Increased Glutathione Synthesis Following Nrf2 Activation by Vanadyl Sulfate in Human Chang Liver Cells
Abstract
:1. Introduction
2. Results and Discussion
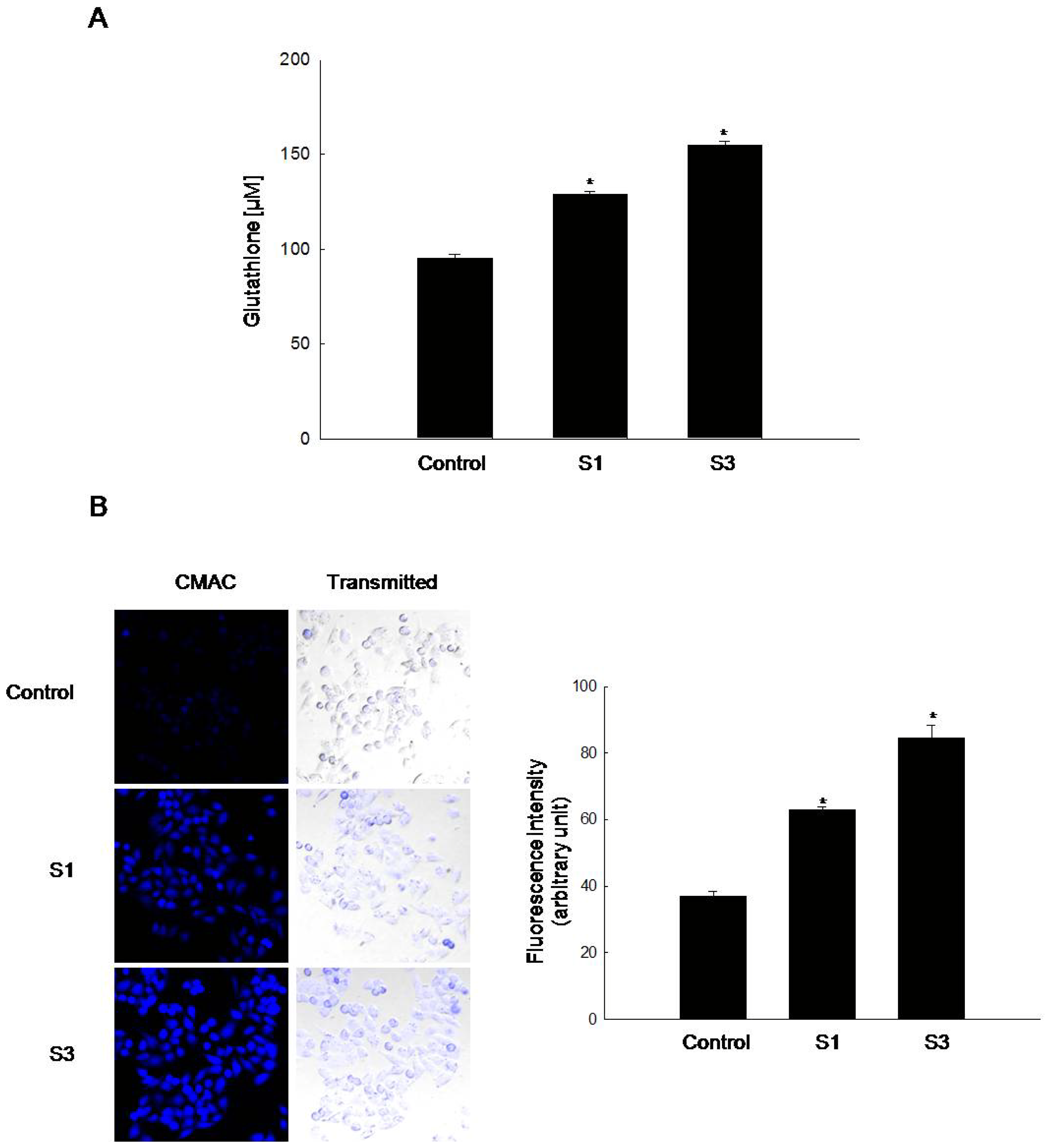
2.1. GSH Amount Is Enhanced by Jeju Ground Water Containing Vanadium Components
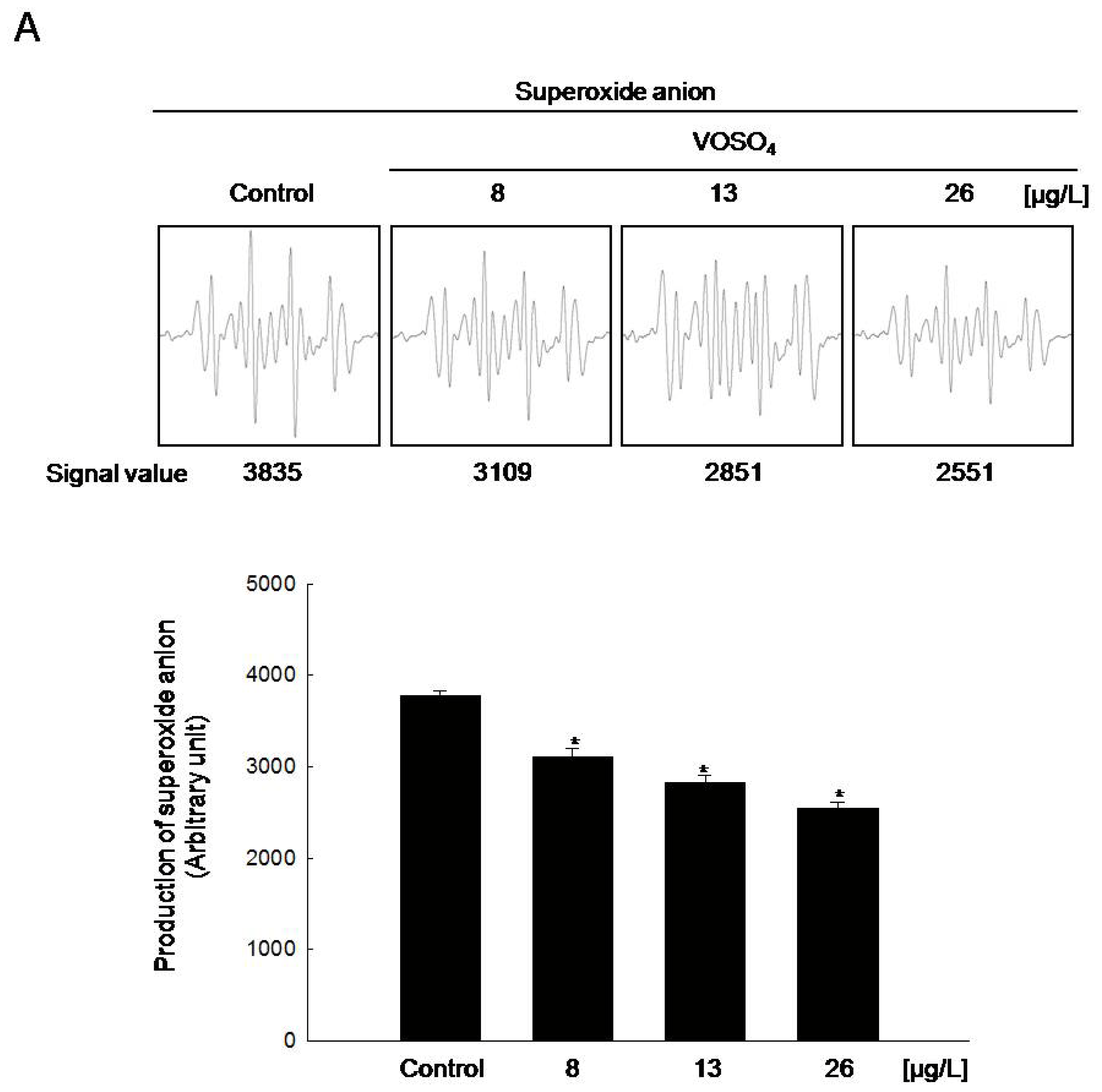
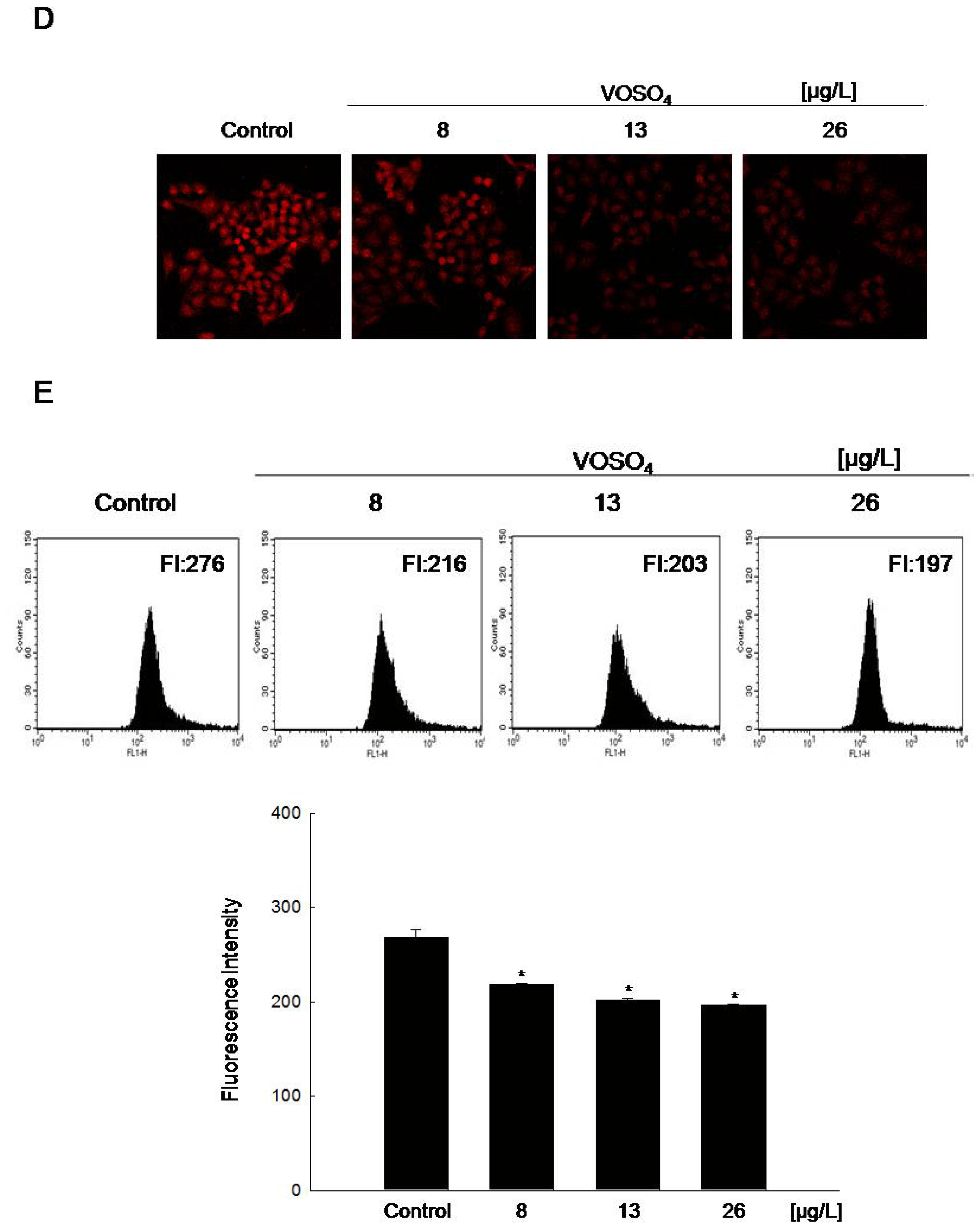
2.2. Radical Scavenging Activity of Vanadyl Sulfate
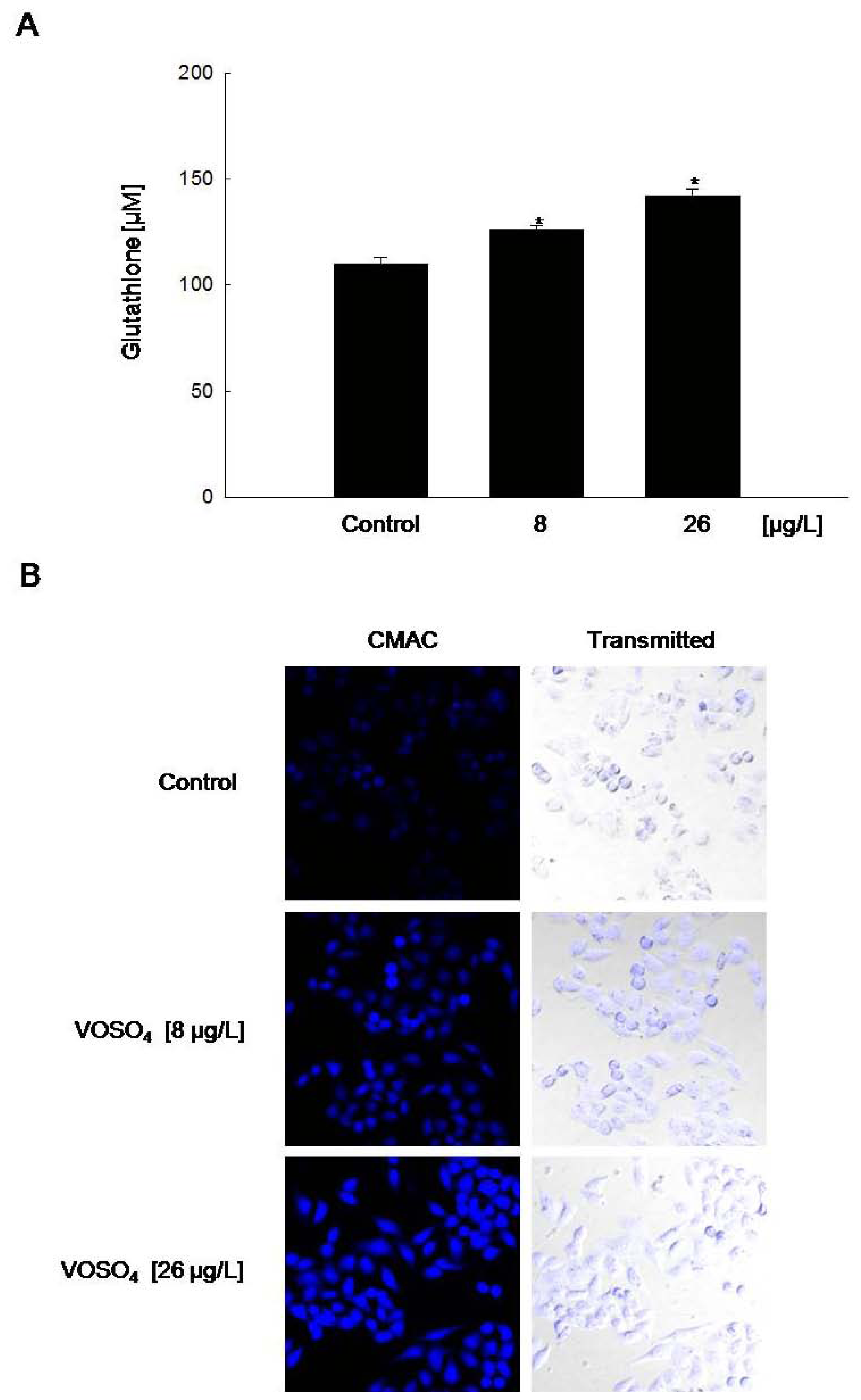
2.3. Induction of GSH Synthesis by Vanadyl Sulfate
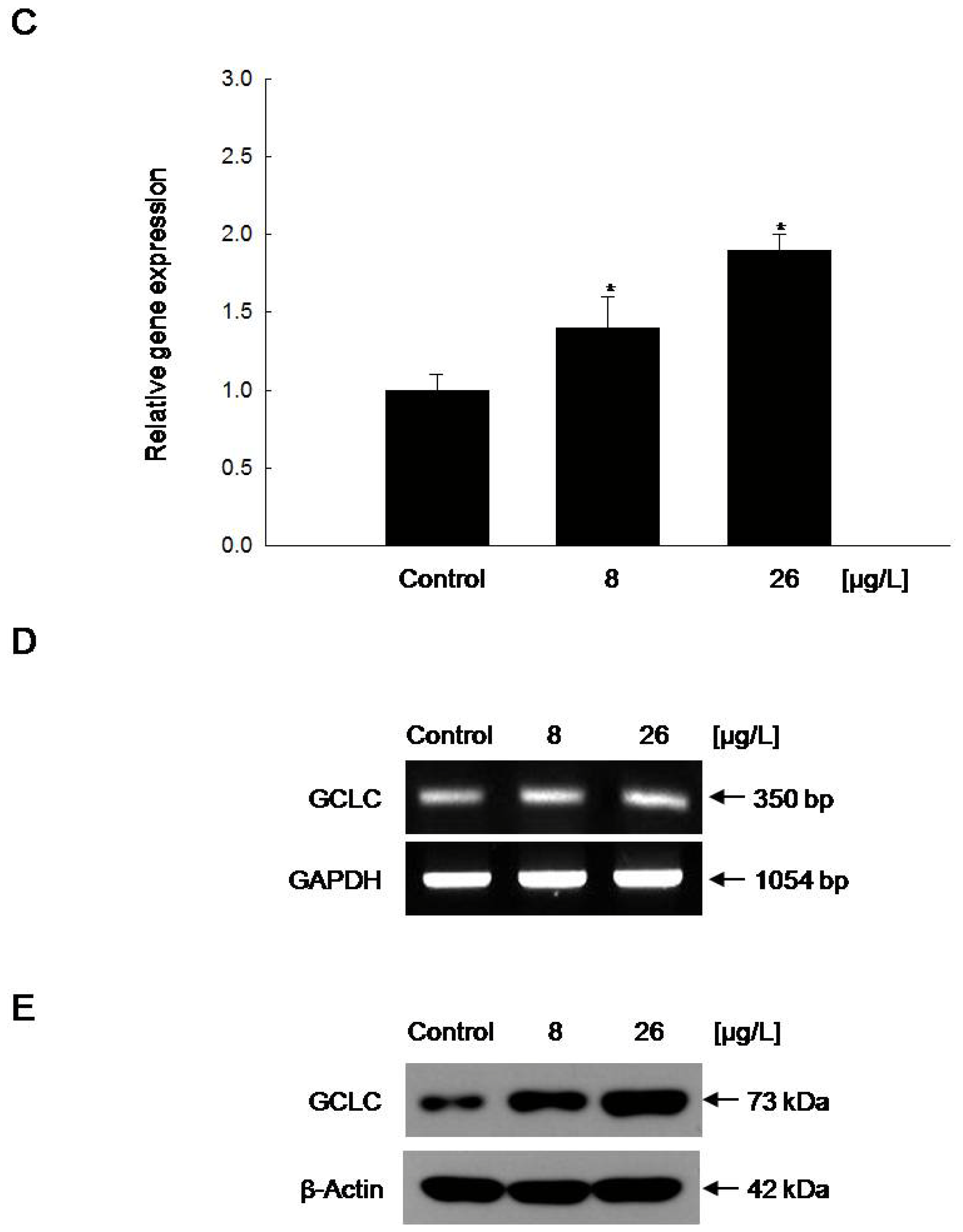
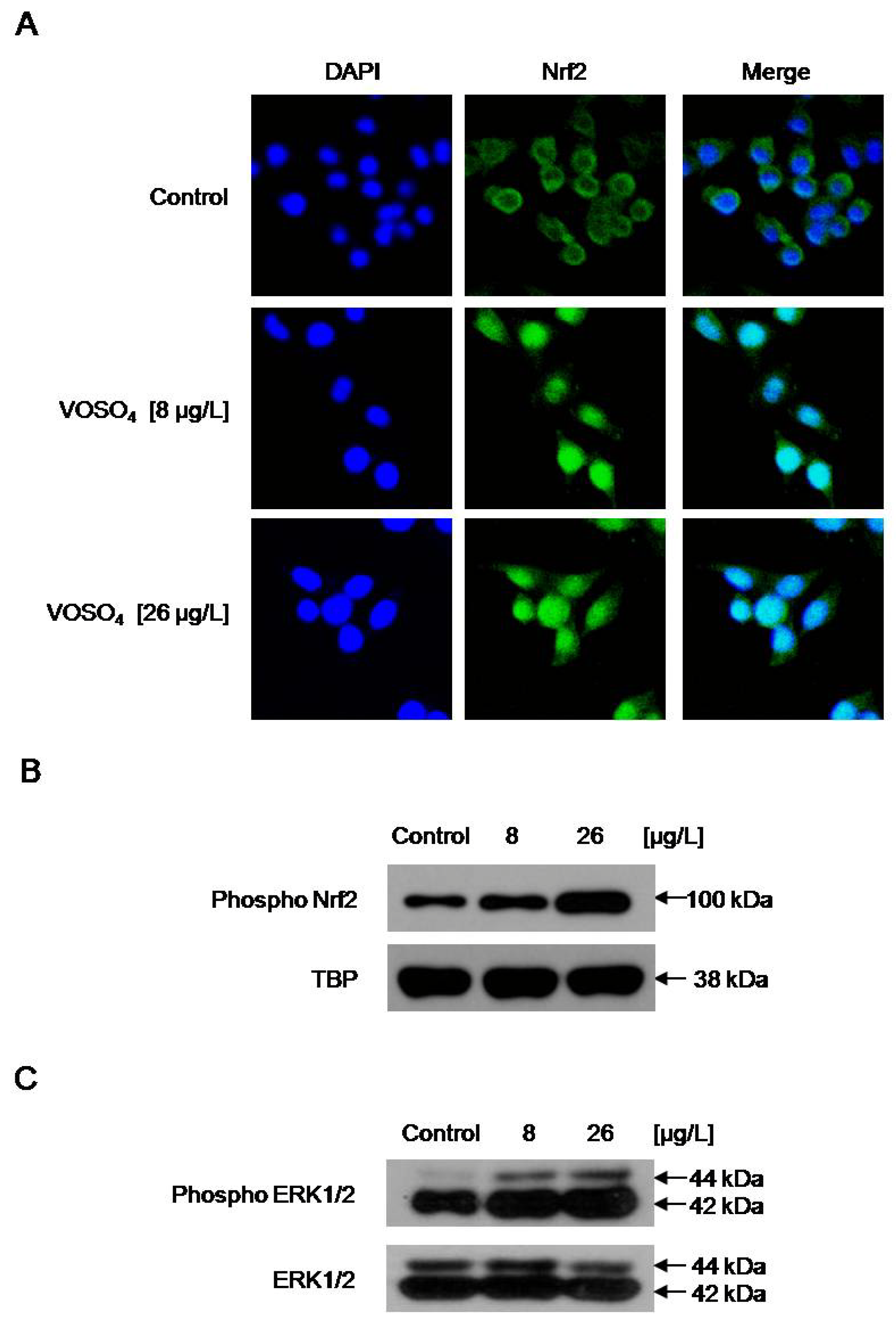
2.4. Increased GCLC Gene Transcription Following the Induction of Nuclear Translocation of Nrf2 by Vanadyl Sulfate
3. Experimental Section
3.1. Reagents
3.2. Cell Culture
3.3. Intracellular GSH Measurement
3.4. Detection of Superoxide Anion
3.5. Detection of Hydroxyl Radical
3.6. Measurement of Intracellular Reactive Oxygen Species (ROS)
3.7. Real Time-Polymerase Chain Reaction (PCR)
3.8. Reverse Transcription Polymerase Chain Reaction (RT-PCR)
3.9. Western Blotting Analysis
3.10. Immunocytochemistry
3.11. Statistical Analysis
4. Conclusions
Acknowledgements
References
- Lu, S.C. Regulation of glutathione synthesis. Mol. Aspects Med 2009, 30, 42–59. [Google Scholar]
- Huang, H.C.; Nguyen, T.; Pickett, C.B. Regulation of the antioxidant response element by protein kinase C-mediated phosphorylation of NF-E2-related factor 2. Proc. Natl. Acad. Sci. USA 2000, 97, 12475–12480. [Google Scholar]
- Moinova, H.R.; Mulcahy, R.T. An electrophile responsive element (EpRE) regulates beta-naphthoflavone induction of the human gamma-glutamylcysteine synthetase regulatory subunit gene. Constitutive expression is mediated by an adjacent AP-1 site. J. Biol. Chem 1998, 273, 14683–14689. [Google Scholar]
- Moinova, H.R.; Mulcahy, R.T. Up-regulation of the human gamma-glutamylcysteine synthetase regulatory subunit gene involves binding of Nrf-2 to an electrophile responsive element. Biochem. Biophys. Res. Commun 1999, 261, 661–668. [Google Scholar]
- Yang, H.; Magilnick, N.; Lee, C.; Kalmaz, D.; Ou, X.; Chan, J.Y.; Lu, S.C. Nrf1 and Nrf2 regulate rat glutamate-cysteine ligase catalytic subunit transcription indirectly via NF-kappaB and AP-1. Mol. Cell. Biol 2005, 25, 5933–5946. [Google Scholar]
- Kim, A.D.; Kang, K.A.; Zhang, R.; Lim, C.M.; Jee, Y.E.; Lee, N.H.; You, H.J.; Ko, K.S.; Hyun, J.W. Reactive oxygen species scavenging effects of Jeju waters containing vanadium components. Cancer Prev. Res (Seoul) 2010, 15, 111–117. [Google Scholar]
- Kim, A.D.; Kang, K.A.; Zhang, R.; Piao, M.J.; Kim, S.M.; Jee, Y.E.; Lee, N.H.; You, H.J.; Ko, K.S.; Hyun, J.W. Effects of Jeju water containing vanadium on antioxidant enzymes in vitro. Cancer Prev. Res (Seoul) 2010, 15, 262–267. [Google Scholar]
- Kim, A.D.; Kang, K.A.; Zhang, R.; Piao, M.J.; Kim, S.M.; Jee, Y.E.; Lee, N.H.; You, H.J.; Ko, K.S.; Hyun, J.W. Antioxidant enzyme-enhancing effects of Jeju water containing vanadium in vivo. Cancer Prev. Res (Seoul) 2011, 16, 58–64. [Google Scholar]
- Ha, D.B.; Kim, M.J.; Joo, H.J.; Cho, J.H.; Bing, S.J.; Lim, Y.K.; Hyun, J.W.; Jee, Y.H. Immune activation of Jeju water containing vanadium on peripheral immunocytes, of Low dose gamma rays-irradiated mice. Kor. J. Vet. Publ. Health 2011, 35, 49–59. [Google Scholar]
- Tunali, S.; Yanardag, R. Effect of vanadyl sulfate on the status of lipid parameters and on stomach and spleen tissues of streptozotocin-induced diabetic rats. Pharmacol. Res 2006, 53, 271–277. [Google Scholar]
- Preet, A.; Gupta, B.L.; Yadava, P.K.; Baquer, N.Z. Efficacy of lower doses of vanadium in restoring altered glucose metabolism and antioxidant status in diabetic rat lenses. J. Biosci 2005, 30, 221–230. [Google Scholar]
- Rehder, D. Biological and medicinal aspects of vanadium. Inorg. Chem. Commun 2003, 6, 604–617. [Google Scholar]
- Jo, M.; Park, M.H.; Choi, D.Y.; Yuk, D.Y.; Lee, Y.M.; Lee, J.M.; Jeong, J.H.; Oh, K.W.; Lee, M.S.; Han, S.B.; et al. Neuroprotective effect of l-theanine on Aβ-induced neurotoxicity through anti-oxidative mechanisms in SK-N-SH and SK-N-MC Cells. Biomol. Ther 2011, 19, 288–295. [Google Scholar]
- Lee, T.D.; Yang, H.; Whang, J.; Lu, S.C. Cloning and characterization of the human glutathione synthetase 5′-flanking region. Biochem. J 2005, 390, 521–528. [Google Scholar]
- Leong, P.K.; Chiu, P.Y.; Chen, N.; Leung, H.; Ko, K.M. Schisandrin B elicits a glutathione antioxidant response and protects against apoptosis via the redox-sensitive ERK/Nrf2 pathway in AML12 hepatocytes. Free Radic. Res 2011, 45, 483–495. [Google Scholar]
- Tauskela, J.S.; Hewitt, K.; Kang, L.P.; Comas, T.; Gendron, T.; Hakim, A.; Hogan, M.; Durkin, J.; Morley, P. Evaluation of glutathione-sensitive fluorescent dyes in cortical culture. Glia 2000, 30, 329–341. [Google Scholar]
- Ueno, I.; Kohno, M.; Yoshihira, K.; Hirono, I. Quantitative determination of the superoxide radicals in the xanthine oxidase reaction by measurement of the electron spin resonance signal of the superoxide radical spin adduct of 5,5-dimethyl-1-pyrroline-1-oxide. J. Pharmacobiodyn 1984, 7, 563–569. [Google Scholar]
- Kohno, M.; Mizuta, Y.; Kusai, M.; Masumizu, T.; Makino, K. Measurements of superoxide anion radical and superoxide anion scavenging activity by electron spin resonance spectroscopy coupled with DMPO spin trapping. Bull. Chem. Soc. Jpn 1994, 67, 1085–1090. [Google Scholar]
- Li, L.; Abe, Y.; Mashino, T.; Mochizuki, M.; Miyata, N. Signal enhancement in ESR spin-trapping for hydroxyl radicals. Anal. Sci 2003, 19, 1083–1084. [Google Scholar]
- Li, L.; Abe, Y.; Kanagawa, K.; Usui, N.; Imai, K.; Mashino, T.; Mochizuki, M.; Miyata, N. Distinguishing the 5,5-dimethyl-1-pyrroline N-oxide (DMPO)-OH radical quenching effect from the hydroxyl radical scavenging effect in the ESR spin-trapping method. Anal. Chim. Acta 2004, 512, 121–124. [Google Scholar]
- Rosenkranz, A.R.; Schmaldienst, S.; Stuhlmeier, K.M.; Chen, W.; Knapp, W.; Zlabinger, G.J. A microplate assay for the detection of oxidative products using 2′,7′-dichlorofluorescin-diacetate. J. Immunol. Methods 1992, 156, 39–45. [Google Scholar]
- Schefe, J.H.; Lehmann, K.E.; Buschmann, I.R.; Unger, T.; Funke-Kaiser, H. Quantitative real-time RT-PCR data analysis: Current concepts and the novel “gene expression’s CT difference” formula. J. Mol. Med (Berlin) 2006, 84, 901–910. [Google Scholar]
- Samanta, S.; Swamy, V.; Suresh, D.; Rajkumar, M.; Rana, B.; Rana, A.; Chatterjee, M. Protective effects of vanadium against DMH-induced genotoxicity and carcinogenesis in rat colon: Removal of O(6)-methylguanine DNA adducts, p53 expression, inducible nitric oxide synthase downregulation and apoptotic induction. Mutat. Res 2008, 650, 123–131. [Google Scholar]
- Sekar, N.; Kanthasamy, A.; William, S.; Balasubramaniyan, N.; Govindasamy, S. Antioxidant effect of vanadate on experimental diabetic rats. Acta Diabetol. Lat 1990, 27, 285–293. [Google Scholar]
- Lapenna, D.; Ciofani, G.; Bruno, C.; Pierdomenico, S.D.; Giuliani, L.; Giamberardino, M.A.; Cuccurullo, F. Vanadyl as a catalyst of human lipoprotein oxidation. Biochem. Pharmacol 2002, 63, 375–380. [Google Scholar]
- Badmaev, V.; Prakash, S.; Majeed, M. Vanadium: A review of its potential role in the fight against diabetes. J. Altern. Complement. Med 1999, 5, 273–291. [Google Scholar]
- Yanardag, R.; Tunali, S. Vanadyl sulfate administration protects the streptozotocin-induced oxidative damage to brain tissue in rats. Mol. Cell. Biochem 2006, 286, 153–159. [Google Scholar]
- Ha, K.N.; Chen, Y.; Cai, J.; Sternberg, P., Jr. Increased glutathione synthesis through an ARE-Nrf2-dependent pathway by zinc in the RPE: Implication for protection against oxidative stress. Invest. Ophthalmol. Vis. Sci. 2006, 47, 2709–2715. [Google Scholar]
- Kode, A.; Rajendrasozhan, S.; Caito, S.; Yang, S.R.; Megson, I.L.; Rahman, I. Resveratrol induces glutathione synthesis by activation of Nrf2 and protects against cigarette smoke-mediated oxidative stress in human lung epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol 2008, 294, L478–L488. [Google Scholar]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev. Pharmacol. Toxicol 2007, 47, 89–116. [Google Scholar]
- Li, M.H.; Jang, J.H.; Na, H.K.; Cha, Y.N.; Surh, Y.J. Carbon monoxide produced by heme oxygenase-1 in response to nitrosative stress induces expression of glutamate-cysteine ligase in PC12 cells via activation of phosphatidylinositol 3-kinase and Nrf2 signaling. J. Biol. Chem 2007, 282, 28577–28586. [Google Scholar]
- Lee, S.E.; Jeong, S.I.; Yang, H.; Park, C.S.; Jin, Y.H.; Park, Y.S. Fisetin induces Nrf2-mediated HO-1 expression through PKC-delta and p38 in human umbilical vein endothelial cells. J. Cell. Biochem 2011, 112, 2352–2360. [Google Scholar]
- Chiu, P.Y.; Chen, N.; Leong, P.K.; Leung, H.Y.; Ko, K.M. Schisandrin B elicits a glutathione antioxidant response and protects against apoptosis via the redox-sensitive ERK/Nrf2 pathway in H9c2 cells. Mol. Cell. Biochem 2011, 350, 237–250. [Google Scholar]

© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kim, A.D.; Zhang, R.; Kang, K.A.; You, H.J.; Hyun, J.W. Increased Glutathione Synthesis Following Nrf2 Activation by Vanadyl Sulfate in Human Chang Liver Cells. Int. J. Mol. Sci. 2011, 12, 8878-8894. https://doi.org/10.3390/ijms12128878
Kim AD, Zhang R, Kang KA, You HJ, Hyun JW. Increased Glutathione Synthesis Following Nrf2 Activation by Vanadyl Sulfate in Human Chang Liver Cells. International Journal of Molecular Sciences. 2011; 12(12):8878-8894. https://doi.org/10.3390/ijms12128878
Chicago/Turabian StyleKim, Areum Daseul, Rui Zhang, Kyoung Ah Kang, Ho Jin You, and Jin Won Hyun. 2011. "Increased Glutathione Synthesis Following Nrf2 Activation by Vanadyl Sulfate in Human Chang Liver Cells" International Journal of Molecular Sciences 12, no. 12: 8878-8894. https://doi.org/10.3390/ijms12128878



