The Effect of Zn-Al-Hydrotalcites Composited with Calcium Stearate and β-Diketone on the Thermal Stability of PVC
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of the Zn-Al-LDHs
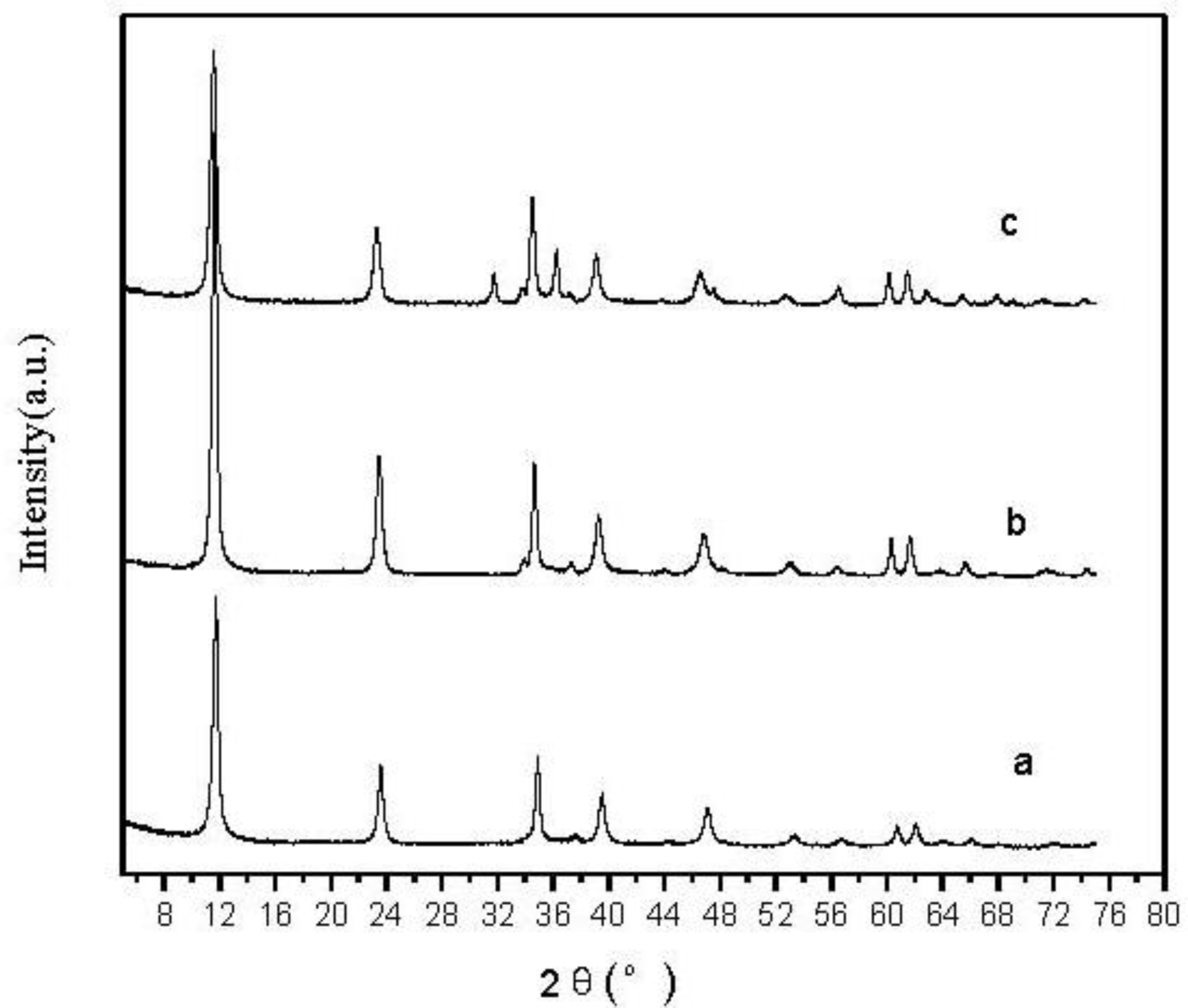
2.1.1. X-ray Diffraction (XRD) Analysis of Zn-Al-LDHs
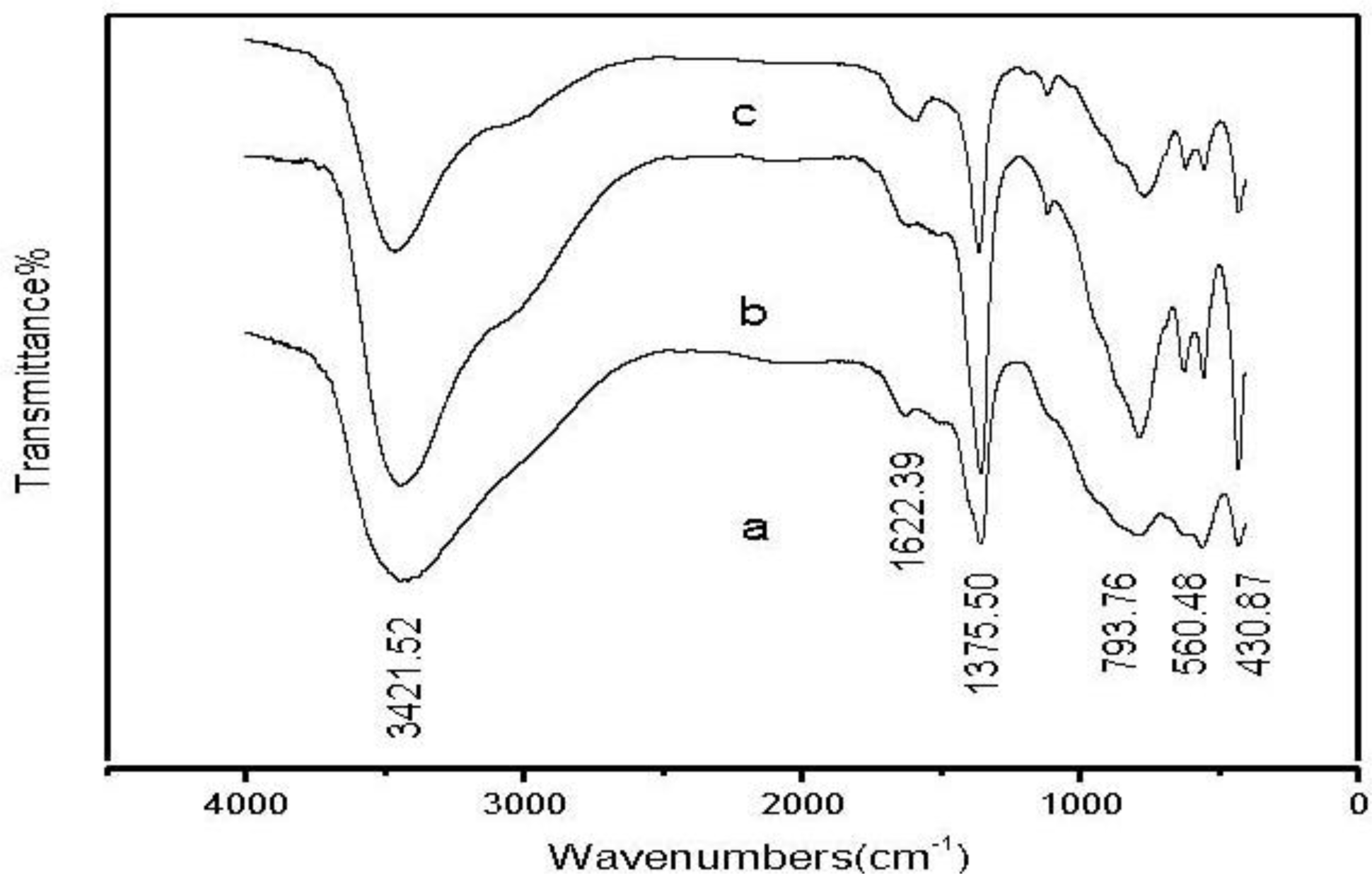
2.1.2. Fourier Transform Infrared Spectroscopy (FT-IR) Analysis of Zn-Al-LDHs
2.1.3. Scanning Electron Microscope (SEM) Micrograph of Zn-Al-LDHs
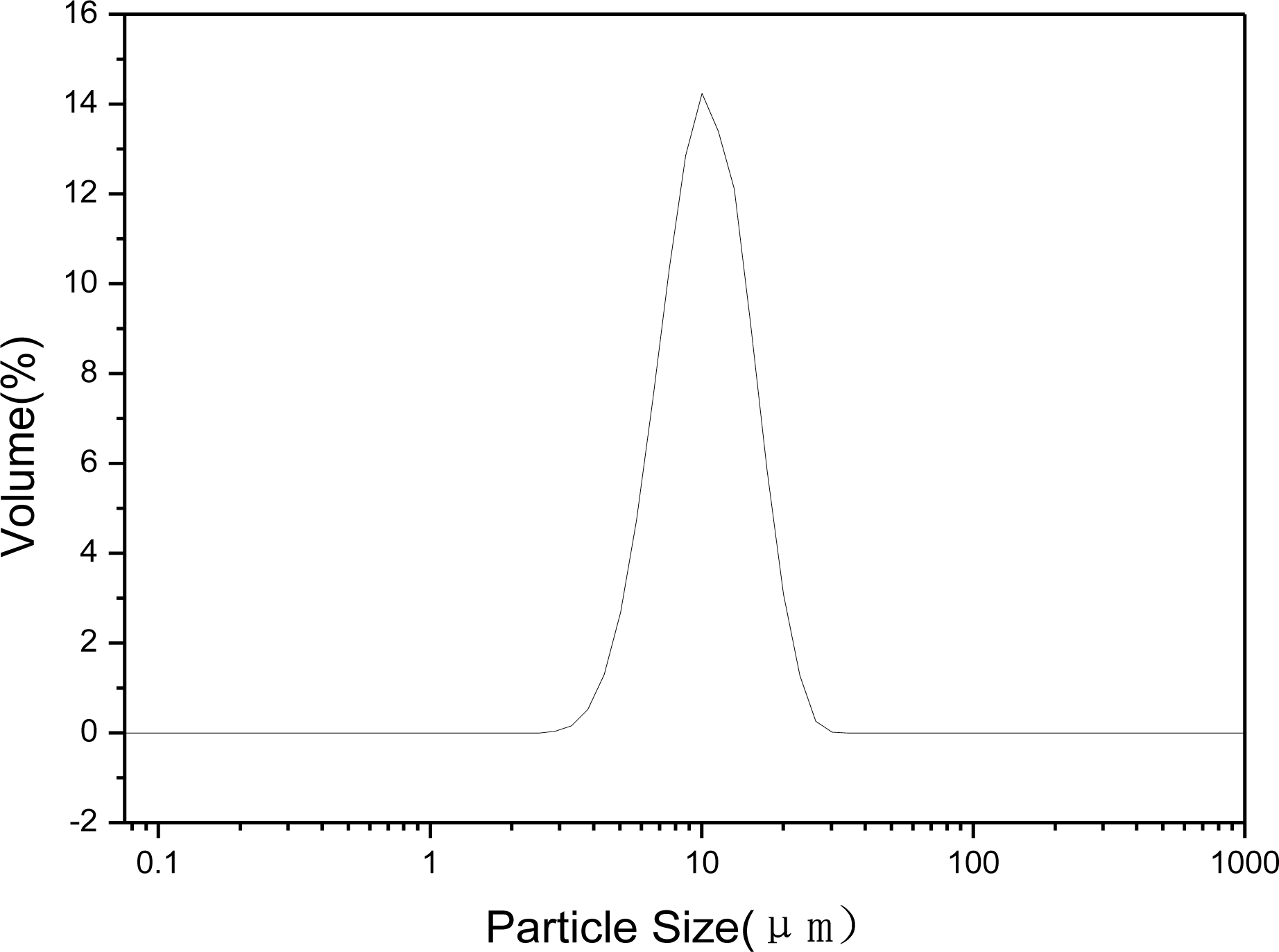
2.1.4. Particle Granularity and Specific Area Analysis of Zn-Al-LDHs
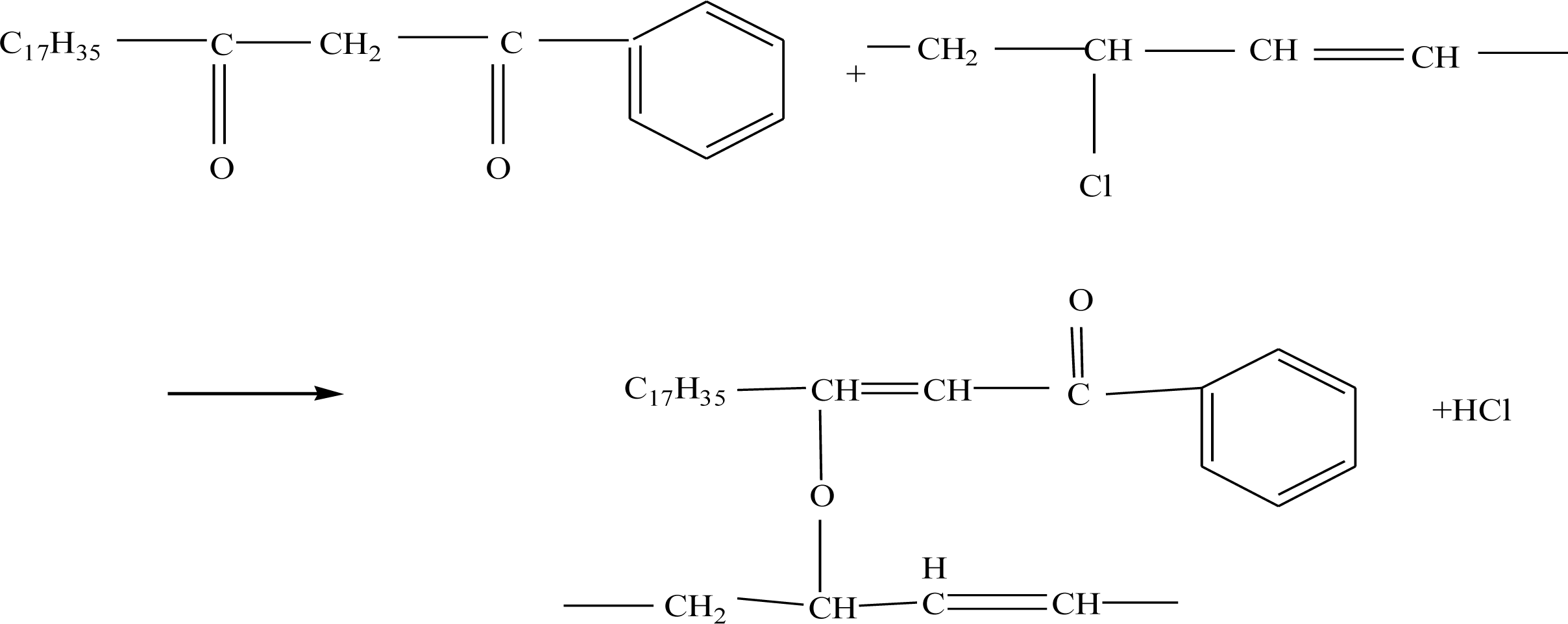
2.2. Synergetic Thermal Stabilizing Effect of Zn-Al-LDHs with Metal Soap and β-Diketone
2.2.1. The Effect of Zn-Al-LDHs, CaSt2 and β-Diketone on the Thermal Stability of PVC
2.2.2. The Effect of Varying the Amount of CaSt2 on the PVC
2.2.3. The Effect of Varying the Amount of β-Diketone on the PVC
3. Experimental Section
3.1. Materials
3.2. Synthesis of Zn-Al-CO3-LDHs
3.3. Characterization Techniques for Hydrotalcites
3.4. Static Thermal Aging Test
4. Conclusions
Acknowledgments
References
- Chen, GM. Preparation of a poly (vinyl chloride)/layered double hydroxide nanocomposite with a reduced heavy-metal thermal stabilizer. J. Appl. Polym. Sci 2007, 106, 817–820. [Google Scholar]
- Mohamed, NA; Yassin, AA; Khalil, KD; Sabaa, MW. Organic thermal stabilizers for rigid poly(vinyl chloride) I. Barbituric and thiobarbituric acids. Polym. Degrad. Stab 2000, 70, 5–10. [Google Scholar]
- Murphy, J. Heat stabilizers. Plastic. Add. Compound 1999, 1, 24–29. [Google Scholar]
- Yang, ZH; Shu, WY; Long, HZ; Li, XH. Influence of rare earth heating stabilizer on heating stabilizing property of PVC. Chin. Rare Earths 1999, 20, 60–62. [Google Scholar]
- Martin, KJ; Pinnavaia, TJ. Layered double hydroxides as supported anionic reagents halied ion reactivity in [Zn2Cr (OH)6] X·nH2O. J. Am. Chem. Soc 1986, 108, 541–542. [Google Scholar]
- Allmann, R. The crystal structure of pyroaurite. Acta. Crystallogr. B 1968, 24, 972–977. [Google Scholar]
- Taylor, HFW. Crystal structures of some double hydroxide minerals. Mineral. Mag 1973, 39, 377–389. [Google Scholar]
- He, J; Wei, M; Li, B; Kang, Y; Evans, DG; Duan, X. Preparation of layered double hydroxides. Struct. Bond 2006, 119, 89–119. [Google Scholar]
- Li, F; Duan, X. Applications of layered double hydroxides. Struct. Bond 2006, 119, 193–223. [Google Scholar]
- Vaccari, A. Preparation and catalytic properties of cationic and anionic clays. Catal. Today 1998, 41, 53–71. [Google Scholar]
- Vaccari, A. Clays and catalysis: A promising future. Appl. Clay Sci 1999, 14, 161–198. [Google Scholar]
- Van der Ven, L; Van Gemert, MLM; Batenburg, LF; Keern, JJ; Gielgens, LH; Koster, TPM; Fischer, HR. On the action of hydrotalcite-like clay materials as stabilizers in polyvinylchloride. Appl. Clay Sci 2000, 17, 25–34. [Google Scholar]
- Wang, X; Zhang, Q. Effect of hydrotalcite on the thermal stability, mechanical properties, rheology and flame retardance of poly (vinyl chloride). Polym. Int 2004, 53, 698–707. [Google Scholar]
- Lin, YJ; Li, DQ; Evans, DG; Duan, X. Modulating effect of Mg-Al-CO3 layered double hydroxides on the thermal stability of PVC resin. Polym. Degrad. Stab 2005, 88, 286–293. [Google Scholar]
- Levchik, SV; Weil, ED. Overview of the recent literature on flame retardancy and smoke suppression in PVC. Polym. Adv. Echnol 2005, 16, 707–716. [Google Scholar]
- Hernandez-Moreno, MJ; Ulibarri, MA; Rendon, JL; Serna, CJ. IR characteristics of hydrotalcite-like compounds. Phys. Chem. Miner 1985, 12, 34–38. [Google Scholar]
- Yi, S; Yang, ZH; Wang, SW; Liu, DR; Wang, SQ; Liu, QY; Chi, WW. Effects of MgAlCe-CO3 layered double hydroxides on the thermal stability of PVC resin. J. Appl. Polym. Sci 2011, 119, 2620–2626. [Google Scholar]
- Xu, ZP; Saha, SK; Braterman, PS; D’Souza, N. The effect of Zn, Al layered double hydroxide on thermal decomposition of poly (vinyl chloride). Polym. Degrad. Stab 2006, 91, 3237–3244. [Google Scholar]
- Gupta, S; Agarwal, DD; Banerjee, S. Synergistic combination of metal stearates and β-diketones with hydrotalcites in poly(vinyl chloride) stabilization. J. Appl. Polym. Sci 2009, 112, 1056–1062. [Google Scholar]





| molar ratio | Wavenumber/cm−1 | ||||
|---|---|---|---|---|---|
| vibration bands of –OH | Antisymmetric stretching of CO32− | flexural vibration of CO32− | lattice vibration of (Zn2+ Al3+) | ||
| 1:1 | 3421 | 1357 | 793 | 560 | 430 |
| 2:1 | 3446 | 1357 | 785 | 555 | 429 |
| 3:1 | 3462 | 1364 | 768 | 553 | 430 |
| Serial number | LDHss/g | Time/min and color stability | ||||||
|---|---|---|---|---|---|---|---|---|
| 10 | 30 | 50 | 70 | 90 | 100 | 110 | ||
| 1 | 0 | 7 | 7 | 7 | 7 | 7 | 7 | 7 |
| 2 | 0.04 | 0 | 1 | 3 | 4 | 5 | 7 | 7 |
| 3 | 0.06 | 0 | 1 | 3 | 4 | 5 | 5 | 7 |
| 4 | 0.08 | 0 | 0 | 0 | 3 | 4 | 5 | 6 |
| 5 | 0.1 | 0 | 0 | 0 | 1 | 1 | 3 | 3 |
| 6 | 0.2 | 0 | 0 | 1 | 4 | 5 | 5 | 6 |
| 7 | 0.3 | 0 | 3 | 3 | 4 | 5 | 6 | 6 |
| Serial number | CaSt2/g | Time/min and color stability | ||||||
|---|---|---|---|---|---|---|---|---|
| 10 | 30 | 50 | 70 | 90 | 100 | 110 | ||
| 1 | 0.45 | 0 | 1 | 1 | 3 | 4 | 4 | 4 |
| 2 | 0.35 | 0 | 1 | 1 | 3 | 3 | 3 | 7 |
| 3 | 0.25 | 0 | 1 | 1 | 3 | 4 | 4 | 6 |
| 4 | 0.2 | 0 | 0 | 0 | 1 | 1 | 3 | 3 |
| 5 | 0.15 | 0 | 0 | 0 | 0 | 1 | 3 | 3 |
| 6 | 0.1 | 0 | 0 | 1 | 3 | 3 | 4 | 5 |
| 7 | 0.05 | 0 | 0 | 1 | 3 | 6 | 7 | 7 |
| Serial number | β-Diketone/g | Time/min and color stability | ||||||
|---|---|---|---|---|---|---|---|---|
| 10 | 30 | 50 | 70 | 90 | 100 | 110 | ||
| 1 | 0.35 | 0 | 1 | 3 | 3 | 5 | 7 | 7 |
| 2 | 0.25 | 0 | 0 | 0 | 0 | 1 | 3 | 3 |
| 3 | 0.15 | 0 | 0 | 1 | 3 | 4 | 5 | 6 |
| 4 | 0.05 | 1 | 1 | 2 | 2 | 3 | 4 | 7 |
| 5 | 0 | 2 | 2 | 2 | 2 | 4 | 6 | 7 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tong, M.; Chen, H.; Yang, Z.; Wen, R. The Effect of Zn-Al-Hydrotalcites Composited with Calcium Stearate and β-Diketone on the Thermal Stability of PVC. Int. J. Mol. Sci. 2011, 12, 1756-1766. https://doi.org/10.3390/ijms12031756
Tong M, Chen H, Yang Z, Wen R. The Effect of Zn-Al-Hydrotalcites Composited with Calcium Stearate and β-Diketone on the Thermal Stability of PVC. International Journal of Molecular Sciences. 2011; 12(3):1756-1766. https://doi.org/10.3390/ijms12031756
Chicago/Turabian StyleTong, Mengliang, Hongyan Chen, Zhanhong Yang, and Runjuan Wen. 2011. "The Effect of Zn-Al-Hydrotalcites Composited with Calcium Stearate and β-Diketone on the Thermal Stability of PVC" International Journal of Molecular Sciences 12, no. 3: 1756-1766. https://doi.org/10.3390/ijms12031756




