Novel Ionic Liquid with Both Lewis and Brønsted Acid Sites for Michael Addition
Abstract
:1. Introduction
2. Results and Discussion
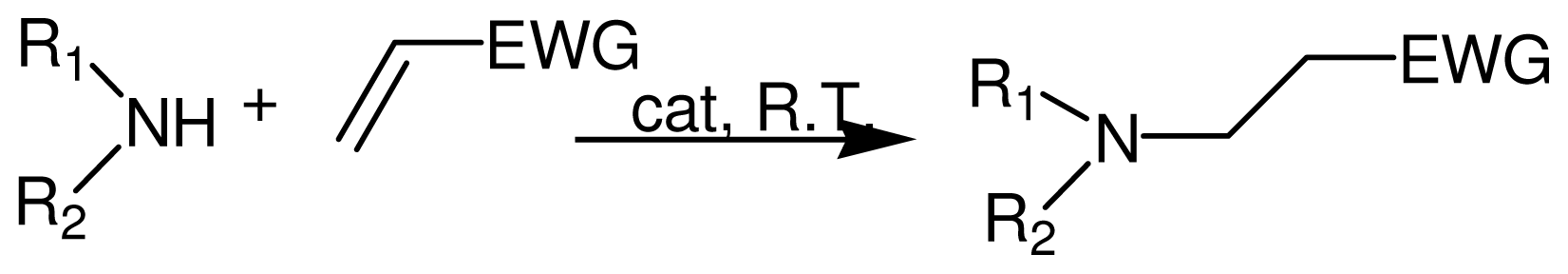
2.1. Catalytic Procedure for the Conjugate Addition
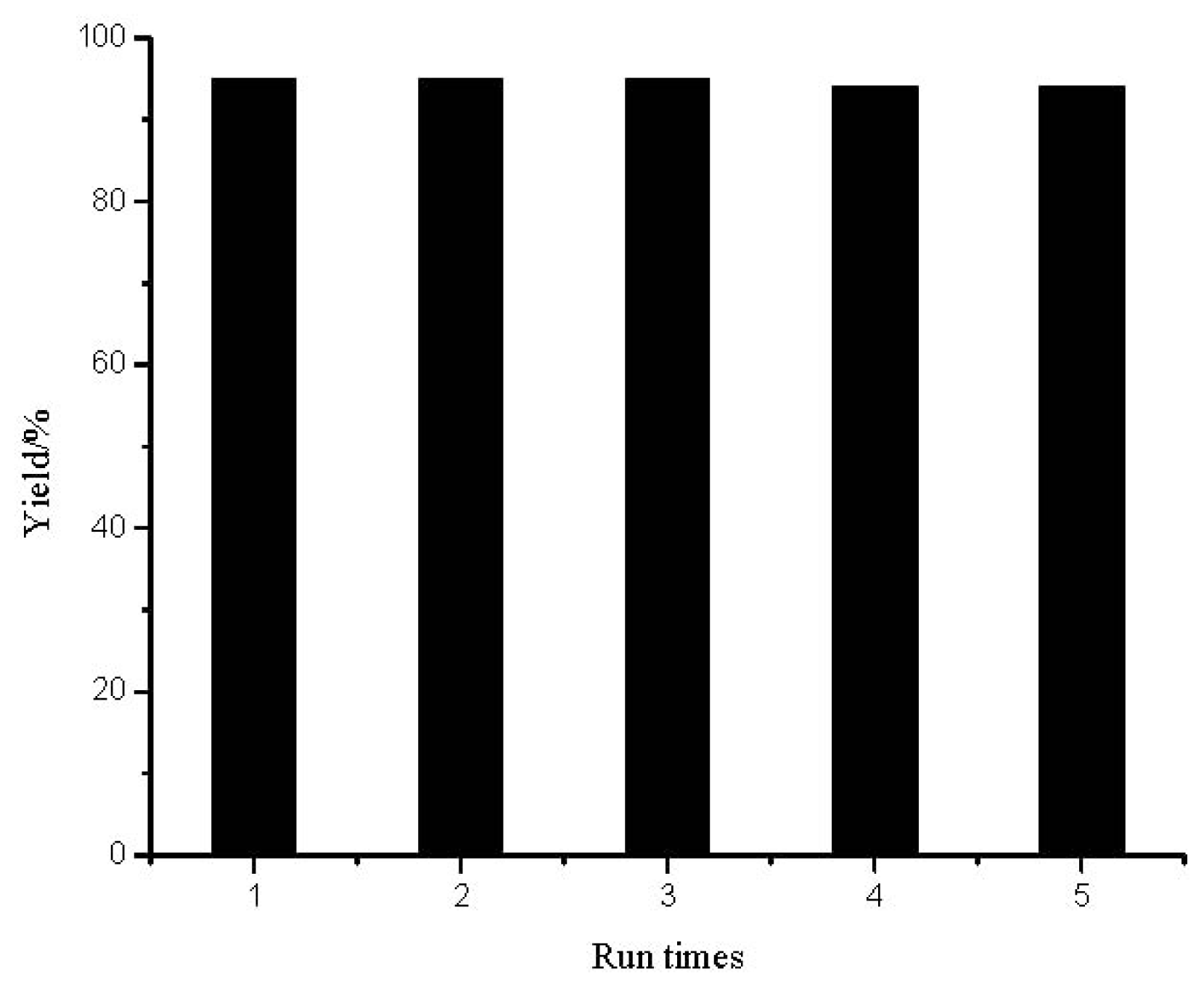
2.2. The Reuse of the Ionic Liquid
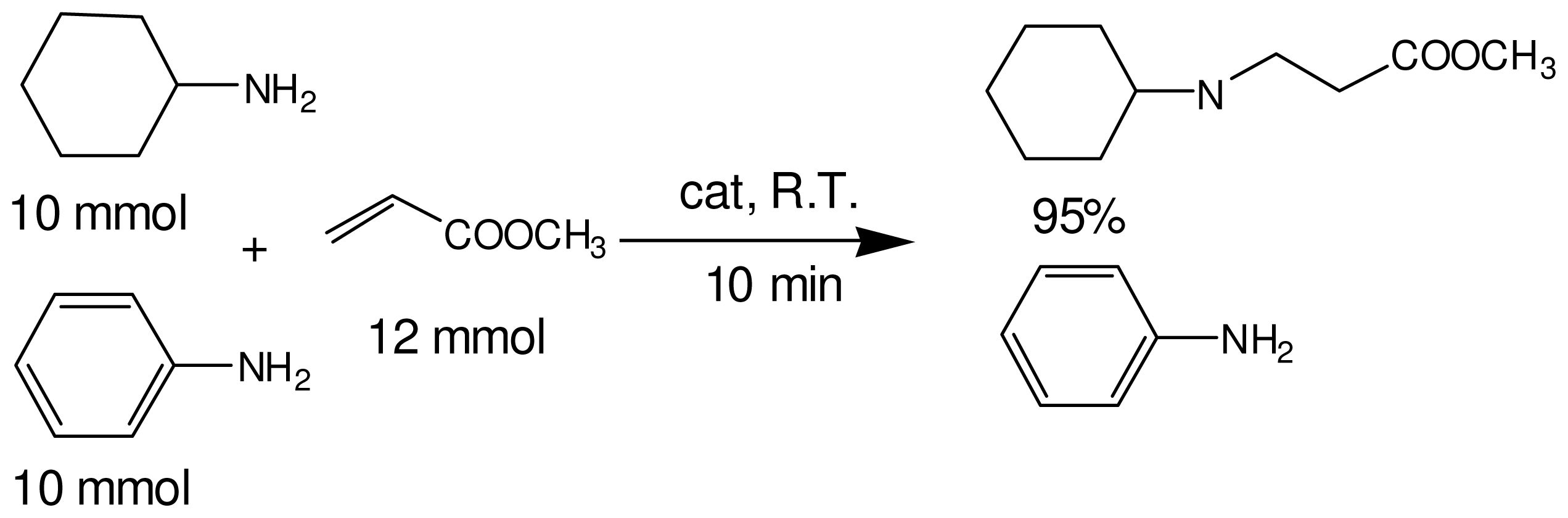
2.3. The Chemoselectivity of the Novel Ionic Liquid
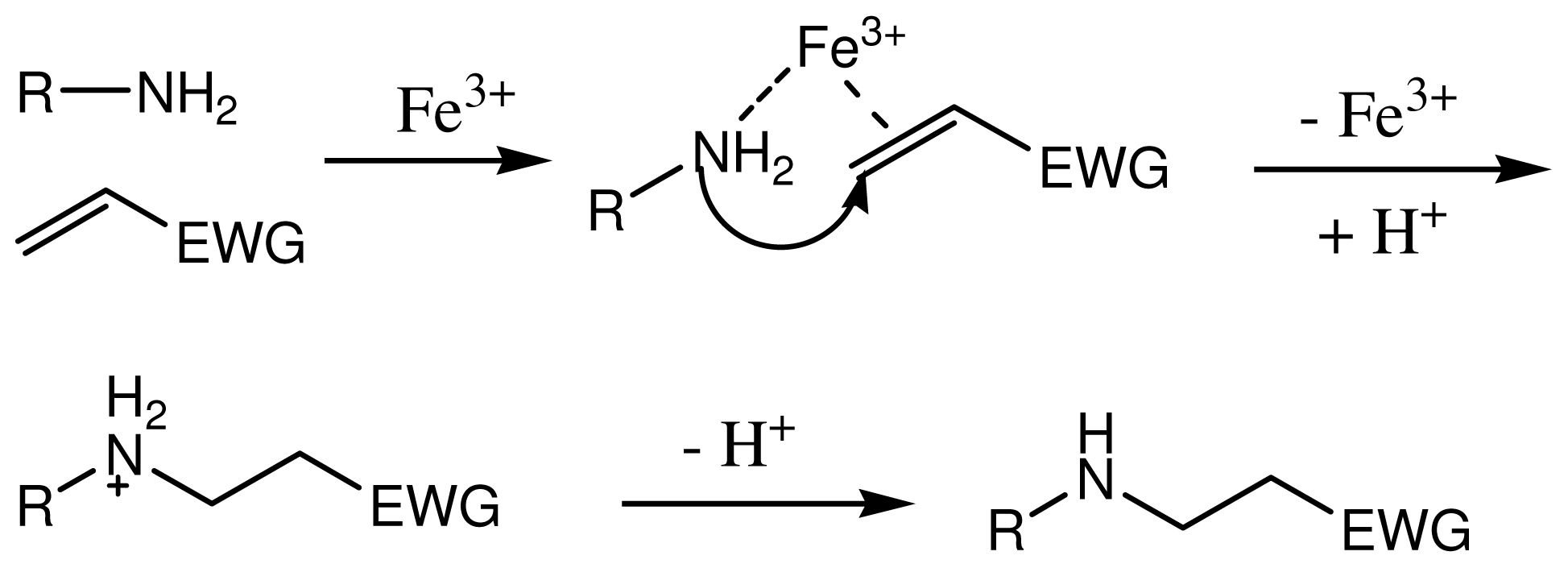
2.4. The Fe3+ Content on the Catalytic Activities of the Novel Ionic Liquid
2.5. The Comparative Study on the Catalytic Activities of the Different Catalysts
3. Experimental Section
3.1. Synthesis of the Novel Ionic Liquid
3.2. Conjugate Addition of Amines to Electron Deficient Alkenes
4. Conclusions
Acknowledgements
References
- Kuang, S.; Uchida, R.; Humphry-Baker, S.M.; Zakeeruddin, M. Organic dye-sensitized ionic liquid based solar cells: Remarkable enhancement in performance through molecular design of indoline sensitizers. Angew. Chem. Int. Ed 2008, 47, 1923–1927. [Google Scholar]
- Gupta, N.S.; Kad, G.L.; Singh, J. Acidic ionic liquid [bmim]HSO4: An efficient catalyst for acetalization and thioacetalization of carbonyl compounds and their subsequent deprotection. Catal. Commun 2007, 8, 1323–1328. [Google Scholar]
- Cole, A.C.; Jensen, J.L.; Ntai, I.; Tran, K.L.T.; Weaver, K.J.; Forbes, D.C.; Davis, J.H. Novel Brønsted acidic ionic liquids and their use as dual solvent-catalysts. J. Am. Chem. Soc 2002, 124, 5962–5963. [Google Scholar]
- Kamal, A.; Chouhan, G. Investigations towards the chemoselective thioacetaliztion of carbonyl compounds by using ionic liquid [bmim]Br as a recyclable catalytic medium. Adv. Synth. Catal 2004, 346, 579–582. [Google Scholar]
- Firouzabadi, H.; Iranpoor, N.; Jafari, A.A.; Jafari, M.R. Tungstophosphoric acid supported on silica gel (H3PW12O40/SiO2) as an eco-friendly, reusable and heterogeneous catalyst for chemoselective oxathioacetalization of carbonyl compounds in solution or under solvent-free conditions. J. Mol. Catal. A 2006, 247, 14–18. [Google Scholar]
- Zhang, D.; Wang, G.; Zhu, R. Insight into the mechanism of the Michael addition of malononitrile to α, β-unsaturated imides catalyzed by bifunctional thiourea catalysts. Tetrahedron Asymmetry 2008, 19, 568–576. [Google Scholar]
- Singh, R.; Goswami, T. Acid catalyzed 1, 2-Michael addition reaction: A viable synthetic route in designing fullerene core starlike macromolecule. J. Phys. Org. Chem 2008, 21, 225–236. [Google Scholar]
- Wang, C.-J.; Zhang, Z.-H.; Dong, X.-Q.; Wu, X.-J. Chiral amine-thioureas bearing multiple hydrogen bonding donors: Highly efficient organocatalysts for asymmetric Michael addition of acetylacetone to nitroolefins. Chem. Commun 2008, 1431–1433. [Google Scholar]

| Entry | Amines | Alkenes | Reaction Time/min | Yield, % a,b |
|---|---|---|---|---|
| 1 | 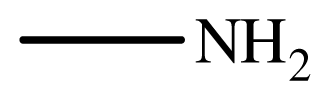 |  | 2 | 99 |
| 2 | 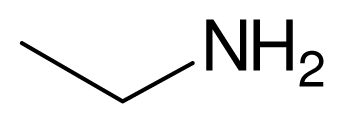 |  | 3 | 99 |
| 3 | 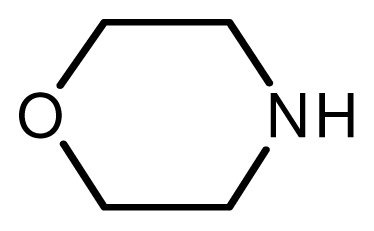 | 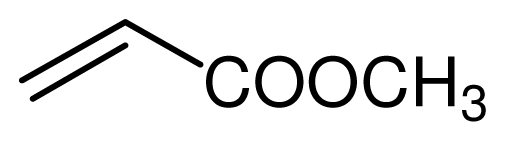 | 7 | 99 |
| 4 | 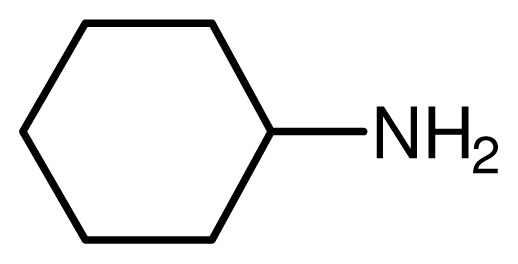 | 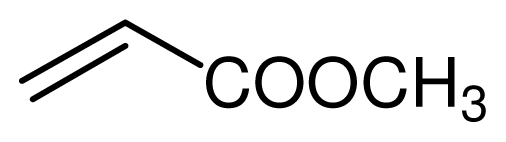 | 10 | 95 |
| 5 |  |  | 4 | 99 |
| 6 |  |  | 7 | 97 |
| 7 | 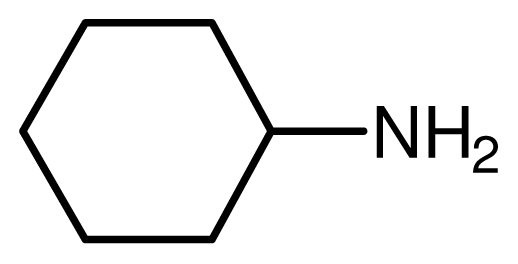 | 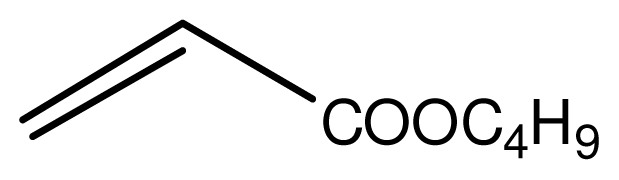 | 13 | 93 |
| 8 |  |  | 5 | 99 |
| 9 | 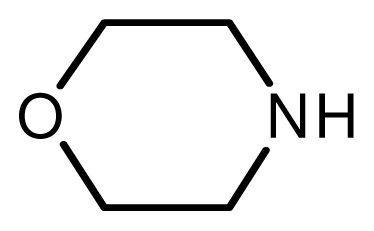 | 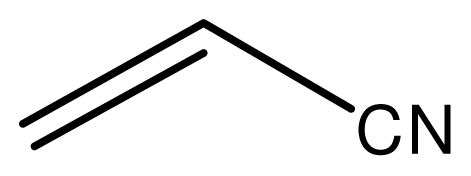 | 6 | 98 |
| 10 |  | 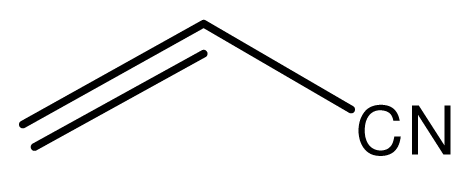 | 12 | 94 |
| Entry | Fe3+ content/% c | Catalyst amount/mmol | Reaction time/min | Yield/% a,b |
|---|---|---|---|---|
| 1 | 0 | 1.00 | 25 | 90 |
| 2 | 25 | 0.50 | 15 | 92 |
| 3 | 50 | 0.14 | 10 | 95 |
| 4 | 75 | 0.75 | 20 | 93 |
| 5 | 100 | 1.20 | 30 | 90 |
| Entry | Catalyst | Catalyst amount/mmol | Reaction time/min | Yield/% a,b |
|---|---|---|---|---|
| 1 | Novel ionic liquid | 0.14 | 10 | 95 |
| 2 | H2SO4 | 1.5 | 15 | 90 |
| 3 | PTSA | 2.0 | 12 | 92 |
| 4 | BF3–OEt2 | 5.5 | 25 | 88 |
| 5 | [mimbSO3H][HSO4] | 1.5 | 15 | 91 |
| 6 | ZnCl2 | 6.5 | 20 | 88 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jiang, X.; Ye, W.; Song, X.; Ma, W.; Lao, X.; Shen, R. Novel Ionic Liquid with Both Lewis and Brønsted Acid Sites for Michael Addition. Int. J. Mol. Sci. 2011, 12, 7438-7444. https://doi.org/10.3390/ijms12117438
Jiang X, Ye W, Song X, Ma W, Lao X, Shen R. Novel Ionic Liquid with Both Lewis and Brønsted Acid Sites for Michael Addition. International Journal of Molecular Sciences. 2011; 12(11):7438-7444. https://doi.org/10.3390/ijms12117438
Chicago/Turabian StyleJiang, Xiaoyue, Weidong Ye, Xiaohua Song, Wenxin Ma, Xuejun Lao, and Runpu Shen. 2011. "Novel Ionic Liquid with Both Lewis and Brønsted Acid Sites for Michael Addition" International Journal of Molecular Sciences 12, no. 11: 7438-7444. https://doi.org/10.3390/ijms12117438




