Kinetic Support for the Generation of a Phenylsulfenium Ion Intermediate
Abstract
:Introduction
Results and Discussion
Kinetic study on the reaction of N-methylbenzenesulfenamide (1) with thioanisole (4) in the presence of trifluoroacetic acid (TFA).
Reaction-order with respect to sulfenamide (1).
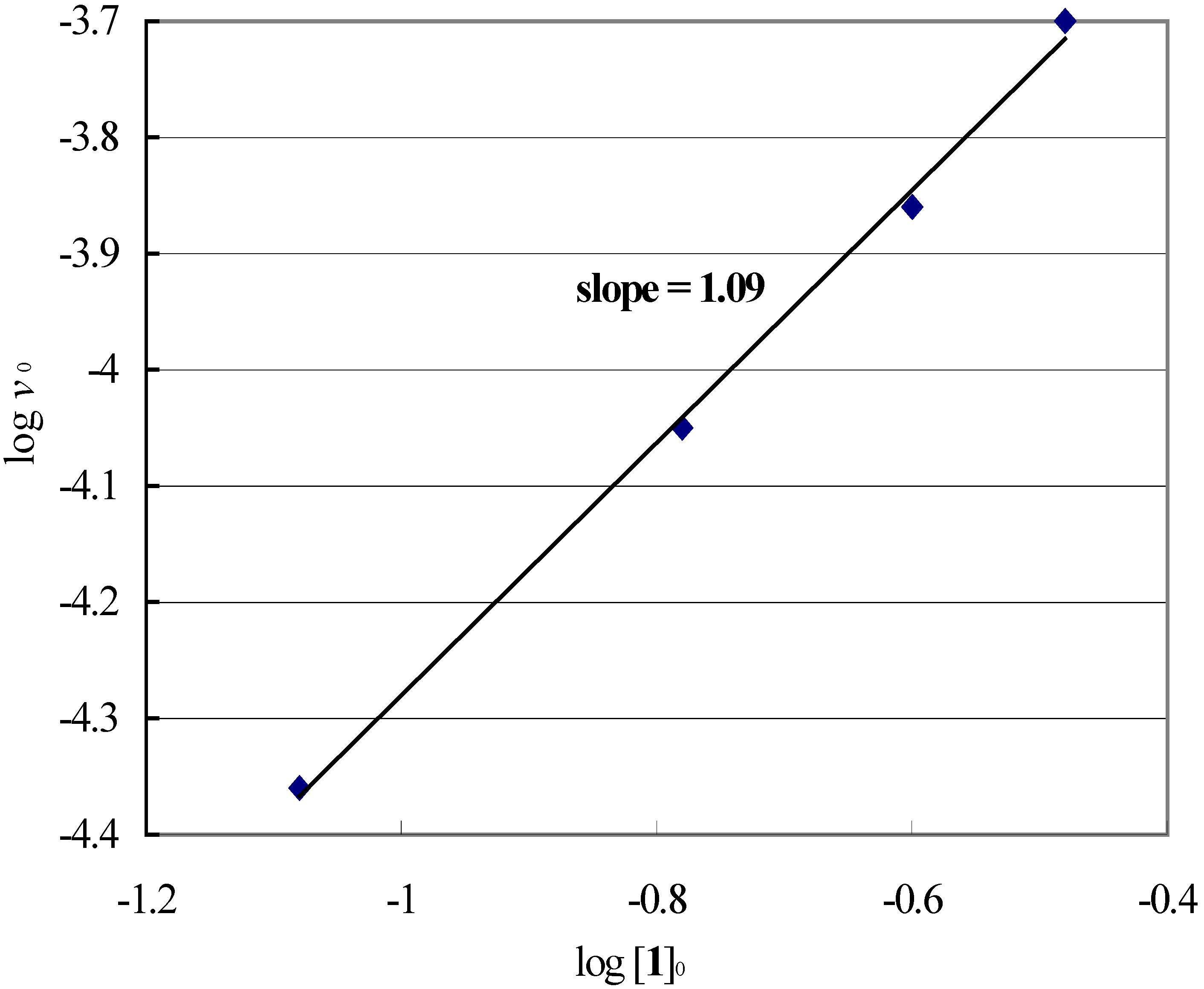
Reaction-order with respect to thioanisole (4).
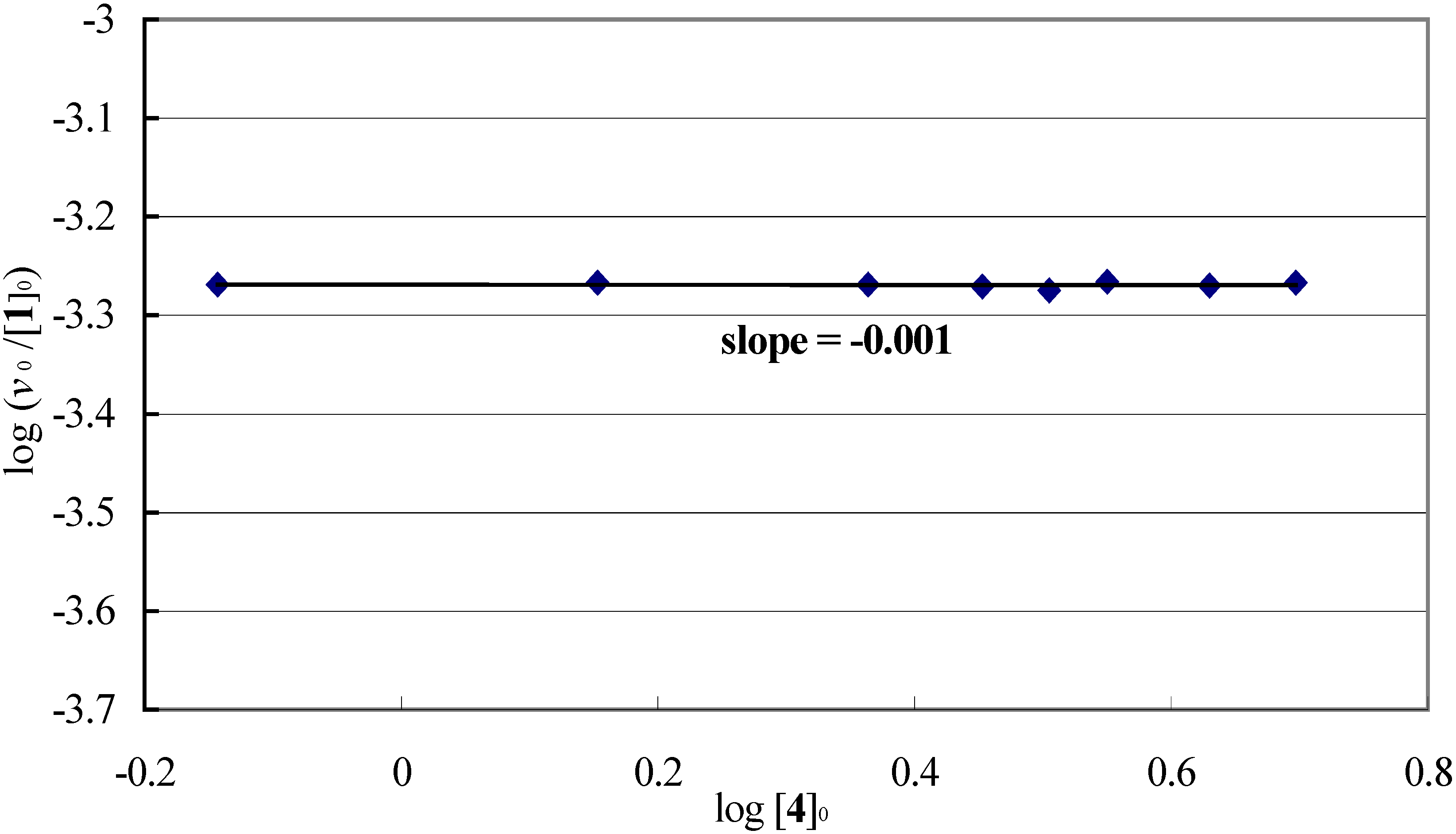
Evaluation of pseudo-first order rate constant kobs.

Evidence for the generation of phenylsulfenium ion
Experimental
General
Kinetic study on generation of phenylsulfenium ion 3
Reaction-order with respect to N-methylbenzenesulfenamide 1.
| [1]0 /10-3 M | Concentration [5]/10-3 M | υ0 /10-6 Msec-1 | α | β | R2 | ||||
| t=150 sec | t=300 sec | t=450 sec | t=600 sec | t=750 sec | |||||
| 83 | 6.2 | 11 | 15 | 16 | 18 | 44 | 3 | 5 | 0.997 |
| 170 | 13 | 24 | 30 | 37 | 42 | 90 | 5 | 5 | 0.998 |
| 250 | 24 | 31 | 45 | 49 | 56 | 137 | 9 | 5 | 0.981 |
| 330 | 31 | 46 | 67 | 77 | 84 | 199 | 1 | 4 | 0.993 |
Reaction-order with respect to thioanisole (4).
| [4]0/M | Concentration [5]/10-3 M | υ0 /10-6 Msec-1 | α | β | R2 | ||||
| t=150 sec | t=300 sec | t=450 sec | t=600 sec | t=750 sec | |||||
| 0.72 | 13 | 22 | 29 | 34 | 38 | 90 | 5 | 5 | 0.998 |
| 1.4 | 13 | 22 | 29 | 34 | 37 | 90 | 5 | 5 | 0.999 |
| 2.1 | 14 | 24 | 30 | 37 | 43 | 90 | 4 | 5 | 0.997 |
| 2.8 | 13 | 23 | 30 | 36 | 41 | 89 | 5 | 5 | 0.999 |
| 3.2 | 13 | 23 | 29 | 38 | 41 | 88 | 5 | 5 | 0.998 |
| 3.6 | 13 | 23 | 30 | 39 | 42 | 90 | 5 | 5 | 0.999 |
| 4.3 | 13 | 24 | 30 | 37 | 42 | 90 | 5 | 5 | 0.998 |
| 5.0 | 13 | 23 | 29 | 35 | 39 | 90 | 5 | 5 | 0.999 |
References and Notes
- Kharasch, N.K.; Bues, C.B.; King, W. J. Am. Chem. Soc. 1953, 75, 6030. Benesch, R.E.; Benesch, R. J. Am. Chem. Soc. 1958, 80, 1666. Parker, A.J.; Kharasch, N. Chem. Rev. 1959, 59, 583. Kharasch, N. Organic Sulfur Compounds; Kharasch, N., Ed.; Pergamon: New York, 1961; Vol. 1, Ch. 31; pp. 375–397. [Google Scholar] Kice, J.L. Acc. Chem. Res. 1968, 1, 58. Owsley, D.C.; Helmkamp, G.H. J. Am. Chem. Soc. 1968, 90, 1635. Russ, C.R.; Douglass, I.B. Sulfur in Organic and Inorganic Chemistry; Senning, A., Ed.; Dekker: New York, 1971; Vol. 1, Ch. 8; pp. 239–259. [Google Scholar] Smith, W.A.; Krimer, M.Z.; Vorobieva, E.A. Tetrahedron Lett. 1975, 2451. Capozzi, G.; Lucchino, V.; Modena, G.; Rivetti, F. J. Chem. Soc., Perkin Trans. 2 1975, 361. Saya, H.; Michida, T. Chem. Pharm. Bull. 1983, 31, 4530. Saya, H.; Michida, T. Chem. Pharm. Bull. 1985, 33, 2541. Bewick, A.; Coe, D.E.; Mellor, J.M.; Owton, W. M. J. Chem. Soc., Perkin Trans., 1 1985, 1033. Edstrom, E.D.; Livinghouse, T. J. Am. Chem. Soc. 1986, 108, 1334. Edstrom, E.D.; Livinghouse, T. J. Org. Chem. 1987, 52, 951. Harring, S.R.; Livinghouse, T. Tetrahedron Lett. 1989, 1499. Harring, S.R.; Livinghouse, T. J. Org. Chem. 1997, 62, 6388.
- Kobayashi, K.; Sato, S.; Horn, E.; Furukawa, N. Tetrahedron Lett. 1998, 39, 2593.
- Modena, G.; Scorrano, G. Mechanisms of Reactions of Sulfur Compounds 1968, 3, 115. Ciuffarin, E.; Fava, A. Prog. Phys. Org. Chem. 1968, 6, 81. Mueller, W.H. Angew. Chem., Int. Ed. Engl. 1969, 8, 482. Kice, J.L. Prog. Inorg. Chem. 1972, 17, 147.
- Gybin, A.S.; Smith, V.A.; Bogdnav, V.S.; Krimer, M.Z.; Kalyan, J.B. Tetrahedron Lett. 1980, 383.
- Brownbridge, P. Tetrahedron Lett. 1984, 3759.
- Takeuchi, H.; Oya, H.; Yanase, T.; Itou, K.; Adachi, T.; Sugiura, H.; Hayashi, N. J. Chem. Soc., Perkin Trans. 2 1994, 827.
- Takeuchi, H.; Takano, K. J. Chem. Soc., Perkin Trans. 1 1986, 611. Takeuchi, H.; Taniguchi, T.; Ueda, T. J. Chem. Soc., Perkin Trans. 2 2000, 296.
- Laiddler, E.J. Chemical Kinetics; McGraw-Hill Book Inc.: New York, 1965; pp. 535–536. [Google Scholar]
- Takeuchi, H. J. Chem. Soc., Chem. Commun. 1987, 961. Takeuchi, H. J. Chem. Soc., Chem. Commun. 1988, 1287. Takeuchi, H.; Hayakawa, S.; Tanahashi, T.; Kobayashi, A.; Adachi, T.; Higuchi, D. J. Chem. Soc., Perkin Trans. 2 1991, 847. Takeuchi, H.; Higuchi, D.; Adachi, T. J. Chem. Soc., Perkin Trans. 1 1991, 1525.
- Craine, L.; Raban, M. Chem. Rev. 1989, 689.
- Unpublished data: under similar conditions at 10 °C, the reaction with PhSPh in place of 4 provided the same first and zero orders for 1 and PhSPh, respectively, with kobs=1.1×10-3 sec-1. The reaction with benzene instead of 4 under similar conditions gave diphenyl disulfide (6) selectively in 70-80% yields. This suggests that the phenylthiolation producing 5 competes with the formation of 6. We assume that the sulfenium ion preferentially reacts with the solvent rather than with benzene, thus leading to undetermined products.
- David, F.A.; Friedman, A.J.; Kluger, E.W.; Skibo, E.B.; Fretz, E.R.; Milicia, A.P.; LeMasters, W.C.; Bentley, M.D.; Lacadie, J.A.; Douglass, I.B. J. Org. Chem. 1977, 42, 967.
- Sample Availability: Available from the authors.
© 2003 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Takeuchi, H.; Tateiwa, J.-i.; Moriguchi, S. Kinetic Support for the Generation of a Phenylsulfenium Ion Intermediate. Molecules 2003, 8, 392-400. https://doi.org/10.3390/80400392
Takeuchi H, Tateiwa J-i, Moriguchi S. Kinetic Support for the Generation of a Phenylsulfenium Ion Intermediate. Molecules. 2003; 8(4):392-400. https://doi.org/10.3390/80400392
Chicago/Turabian StyleTakeuchi, Hiroshi, Jun-ichi Tateiwa, and Shintaro Moriguchi. 2003. "Kinetic Support for the Generation of a Phenylsulfenium Ion Intermediate" Molecules 8, no. 4: 392-400. https://doi.org/10.3390/80400392




