Chemically Induced Breast Tumors in Rats Are Detectable in Early Stages by Contrast Enhanced Magnetic Resonance Imaging but Not by Changes in the Acute-Phase Reactants in Serum
Abstract
:1. Introduction
2. Results
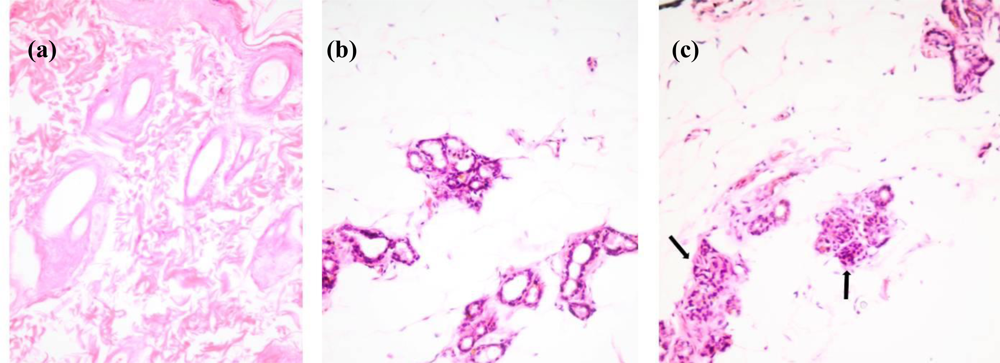
2.1. Histological Staining of Rat Mammary Sections
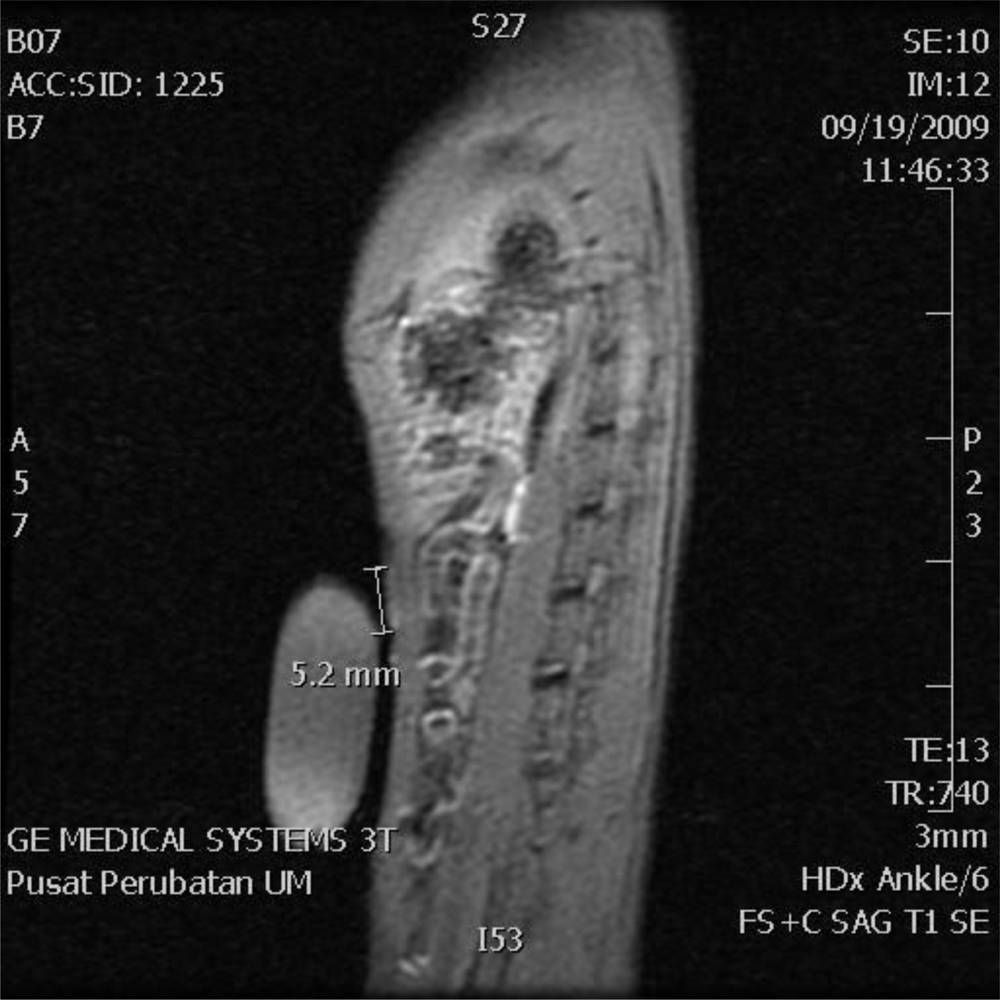
2.2. MRI Analysis
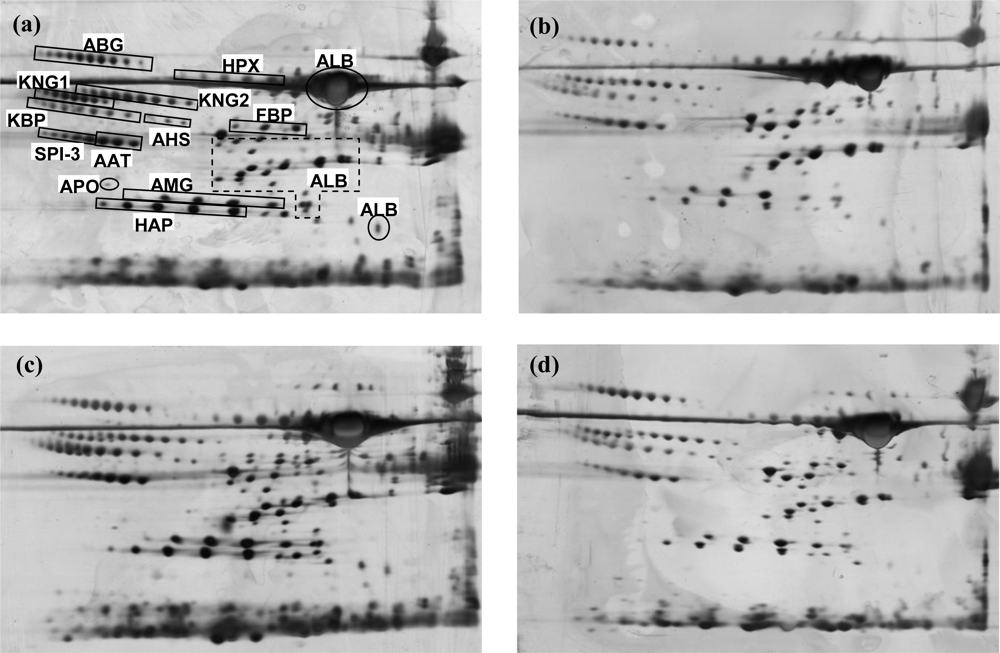
2.3. 2-DE Serum Protein Profiles
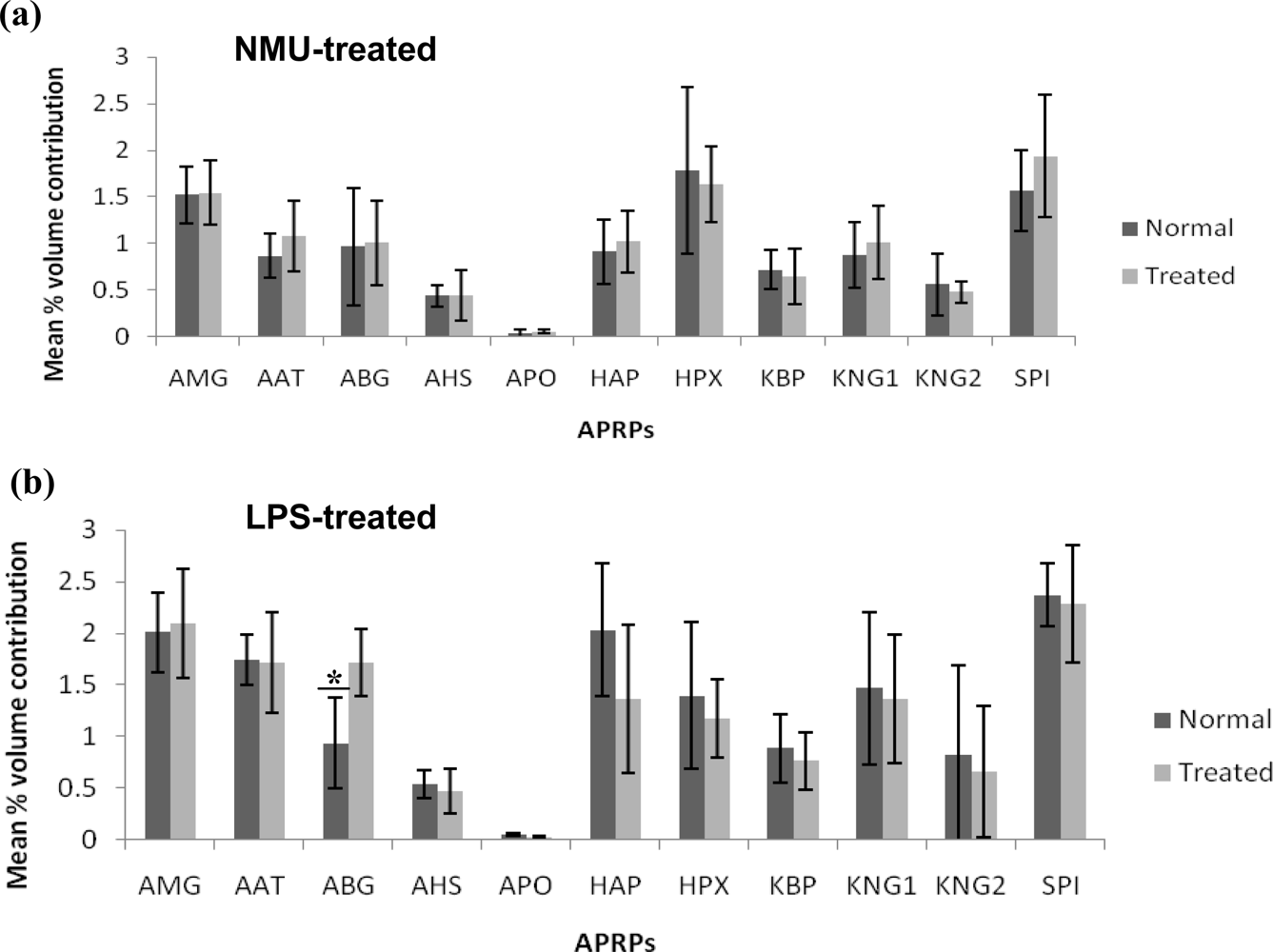
2.4. Analysis of Spot Volumes
3. Discussion
4. Experimental Section
4.1. Rat Serum Samples
4.2. Chemical Treatment
4.3. Histological Staining
4.4. Contrast Enhanced MRI
4.5. 2-DE and Mass Spectrometry
4.6. Image and Statistical Analyses
5. Conclusions
Acknowledgments
References
- Ferlay, J; Shin, H; Bray, F; Forman, D; Mathers, C; Parkin, D. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010. [Google Scholar] [CrossRef]
- Jansen, SA; Conzen, SD; Fan, X; Krausz, T; Zamora, M; Foxley, S; River, J; Newstead, GM; Karczmar, GS. Detection of in situ mammary cancer in a transgenic mouse model: In vitro and in vivo MRI studies demonstrate histopathologic correlation. Phys. Med. Biol 2008, 53, 5481–5493. [Google Scholar]
- Frangioni, J. New technologies for human cancer imaging. J. Clin. Oncol 2008, 26, 4012–4021. [Google Scholar]
- Hara, N; Okuizumi, M; Koike, H; Kawaguchi, M; Bilim, V. Dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) is a useful modality for the precise detection and staging of early prostate cancer. Prostate 2005, 62, 140–147. [Google Scholar]
- Turnbull, L. Dynamic contrast-enhanced MRI in the diagnosis and management of breast cancer. NMR Biomed 2009, 22, 28–39. [Google Scholar]
- Turetschek, K; Huber, S; Floyd, E; Helbich, T; Roberts, TP; Shames, DM; Tarlo, KS; Wendland, MF; Brasch, RC. MR imaging characterization of microvessels in experimental breast tumors by using a particulate contrast agent with histopathologic correlation. Radiology 2001, 218, 562–569. [Google Scholar]
- Suffredini, A; Fantuzzi, G; Badolato, R; Oppenheim, J; O’Grady, N. New insights into the biology of the acute phase response. J. Clin. Immunol 1999, 19, 203–214. [Google Scholar]
- Doustjalali, SR; Yusof, R; Yip, CH; Looi, LM; Pillay, B; Hashim, OH. Aberrant expression of acute-phase reactant proteins in sera and breast lesions of patients with malignant and benign breast tumors. Electrophoresis 2004, 25, 2392–2401. [Google Scholar]
- Doustjalali, SR; Yusof, R; Govindasamy, G; Bustam, AZ; Pillay, B; Hashim, OH. Patients with nasopharyngeal carcinoma demonstrate enhanced serum and tissue ceruloplasmin expression. J. Med. Invest 2006, 53, 20–28. [Google Scholar]
- Abdul-Rahman, PS; Lim, BK; Hashim, OH. Expression of high-abundance proteins in sera of patients with endometrial and cervical cancers: Analysis using 2-DE with silver staining and lectin detection methods. Electrophoresis 2007, 28, 1989–1996. [Google Scholar] [Green Version]
- Chen, Y; Lim, BK; Peh, SC; Abdul-Rahman, PS; Hashim, OH. Profiling of serum and tissue high abundance acute-phase proteins of patients with epithelial and germ line ovarian carcinoma. Proteome Sci 2008, 6, 20. [Google Scholar]
- Ahmed, N; Barker, G; Oliva, KT; Hoffmann, P; Riley, C; Reeve, S; Smith, AI; Kemp, BE; Quinn, MA; Rice, GE. Proteomic-based identification of haptoglobin-1 precursor as a novel circulating biomarker of ovarian cancer. Br. J. Cancer 2004, 91, 129–140. [Google Scholar]
- Koomen, J; Shih, L; Coombes, K; Li, D; Xiao, L; Fidler, I; Abbruzzese, J; Kobayashi, R. Plasma protein profiling for diagnosis of pancreatic cancer reveals the presence of host response proteins. Clin. Cancer Res 2005, 11, 1110–1118. [Google Scholar]
- Yu, K; Rustgi, A; Blair, I. Characterization of proteins in human pancreatic cancer serum using differential gel electrophoresis and tandem mass spectrometry. J. Proteome Res 2005, 4, 1742–1751. [Google Scholar]
- Dowling, P; O’Driscoll, L; Meleady, P; Henry, M; Roy, S; Ballot, J; Moriarty, M; Crown, J; Clynes, M. 2-D difference gel electrophoresis of the lung squamous cell carcinoma versus normal sera demonstrates consistent alterations in the levels of ten specific proteins. Electrophoresis 2007, 28, 4302–4310. [Google Scholar]
- Pang, WW; Abdul-Rahman, PS; Wan-Ibrahim, WI; Hashim, OH. Can the acute-phase reactant proteins be used as cancer biomarkers? Int. J. Biol. Markers 2010, 25, 1–11. [Google Scholar]
- Chen, Y; Lim, BK; Hashim, OH. Different altered stage correlative expression of high abundance acute-phase proteins in sera of patients with epithelial ovarian carcinoma. J. Hematol. Oncol 2009, 2, 37. [Google Scholar]
- Murray, TJ; Ucci, AA; Maffini, MV; Sonnenschein, C; Soto, AM. Histological analysis of low dose NMU effects in the rat mammary gland. BMC Cancer 2009, 9, 267. [Google Scholar]
- Haynes, P; Miller, I; Aebersold, R; Gemeiner, M; Eberini, I; Lovati, M; Manzoni, C; Vignati, M; Gianazza, E. Proteins of rat serum: I. establishing a reference two-dimensional electrophoresis map by immunodetection and microbore high performance liquid chromatography-electrospray mass spectrometry. Electrophoresis 1998, 19, 1484–1492. [Google Scholar]
- McCormick, DL; Adamowski, CB; Fiks, A; Moon, RC. Lifetime dose-response relationships for mammary tumor induction by a single administration of N-methyl-N-nitrosourea. Cancer Res 1981, 41, 1690–1694. [Google Scholar]
- Roomi, MW; Roomi, NW; Ivanov, V; Kalinovsky, T; Niedzwiecki, A; Rath, M. Modulation of N-methyl-N-nitrosourea induced mammary tumors in Sprague-Dawley rats by combination of lysine, proline, arginine, ascorbic acid and green tea extract. Breast Cancer Res 2005, 7, R291–R295. [Google Scholar]
- Schepkin, VD; Ross, BD; Chenevert, TL; Rehemtulla, A; Sharma, S; Kumar, M; Stojanovska, J. Sodium magnetic resonance imaging of chemotherapeutic response in a rat glioma. Magn. Reson. Med 2005, 53, 85–92. [Google Scholar]
- Driehuys, B; Nouls, J; Badea, A; Bucholz, E; Ghaghada, K; Petiet, A; Hedlund, LW. Small animal imaging with magnetic resonance microscopy. ILAR J 2008, 49, 35–53. [Google Scholar]
- Seriramalu, R; Pang, WW; Jayapalan, JJ; Mohamed, E; Abdul-Rahman, PS; Bustam, AZ; Khoo, AS; Hashim, OH. Application of champedak mannose-binding lectin in the glycoproteomic profiling of serum samples unmasks reduced expression of alpha-2 macroglobulin and complement factor B in patients with nasopharyngeal carcinoma. Electrophoresis 2010, 31, 2388–2395. [Google Scholar]
| APRP# | Accession Number (Swiss-Prot) | Theoretical Mass | Mascot Score | Theoretical pI | No of Peptide Hits | Sequence Coverage (%) |
|---|---|---|---|---|---|---|
| ALB (serum albumin) | P02770 | 68686 | 739 | 6.46 | 12 | 27 |
| AHS (α2-HS glycoprotein) | P24090 | 37958 | 263 | 6.05 | 4 | 22 |
| ABG (α1B-glycoprotein) | Q9EPH1 | 56443 | 272 | 6.89 | 7 | 14 |
| KNG1 (T-kininogen) | P01048 | 47745 | 296 | 6.08 | 6 | 20 |
| KBP (kallikrein-binding protein) | P05545 | 46532 | 327 | 5.31 | 8 | 22 |
| SPI-3 (serine protease inhibitor) | P09006 | 46622 | 384 | 5.33 | 7 | 21 |
| APO (apolipoprotein A-IV) | P02651 | 44429 | 770 | 5.12 | 15 | 45 |
| AMG (α1-macroglobulin) | Q63041 | 167019 | 643 | 6.46 | 10 | 6 |
| HAP (haptoglobin) | P06866 | 38539 | 508 | 6.10 | 12 | 32 |
| AAT (α1-antitrypsin) | P17475 | 46107 | 720 | 5.70 | 13 | 34 |
| HPX (hemopexin) | P20059 | 51318 | 498 | 7.58 | 9 | 19 |
| FBP (fetuin-B precursor) | Q9QX79 | 41506 | 451 | 6.71 | 9 | 40 |
| KNG2 (T-kininogen) | P01048 | 47745 | 424 | 6.08 | 8 | 18 |
| Parameters | AX FSE PD | AX T1 SE FAT SAT | AX T1 SE FAT SAT + CONTRAST | SAG T1 FAT SAT + CONTRAST |
|---|---|---|---|---|
| BANDWITH: | 62.5 | 11.9 | 11.9 | 11.9 |
| FOV | 8 × 8 | 8 × 8 | 8 × 8 | 8 × 8 |
| FREQ: | 512 × 224 | 192 × 160 | 192 × 160 | 192 × 160 |
| IMAGING OPTIONS | FAST, NPW, TRF | NPW, ZIP512, StFCl | NPW, ZIP512, StFCl | NPW, ZIP512, StFCl |
| MODE: | 2D | 2D | 2D | 2D |
| NEX: | 1 | 2 | 2 | 1 |
| PULSE | SEQ:FSE-XL | SEQ:FSE-XL | FSE-XL | FSE-XL |
| SHIM: | AUTO | AUTO | AUTO | AUTO |
| SLICE THICK: | 2 | 2 | 2 | 3 |
| SPACING: | 0 | 0 | 0 | 0 |
| TE: | 33.5 | 13 | 13 | 13 |
| TR: | 7660 | 900 | 900 | 740 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Golbabapour, S.; Pang, W.W.; George, J.; Pasupati, T.; Abdul-Rahman, P.S.; Hashim, O.H. Chemically Induced Breast Tumors in Rats Are Detectable in Early Stages by Contrast Enhanced Magnetic Resonance Imaging but Not by Changes in the Acute-Phase Reactants in Serum. Int. J. Mol. Sci. 2011, 12, 1030-1040. https://doi.org/10.3390/ijms12021030
Golbabapour S, Pang WW, George J, Pasupati T, Abdul-Rahman PS, Hashim OH. Chemically Induced Breast Tumors in Rats Are Detectable in Early Stages by Contrast Enhanced Magnetic Resonance Imaging but Not by Changes in the Acute-Phase Reactants in Serum. International Journal of Molecular Sciences. 2011; 12(2):1030-1040. https://doi.org/10.3390/ijms12021030
Chicago/Turabian StyleGolbabapour, Shahram, Wei Wei Pang, John George, Thanikachalam Pasupati, Puteri Shafinaz Abdul-Rahman, and Onn Haji Hashim. 2011. "Chemically Induced Breast Tumors in Rats Are Detectable in Early Stages by Contrast Enhanced Magnetic Resonance Imaging but Not by Changes in the Acute-Phase Reactants in Serum" International Journal of Molecular Sciences 12, no. 2: 1030-1040. https://doi.org/10.3390/ijms12021030




