Deleted in Malignant Brain Tumors-1 Protein (DMBT1): A Pattern Recognition Receptor with Multiple Binding Sites
Abstract
:1. Introduction: Saliva as a Protective Fluid
2. Adherence and Oral Infections
3. Deleted in Malignant Brain Tumors-1 (DMBT1), Salivary Agglutinin (DMBT1SAG) and Glycoprotein 340 (DMBT1GP340)
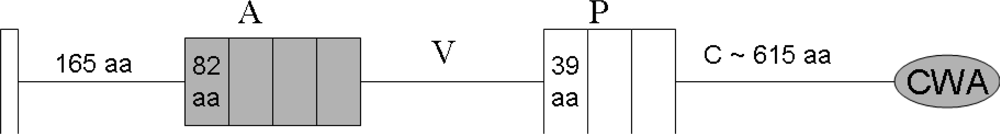
4. The Domain Organization of DMBT1
5. SRCR Domains
6. CUB Domains
7. Zona Pellucida Domains
8. Glycosylation Patterns of DMBT1SAG
9. Bacteria Binding Sites on DMBT1SAG
9.1. Binding Sites on the SRCR Domains
9.2. Binding Sites on the Glycan Chains
10. DMBT1SAG Binding Sites on Bacteria
10.1. Antigen I/II Polypeptides
10.2. Pathogen Associated Molecular Patterns
11. Interaction with Viruses
12. Interaction with Endogenous Protein Ligands
13. Role of DMBT1 in Innate Immunity
14. Conclusions
References
- Tlaskalova-Hogenova, H; Tuckova, L; Mestecky, J; Kolinska, J; Rossmann, P; Stepankova, R; Kozakova, H; Hudcovic, T; Hrncir, T; Frolova, L; Kverka, M. Interaction of mucosal microbiota with the innate immune system. Scand. J. Immunol 2005, 62, 106–113. [Google Scholar]
- Schenkels, LCPM; Veerman, ECI; Nieuw Amerongen, AV. Biochemical composition of human saliva in relation to other mucosal fluids. Crit. Rev. Oral Biol. Med 1995, 6, 161–175. [Google Scholar]
- Valdez, IH; Fox, PC. Diagnosis and management of salivary dysfunction. Critical Reviews in Oral Biol. Med 1993, 4, 271–277. [Google Scholar]
- Daniels, TE. Evaluation, differential diagnosis, and treatment of xerostomia. J. Rheumatol 2000, 27, 6–10. [Google Scholar]
- Almstahl, A; Wikstrom, M; Stenberg, I; Jakobsson, A; Fagerberg-Mohlin, B. Oral microbiota associated with hyposalivation of different origins. Oral Microbiol. Immunol 2003, 18, 1–8. [Google Scholar]
- Scannapieco, FA. Role of oral bacteria in respiratory infection. J. Periodontol 1999, 70, 793–802. [Google Scholar]
- Kolenbrander, PE; London, J. Adhere today, here tomorrow: Oral bacterial adherence. J. Bacteriol 1993, 175, 3247–3252. [Google Scholar]
- Gibbons, RJ. Adherent interactions which may affect microbial ecology in the mouth. J. Dent. Res 1984, 63, 378–385. [Google Scholar]
- Emilson, CG; Ciardi, JE; Olsson, J; Bowen, WH. The influence of saliva on infection of the human mouth by mutans streptococci. Arch. Oral Biol 1989, 34, 335–340. [Google Scholar]
- Rudney, JD; Pan, Y; Chen, R. Streptococcal diversity in oral biofilms with respect to salivary function. Arch. Oral Biol 2003, 48, 475–493. [Google Scholar]
- Rudney, JD. Does variability in salivary protein concentrations influence oral microbial ecology and oral health? Crit. Rev. Oral Biol. Med 1995, 6, 343–367. [Google Scholar]
- Ericson, T; Rundegren, J. Characterization of a salivary agglutinin reacting with a serotype c strain of Streptococcus mutans. Eur. J. Biochem 1983, 133, 255–261. [Google Scholar]
- Ligtenberg, AJ; Veerman, EC; Nieuw Amerongen, AV. A role for Lewis a antigens on salivary agglutinin in binding to Streptococcus mutans. Antonie van Leeuwenhoek 2000, 77, 21–30. [Google Scholar]
- Rundegren, J. Calcium-dependent salivary agglutinin with reactivity to various oral bacterial species. Infect. Immun 1986, 53, 173–178. [Google Scholar]
- Rundegren, J; Arnold, RR. Differentiation and interaction of secretory immunoglobulin A and a calcium-dependent parotid agglutinin for several bacterial strains. Infect. Immun 1987, 55, 288–292. [Google Scholar]
- Boackle, RJ; Connor, MH; Vesely, J. High molecular weight non-immunoglobulin salivary agglutinins (NIA) bind C1Q globular heads and have the potential to activate the first complement component. Mol. Immunol 1993, 30, 309–319. [Google Scholar]
- Mitoma, M; Oho, T; Shimazaki, Y; Koga, T. Inhibitory effect of bovine milk lactoferrin on the interaction between a streptococcal surface protein antigen and human salivary agglutinin. J. Biol. Chem 2001, 276, 18060–18065. [Google Scholar]
- Ligtenberg, AJM; Bikker, FJ; de Blieck-Hogervorst, JMA; Veerman, ECI; Nieuw Amerongen, AV. Binding of salivary agglutinin to IgA. Biochem. J 2004, 383, 159–164. [Google Scholar]
- End, C; Lyer, S; Renner, M; Stahl, C; Ditzer, J; Holloschi, A; Kuhn, HM; Flammann, HT; Poustka, A; Hafner, M; Mollenhauer, J; Kioschis, P. Generation of a vector system facilitating cloning of DMBT1 variants and recombinant expression of functional full-length DMBT1. Protein Expres. Purif 2005, 41, 275–286. [Google Scholar]
- Holmskov, U; Lawson, P; Teisner, B; Tornoe, I; Willis, AC; Morgan, C; Koch, C; Reid, KBM. Isolation and characterization of a new member of the Scavenger receptor superfamily, glycoprotein-340 (GP340), as a lung surfactant protein D binding molecule. J. Biol. Chem 1997, 272, 13743–13749. [Google Scholar]
- Ligtenberg, TJM; Bikker, FJ; Groenink, J; Tornoe, I; Leth-Larsen, R; Veerman, ECI; Amerongen, AVN; Holmskov, U. Human salivary agglutinin binds to lung surfactant protein-D and is identical with scavenger receptor protein gp-340. Biochem. J 2001, 359, 243–248. [Google Scholar]
- Prakobphol, A; Xu, F; Hoang, VM; Larsson, T; Bergstrom, J; Johansson, I; Frangsmyr, L; Holmskov, U; Leffler, H; Nilsson, C; Boren, T; Wright, JR; Stromberg, N; Fisher, SJ. Salivary agglutinin, which binds Streptococcus mutans and Helicobacter pylori, is the lung scavenger receptor cysteine-rich protein gp-340. J. Biol. Chem 2000, 275, 39860–39866. [Google Scholar]
- Holmskov, U; Mollenhauer, J; Madsen, J; Vitved, L; Gronlund, J; Tornoe, I; Kliem, A; Reid, KBM; Poustka, A; Skjodt, K. Cloning of GP-340, a putative opsonin receptor for lung surfactant protein D. Proc. Natl. Acad. Sci. USA 1999, 96, 10794–10799. [Google Scholar]
- Mollenhauer, J; Wiemann, S; Scheurlen, W; Korn, B; Hayashi, Y; Wilgenbus, KK; vonDeimling, A; Poustka, A. DMBT1, a new member of the SRCR superfamily, on chromosome 10q25.3-26.1 is deleted in malignant brain tumours. Nat. Genet 1997, 17, 32–39. [Google Scholar]
- Tino, MJ; Wright, JR. Glycoprotein-340 binds surfactant protein-A (SP-A) and stimulates alveolar macrophage migration in an SP-A-independent manner. Am. J. Respirat. Cell Mol. Biol 1999, 20, 759–768. [Google Scholar]
- Crouch, E; Hartshorn, K; Ofek, I. Collectins and pulmonary innate immunity. Immunol. Rev 2000, 173, 52–65. [Google Scholar]
- Mori, M; Shiraishi, T; Tanaka, S; Yamagata, M; Mafune, K; Tanaka, Y; Ueo, H; Barnard, GF; Sugimachi, K. Lack of DMBT1 expression in oesophageal, gastric and colon cancers. Br. J. Cancer 1999, 79, 211–213. [Google Scholar]
- Somerville, RPT; Shoshan, Y; Eng, C; Barnett, G; Miller, D; Cowell, JK. Molecular analysis of two putative tumour suppressor genes, PTEN and DMBT, which have been implicated in glioblastoma multiforme disease progression. Oncogene 1998, 17, 1755–1757. [Google Scholar]
- Wu, WG; Kemp, BL; Proctor, ML; Gazdar, AF; Minna, JD; Hong, WK; Mao, L. Expression of DMBT1, a candidate tumor suppressor gene, is frequently lost in lung cancer. Cancer Res 1999, 59, 1846–1851. [Google Scholar]
- Mollenhauer, J; Holmskov, U; Wiemann, S; Krebs, I; Herbertz, S; Madsen, J; Kioschis, P; Coy, JF; Poustka, A. The genomic structure of the DMBT1 gene: Evidence for a region with susceptibility to genomic instability. Oncogene 1999, 18, 6233–6240. [Google Scholar]
- Braidotti, P; Nuciforo, PG; Mollenhauer, J; Poustka, A; Pellegrini, C; Moro, A; Bulfamante, G; Coggi, G; Bosari, S; Pietra, GG. DMBT1 expression is down-regulated in breast cancer. Bmc Cancer 2004, 4. [Google Scholar]
- Mollenhauer, J; Muller, H; Kollender, G; Lyer, S; Diedrichs, L; Helmke, B; Holmskov, U; Ligtenberg, T; Herbertz, S; Krebs, I; Madsen, J; Bikker, F; Schmitt, L; Wiemann, S; Scheurlen, W; Otto, HF; von Deimling, A; Poustka, A. The SRCR/SID region of DMBT1 defines a complex multi-allele system representing the major basis for its variability in cancer. Genes Chromosomes Cancer 2002, 35, 242–255. [Google Scholar]
- Mollenhauer, J; Helmke, B; Muller, H; Kollender, G; Lyer, S; Diedrichs, L; Holmskov, U; Ligtenberg, T; Herbertz, S; Krebs, I; Wiemann, S; Madsen, J; Bikker, F; Schmitt, L; Otto, HF; Poustka, A. Sequential changes of the DMBT1 expression and location in normal lung tissue and lung carcinomas. Genes Chromosomes Cancer 2002, 35, 164–169. [Google Scholar]
- Mollenhauer, J; Deichmann, M; Helmke, B; Muller, H; Kollender, G; Holmskov, U; Ligtenberg, T; Krebs, I; Wiemann, S; Bantel-Schaal, U; Madsen, J; Bikker, F; Klauck, SM; Otto, HF; Moldenhauer, G; Poustka, A. Frequent downregulation of DMBT1 and galectin-3 in epithelial skin cancer. Int. J. Cancer 2003, 105, 149–157. [Google Scholar]
- Mollenhauer, J; Herbertz, S; Helmke, B; Kollender, G; Krebs, I; Madsen, J; Holmskov, U; Sorger, K; Schmitt, L; Wiemann, S; Otto, HF; Grone, HJ; Poustka, A. Deleted in Malignant Brain Tumors 1 is a versatile mucin-like molecule likely to play a differential role in digestive tract cancer. Cancer Res 2001, 61, 8880–8886. [Google Scholar]
- Mollenhauer, J; Helmke, B; Medina, D; Bergmann, G; Gassler, N; Muller, H; Lyer, S; Diedrichs, L; Renner, M; Wittig, R; Blaich, S; Hamann, U; Madsen, J; Holmskov, U; Bikker, F; Ligtenberg, A; Carlen, A; Olsson, J; Otto, HF; O’Malley, B; Poustka, A. Carcinogen inducibility in vivo and down-regulation of DMBTI during breast carcinogenesis. Genes Chromosomes Cancer 2004, 39, 185–194. [Google Scholar]
- Aruffo, A; Bowen, MA; Patel, DD; Haynes, BF; Starling, GC; Gebe, JA; Bajorath, J. CD6-ligand interactions: A paradigm for SRCR domain function? Immunol. Today 1997, 18, 498–504. [Google Scholar]
- Resnick, D; Pearson, A; Krieger, M. The SRCR superfamily: A family reminiscent of the Ig superfamily. Trends Biochem. Sci 1994, 19, 5–8. [Google Scholar]
- Bork, P; Beckmann, G. The CUB Domain - a widespread module in developmentally-regulated proteins. J. Mol. Biol 1993, 231, 539–545. [Google Scholar]
- Sarrias, MR; Gronlund, J; Padilla, O; Madsen, J; Holmskov, U; Lozano, F. The scavenger receptor cysteine-rich (SRCR) domain: An ancient and highly conserved protein module of the innate immune system. Crit. Rev. Immunol 2004, 24, 1–37. [Google Scholar]
- Muller, WEG. Origin of metazoan adhesion molecules and adhesion receptors as deduced from cDNA analysis in the marine sponge Geodia cydonium: A review. Cell Tissue Res 2003, 289, 383–395. [Google Scholar]
- Hohenester, E; Sasaki, T; Timpl, R. Crystal structure of a scavenger receptor cysteine-rich domain sheds light on an ancient superfamily. Nat. Struct. Biol 1999, 6, 228–232. [Google Scholar]
- Claudianos, C; Dessens, JT; Trueman, HE; Arai, M; Mendoza, J; Butcher, GA; Crompton, T; Sinden, RE. A malaria scavenger receptor-like protein essential for parasite development. Mol. Microbiol 2002, 45, 1473–1484. [Google Scholar]
- Abrahamsen, MS; Templeton, TJ; Enomoto, S; Abrahante, JE; Zhu, G; Lancto, CA; Deng, MQ; Liu, C; Widmer, G; Tzipori, S; Buck, GA; Xu, P; Bankier, AT; Dear, PH; Konfortov, BA; Spriggs, HF; Iyer, L; Anantharaman, V; Aravind, L; Kapur, V. Complete genome sequence of the apicomplexan, Cryptosporidium parvum. Science 2004, 304, 441–445. [Google Scholar]
- Rast, JP; Smith, LC; Loza-Coll, M; Hibino, T; Litman, GW. Genomic insights into the immune system of the sea urchin. Science 2006, 314, 952–956. [Google Scholar]
- Romero, A; Romao, MJ; Varela, PF; Kolln, I; Dias, J; Carvalho, AL; Sanz, L; Topfer-Petersen, E; Calvette, JJ. The crystal structures of two spermadhesins reveal the CUB domain fold. Nat. Struct. Biol 1997, 4, 783–788. [Google Scholar]
- Bork, P; Sander, C. A large domain common to sperm receptors (ZP2 and ZP3) and TGF-beta type-III receptor. Febs Lett 1992, 300, 237–240. [Google Scholar]
- Jovine, L; Qi, HY; Williams, Z; Litscher, ES; Wassarman, PM. A duplicated motif controls assembly of zona pellucida domain proteins. Proc. Natl. Acad. Sci. USA 2004, 101, 5922–5927. [Google Scholar]
- Jovine, L; Qi, HY; Williams, Z; Litscher, E; Wassarman, PM. The ZP domain is a conserved module for polymerization of extracellular proteins. Nat. Cell Biol 2002, 4, 457–461. [Google Scholar]
- Jovine, L; Darie, CC; Litscher, ES; Wassarman, PM. Zona pellucida domain proteins. Annu. Rev. Biochem 2005, 74, 83–114. [Google Scholar]
- Young, A; Rykke, M; Smistad, G; Rolla, G. On the role of human salivary micelle-like globules in bacterial agglutination. Eur. J. Oral Sci 1997, 105, 485–494. [Google Scholar]
- Oho, T; Yu, H; Yamashita, Y; Koga, T. Binding of salivary glycoprotein-secretory immunoglobulin a complex to the surface protein antigen of Streptococcus mutans. Infect. Immun 1998, 66, 115–121. [Google Scholar]
- Rose, MC; Voter, WA; Sage, H; Brown, CF; Kaufman, B. Effects of deglycosylation on the architecture of ovine submaxillary mucin glycoprotein. J. Biol. Chem 1984, 259, 3167–3172. [Google Scholar]
- Amado, M; Almeida, R; Schwientek, T; Clausen, H. Identification and characterization of large galactosyltransferase gene families: Galactosyltransferases for all functions. Biochim. Biophys. Acta-Gen. Subjects 1999, 1473, 35–53. [Google Scholar]
- Eriksson, C; Frangsmyr, L; Niemi, LD; Loimaranta, V; Holmskov, U; Bergman, T; Leffler, H; Jenkinson, HF; Stromberg, N. Variant size- and glycoforms of the scavenger receptor cysteine-rich protein gp-340 with differential bacterial aggregation. Glycoconj. J 2007, 24, 131–142. [Google Scholar]
- Ciopraga, J; Motas, C; Doyle, RJ. Inhibition of saliva-induced oral streptococcal aggregation by blood-group glycoproteins. Fems Immun. Med. Microbiol 1995, 10, 145–149. [Google Scholar]
- Ligtenberg, AJM; Veerman, ECI; de Graaff, J; Nieuw Amerongen, AV. Influence of the blood-group reactive substances in saliva on the aggregation of Streptococcus rattus. Antonie Van Leeuwenhoek 1990, 57, 97–107. [Google Scholar]
- Ligtenberg, AJM; Veerman, ECI; de Graaff, J; Nieuw Amerongen, AV. Saliva-induced aggregation of oral streptococci and the influence of blood-group reactive substances. Arch. Oral Biol 1990, 35, S141–S143. [Google Scholar]
- Arneberg, P; Kornstad, L; Nordbo, H; Gjermo, P. Less dental-caries among secretors than among non-secretors of blood-group substance. Scand. J. Dent. Res 1976, 84, 362–366. [Google Scholar]
- Hartshorn, KL; Ligtenberg, A; White, MR; van Eijk, M; Hartshorn, M; Pemberton, L; Holmskov, U; Crouch, E. Salivary agglutinin and lung scavenger receptor cysteine-rich glycoprotein 340 have broad anti-influenza activities and interactions with surfactant protein D that vary according to donor source and sialylation. Biochem. J 2006, 393, 545–553. [Google Scholar]
- Schulz, BL; Oxley, D; Packer, NH; Karlsson, NG. Identification of two highly sialylated human tear-fluid DMBT1 isoforms: The major high-molecular-mass glycoproteins in human tears. Biochem. J 2002, 366, 511–520. [Google Scholar]
- Yeh, JC; Hiraoka, N; Petryniak, B; Nakayama, J; Ellies, LG; Rabuka, D; Hindsgaul, O; Marth, JD; Lowe, JB; Fukuda, M. Novel sulfated lymphocyte homing receptors and their control by a core1 extension beta 1, 3-N-acetylglucosaminyltransferase. Cell 2001, 105, 957–969. [Google Scholar]
- Prakobphol, A; Boren, T; Ma, W; Peng, ZX; Fisher, SJ. Highly glycosylated human salivary molecules present oligosaccharides that mediate adhesion of leukocytes and Helicobacter pylori. Biochemistry 2005, 44, 2216–2224. [Google Scholar]
- Issa, S; Moran, AP; Ustinov, SN; Lin, JH; Ligtenberg, AJ; Karlsson, NG. O-linked oligosaccharides from salivary agglutinin: Helicobacter pylori binding sialyl-Lewis x and Lewis b are terminating moieties on hyperfucosylated oligo-N-acetyllactosamine. Glycobiology 2010, 20, 1046–1057. [Google Scholar]
- Bikker, FJ; Ligtenberg, AJ; Nazmi, K; Veerman, EC; van’t Hof, W; Bolscher, JG; Poustka, A; Nieuw Amerongen, AV; Mollenhauer, J. Identification of the bacteria-binding peptide domain on salivary agglutinin (gp-340/DMBT1), a member of the scavenger receptor cysteine-rich superfamily. J. Biol. Chem 2002, 277, 32109–32115. [Google Scholar]
- Bikker, FJ; Ligtenberg, AJM; End, C; Renner, M; Blaich, S; Lyer, S; Wittig, R; van’t Hof, W; Veerman, ECI; Nazmi, K; de Blieck-Hogervorst, JMA; Kioschis, P; Nieuw Amerongen, AV; Poustka, A; Mollenhauer, J. Bacteria binding by DMBT1/SAG/gp-340 is confined to the VEVLXXXXW motif in its scavenger receptor cysteine-rich domains. J. Biol. Chem 2004, 279, 47699–47703. [Google Scholar]
- Leito, JTD; Ligtenberg, AJM; Nazmi, K; de Blieck-Hogervorst, JMA; Veerman, ECI; Nieuw Amerongen, AV. A common binding motif for various bacteria of the bacteria-binding peptide SRCRP2 of DMBT1/gp-340/salivary agglutinin. Biol. Chem 2008, 389, 1193–1200. [Google Scholar]
- Elomaa, O; Kangas, M; Sahlberg, C; Tuukkanen, J; Sormunen, R; Liakka, A; Thesleff, I; Kraal, G; Tryggvason, K. Cloning of a novel bacteria-binding receptor structurally related to scavenger receptors and expressed in a subset of macrophages. Cell 1995, 80, 603–609. [Google Scholar]
- Elomaa, O; Sankala, M; Pikkarainen, T; Bergmann, U; Tuuttila, A; Raatikainen-Ahokas, A; Sariola, H; Tryggvason, K. Structure of the human macrophage MARCO receptor and characterization of its bacteria-binding region. J. Biol. Chem 1998, 273, 4530–4538. [Google Scholar]
- Kraal, G; van der Laan, LJ; Elomaa, O; Tryggvason, K. The macrophage receptor MARCO. Microb. Infect 2000, 2, 313–316. [Google Scholar]
- Brannstrom, A; Sankala, M; Tryggvason, K; Pikkarainen, T. Arginine residues in domain V have a central role for bacteria-binding activity of macrophage scavenger receptor MARCO. Biochem. Biophys. Res. Commun 2002, 290, 1462–1469. [Google Scholar]
- Sankala, M; Brannstrom, A; Schulthess, T; Bergmann, U; Morgunova, E; Engel, J; Tryggvason, K; Pikkarainen, T. Characterization of recombinant soluble macrophage scavenger receptor MARCO. J. Biol. Chem 2002, 277, 33378–33385. [Google Scholar]
- Sarrias, MR; Rosello, S; Sanchez-Barbero, F; Sierra, JM; Vila, J; Yelamos, J; Vives, J; Casals, C; Lozano, F. A role for human SP alpha as a pattern recognition receptor. J. Biol. Chem 2005, 280, 35391–35398. [Google Scholar]
- Sarrias, MR; Farnos, M; Mota, R; Sanchez-Barbero, F; Ibanez, A; Gimferrer, I; Vera, J; Fenutria, R; Casals, C; Yelamos, J; Lozano, F. CD6 binds to pathogen-associated molecular patterns and protects from LPS-induced septic shock. Proc. Natl. Acad. Sci. USA 2007, 104, 11724–11729. [Google Scholar]
- Fabriek, BO; van, BR; Deng, DM; Ligtenberg, AJ; Nazmi, K; Schornagel, K; Vloet, RP; Dijkstra, CD; van den Berg, TK. The macrophage scavenger receptor CD163 functions as an innate immune sensor for bacteria. Blood 2009, 113, 887–892. [Google Scholar]
- Schengrund, CL. “Multivalent” saccharides: Development of new approaches for inhibiting the effects of glycosphingolipid-binding pathogens. Biochem. Pharmacol 2003, 65, 699–707. [Google Scholar]
- Boren, T; Falk, P. Helicobacter pylori binds to blood group antigens. Scientific Am 1994, 28–37. [Google Scholar]
- Demuth, DR; Lammey, MS; Huck, M; Lally, ET; Malamud, D. Comparison of Streptococcus mutans and Streptococcus sanguis receptors for human salivary agglutinin. Microb. Pathogenesis 1990, 9, 199–211. [Google Scholar]
- Stenudd, C; Nordlund, A; Ryberg, M; Johansson, I; Kallestal, C; Stromberg, N. The association of bacterial adhesion with dental caries. J. Dent. Res 2001, 80, 2005–2010. [Google Scholar]
- Carlen, A; Bratt, P; Stenudd, C; Olsson, J; Stromberg, N. Agglutinin and acidic proline-rich protein receptor patterns may modulate bacterial adherence and colonization on tooth surfaces. J. Dent. Res 1998, 77, 81–90. [Google Scholar]
- Rosan, B; Appelbaum, B; Golub, E; Malamud, D; Mandel, ID. Enhanced saliva-mediated bacterial aggregation and decreased bacterial adhesion in caries-resistant versus caries-susceptible individuals. Infect. Immun 1982, 38, 1056–1059. [Google Scholar]
- Jenkinson, HF; Demuth, DR. Structure, function and immunogenicity of streptococcal antigen I/II polypeptides [review]. Mol. Microbiol 1997, 23, 183–190. [Google Scholar]
- brady, LJ; Maddocks, SE; Larson, MR; Forsgren, N; Persson, K; Deivanayagam, CC; Jenkinson, HF. The changing faces of Streptococcus antigen I/II polypeptide family adhesins. Mol. Microbiol 2010, 77, 276–286. [Google Scholar]
- Kelly, CG; Todryk, S; Kendal, HL; Munro, GH; Lehner, T. T-cell, adhesion, and B-cell epitopes of the cell surface Streptococcus mutans protein antigen I/II. Infect. Immun 1995, 63, 3649–3658. [Google Scholar]
- Kelly, CG; Younson, JS; Hikmat, BY; Todryk, SM; Czisch, M; Haris, PI; Flindall, IR; Newby, C; Mallet, AI; Ma, JK; Lehner, T. A synthetic peptide adhesion epitope as a novel antimicrobial agent. Nat. Biotechnol 1999, 17, 42–47. [Google Scholar]
- Jakubovics, NS; Stromberg, N; van Dolleweerd, CJ; Kelly, CG; Jenkinson, HF. Differential binding specificities of oral streptococcal antigen I/II family adhesins for human or bacterial ligands. Mol. Microbiol 2005, 55, 1591–1605. [Google Scholar]
- Hamada, T; Kawashima, M; Watanabe, H; Tagami, J; Senpuku, H. Molecular interactions of surface protein peptides of Streptococcus gordonii with human salivary components. Infect. Immun 2004, 72, 4819–4826. [Google Scholar]
- Koba, H; Okuda, K; Watanabe, H; Tagami, J; Senpuku, H. Role of lysine in interaction between surface protein peptides of Streptococcus gordonii and agglutinin peptide. Oral Microbiol. Immunol 2009, 24, 162–169. [Google Scholar]
- Oho, T; Bikker, FJ; Nieuw Amerongen, AV; Groenink, J. A peptide domain of bovine milk lactoferrin inhibits the interaction between streptococcal surface protein antigen and a salivary agglutinin peptide domain. Infect. Immun 2004, 72, 6181–6184. [Google Scholar]
- Niemi, LD; Johansson, I. Salivary statherin peptide-binding epitopes of commensal and potentially infectious Actinomyces spp. delineated by a hybrid peptide construct. Infect. Immun 2004, 72, 782–787. [Google Scholar]
- Niemi, LD; Hernell, O; Johansson, I. Human milk compounds inhibiting adhesion of mutans streptococci to host ligand-coated hydroxyapatite in vitro. Caries Res 2009, 43, 171–178. [Google Scholar]
- Wu, ZW; Golub, E; Abrams, WR; Malamud, D. gp340 (SAG) binds to the V3 sequence of gp120 important for chemokine receptor interaction. Aids Res. Hum. Retroviruses 2004, 20, 600–607. [Google Scholar]
- Larson, MR; Rajashankar, KR; Patel, MH; Robinette, RA; Crowley, PJ; Michalek, S; Brady, LJ; Deivanayagam, C. Elongated fibrillar structure of a streptococcal adhesin assembled by the high-affinity association of alpha- and PPII-helices. Proc. Natl. Acad. Sci. USA 2010, 107, 5983–5988. [Google Scholar]
- Troffer-Charlier, N; Ogier, J; Moras, D; Cavarelli, J. Crystal structure of the V-region of Streptococcus mutans antigen I/II at 2.4 angstrom resolution suggests a sugar preformed binding site. J. Mol. Biol 2002, 318, 179–188. [Google Scholar]
- Loimaranta, V; Jakubovics, NS; Hytonen, J; Finne, J; Jenkinson, HF; Stromberg, N. Fluid- or surface-phase human salivary scavenger protein gp340 exposes different bacterial recognition properties. Infect. Immun 2005, 73, 2245–2252. [Google Scholar]
- Brady, LJ; Piacentini, DA; Crowley, PJ; Oyston, PCF; Bleiweis, AS. Differentiation of salivary agglutinin-mediated adherence and aggregation of mutans streptococci by use of monoclonal-antibodies against the major surface adhesin-P1. Infect. Immun 1992, 60, 1008–1017. [Google Scholar]
- Zhang, Y; Lei, Y; Nobbs, A; Khammanivong, A; Herzberg, MC. Inactivation of Streptococcus gordonii SspAB alters expression of multiple adhesin genes. Infect. Immun 2005, 73, 3351–3357. [Google Scholar]
- Gibbons, RJ; Hay, DI; Childs, WC; Davis, G. Role of cryptic receptors (cryptitopes) in bacterial adhesion to oral-surfaces. Arch. Oral Biol 1990, 35, S107–S114. [Google Scholar]
- Gibbons, RJ; Hay, DI. Human salivary acidic proline-rich proteins and statherin promote the attachment of Actinomyces viscosus LY7 to apatitic surfaces. Infect. Immun 1988, 56, 439–445. [Google Scholar]
- Goobes, G; Goobes, R; Schueler-Furman, O; Baker, D; Stayton, PS; Drobny, GP. Folding of the C-terminal bacterial binding domain in statherin upon adsorption onto hydroxyapatite crystals. Proc. Natl. Acad. Sci. USA 2006, 103, 16083–16088. [Google Scholar]
- Madsen, J; Tornoe, I; Nielsen, O; Lausen, M; Krebs, I; Mollenhauer, J; Kollender, G; Poustka, A; Skjodt, K; Holmskov, U. CRP-ductin, the mouse homologue of gp-340/deleted in malignant brain tumors 1 (DMBT1), binds gram-positive and gram-negative bacteria and interacts with lung surfactant protein D. Eur. J. Immunol 2003, 33, 2327–2336. [Google Scholar]
- End, C; Bikker, F; Renner, M; Bergmann, G; Lyer, S; Blaich, S; Hudler, M; Helmke, B; Gassler, N; Autschbach, F; Ligtenberg, AJ; Benner, A; Holmskov, U; Schirmacher, P; Nieuw Amerongen, AV; Rosenstiel, P; Sina, C; Franke, A; Hafner, M; Kioschis, P; Schreiber, S; Poustka, A; Mollenhauer, J. DMBT1 functions as pattern-recognition molecule for poly-sulfated and poly-phosphorylated ligands. Eur. J. Immunol 2009, 39, 833–842. [Google Scholar]
- Wu, Z; Van Ryk, D; Davis, C; Abrams, WR; Chaiken, I; Magnani, J; Malamud, D. Salivary agglutinin inhibits HIV type 1 infectivity through interaction with viral glycoprotein 120. Aids Res. Hum. Retroviruses 2003, 19, 201–209. [Google Scholar]
- Hartshorn, KL; White, MR; Mogues, T; Ligtenberg, T; Crouch, E; Holmskov, U. Lung and salivary scavenger receptor glycoprotein-340 contribute to the host defense against influenza A viruses. Am. J. Physiol.-Lung Cell. Mol. Physiol 2003, 285, L1066–L1076. [Google Scholar]
- Sattentau, QJ; Moore, JP. Conformational changes induced in the human-immunodeficiency-virus envelope glycoprotein by soluble CD4 binding. J. Exp. Med 1991, 174, 407–415. [Google Scholar]
- Trkola, A; Dragic, T; Arthos, J; Binley, JM; Olson, WC; Allaway, GP; ChengMayer, C; Robinson, J; Maddon, PJ; Moore, JP. CD4-dependent, antibody-sensitive interactions between HIV-1 and its co-receptor CCR-5. Nature 1996, 384, 184–187. [Google Scholar]
- Wu, ZW; Lee, S; Abrams, W; Weissman, D; Malamud, D. The N-terminal SRCR-SID domain of gp-340 interacts with HIV type 1 gp120 sequences and inhibits viral infection. Aids Res. Hum. Retroviruses 2006, 22, 508–515. [Google Scholar]
- Thornton, DJ; Davies, JR; Kirkham, S; Gautrey, A; Khan, N; Richardson, PS; Sheehan, JK. Identification of a nonmucin glycoprotein (gp-340) from a purified respiratory mucin preparation: Evidence for an association involving the MUC5B mucin. Glycobiology 2001, 11, 969–977. [Google Scholar]
- Kojouharova, MS; Tsacheva, IG; Tchorbadjieva, MI; Reid, KBM; Kishore, U. Localization of ligand-binding sites on human C1q globular head region using recombinant globular head fragments and single-chain antibodies. Biochim. Biophys. Acta-Proteins Proteomics 2003, 1652, 64–74. [Google Scholar]
- Armstrong, EA; Ziola, B; Habbick, BF; Komiyama, K. Role of cations and IgA in saliva-mediated aggregation of Pseudomonas aeruginosa in cystic-fibrosis patients. J. Oral Pathol. Med 1993, 22, 207–213. [Google Scholar]
- Singh, PK; Parsek, MR; Greenberg, EP; Welsh, MJ. A component of innate immunity prevents bacterial biofilm development. Nature 2002, 417, 552–555. [Google Scholar]
- Groenink, J; Walgreen-Weterings, E; van ’t Hof, W; Veerman, ECI; Nieuw Amerongen, AV. Cationic amphipatic peptides, derived from bovine and human lactoferrins, with antimicrobial activity against oral pathogens. FEMS Microbiol. Lett 1999, 179, 217–222. [Google Scholar]
- Kishore, U; Greenhough, TJ; Waters, P; Shrive, AK; Ghai, R; Kamran, MF; Bernal, AL; Reid, KBM; Madan, T; Chakraborty, T. Surfactant proteins SP-A and SP-D: Structure, function and receptors. Mol. Immunol 2006, 43, 1293–1315. [Google Scholar]
- Hoffmann, JA; Kafatos, FC; Janeway, CA; Ezekowitz, RA. Phylogenetic perspectives in innate immunity. Science 1999, 1313–1318. [Google Scholar]
- Janeway, C, Jr; Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol 2003, 20, 197–216. [Google Scholar]
- Bainbridge, BW; Darveau, RP. Porphyromonas gingivalis lipopolysaccharide: An unusual pattern recognition receptor ligand for the innate host defense system. Acta Odontol. Scand 2001, 59, 131–138. [Google Scholar]
- Ulevitch, RJ; Mathison, JC; Correia, JD. Innate immune responses during infection. Vaccine 2004, 22, S25–S30. [Google Scholar]
- Medzhitov, R; Janeway, C, Jr. Innate immunity. N. Engl. J. Med 2000, 343, 338–343. [Google Scholar]
- Aderem, A; Ulevitch, RJ. Toll-like receptors in the induction of the innate immune response. Nature 2000, 406, 782–787. [Google Scholar]
- Greaves, DR; Gordon, S. Recent insights into the biology of macrophage scavenger receptors. J. Lipid Res 2005, 46, 11–20. [Google Scholar]


| Organism | Name | Function |
|---|---|---|
| Human | DMBT1 | Tumor suppression |
| gp-340 | Epithelial cell differentiation | |
| SAG | Innate immunity | |
| Mouse | CRP-ductin | Mucosal defense |
| Vomeroglandin | Epithelial differentiation | |
| Muclin | Pheromone perception | |
| apactin | Sorting receptor | |
| Rabbit | Hensin | Terminal differentiation of kidney epithelial intercalated cells and embryonic stem cells |
| Rat | Ebnerin | Liver regeneration |
| Pancrin | Taste perception | |
| Pig | Porcine dmbt1 | |
| Cow | Bovine gallbladder | Cholesterol-binding |
| mucin | Gallstone formation | |
| Rhesus monkey | H3 | Hormone-responsive |
| Endometrial regeneration in ovulation cycle | ||
| Oligosaccharide | Structure | Method | Source | Reference: |
|---|---|---|---|---|
| core 1 | Galβ1-3GalNAcα1-Ser/Thr | |||
| sialylated-core 1 | NeuAcα2-3 Galβ1-3GalNAcα1-Ser/Thr | LC-MS2 | Tears/Saliva | [61,64] |
| disialylated-core 1 | NeuAcα2-3Galβ1-3(NeuAcα2-6)GalNAcα1-Ser/Thr | |||
| core 2 | Galβ1-3(GlcNAcβ1-6)GalNAcα1-Ser/Thr | LC-MS2 | Tears/Saliva | [61,64] |
| sialylated-core 2 | NeuAcα2-3 Galβ1-3(GlcNAcβ1-6)GalNAcα1-Ser/Thr | |||
| (Le-a) | Galβ1-4(Fucα1-3)GlcNAc | immunoblotting | Saliva | [13] |
| (Le-x) | Galβ1-3(Fucα1-4)GlcNAc | immunoblotting | Saliva | [13,22] |
| (Le-b) | Fucα1-2 Galβ1-4(Fucα1-3)GlcNAc | immunoblotting/LC-MS2 | Saliva | [13,64] |
| (Le-y) | Fucα1-2Galβ1-3(Fucα1-4)GlcNAc | immunoblotting/LC-MS2 | Saliva | [13,64] |
| H antigen | Fucα1-2 Galβ1-4GlcNAc | immunoblotting/LC-MS2 | Saliva | [13,64] |
| A antigen | GalNAcα1-3(Fucα1-2)Galβ1-4GlcNAc | immunoblotting | Saliva | [13] |
| B antigen | Galα1-3(Fucα1-2)Galβ1-4GlcNAc | immunoblotting | Saliva | [13] |
| (Sialyl-Lex) | NeuA-α2-3Galβ1-3(Fucα1-4)GlcNAc | immunoblotting | saliva, not lung | [22] |
| Sialyl-Lea | NeuAcα2-3 Galβ1-4(Fucα1-3)GlcNAc | Immunoblotting/LC-MS2 | Tears | [61]] |
| NeuAcα2-3 | lectin blotting | Saliva, not lung | [60] | |
| NeuAcα2-6 | lectin blotting | lung, saliva | [60] | |
| SO3-6GlcNAc | Immunoblotting | Saliva | [63] | |
| Protein | Sequence | SRCRP2 binding | Ref. |
|---|---|---|---|
| S.mutans antigen I/II | PQLKTADLPAGRDETTSFVLV | [85] | |
| S.gordonii antigen I/II | ELKTEALTAGRPKTTSFVLV | [86] | |
| S.mutans antigen I/II | QLKTADLPAGRDETTSFVLV | ||
| S.gordonii SspB 390-402 | DYQAKLAAYQTEL DYQAKLAAYQKEL | + | [87] |
| S.gordonii SspB 390-T400K-402 | + | ||
| SsP(A4K-A11K) | DYQKKLAAYQKEL | + | [88] |
| Consensus SspA and B | DYQAKLAAYQAEL | + | |
| Bovine lactoferrin 480-492 | SCAFDEFFSQSCA | + | [89,90] |
| Human lactoferrin | DCKFDEYFSQSCA | [91] | |
| Human casein | LLNQELLNPTHQIYPVTQPLAPVHNPISV | [91] | |
| HIV V3 region | CTRPNYNKRKR | [92] | |
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ligtenberg, A.J.M.; Karlsson, N.G.; Veerman, E.C.I. Deleted in Malignant Brain Tumors-1 Protein (DMBT1): A Pattern Recognition Receptor with Multiple Binding Sites. Int. J. Mol. Sci. 2010, 11, 5212-5233. https://doi.org/10.3390/ijms1112521
Ligtenberg AJM, Karlsson NG, Veerman ECI. Deleted in Malignant Brain Tumors-1 Protein (DMBT1): A Pattern Recognition Receptor with Multiple Binding Sites. International Journal of Molecular Sciences. 2010; 11(12):5212-5233. https://doi.org/10.3390/ijms1112521
Chicago/Turabian StyleLigtenberg, Antoon J. M., Niclas G. Karlsson, and Enno C. I. Veerman. 2010. "Deleted in Malignant Brain Tumors-1 Protein (DMBT1): A Pattern Recognition Receptor with Multiple Binding Sites" International Journal of Molecular Sciences 11, no. 12: 5212-5233. https://doi.org/10.3390/ijms1112521




