1. Introduction
Bacteriocins (ribosomally synthesized antimicrobial peptides) and bacteriocin-like inhibitory substances (BLIS) are produced by several species of bacteria, including lactic acid bacteria that are generally recognized as safe [
1–
3]. Bacteriocins are diverse peptides and can be divided into the Class I (lanthionine-containing post-translationally modified bacteriocins), Class II (non-lanthionine-containing bacteriocins that do not undergo extensive post-translational modification) and the bacteriolysins [
1]. Each of these groups can be further sub-divided. Bacteriocins are membrane active peptides and most form pores in the cell membranes of sensitive bacteria due to the cationic and amphiphilic nature of these peptides, however there are exceptions [
4]. Pore formation leads to the disruption of the proton motive force (PMF) and the efflux of intracellular material [
5]. Bacteriocins can have a narrow (active against closely related species) or broad spectrum of activity [
6,
7]. Class Ia bacteriocins, or lantibiotics, use lipid II as target for their mode of action and kill sensitive bacteria by pore formation or by hampering cell wall formation [
8]. Lantibiotics can also inhibit the germination of spores [
8] Class IIa, or pediocin-like bacteriocins, mainly form pores in the membranes of sensitive cells [
1]. By disrupting the PMF, cells cannot secrete antibiotics via transport systems located in their cell membranes. This could be an important answer to our search towards more effective ways of infection control. More than 95% of
Staphylococcus aureus isolates are resistant to penicillin. Of these, 60 to 70% are resistant to methicillin [
9].
A few bacteriocins are active against methicillin-resistant strains of
Staphylococcus aureus (MRSA), and
versus other bacteria of interest in human health, such as
Pseudomonas aeruginosa,
Streptococcus pneumonia and vancomycin-resistant enterococci [
10,
11]. Bacteriocin ST4SA, produced by
Enterococcus mundtii, is a Class IIa bacteriocin and is active against several pathogens, including
Acinetobacter baumannii,
Pseudomonas aeruginosa,
Streptococcus pneumonia,
Staphylococcus aureus and
Enterococcus faecium [
5]. Bacteriocin ST4SA survived conditions in the human and animal gastro-intestinal tract [
12,
13] and was constitutively expressed [
14]. Nisin F, produced by
Lactococcus lactis, was effective in the treatment of
Staphylococcus aureus in respiratory tract infections [
15]. Plantaricin 423, produced by
Lactobacillus plantarum, displaced
Clostridium sporogenes and
Enterococcus faecalis from Caco-2 cells [
16]. Plantaricin 423 is also a Class IIa bacteriocin and shows activity against
Bacillus cereus,
Clostridium sporogenes,
Enterococcus faecalis,
Lactobacillus brevis (
Lactobacillus spp.),
Listeria innocua, Listeria monocytogenes,
Propionibacterium sp.,
Propionibacterium acidipropionic and
Staphylococcus carnosum [
17]. Bacteriocins may thus offer an alternative to control bacterial infections, administered alone or in combination with antibiotics [
5]. The challenge would be to protect bacteriocins from proteolytic enzymes, keep the peptides in contact with the infected surface and extend the contact time for as long as possible.
Electrospun nanofibers, prepared from selected polymers, may be used as carrier matrix for bacteriocins and other antimicrobial peptides [
18]. Nanofibers have been used to release antibiotics, proteins, growth factors, silver nanoparticles, plasmid DNA, as well as viable cells [
18–
26]. Specific structures can be designed, such as oxygen permeable membranes, matrices with a high surface to volume ratio and scaffolds that are morphologically similar to the extracellular matrix of skin [
27–
31].
In this paper we studied the feasibility of electrospinning bacteriocins into nanofibers, as well as investigating the nanofibers as a delivery system. Plantaricin 423 and bacteriocin ST4SA were used as model bacteriocins (Class IIa) in this study. These bacteriocins were dissolved in N,N-dimethylformamide (DMF) and electrospun into biodegradable nanofiber blends of poly(d,l-lactide) (PDLLA) and poly(ethylene oxide) (PEO). The nanofiber scaffolds were evaluated (in vitro) for the release of the peptides and antimicrobial activity.
3. Experimental Section
3.1. Isolation of Antimicrobial Peptides
Lactobacillus plantarum 423 and
Enterococcus mundtii ST4SA were cultured, separately, in MRS broth (Biolab, Biolab Diagnostics, Midrand, South Africa) for 24 h at 37 °C. Cells were harvested (8000× g, 10 min, 4 °C), the pH of the cell-free supernatant adjusted to between pH 6.5–7.0 with 10 M NaOH and then heated at 80 °C for 10 min to inactivate proteolytic enzymes. Peptides were precipitated from the cell-free supernatant with 70% saturated ammonium sulfate [
32] and desalted against distilled water by using a 1000 Da cut-off dialysis membrane (Spectrum Inc., CA, USA). Protein concentrations were determined by using the Micro-bicinchoninic Acid (BCA) kit (Pierce, Rockford, IL). Readings were recorded at 562 nm. Samples were freeze-dried and stored at −20 °C.
3.2. Electrospinning of Nanofibers
Freeze-dried peptides were dissolved in
N,
N-dimethylformamide (DMF) and centrifuged (6000× g, 1 min) to obtain the supernatants. From each of these supernatants, 2 mL was used as solvent for PDLLA (20–24%, w/v, Sigma-Aldrich) blended with poly(ethylene oxide) (PEO, 200,000 Da, Sigma-Aldrich) at a ratio of 10:90, 30:70 and 50:50, respectively. Solutions were heated to 40 °C on a hot plate and then electrospun using a gravity system described by [
20]. A constant electric field of +10 kV was applied to the polymer solution and −5 kV to the collector. The collector was placed 15 cm from the polymer solution. The relative humidity was kept constant between 50–55%.
3.3. Scanning Electron Microscopy of Electrospun Fibers
Images of the nanofibers were recorded with a Leo® 1430VP Scanning Electron Microscope (SEM). Samples were coated with a thin layer of gold to increase conductivity. Fiber diameter was determined by using the SEM Image Studio Software (version 10.1).
3.4. Fluorescent Labeling of Antimicrobial Peptides and Polymers
Freeze-dried plantaricin 423 (20 mg) was suspended in 1 mL DMF and the supernatant was reacted with 20 mg rhodamine B isothiocyanate (RBITC) or fluorescein isothiocyanate (FITC) from Sigma-Aldrich, dissolved in the same volume. The solution was placed on an orbital shaker and incubated in the dark at 4 °C for 48 h. Unbound fluorescent marker was removed by dialyzing against sterile distilled water for 3 weeks at 4 °C. Dialyses bags with 1000 Da cut-off (Spectrum Inc., CA, USA) were used. Labeled peptides were freeze-dried, dissolved in DMF and electrospun into nanofibers as described before.
PEO (0.2048 mg) was dissolved in 2 mL sterile distilled water containing RBITC (20 mg/mL). The polymer solution was placed on an orbital shaker and incubated in the dark at 25 °C for 48 h. Unbound fluorescent marker was removed through dialysis as described before and freeze-dried. PDLLA (0.2048 mg) was dissolved in 2 mL DMF and was labeled with FITC (20 mg/mL) for 48h on an orbital shaker and incubated in the dark at 25 °C.
The labeled peptides and polymer solution were electrospun into nanofibers as described before. Nanofibers were excited at 472 nm and 572 nm, respectively, by using a Xenon-Arc burner light source (Olympus Biosystems GMBH). Fluorescent images were captured with an Olympus Cell^R system attached to an IX-81 inverted fluorescent microscope. An Olympus Plan Apo N 60×/1.4 Oil objective was used. Images were taken with an F-view-II cooled CCD camera (Soft Imaging Systems) and the background subtracted using the Cell^R imaging software.
3.5. Differential Scanning Calorimetry (DSC)
Thermal analysis was carried out with a TA Q100 Differential Scanning Calorimeter (DSC) (TA Instruments) which was calibrated against an indium standard. An empty aluminium pan was the reference. Fiber samples (15–18 mg) were run at a 10 °C/min in three cycles under nitrogen atmosphere (flow rate 50 mL/min). In the first cycle, samples were heated from −80 °C to 180 °C and isothermed for 1 min; cooled back to −80 °C and isothermed for 5 min. Finally, the samples were heated for a second time at 10 °C/min to 180 °C. The crystalline melting temperature (Tm) and the glass transition temperature (Tg) were recorded in the second heating cycle.
3.6. Release of Antimicrobial Peptides and Degradation of Nanofibers
Nanofibers (25 mg) were placed in 2 mL sterile phosphate buffered saline (PBS, pH 7.4) and incubated at 37 °C on an orbital shaker (30 rpm). At pre-determined time intervals the PBS was replaced with 2 mL sterile PBS (pH 7.4). The protein concentration in each sample was determined by using the BCA protein assay kit as described before. The concentration of bacteriocin in the nanofibers was determined by dissolving 25 mg fibers in DMF. This suspension was diluted in PBS 7.4 before the protein concentration was determined by using the BCA protein assay kit as described before. Nanofibers were freeze-dried and then visualized by SEM. Changes in the weight of nanofibers were recorded.
3.7. Antimicrobial Activity Tests
In a separate experiment, 25 mg of PEO 50:PDLLA 50 nanofibers containing plantaricin 423 was placed in 50 mL MRS broth, inoculated with Enterococcus faecium HKLHS (105 cfu/mL) and incubated at 37 °C for 12 h. Changes in optical density readings were recorded at 600 nm.
4. Discussion
Electrospinning is a versatile technique to produce large amounts of fibers, in the micrometer to nanometer range, in a short amount of time. A variety of natural and synthetic polymers have been electrospun into fibers and some have been used to control the release of various compounds [
13,
19,
22,
26,
31,
33]. Blending hydrophilic and hydrophobic polymers directly in a suitable solvent before electrospinning, is an easy way to change the hydrophobicity, thermal stability and mechanical strength of the fibers [
34]. Since blending is an attractive way to modify polymer properties, this method was chosen to modify the hydrophobicity/hydrophilicity of the electrospun fibers by the addition of PEO, to help facilitate the release of the bacteriocin from the nanofibers.
Fibers were all in the nanometer range and addition of bacteriocin did not influence the fiber structure as can be seen in
Figure 1.
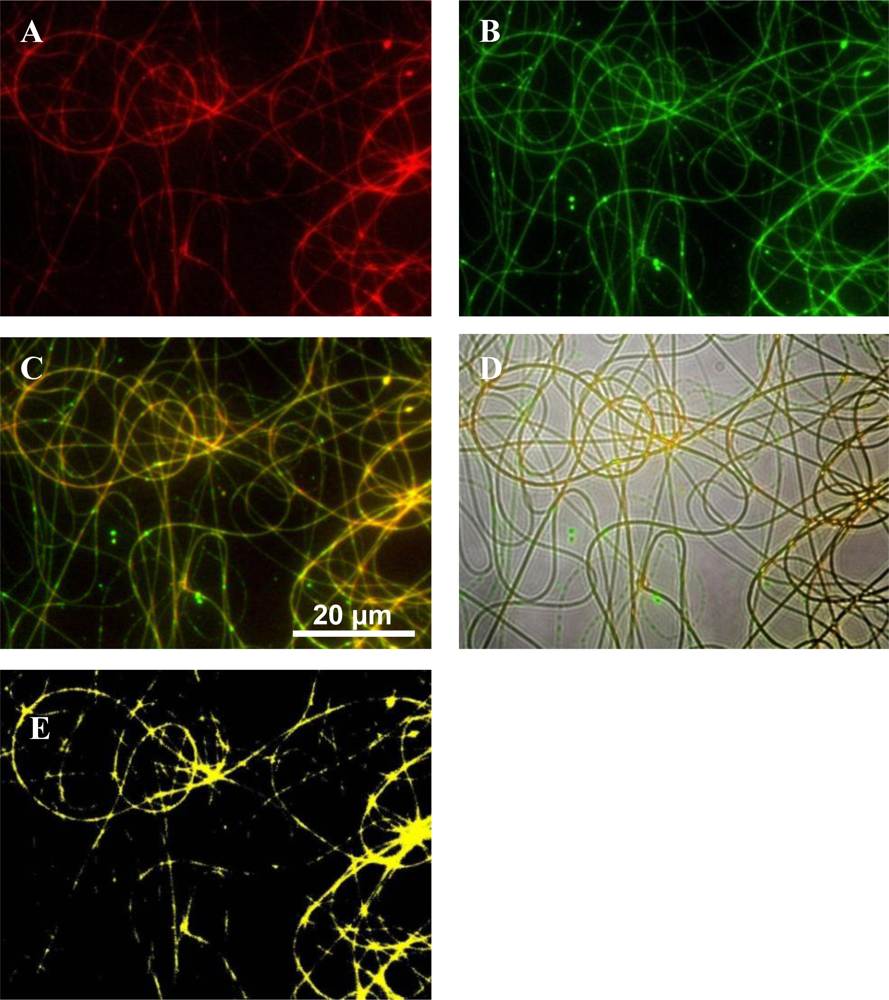
Polymers were labeled to elucidate fiber morphologies after electrospinning. Fluorescent microscopy images showed that there is a large degree of miscibility between the PEO and PDLLA when these polymers are used in a 50:50 combination (
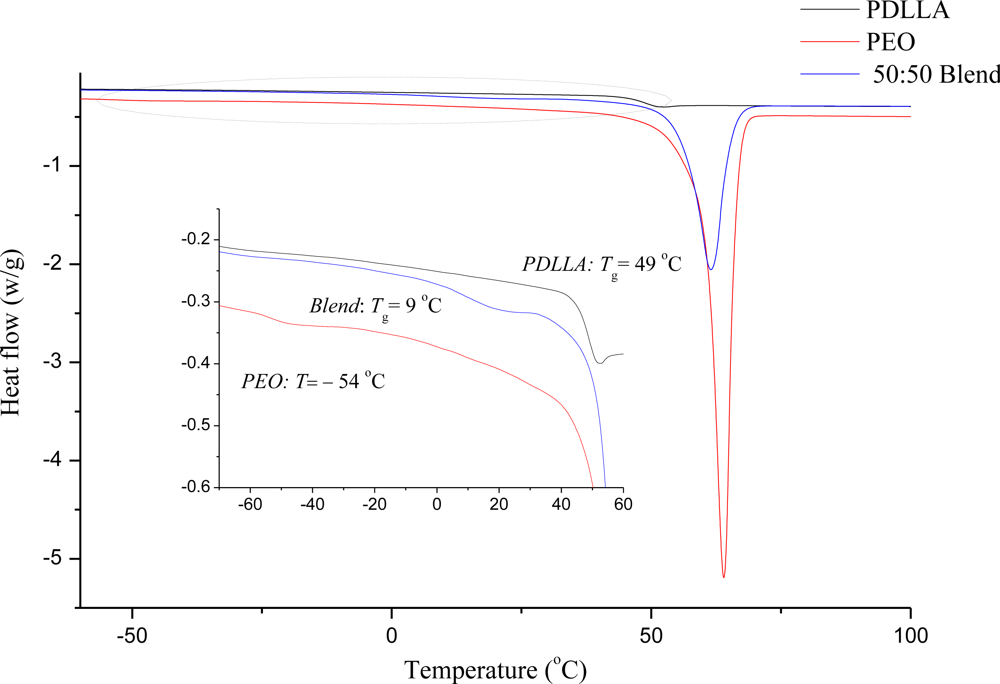
Figure 2). However, to confirm these results and to determine the miscibility of the polymers, DSC analysis was performed on the PEO 50:PDLLA 50 fiber blend, see
Figure 3. This fiber blend was chosen as it retained its structure the best, had the least amount of weight loss and still released enough bacteriocin to inhibit the growth of a sensitive strain. Nijenhuis
et al. [
34] have studied thermal properties using DSC of polymer blends containing various ratios of PEO and the poly
l-lactide (PLLA) isomer. The results showed that blends containing up to 50 wt% PEO were miscible in the amorphous phase. In this work, PDLLA which is an amorphous grade unlike the semi crystalline poly (
d-lactide) PDLA was used. A good indication of a miscible polymer blend is the shift of the
Tg to a value between the
Tgs of the pure polymers. The
Tg value of a mixed phase is determined by the weight fraction of each polymer in that phase, and by the
Tgs of the pure polymers. DSC curves of the second heating cycle of the PEO 50:PDLLA 50 blend and the corresponding homopolymers are shown in
Figure 3. A single
Tg (9 °C) can be seen that is between the
Tgs values for PEO (−54 °C) and PDLLA (49 °C) which suggests that the PEO 50:PDLLA 50 blend is miscible in the amorphous phase. DSC analysis performed on the PEO 50:PDLLA 50 blend, containing plantaricin 423, revealed that the
Tg value still fell between the
Tg of the component polymers and is therefore still miscible to an extent. It could be due to the peptide that is more associated with the PEO domains or simply restricting chain mobility. Investigation of the distribution of bacteriocins in the final fiber structure after electrospinning could yield important information in designing polymeric drug delivery systems for these peptides.
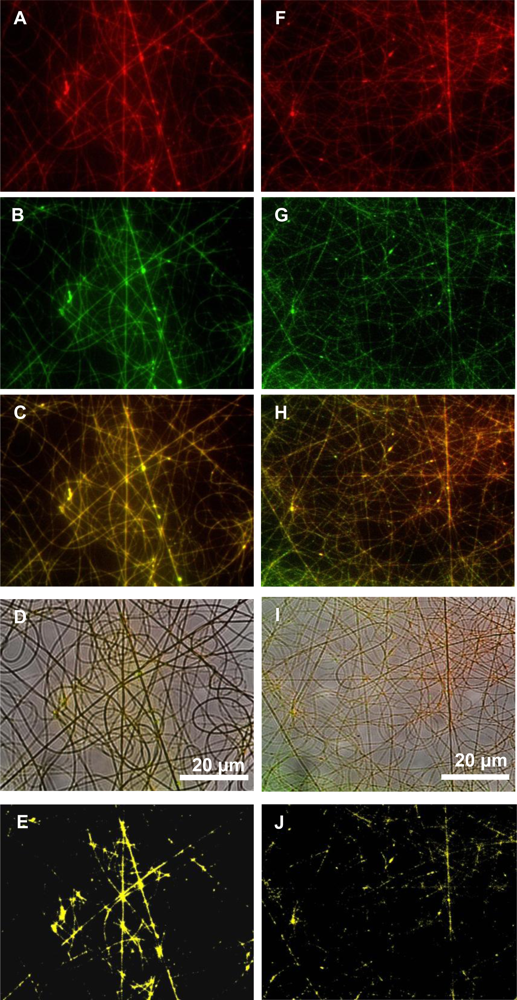
Fluorescent microscopy images revealed that the bacteriocin was better associated with PEO than with PDLLA, even though the peptide was associated with both these polymers (
Figure 4). Bacteriocins are amphiphilic peptides with both hydrophilic and hydrophobic residues [
1]. Because of this amphiphilic nature it is possible for the peptide to interact better with one polymer than with the other. These results indicate that the bacteriocin is better associated with the hydrophilic polymer, in this case PEO, but were also associated with the PDLLA to a lesser extent. This would explain the high initial burst release within the first few hours.
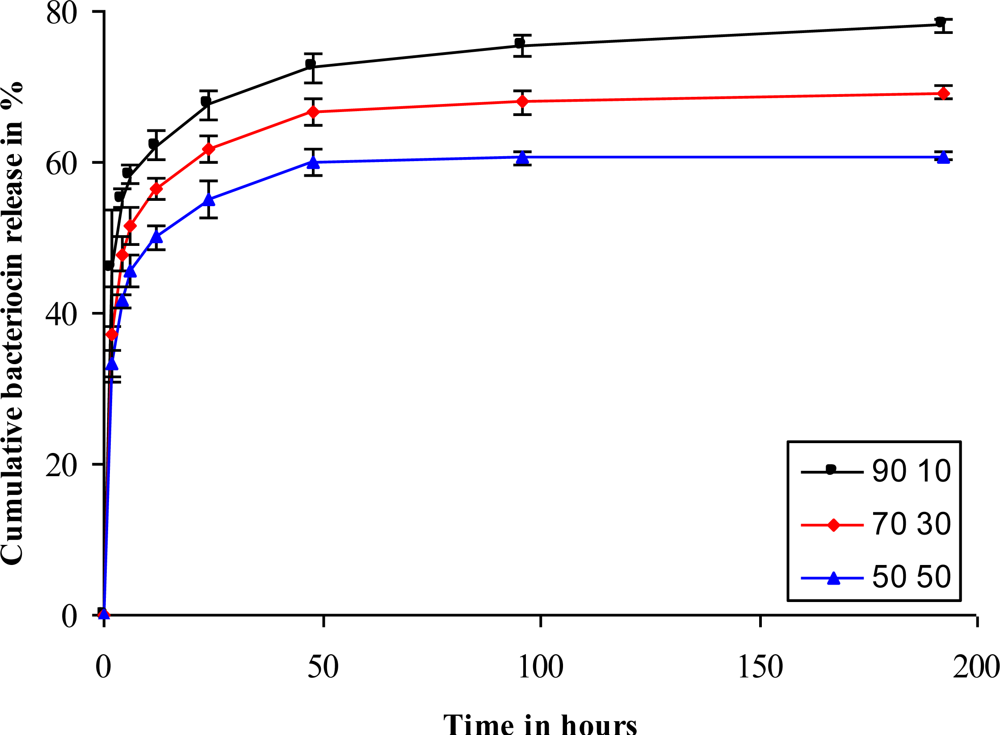
Cumulative release studies revealed that fibers with a higher PEO content showed a higher release of the bacteriocin than those with a lower PEO content (
Figure 5). An initial burst release was seen within 2 h, after which a more sustained release was seen. This could be an ideal release profile in the control of infections. A rapid release of antimicrobial peptides during the first 2 h would kill most of the pathogens, whereas a slow and constant release over the following few days would keep infection under control. The PEO 70:PDLLA 30 and PEO 50:PDLLA 50 blends released lower amounts of the peptide, with the PEO 70:PDLLA 30 and PEO 50:PDLLA 50 blends releasing 69% and 60% of the peptides, respectively (
Figure 5). Blends of different polymers have earlier shown to facilitate the release of molecules from electrospun fibers. Lysozyme release from nanofibers was facilitated by blending PEO and poly(ɛ-caprolactone) (PCL), with a 90:10 blend releasing most of the lysozyme with a high initial burst release [
22]. Higher concentrations of cytochrome C were also released from fibers consisting of poly(
l-lactide) PLLA and high concentrations of both poly(
l-lysine) (PLL) and poly(ethylene imine) (PEI), respectively [
26]. Fibers containing 50% PLL released most of the protein (75%) with a high initial burst release.
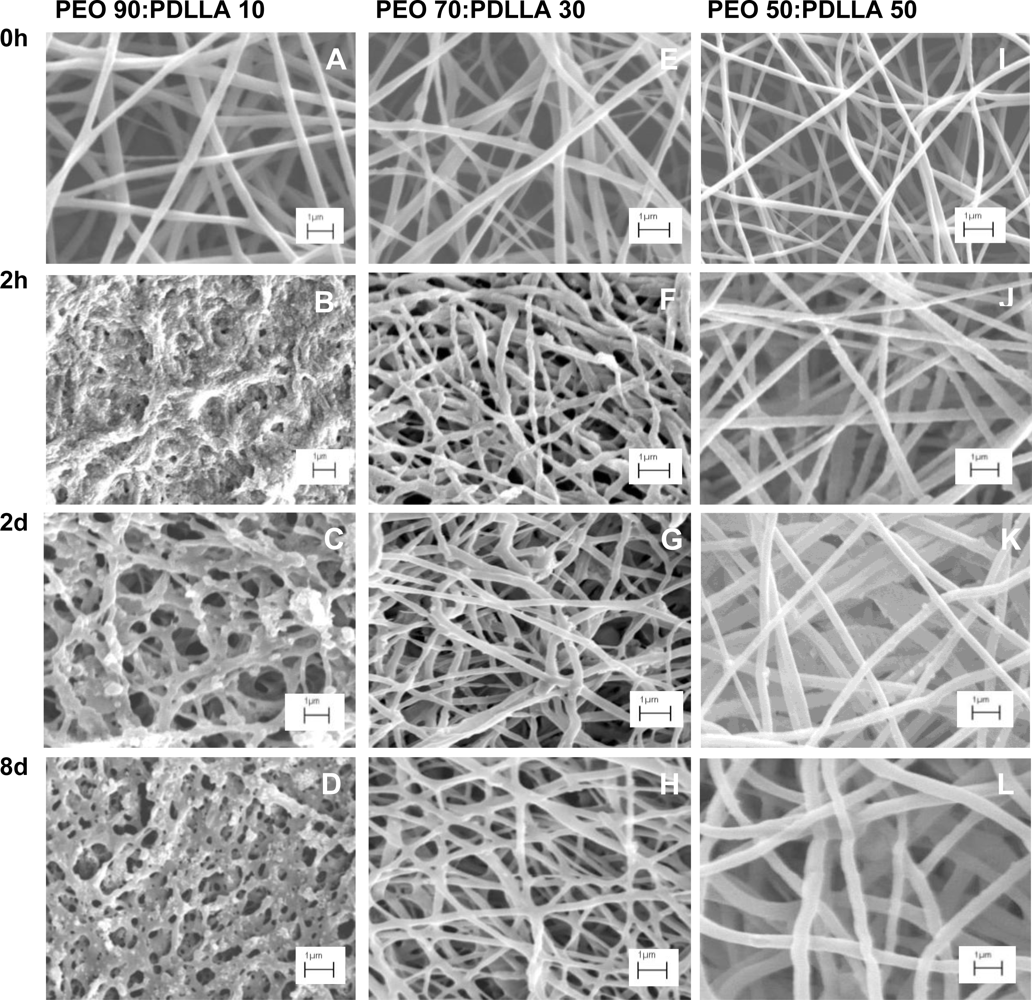
SEM images revealed that as the PEO content increased, significant morphological changes can be seen as the incubation time increased (
Figure 6) The PEO 90:PDLLA 10 blend showed the most significant morphological changes over the 8 day period. This could be due to the high content of PEO in the fibers as PEO swells readily in an aqueous environment and dissolves easily. The resulting destruction of the nanofiber scaffold explains the higher release of antimicrobial peptide as observed over 8 days (
Figure 5). Fibers retained their structure better in the PBS buffer (pH 7.4), as the PEO content in the fibers decreased. This can be observed in the PEO 50:PDLLA 50 blend, with this blend showing no significant signs of structural changes or deterioration in the buffer medium (
Figure 6I–L). Slight structural changes were seen for the PEO 70:PDLLA 30 blend after 8 days (
Figure 6E–H). These results suggest that the PEO 50:PDLLA 50 blend nanofibers would be stable for at least 8 days and show no significant signs of degradation. The ideal nanofiber drug delivery systems has to retain its structure, have high oxygen permeability, variable pore size and a high surface to volume ratio. Nanofibers prepared from the PEO 90:PDLLA 10 blend showed very high weight loss over the 8 days period, which is not very desirable. The PEO 70:PDLLA 30 showed lower weight loss, however the PEO 50:PDLLA 50 showed the best results and lost the least amount of its original weight. It is very important that the nanofiber scaffolds retain their integrity and with this in mind, the PEO 50:PDLLA 50 would be the preferred choice as drug delivery system in this work.
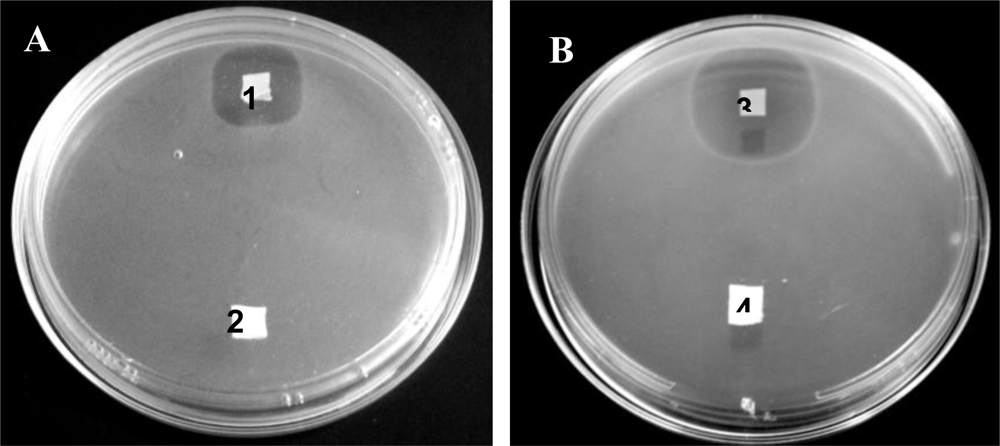
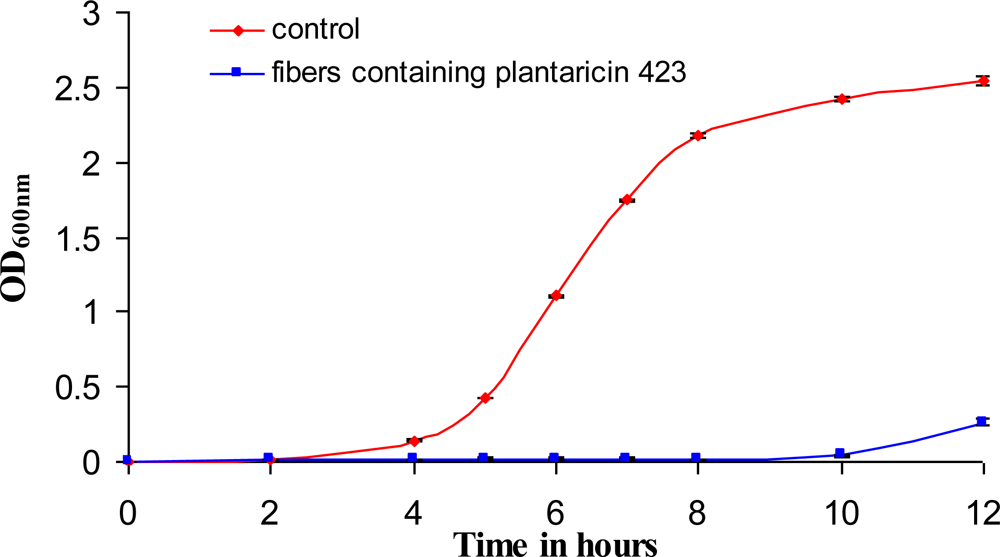
Bacteriocins retained their activity after electrospinning and were able to inhibit the growth of a sensitive strain on solid media as well as in liquid broth (
Figures 7 and
8). The bacteriocins showed activity on the solid media for at least 6 days of incubation. Growth inhibition in liquid broth was observed and may be obtained over longer periods of time by electrospinning higher concentrations of bacteriocin in the fibers, by using more fibers (weight), or by replacing the fibers in the broth with new fibers at certain time intervals. Only 25 mg of fibers, containing plantaricin 423, was able to inhibit actively growing cells of
Enterococcus faecium HKLHS, indicating the potent antimicrobial effect of this bacteriocin. The difference in antimicrobial activity and release on solid media and in liquid broth gives an indication that there is a big difference in release profile on solid media and in a buffer/liquid medium. During release studies in a buffer medium, a high concentration gradient is created (such as in the case of the method used for determining release) by replacing the old buffer with new buffer at each sampling point. This can increase the rate of diffusion of the peptide from the nanofiber scaffold to the buffer medium. The detection limit of the protein assay is also in μg/mL, whereas the bacteriocins can still be active at lower concentrations of ng/mL. However, the concentration gradient can be much lower on solid media, resulting in lower release profiles and thus leads to increased periods of antimicrobial activity.