2.1. Seed Quality-Related Traits
The results of analysis of variance showed a significant difference among safflower genotypes for protein, oil, ash, fiber and fatty acid contents of seed (
Table 1).
Seed oil content of the genotypes ranged from 21% in Wht-ISF to 33.5% in Mex.2-138 (
Table 2). Regarding protein content, K
21 (25.6%) and Mex.13-216 (13.5%) possessed the highest and the lowest protein content, respectively.
Palmitic acid of safflower genotypes ranged from 6.49% in Mex.13-216 to 11.07% in ISF
14 (
Table 3). Mean of stearic acid content (%) varied from 1.43 in Mex.13-216 to 2.94 in GE
62918 genotype (
Table 2). With considering the breeding aims in decreasing these two saturated fatty acids, using Mex.13-216 genotype could be recommended. Oleic acid content showed the highest variation in the studied genotypes. Oleic acid ranged from 14.1 (ISF
14) to 35.26% (Mex.22-191) (
Table 2). Linoleic acid content varied from 55.8% belonging to Mex.22-191 to 75.5% belonging to A
2 genotype (
Table 2). The ranges of 2–4%, 6–8%, 16–20% and 70–75% for stearic acid, palmitic acid, oleic acid and linoleic acid for safflower cultivars have been reported by other workers [
32–
34]. These discrepancies in protein, oil and fatty acid composition of seed of the safflower genotypes reported here and elsewhere were mainly due to the genes and environmental effects that could jointly influence these traits in safflower [
4]. Further research to resolve these discrepancies is needed to understand the direction of gene effects on the one hand, and the knowledge of interplay between the genes and environments on the other.
Seed ash content of the genotypes ranged from 3.10% belonging to ISF
28 to 0.92% belonging to Mex.13-216 (
Table 2). The safflower genotypes also varied significantly for fiber content ranging from 33% in Mex.17–45 to 42.4% in K
21 (
Table 2). The high variation observed for the biochemical traits of the seed of safflower genotypes could be attributed to the influences of these traits by genotype, environment and their interactions [
33].
Calculation of genetic coefficient of variation revealed that the highest and the lowest of genetic variations belong to seed ash and fiber content, respectively (
Table 2). Based on phenotypic coefficient of variation, the highest and the lowest values belonged to oleic acid content (%) and fiber content (%), respectively (
Table 2). This result showed that fiber content is the least environmentally affected seed quality trait.
Pearson correlation coefficient was calculated for the seed quality-related traits (
Table 3). The highest negative relationship was observed between oleic acid and linoleic acid content (
r = −0.98 **). The negative association between oleic acid and linoleic acid was also reported in safflower by Fernandez-Martinez
et al. [
32], and Mahasi
et al. [
29]. A negative significant correlation was observed between oil content and stearic acid (
r = −0.50 *). This result is in agreement with that of Johnson
et al. [
34], who observed a significant and negative correlation between stearic acid and oil content (%) in safflower genotypes. Palmitic acid correlated with oleic acid significantly and negatively (
r = −0.51 *). A positive significant correlation (
r = 0.54 *) was observed between protein content and palmitic acid. Strong positive relationship was found between protein content and ash content (
r = 0.66 **). Protein content and fiber content was also correlated significantly (
r = 0.54 *). Revealing the relationships between seed quality-related traits could help in planning effective breeding strategies for simultaneous improvement of these traits in safflower.
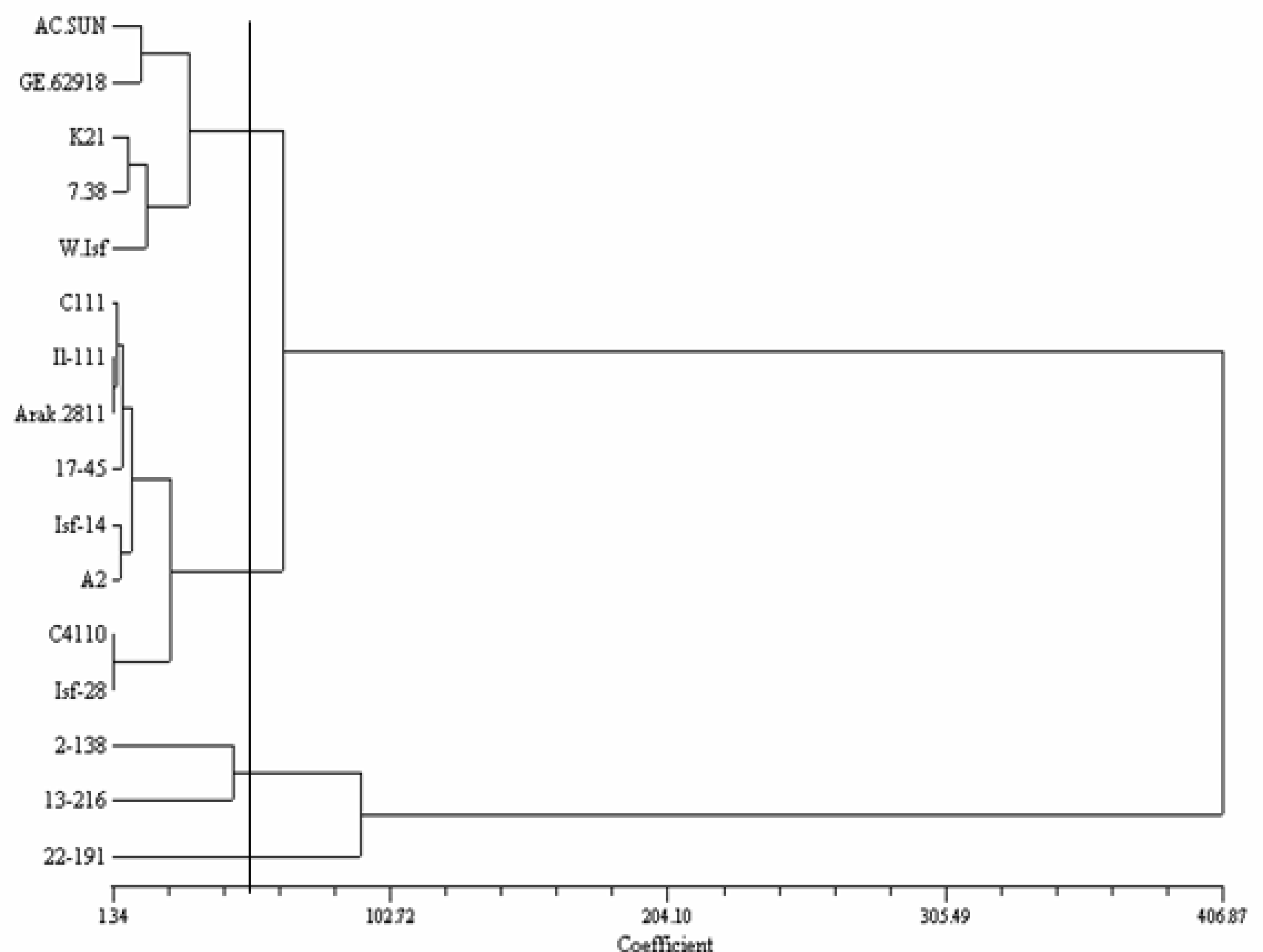
Clustering based on seed quality-related traits divided the genotypes into four groups (
Figure 1). Group 1 includes GE
62918 from Germany, AC-Sunset from Canada, Mex.7–38 from Mexico and two Iranian lines (K
21 and Wht-ISF). Group 2 was the largest group and includes 5 Iranian lines (IL.111, Arak-2811, ISF
14 and A
2) and a Mexican cultivar (Mex.17-45). Group 3 only possesses two Mexican cultivars (Mex. 2-138 and Mex.13-216). Group 4 includes a Mexican cultivar (Mex.22-191).
There was a broad genetic variation among safflower genotypes for fatty acids, oil and protein content. These variations in the seed quality-related traits imply a considerable potential of the studied genotypes for safflower improvement. Also, the present study showed oleic type genotypes including Mex.7-38 and Mex.22-191 (
Table 3). An Iranian line (A
2) originated from a cold climate of Iran (Azarbayjan province) has an elevated linoleic content (75.5%). This finding might be related to the impact of temperature and geographical origin in the diversity of fatty acid contents [
33]. Genetic variation for fatty acids and biochemical traits was also reported in safflower by other studies [
8,
22,
34]. Variation of unsaturated fatty acids in safflower oil points out the possibility of improving oil quality through the breeding programs [
10]. Although, genotypes from different geographical regions clustered in a same group (
Figure 1), two Mexican genotypes created a separate cluster. Moreover, Mex.22-191, as an oleic type genotype, separated from the other genotypes. Considering the results of cluster analysis, genotypes from distant clusters, could be used to produce superior hybrid with elevated seed quality-related traits. In this study, a clear relationship between diversity patterns and geographical origin was not revealed. The lack of relationship of the genotypic clusters for seed quality-related traits with the geographical origins was also reported by others [
11].
2.2. ISSR Analysis
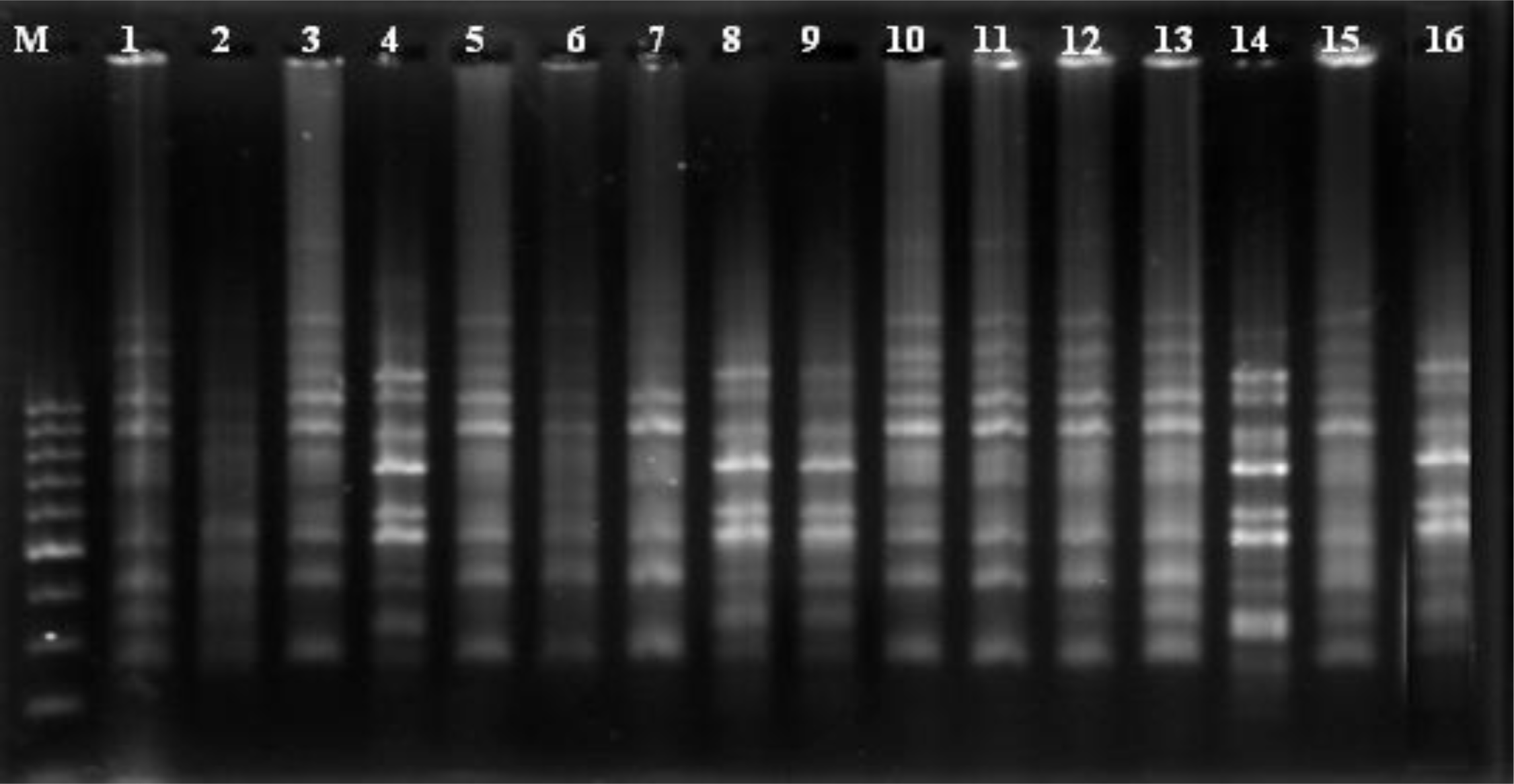
Out of 30 ISSR primers used, 20 showed polymorphism.
Figure 2 shows the polymorphic fingerprinting pattern of the 16 safflower genotypes generated by ISSR primer number 16.
A total of 204 bands were scored for 20 ISSR primers (
Table 4). With an average of 10.2 bands per primer, a total of 204 bands generated and 149 bands (73%) of these were polymorphic. Total generated band ranged from 5 to 16 and the polymorphic bands of the primers ranged from 3 to14 bands. The mean of polymorphic bands for (GA)
n, (AC)
n, (AG)
n, (CT)
n, (TCC)
5, (TCC)
7 and (TG)
n primers was 12, 11, 10.4, 9.3, 8.5 and 8, respectively. The mean of polymorphism for (CT)
n, (AC)
n, (TCC)
7, (TCC)
5 and (TG)
n was 83%, 76.5%, 69.5%, 58.66% and 50%, respectively. Two motif primers of (CT)
n, (AC)
n and (AG)
n types produced the highest mean of polymorphism. The un-weighted pair-group method with arithmetic mean (UPGMA) cluster analysis showed that 16 safflower genotypes were grouped into four marker-based groups (
Figure 3). Cluster 1 comprises AC-Sunset, a Canadian genotype. Cluster 2 includes 9 Iranian lines from different geographical regions that compromised from Northern, Western and Central regions of Iran. Cluster 3 possessed 3 Mexican genotypes includes Mex.7-38, Mex.2-138 and Mex.22-191. Finally, cluster 4 contains 2 Mexican genotypes (Mex.13-216 and Mex.17-45) accompanied with GE
62918 from Germany.
Principal coordinate analysis (PCoA) for displaying the relationships among the safflower genotypes was performed. The first three principal coordinates explained 75.56% of the total variation, with 59.82% explained by the first, 9.48% by the second and 6.25 by the third at the DNA sequence level (
Table 5).
The ISSR primers used in this study revealed an acceptable genomic variation among the selected genotypes. The relationships between the genetic distances estimated based on seed quality traits and molecular markers was not statistically significant (r = 0.13).
Analysis of molecular variance (AMOVA) revealed a non-significant difference between two groups of genotypes (native vs. exotic). AMOVA for ISSR data also indicated significant variations within the genotypic groups.
Similar to seed quality-related traits, high molecular genetic variation was observed which is in agreement with the observation of high efficiency of ISSR marker in detecting genetic variation in safflower by Yang
et al. [
27]. Assessment of genetic diversity, based on both molecular markers and seed quality-related traits including biochemical traits and fatty acid composition, has only been employed in a few tree species [
12,
13].
In this study, the most polymorphic bands was related to (CT)
n, (AC)
n, and (AG)
n primers. Similar to our results, Yang
et al. [
27], has reported that (AC)
n and (CT)
n ISSR primers produced the highest polymorphic bands in safflower. In this study, (GT)
n primers have not produced any band, this finding was in agreement with Yang
et al. [
27]. On the other hand, in rice Blair
et al. [
15], have reported that (GT)
n primers have produced a high frequency of banding pattern because of additional nucleotide in 5′ terminal sequence. Because of the high frequency of polymorphic band generated by (GA)
n, (CT)
n and (AC)
n primers, it can be deduced that these motifs have a significant contribution in the safflower genome.
The cluster analysis based on ISSR data showed that there was a considerable agreement between geographic origin and
their genomic similarities. However, in viewpoint of molecular clustering, similarities in genotypes that grouped in the same cluster could also arise because of sharing a common parentage, convergent evolution and selection. According to
Figure 3, GE
62918 (from Germany) and AC-Sunset (from Canada) have clustered in a same group with two Mexican cultivars. They could probably have originated from a common ancestor or probability of being duplicates [
11]. A non-significant correlation between seed quality-related traits and ISSR markers in the Mantel test [
35] suggested there was a slight similarity between these clusterings. This could be due to the exchange of plant material across the regions during the evolution of safflower [
11]. Another reason could be that ISSR markers detect polymorphism in coding and non-coding regions of genome, but seed quality-related traits are the results of expressed sections of the genome [
11].
Although some work based on morphological traits has been conducted to assess genetic variation in safflower genotypes, the results are still ambiguous, because phenotypic traits are affected by developmental stages and environmental conditions. On the other hand, ISSR markers overcome those disadvantages to a certain extent. The results of the present study are consistent with those of Yang
et al. [
27], who emphasized that ISSR, is an effective marker system for detecting genetic diversity among safflower genotypes and provides useful information about the phylogenic relationships.