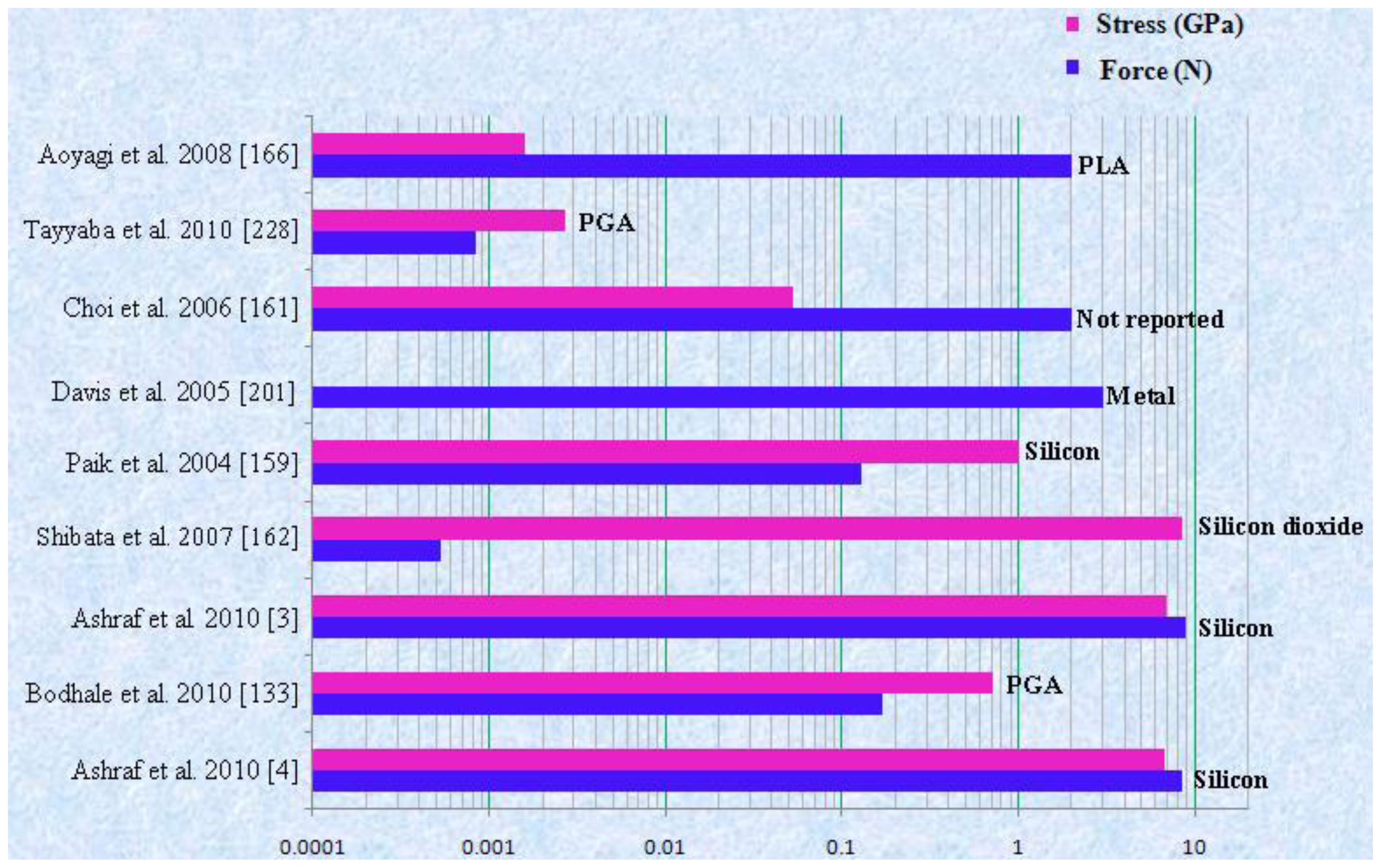
| Waseem et al. 2010 [4] | Silicon | Hollow/Out-of plane | Cylindrical | L = 200 μm Di = 60 μm Do = 150 μm | 5 × 5 | Structural/CFD (Static/Transient) | Not reported | ICP etching | Transdermal drug delivery |
| Chen et al. 2010 [173] | Silicon | Hollow/Out-of plane | Cylindrical base | L = 100 μm D = 80 μm | 30 × 30 | Fluidic analysis | Pig Skin | Deep reactive ion etching (DRIE) | Transdermal drug delivery |
| Zhang et al. 2010 [174] | Silicon | Solid/Out-of plane | Star shape | L = 200 μm | 10 × 10 | PLGA nano Particles distribution | Human skin | RIE/Thin film deposition Photolithography | Transdermal drug delivery |
| Waseem et al. 2010 [3] | Silicon | Hollow/Out-of plane | Cylindrical base tapered tip | L = 200 μm Di = 40 μm Do = 425 μm | 6 × 6 | Structural/Fluidic (Static/Transient) | Not reported | ICP etching | Transdermal drug delivery |
| Zhang et al. 2009 [175] | Silicon | Hollow/Out-of plane | Cylindrical/Side opened at tip | L = 200 μm Di = 40 μm Dt = 450 nm | 10 × 11 | Fluidic analysis | Potato skin/Chicken skin | Bi-mask technique | Drug delivery/fluid sampling |
| Ding et al. 2009 [172] | Silicon | Solid/Hollow | Tangentially cut tip | L1 = 300 μm to 900 μm L2 = 300 μm D2 = 200 μm L3 = 245 μm | 4 × 4 9 × 9 | Fluidic analysis/Statistical | Mouse skin | Surface micromachining/Etching | Dermal diphtheria/influenza vaccination |
| Haq et al. 2009 [155] | Silicon | Hollow/Out-of- plane | Pyramidal | L1 = 180 μm L2 = 280 μm Db = 180 μm | 6 × 6 | Fluidic analysis | Human skin | Wet etching | Transcutaneous drug delivery |
| Yu et al. 2009 [176] | Silicon | Hollow/Out-of plane | Cylindrical | DP = 200 μm D = 100 μm | Not reported | Structural analysis | One-lead ECG recording system | DRIE | ECG measurement |
| Coulman et al. 2009 [154] | Silicon | Solid | Pyramidal shape/Pointed/Frustum tip | L = 280 μm, Db = 200 μm | 16 needles | Diffusion of nano particles | Human epidermal membrane | Wet etching | Transdermal/Intradermal drug delivery |
| Chen et al. 2008 [177] | Silicon | Out-of plane | Macro porous tip | Not reported | Not reported | Fluidic analysis | Pig skin | DRIE | Transdermal drug delivery |
| Roxhed et al. 2008 [178] | Silicon | Out-of- plane/Hollow | Cross/Circler | L1 = 310 μm, L2 = 400 μm | 25 needles | Fluidic analysis | Human skin | DRIE | Transdermal drug delivery |
| Bhandari et al. 2008 [179] | Silicon | Hollow/Out- of-plane | Square base canonical | Not reported | 10 × 10 | Not reported | Not reported | Laser micromachining/Dicing/Etching | Blood sampling |
| Donnelly et al. 2008 [180] | Silicon | Not reported | Sharp 3D Tip/Grooves- embedded shaft | L = 270 μm, Db = 240 μm | Not reported | Fluidic analysis/Statistical | Mouse skin/Porcine skin of piglets | Wet etching | Photodynamic therapy |
| Lee et al. 2008 [181] | Silicon | Solid/Out- of plane | Conical/Pyramidal | L1 = 800 μm, Db = 200 μm, Dt = 20 μm, L2 = 600 μm, Wb = 300 μm | 3 × 3 | Structural analysis | Not reported | Micromolding | Drug delivery |
| Reference | Materials | Structure of microneedles | Shapes of microneedles | Dimensions | Array size/Needles | Analysis type | Testing | Fabrication techniques | Application |
|---|---|---|---|---|---|---|---|---|---|
| Park et al. 2010 [182] | PLA | Solid/Out-of- plane | Canonical/Square base | L = 600 μm Wb = 250 μm | 10 × 10 | Diffusion of trypan blue | Human/Porcine cadaver skin | UV lithography | Transdermal drug delivery |
| Gomaa et al. 2010 [183] | PMVE/MA | Solid/Out-of- plane | Canonical | L1 = 400 μm L2 = 600 μm L3 = 100 μm | 11 × 11 14 × 14 19 × 19 | Effect of Skin Permeability with microneedle density | Human skin | Laser micromachining | Drug delivery |
| Donnelly et al. 2010 [184] | Polymeric (Gantrez) | Solid/Out-of- plane | Canonical | Not reported | Not reported | Statistical | Porcine skin | Molding process | Intradermal delivery |
| Bodhale et al. 2010 [133] | PGA | Hollow/Out-of plane | Side opened/Sharp tip | L = 200 μm Di = 30 μm Do = 150 μm Db = 300 μm | 25 × 25 | Structural/Fluidic | Not reported | Hot embossing/UV excimer laser(Proposed) | Drug delivery |
| Matteucci et al. 2009 [185] | PMMA | Hollow/Out-of plane | Rounded tip/Sharp tip | L = 500 to 1100 μm Bevel angle = 30° to 40° | 10 arrays | Not reported | Not reported | DXRL | Not reported |
| Han et al. 2009 [186] | PLLA | Solid/Out-of plane | Sharp 3D Tip/Grooves-embedded shaft | L = 880 ± 20 μm, Wb = 710 ± 15 μm T = 145 ± 15 μm | Not reported | Protein transportation analysis | Mouse skin/Serum | Lithography/Ni electroplating/PDMS replication/Hot embossing | Intradermal immunization |
| Jin et al. 2009 [187] | PMMA/PC | Solid/In-plane | Quadrangular/Pyramidal | L = 200–1500 mm | Not reported | Drug transportation | Mouse skin and serum | DXRL/Hot embossing | Transdermal drug delivery |
| Oh et al. 2008 [156] | PC | Solid/Out-of plane | Sharp tip/Spear | L = 200–500 μm | Not reported | Hydrophilic molecules transportation | Mouse skin | Molding/Hot embossing | To improve skin permeability for hydrophilic molecules |
| Emam et al. 2008 [188] | SU-8 | Out-of-plane/Hollow | Sharp tip | L = 500 μm, Wb = 100 μm | Not reported | Fluid analysis | Not reported | Deposition/Lithography/Etching | Treatment of hydrocephalus |
| Aoyagi et al. 2007[166] | PLA | Solid/Out-of plane | Straight/Harpoon shape/Sharp tip | L = 400 μm Wb = 90, 120, 150, 230 μm T = 115 μm Tip angle = 10°, 20°, 30°, 40° | Not reported | Structural | Artificial skin of silicone rubber | Etching/Injection molding | Drug delivery |
| Hsu et al. 2007 [189] | SU-8 2050 | Out-of-plane | V-groove | L1 = 236 μm L2 = 350 μm | Not reported | Not reported | Not reported | Molding/KOH etching | Biomedicine technology |
| Reference | Materials | Structure of Microneedles | Shapes of microneedles | Dimensions | Array size/Needles | Analysis Type | Testing | Fabrication Techniques | Applications |
|---|---|---|---|---|---|---|---|---|---|
| Kim et al. 2010 [190] | Stainless steel | Solid/In-plane | Spear/Sharp tip | L = 700 μm Wb = 160 μm T = 50 μm | 5 microneedles | Drug transportation/Statistical analysis | Mouse skin | Infrared Laser | Vaccine delivery |
| Kato et al. 2010 [191] | SiO2 | Hollow/Out- of-Plane | Circular Tip | L = 77 μm Do = 5.5 μm Di = 3.5 μm | Not reported | Structural (Panitration) | Gelatin | DRIE/Micromachining | Cellular function analysis |
| Ding et al. 2009 [172] | Stainless steel | Solid/Hollow/Out-of-Plane | Tangentially cut tip | L = 245, 300–900 μm D2 = 200 μm | 4 × 4 9 × 9 | Drug transportation/Statistical | Mouse skin | Surface micromachining/Etching | Dermal diphtheria/Influenza vaccination |
| Jiang et al. 2009 [167] | Glass | Hollow | Elliptical tip opening | L = 3–4 cm | Not reported | Histological/Microscopic image analysis | Human cadaver eyes | Micropipette pulling technique | Intrascleral delivery |
| Jin et al. 2009 [187] | Ni | Solid/In-plane | Triangular/Pyramidal | L= 200–1500 mm | Not reported | Drug transportation | Mouse skin and serum | DXRL/Hot embossing | Transdermal drug delivery |
| Hou et al. 2008 [192] | Ti-alloy | Hollow/Out- of-Plane | Not reported | L = 120 μm | 10 × 10 | Fluidic analysis | Not reported | Not reported | Transdermal drug delivery |
| Kolli and Banga 2008 [193] | Maltose | Solid/In-plane | Tetrahedron/Sharp tip | L = 500 μm Dt = 6 μm | 27 needle per array | Drug transportation | Mouse skin/Jacketed Franz diffusion cells | Micro-molding | Transdermal drug delivery |
| Verbaan et al. 2008 [194] | Metal | Solid/Hollow | Triangular tip/Tapered shaft | L = 245, 300 μm D = 200, 300 μm Beveled angle = 45° Db = 250 μm | 4 × 4 6 × 6 9 × 9 | Waters HPLC System | Human skin | Surface micromachining/Etching | Transdermal drug delivery |
| Parker et al. 2007 [168] | Ti | Hollow/In- plane | Spare/Sharp tip | L = 500, 750, 1000 μm Wb = 100 μm Tip taper angle = 60° | 10 needles | Fluidic/Structural analysis | Pressure Testing apparatus | Bulk micromachining/Multilayer lamination | Drug delivery |
| Shibata et al. 2007 [162] | SiO2 | Hollow/Out-of- plane | Circler tip/Cylindrical | L = 77 μm, Do = 5.5 μm Di = 3.5 μm | Not reported | Structural analysis | Gelatin | Photolithography/DRIE | Cell surgery |
| Kim and Lee 2007 [195] | Metallic | Hollow/Out-of- plane | Tapered tip | L1 = 200 μm T = 10 μm L2 = 400 μm T = 20 μm Tapering angle < 5° | 10 × 10 | Fluidic analysis | Not reported | SU-8 based UV LIGA | Drug delivery/Body fluid sampling |
| Tsuchiya et al. 2005 [171] | Ti | Hollow/Out-of- plane | Cylindrical | L = 1 mm Di = 25 μm Do = 60 μm | Not reported | Fluidic analysis | Not reported | Sputter deposition | Blood extraction |
© 2011 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ashraf, M.W.; Tayyaba, S.; Afzulpurkar, N. Micro Electromechanical Systems (MEMS) Based Microfluidic Devices for Biomedical Applications. Int. J. Mol. Sci. 2011, 12, 3648-3704. https://doi.org/10.3390/ijms12063648
Ashraf MW, Tayyaba S, Afzulpurkar N. Micro Electromechanical Systems (MEMS) Based Microfluidic Devices for Biomedical Applications. International Journal of Molecular Sciences. 2011; 12(6):3648-3704. https://doi.org/10.3390/ijms12063648
Chicago/Turabian StyleAshraf, Muhammad Waseem, Shahzadi Tayyaba, and Nitin Afzulpurkar. 2011. "Micro Electromechanical Systems (MEMS) Based Microfluidic Devices for Biomedical Applications" International Journal of Molecular Sciences 12, no. 6: 3648-3704. https://doi.org/10.3390/ijms12063648




