HE4 in the Differential Diagnosis of a Pelvic Mass: A Case Report
Abstract
:1. Introduction
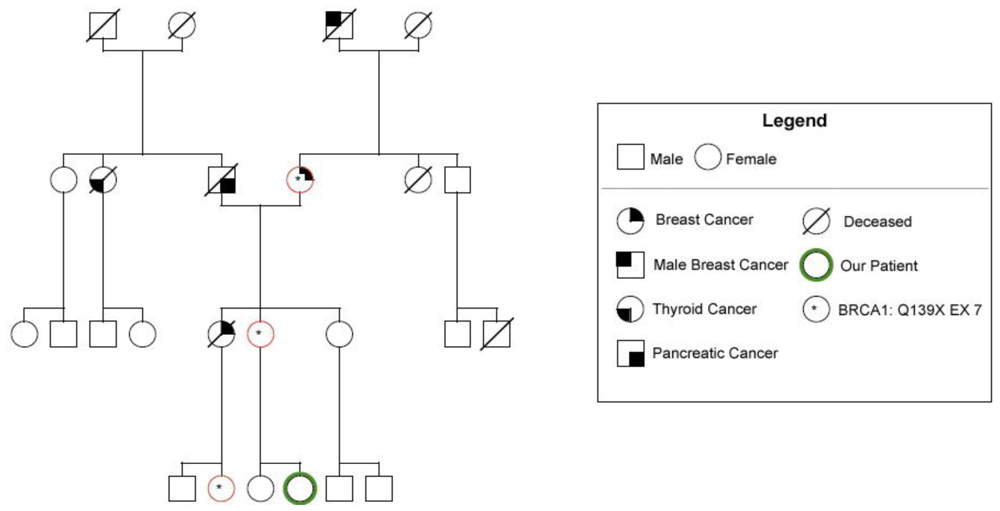
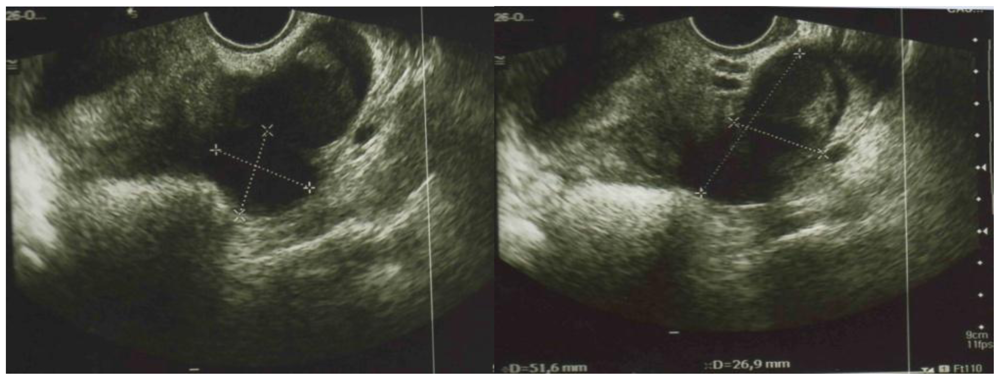
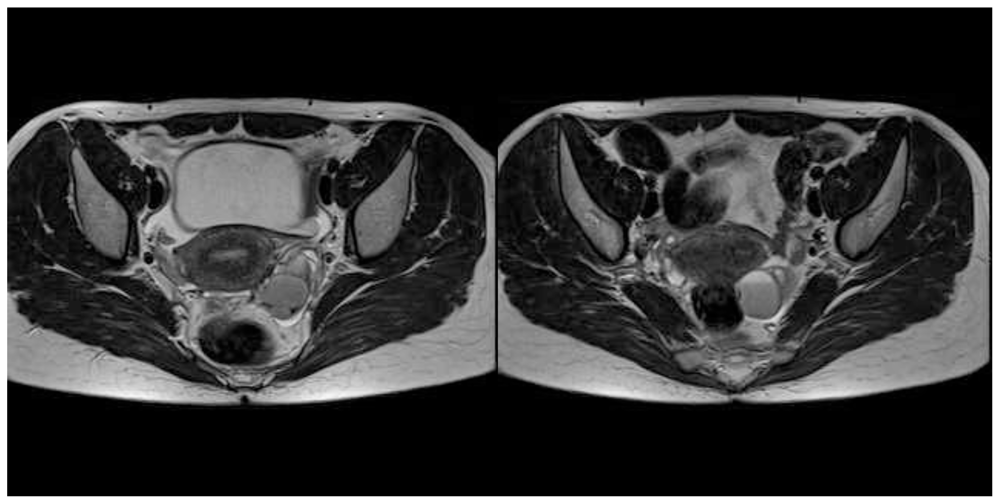
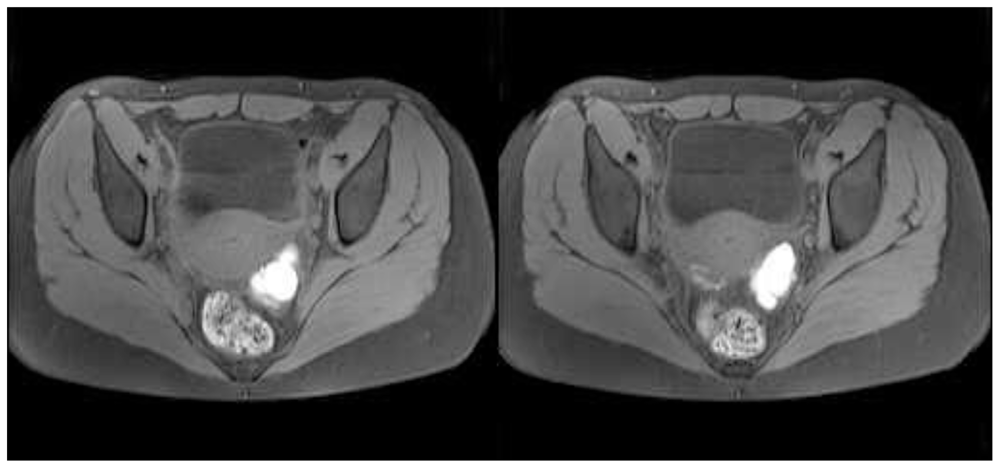
2. Case Presentation and Discussion
3. Conclusions
Acknowledgements
References
- Russo, A; Calò, V; Bruno, L; Rizzo, S; Bazan, V; Di Fede, G. Hereditary ovarian cancer. Crit. Rev. Oncol. Hematol 2009, 69, 28–44. [Google Scholar]
- Palma, M; Ristori, E; Ricevuto, E; Giannini, G; Gulino, A. BRAC1 and BRAC2: the genetic testing and the current management options for mutation carriers. Crit. Rev. Oncol. Hematol 2006, 57, 1–23. [Google Scholar]
- Schink, JC. Current initial therapy of stage II and IV ovarian cancer: challenges for managed care. Sem. Oncol 1999, 26, 2–7. [Google Scholar]
- Hellerstrom, I; Hellerstron, KE. SMRP and HE4 as biomarkers for ovarian carcinoma when used alone and in combination with CA125 and/or each other. Adv. Exp. Med. Biol 2008, 622, 15–21. [Google Scholar]
- Kirchhoff, C. Molecular characterization of epididymal proteins. Rev. Reprod 1998, 3, 86–95. [Google Scholar]
- Bingle, L; Singleton, V; Bingle, CD. The putative ovarian tumor marker gene HE4 (WFDC2), is expressed in normal tissues and undergoes complex alternative splicing to yeld multiple protein isoforms. Oncogene 2002, 21, 2768–2773. [Google Scholar]
- Clauss, A; Lilja, H; Lundwall, A. The evolution of a genetic locus encoding small serine proteinase inhibitors. Biochem. Biophys. Res. Commun 2005, 333, 383–389. [Google Scholar]
- Anastasi, E; Granato, T; Marchei, GG; Viggiani, V; Colaprisca, B; Comploj, S; Reale, MG; Frati, L; Midulla, C. Ovarian tumor marker HE4 is differently expressed during the phases of the menstrual cycle in healthy young women. Tumor Biol 2010, 31, 411–415. [Google Scholar]
- Anastasi, E; Marchei, GG; Viggiani, V; Gennarini, G; Frati, L; Reale, MG. HE4: A new potential early biomarker for the recurrence of ovarian cancer. Tumor Biol 2010, 31, 113–119. [Google Scholar]
- Chamiè, LP; Blasbalg, R; Goncalves, MO; Carvalho, FM; Abrao, MS; de Oliveira, IS. Accuracy of magnetic resonance imaging for siagnosis and preoperative assessement of deeply infiltrating endometriosis. Int. J. Gynecol. Obstet 2009, 106, 198–201. [Google Scholar]
- Guerra, A; Cunha, TM; Félix, A. Magnetic resonance evaluation of adnexal masses. Acta Radiol 2008, 49, 700–709. [Google Scholar]
- Anderson, GL; Mcintosh, M; Wu, L; Barnett, M; Goodman, G; Thorpe, JD; Bergan, L; Thornnquist, MD; Scholler, N; Kim, N; O’Briant, K; Drescher, C; Urban, N. Assessing lead time of selected ovarian cancer biomarkers. A nested case-control study. J. Natl. Cancer Inst 2010, 102, 26–38. [Google Scholar]
| Sample 1 | Sample 2 | |
|---|---|---|
| CA 125 (U/mL) | 185 | 330 |
| HE4 (pmol/L) | 44 | 40 |
© 2011 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Anastasi, E.; Granato, T.; Coppa, A.; Manganaro, L.; Giannini, G.; Comploj, S.; Frati, L.; Midulla, C. HE4 in the Differential Diagnosis of a Pelvic Mass: A Case Report. Int. J. Mol. Sci. 2011, 12, 627-632. https://doi.org/10.3390/ijms12010627
Anastasi E, Granato T, Coppa A, Manganaro L, Giannini G, Comploj S, Frati L, Midulla C. HE4 in the Differential Diagnosis of a Pelvic Mass: A Case Report. International Journal of Molecular Sciences. 2011; 12(1):627-632. https://doi.org/10.3390/ijms12010627
Chicago/Turabian StyleAnastasi, Emanuela, Teresa Granato, Anna Coppa, Lucia Manganaro, Giuseppe Giannini, Sara Comploj, Luigi Frati, and Cecilia Midulla. 2011. "HE4 in the Differential Diagnosis of a Pelvic Mass: A Case Report" International Journal of Molecular Sciences 12, no. 1: 627-632. https://doi.org/10.3390/ijms12010627





