Bioactive Compounds, Antioxidant, Xanthine Oxidase Inhibitory, Tyrosinase Inhibitory and Anti-Inflammatory Activities of Selected Agro-Industrial By-products
Abstract
:1. Introduction
2. Results and Discussion
2.1. Total Phenolic and Flavonoid Compounds
2.2. Bioactive Compounds
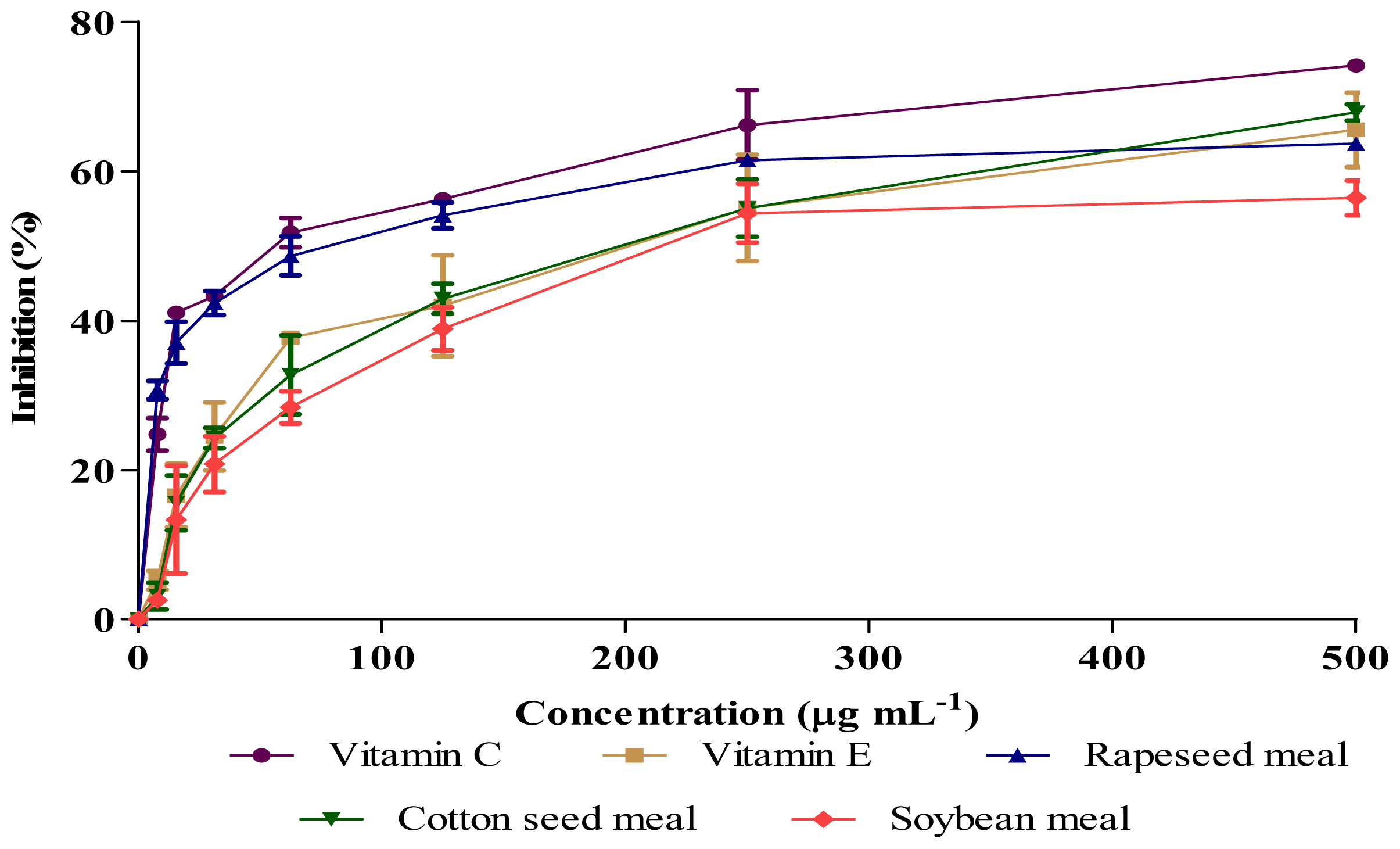
2.3. Antioxidant Activity (DPPH and NO Scavenging)
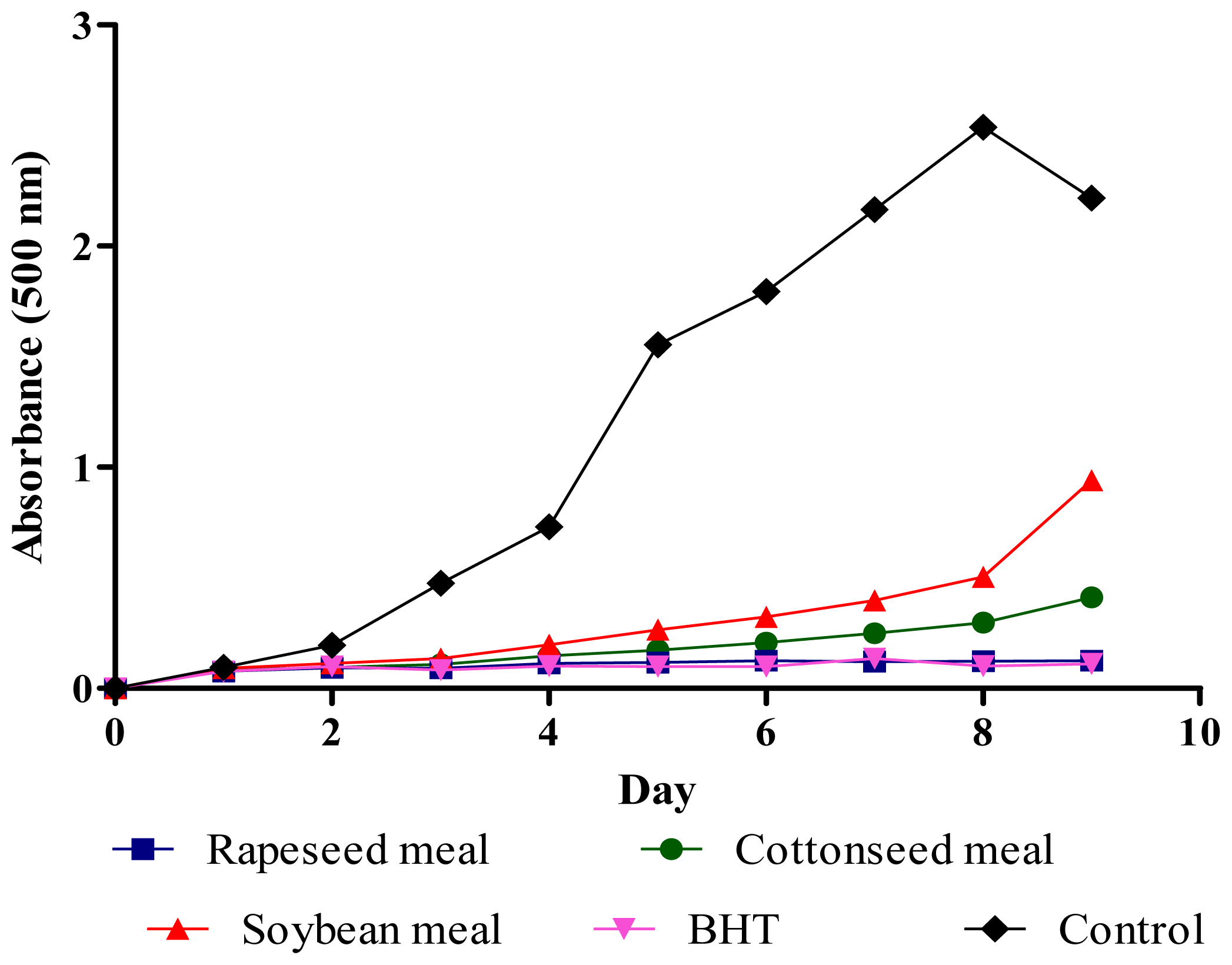
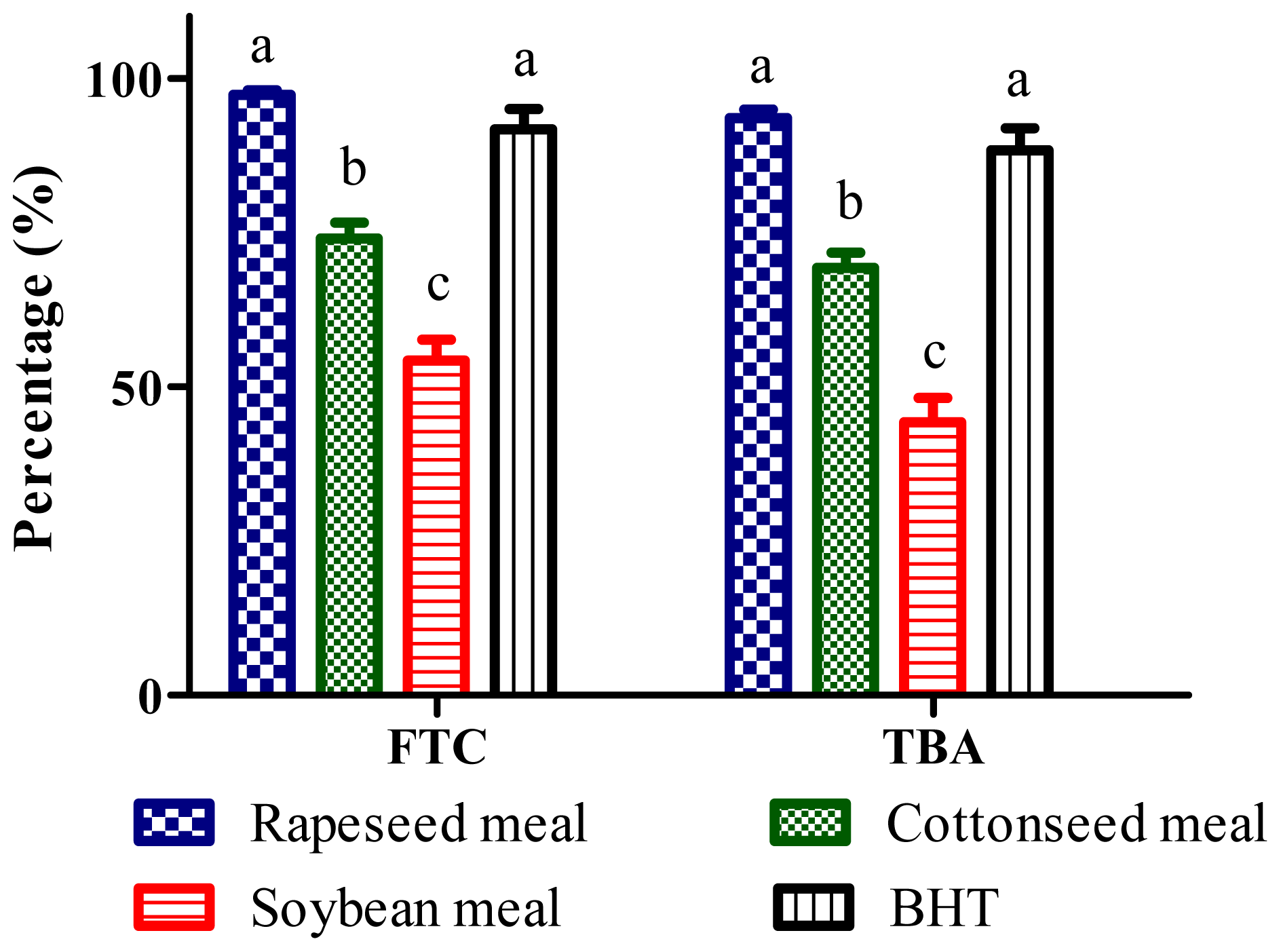
2.4. Total Antioxidant Activity (FTC and TBA Tests)
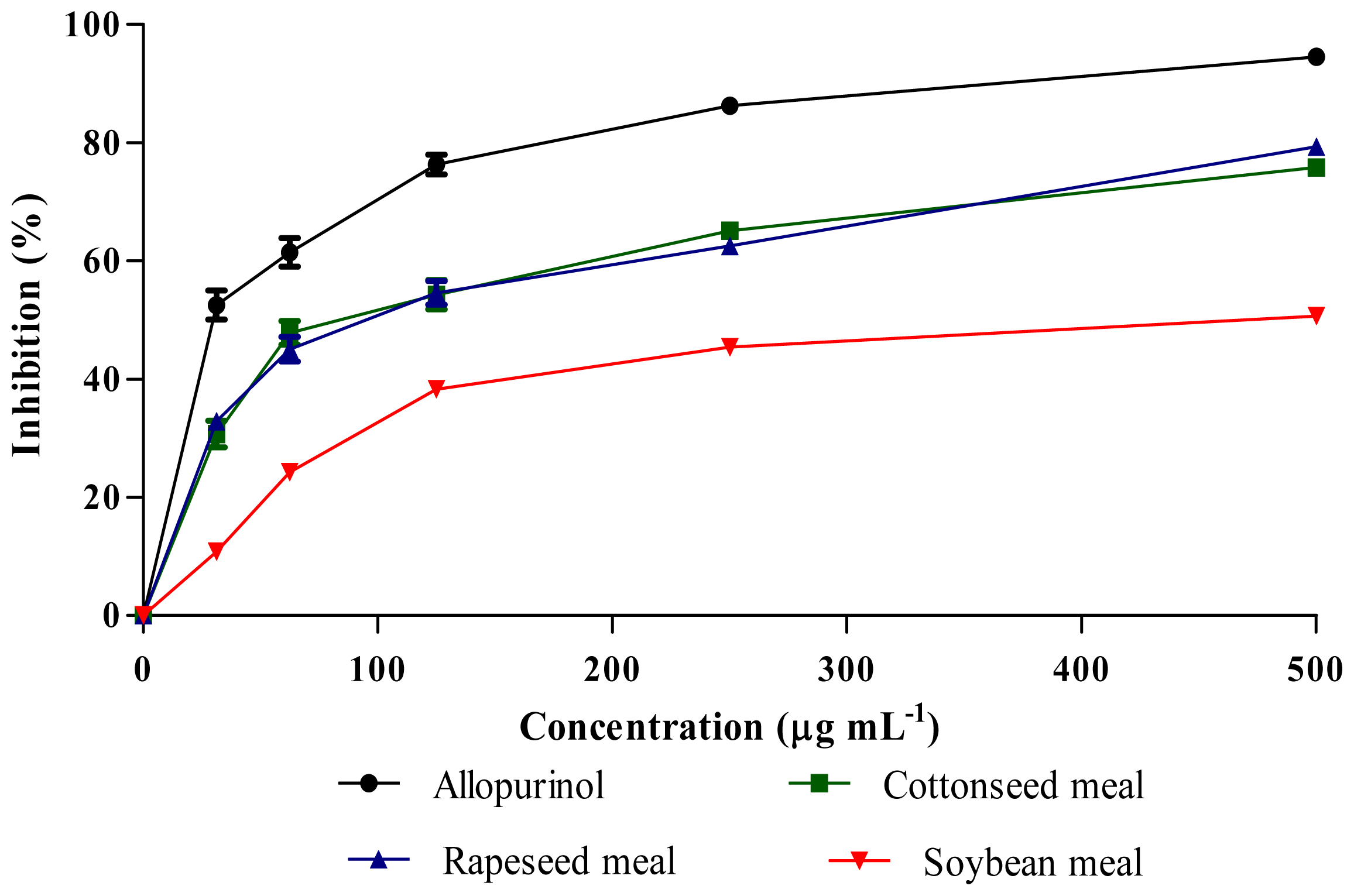
2.5. Xanthine Oxidase (XO) Inhibitory Activity
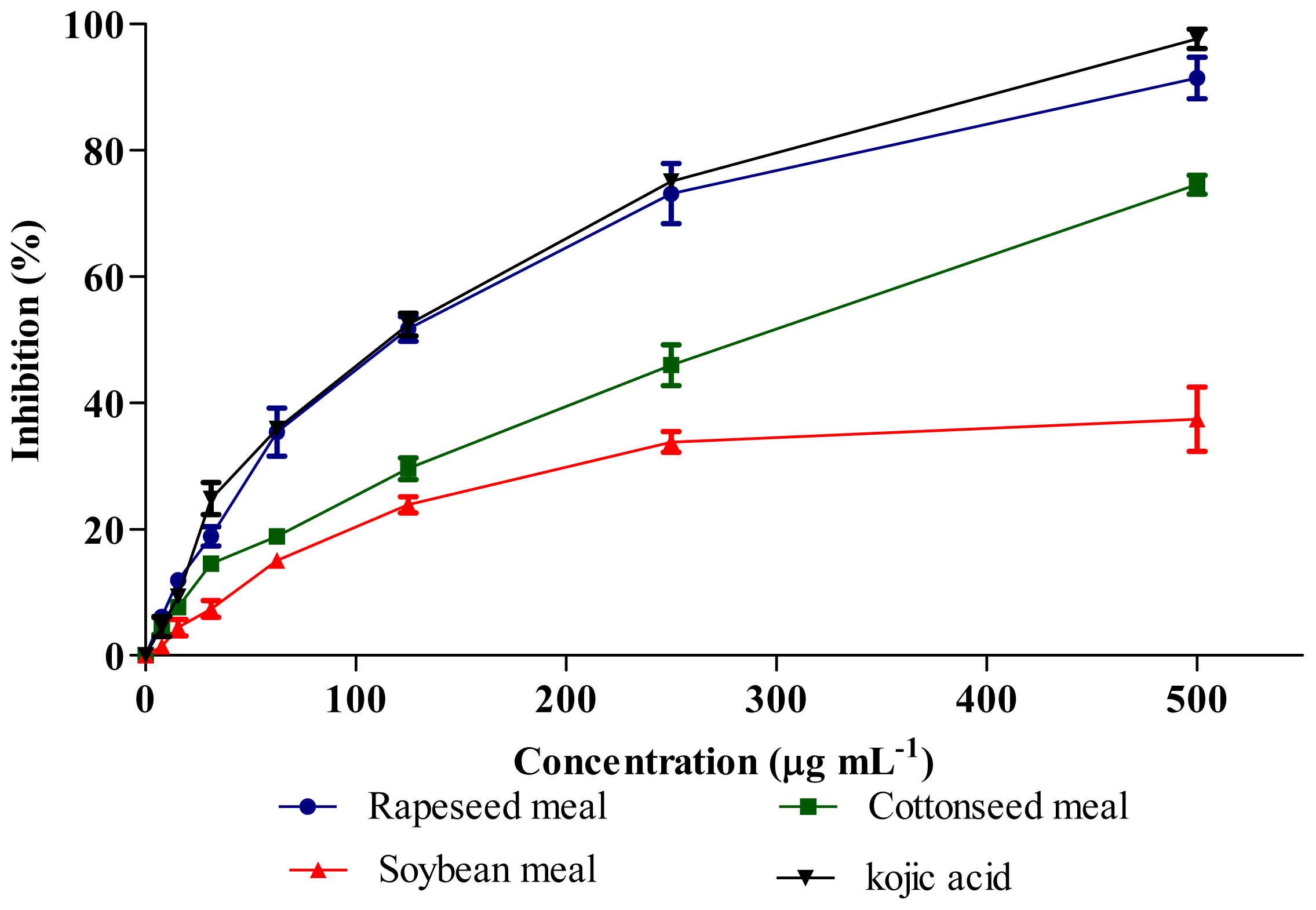
2.6. Tyrosinase Inhibitory Activity
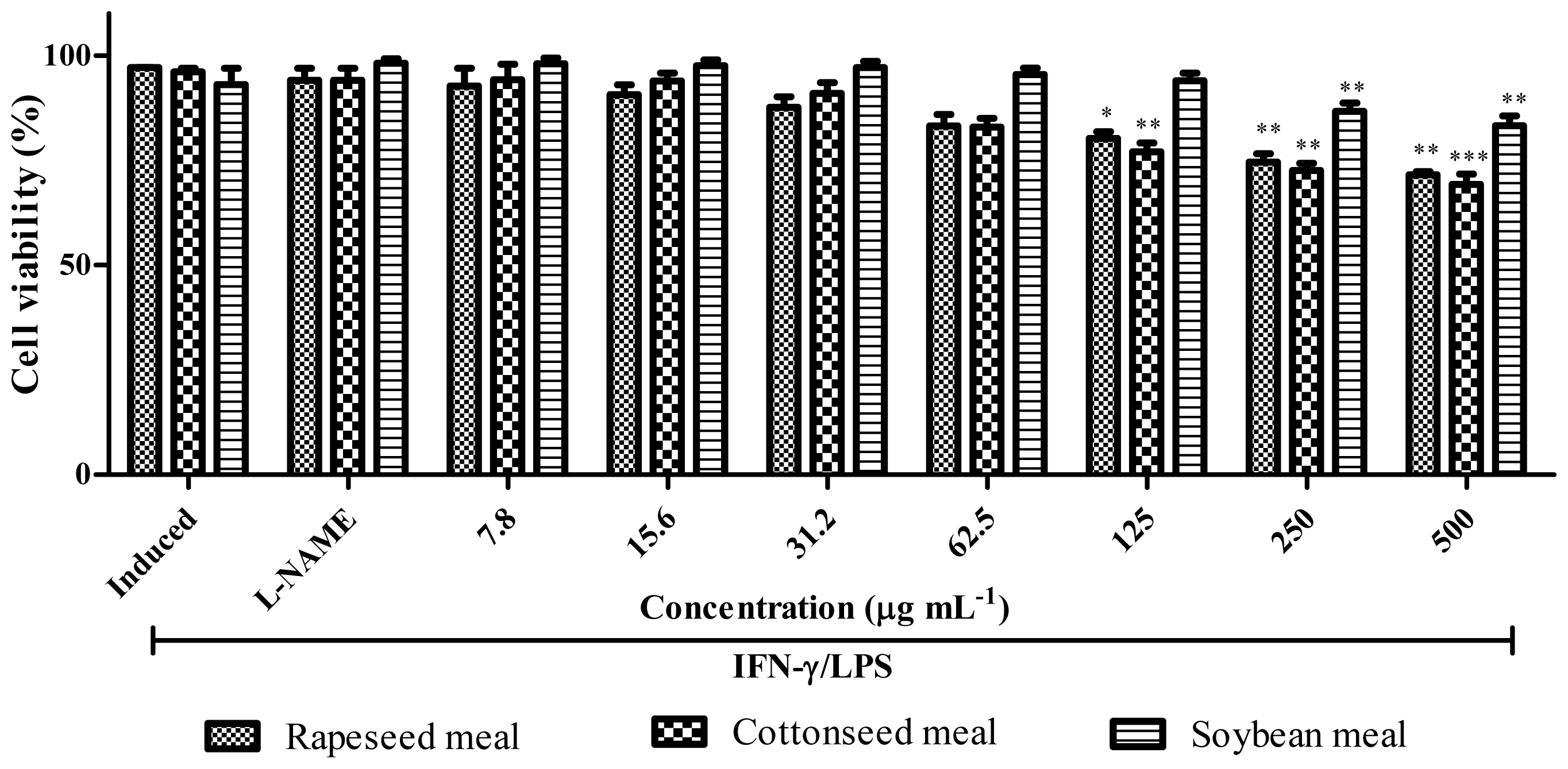
2.7. Anti-Inflammatory Activity
3. Experimental Section
3.1. Samples and Microwave Extract Preparation
3.2. Total Phenolic and Flavonoid Compounds
3.2.1. Chemicals
3.2.2. Total Phenolic Compounds
3.2.3. Total Flavonoid Compounds
3.2.4. Analyses of Phenolic and Flavonoid Compounds by HPLC
3.3. Antioxidant Activity
3.3.1. Free Radical Scavenging Activity
3.3.2. Nitric Oxide Scavenging Activity
3.3.3. Total Antioxidant Activity Assay
3.4. Xanthine Oxidase Inhibitory Activity
3.5. Tyrosinase Inhibitory Activity
3.6. Anti-Inflammatory Activity
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Jurgens, M.H. Animal Feeding and Nutrition, 8th ed; Hunt Publishing Company: Dubuque, IA, USA, 1996; pp. 135–143. [Google Scholar]
- Vollmann, J.; Rajcan, I. Oil Crops, 1st ed.; Springer: Berlin, Germany, 2009; pp. 14–25. [Google Scholar]
- Oskoueian, E.; Abdullah, N.; Saad, W.Z.; Omar, A.R.; Ahmad, S.; Kuan, W.B.; Zolkifli, N.A.; Hendra, R.; Ho, Y.W. Antioxidant, anti-inflammatory and anticancer activities of methanolic extracts from Jatropha curcas Linn. J. Med. Plants Res 2011, 5, 49–57. [Google Scholar]
- Oskoueian, E.; Abdullah, N.; Ahmad, S.; Saad, W.Z.; Omar, A.R.; Ho, Y.W. Bioactive compounds and biological activities of Jatropha curcas L. kernel meal extract. Int. J. Mol. Sci 2011, 12, 5955–5970. [Google Scholar]
- Namuli, A.; Abdullah, N.; Sieo, C.; Zuhainis, S.; Oskoueian, E. Phytochemical compounds and antibacterial activity of Jatropha curcas Linn. extracts. J. Med. Plants Res 2011, 5, 3982–3990. [Google Scholar]
- Hendra, R.; Ahmad, S.; Sukari, A.; Shukor, M.Y.; Oskoueian, E. Flavonoid analyses and antimicrobial activity of various parts of Phaleria macrocarpa (Scheff.) boerl Fruit. Int. J. Mol. Sci 2011, 12, 3422–3431. [Google Scholar]
- Moure, A.; Cruz, J.M.; Franco, D.; Dominguez, J.M.; Sineiro, J.; Dominguez, H.; Nunez, M.J.; Parajo, J.C. Natural antioxidants from residual sources. Food Chem 2001, 72, 145–171. [Google Scholar]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem 2006, 99, 191–203. [Google Scholar]
- Nigam, P.S.; Pandey, A. Biotechnology for Agro-Industrial Residues Utilisation: Utilisation of Agro-Residues, 1st ed.; Springer: Berlin, Germany, 2009; pp. 3–129. [Google Scholar]
- Bilusic Vundac, V.; Brantner, A.H.; Plazibat, M. Content of polyphenolic constituents and antioxidant activity of some Stachys taxa. Food Chem 2007, 104, 1277–1281. [Google Scholar]
- Vuorela, S.; Meyer, A.S.; Heinonen, M. Quantitative analysis of the main phenolics in rapeseed meal and oils processed differently using enzymatic hydrolysis and HPLC. Eur. Food Res. Tech 2003, 217, 517–523. [Google Scholar]
- Xiao, Z.P.; Peng, Z.Y.; Peng, M.J.; Yan, W.B.; Ouyang, Y.Z.; Zhu, H.L. Flavonoids health benefits and their molecular mechanism. Mini Rev. Med. Chem 2011, 11, 169–177. [Google Scholar]
- Prochazkova, D.; Bousova, I.; Wilhelmova, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar]
- Umamaheswari, M.; Asokkumar, K.; Sivashanmugam, A.T.; Remyaraju, A.; Subhadradevi, V.; Ravi, T.K. In vitro xanthine oxidase inhibitory activity of the fractions of Erythrina stricta Roxb. J. Ethnopharmacol 2009, 124, 646–648. [Google Scholar]
- Kubo, I.; Chen, Q.X.; Nihei, K.I. Molecular design of antibrowning agents: Antioxidative tyrosinase inhibitors. Food Chem 2003, 81, 241–247. [Google Scholar]
- Kim, Y.J.; Kang, K.S.; Yokozawa, T. The anti-melanogenic effect of pycnogenol by its anti-oxidative actions. Food Chem. Toxicol 2008, 46, 2466–2471. [Google Scholar]
- Tsai, P.J.; Tsai, T.H.; Yu, C.H.; Ho, S.C. Comparison of NO-scavenging and NO-suppressing activities of different herbal teas with those of green tea. Food Chem 2007, 103, 181–187. [Google Scholar]
- Kang, J.; Xie, C.; Li, Z.; Nagarajan, S.; Schauss, A.G.; Wu, T.; Wu, X. Flavonoids from acai (Euterpe oleracea Mart.) pulp and their antioxidant and anti-inflammatory activities. Food Chem 2011, 128, 152–157. [Google Scholar]
- Nwosu, F.; Morris, J.; Lund, V.A.; Stewart, D.; Ross, H.A.; McDougall, G.J. Anti-proliferative and potential anti-diabetic effects of phenolic-rich extracts from edible marine algae. Food Chem 2011, 126, 1006–1012. [Google Scholar]
- Salvati, S.; Attorri, L.; di Benedetto, R.; Fortuna, S.; di Biase, A. Micronutrient-Enriched rapeseed oils improve the brain oxidant/antioxidant system in rats fed a high-fat diet. J. Agric. Food Chem 2011, 59, 4483–4488. [Google Scholar]
- Liu, M.; Wallmon, A.; Olsson-Mortlock, C.; Wallin, R.; Saldeen, T. Mixed tocopherols inhibit platelet aggregation in humans: Potential mechanisms. Am. J. Clin. Nutr 2003, 77, 700–706. [Google Scholar]
- Chen, C.Y.; Milbury, P.E.; Lapsley, K.; Blumberg, J.B. Flavonoids from almond skins are bioavailable and act synergistically with vitamins C and E to enhance hamster and human LDL resistance to oxidation. J. Nutr 2005, 135, 1366–1373. [Google Scholar]
- Henry, G.E.; Momin, R.A.; Nair, M.G.; Dewitt, D.L. Antioxidant and cyclooxygenase activities of fatty acids found in food. J. Agric. Food Chem 2002, 50, 2231–2234. [Google Scholar]
- Muanda, F.N.; Dicko, A.; Soulimani, R. Assessment of polyphenolic compounds, in vitro antioxidant and anti-inflammation properties of Securidaca longepedunculata root barks. C. R. Biol 2010, 333, 663–669. [Google Scholar]
- Lee, C.J.; Chen, L.G.; Chang, T.L.; Ke, W.M.; Lo, Y.F.; Wang, C.C. The correlation between skin-care effects and phytochemical contents in Lamiaceae plants. Food Chem 2011, 124, 833–841. [Google Scholar]
- Rao, K.S.; Chaudhury, P.K.; Pradhan, A. Evaluation of anti-oxidant activities and total phenolic content of Chromolaena odorata. Food Chem. Toxicol 2010, 48, 729–732. [Google Scholar]
- Owen, P.L.; Johns, T. Xanthine oxidase inhibitory activity of northeastern North American plant remedies used for gout. J. Ethnopharmacol 1999, 64, 149–160. [Google Scholar]
- Wang, C.Y.; Ng, C.C.; Lin, H.T.; Shyu, Y.T. Free radical-scavenging and tyrosinase-inhibiting activities of extracts from sorghum distillery residue. J. Biosci. Bioeng 2011, 111, 554–556. [Google Scholar]
- Halici, M.; Odabasoglu, F.; Suleyman, H.; Cakir, A.; Aslan, A.; Bayir, Y. Effects of water extract of Usnea longissima on antioxidant enzyme activity and mucosal damage caused by indomethacin in rats. Phytomedicine 2005, 12, 656–662. [Google Scholar]
- Ismail, H.I.; Chan, K.W.; Mariod, A.A.; Ismail, M. Phenolic content and antioxidant activity of cantaloupe (cucumis melo) methanolic extracts. Food Chem 2010, 119, 643–647. [Google Scholar]
- Crozier, A.; Jensen, E.; Lean, M.; McDonald, M. Quantitative analysis of flavonoids by reversed-phase high-performance liquid chromatography. J. Chromatogr. A 1997, 761, 315–321. [Google Scholar]
- Gulcin, I.; Gungor Sat, I.; Beydemir, S.; Elmastas, M.; Kufrevioglu, O.I. Comparison of antioxidant activity of clove (Eugenia caryophylata Thunb) buds and lavender (Lavandula stoechas L.). Food Chem 2004, 87, 393–400. [Google Scholar]
- Orhan, I.; Kartal, M.; Naz, Q.; Ejaz, A.; Yilmaz, G.; Kan, Y.; Konuklugil, B.; Sener, B.; Choudhary, M.I. Antioxidant and anticholinesterase evaluation of selected Turkish Salvia species. Food Chem 2007, 103, 1247–1254. [Google Scholar]
- Lee, K.H.; Aziz, F.H.; Syahida, A.; Abas, F.; Shaari, K.; Israf, D.A.; Lajis, N.H. Synthesis and biological evaluation of curcumin-like diarylpentanoid analogues for anti-inflammatory, antioxidant and anti-tyrosinase activities. Eur. J. Medic. Chem 2009, 44, 3195–3200. [Google Scholar]
- Ahmad, R.; Ali, A.M.; Israf, D.A.; Ismail, N.H.; Shaari, K.; Lajis, N.H. Antioxidant, radical-scavenging, anti-inflammatory, cytotoxic and antibacterial activities of methanolic extracts of some Hedyotis species. Life Sci 2005, 76, 1953–1964. [Google Scholar]
- SAS, version 9.1.3; SAS Institute Inc: Cary, NC, USA, 2003.
- GraphPad Prism, version 5.01; GraphPad Software Inc.: San Diego, CA, USA, 2007.


| Samples | Total Phenolic Compounds * | Total Flavonoid Compounds ** |
|---|---|---|
| Rapeseed meal | 5.3 a | 2.3 a |
| Cottonseed meal | 1.5 b | 1.2 b |
| Soybean meal | 0.9 c | 0.8 c |
| SEM | 0.09 | 0.07 |
| P value | 0.01 | 0.01 |
| Compounds | Phenolic, Flavonoid and Isoflavonoid (μg g−1 DW)
| ||
|---|---|---|---|
| Rapeseed Meal | Cottonseed Meal | Soybean Meal | |
| Phenolics | |||
| Gallic acid | 419.5 ± 3.32 | nd | nd |
| Salicylic acid | nd | nd | nd |
| Caffeic acid | nd | nd | 295.8 ± 2.73 |
| Vanillic acid | nd | nd | nd |
| Syringic acid | 177.6 ± 2.82 | nd | nd |
| Flavonoids | |||
| Apigenin | 64.9 ± 2.48 | nd | nd |
| Kaempferol | 494.0 ± 1.71 | 113.7 ± 4.59 | nd |
| Myricetin | nd | nd | nd |
| Naringenin | 793.5 ± 5.90 | 178.9 ± 6.88 | 352.3 ± 3.66 |
| Quercetin | nd | nd | nd |
| Rutin | nd | 209.1 ± 2.45 | nd |
| Isoflavonoids | |||
| Daidzein | nd | nd | 521.4 ± 6.58 |
| Genistein | nd | nd | nd |
| Samples | IC50 (μg mL−1)
| |
|---|---|---|
| DPPH Scavenging Activity | Nitric Oxide Scavenging Activity | |
| Rapeseed meal | 87.9 c | 108.2 b |
| Cottonseed meal | 191.1 b | 191.3 a |
| Soybean meal | 466.3 a | 207.6 a |
| Vitamin C | 20.4 d | 52.7 c |
| Vitamin E | 45.4 d | 72.9 c |
| SEM | 7.98 | 7.12 |
| P value | P < 0.01 | P < 0.01 |
| Samples | IC50 μg mL−1 | |
|---|---|---|
| Xanthine Oxidase Inhibitory Activity | Tyrosinase Inhibitory Activity | |
| Rapeseed meal | 84.8 b | 120.3 c |
| Cottonseed meal | 86.8 b | 286.0 b |
| Soybean meal | 464.2 a | >500 a |
| Allopurinol | 29.7 c | - |
| Kojic acid | - | 116.2 c |
| SEM | 7.55 | 5.43 |
| P value | P < 0.01 | P < 0.01 |
| Correlation (r2)
| |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Parameters | TPC | TFC | DPPH | NO | FTC | TBA | XO | TI | iNOS |
| TPC | - | 0.987 | 0.637 | 0.998 | 0.877 | 0.839 | 0.373 | 0.702 | 0.854 |
| TFC | - | - | 0.734 | 0.993 | 0.933 | 0.911 | 0.482 | 0.596 | 0.922 |
| DPPH | - | - | - | 0.656 | 0.911 | 0.939 | 0.933 | 0.111 | 0.932 |
| NO | - | - | - | - | 0.898 | 0.863 | 0.397 | 0.677 | 0.873 |
| FTC | - | - | - | - | - | 0.997 | 0.714 | 0.359 | 0.998 |
| TBA | - | - | - | - | - | - | 0.762 | 0.307 | 0.999 |
| XO | - | - | - | - | - | - | - | 0.006 | 0.749 |
| TI | - | - | - | - | - | - | - | - | 0.321 |
| iNOS | - | - | - | - | - | - | - | - | - |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Oskoueian, E.; Abdullah, N.; Hendra, R.; Karimi, E. Bioactive Compounds, Antioxidant, Xanthine Oxidase Inhibitory, Tyrosinase Inhibitory and Anti-Inflammatory Activities of Selected Agro-Industrial By-products. Int. J. Mol. Sci. 2011, 12, 8610-8625. https://doi.org/10.3390/ijms12128610
Oskoueian E, Abdullah N, Hendra R, Karimi E. Bioactive Compounds, Antioxidant, Xanthine Oxidase Inhibitory, Tyrosinase Inhibitory and Anti-Inflammatory Activities of Selected Agro-Industrial By-products. International Journal of Molecular Sciences. 2011; 12(12):8610-8625. https://doi.org/10.3390/ijms12128610
Chicago/Turabian StyleOskoueian, Ehsan, Norhani Abdullah, Rudi Hendra, and Ehsan Karimi. 2011. "Bioactive Compounds, Antioxidant, Xanthine Oxidase Inhibitory, Tyrosinase Inhibitory and Anti-Inflammatory Activities of Selected Agro-Industrial By-products" International Journal of Molecular Sciences 12, no. 12: 8610-8625. https://doi.org/10.3390/ijms12128610




