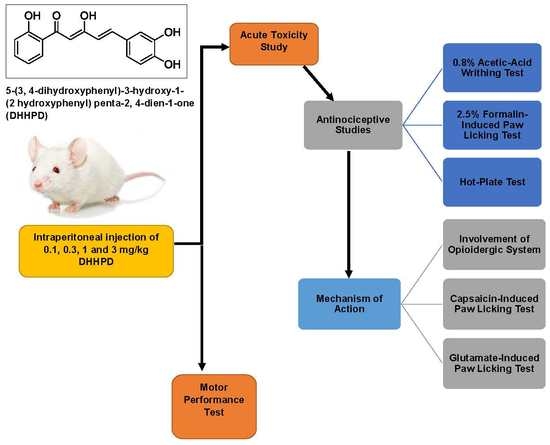
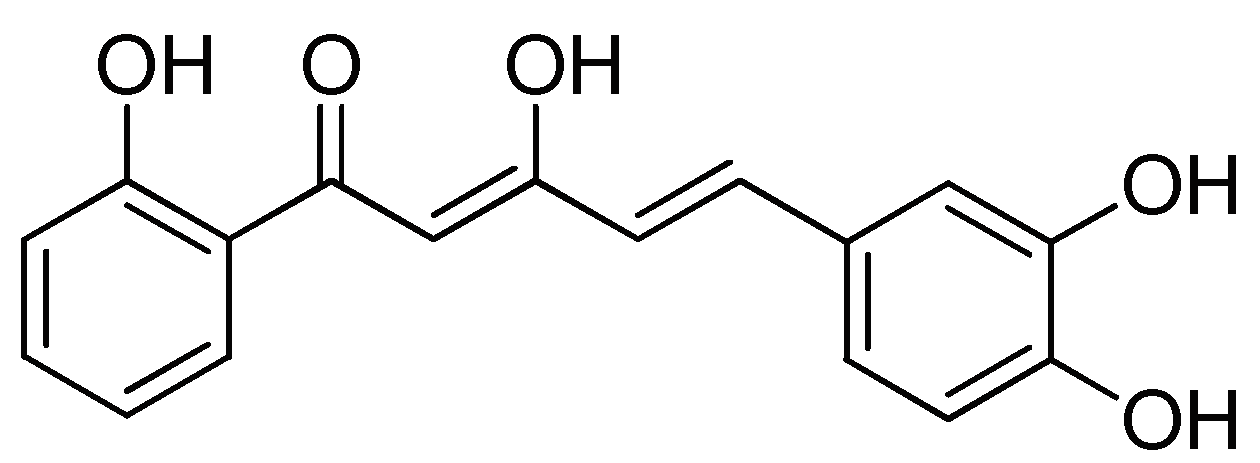
Analgesic Effect of 5-(3,4-Dihydroxyphenyl)-3-hydroxy-1-(2-hydroxyphenyl)penta-2,4-dien-1-one in Experimental Animal Models of Nociception
Abstract
:1. Introduction
2. Results
2.1. Acute Toxicity Study
2.2. Antinociceptive Studies
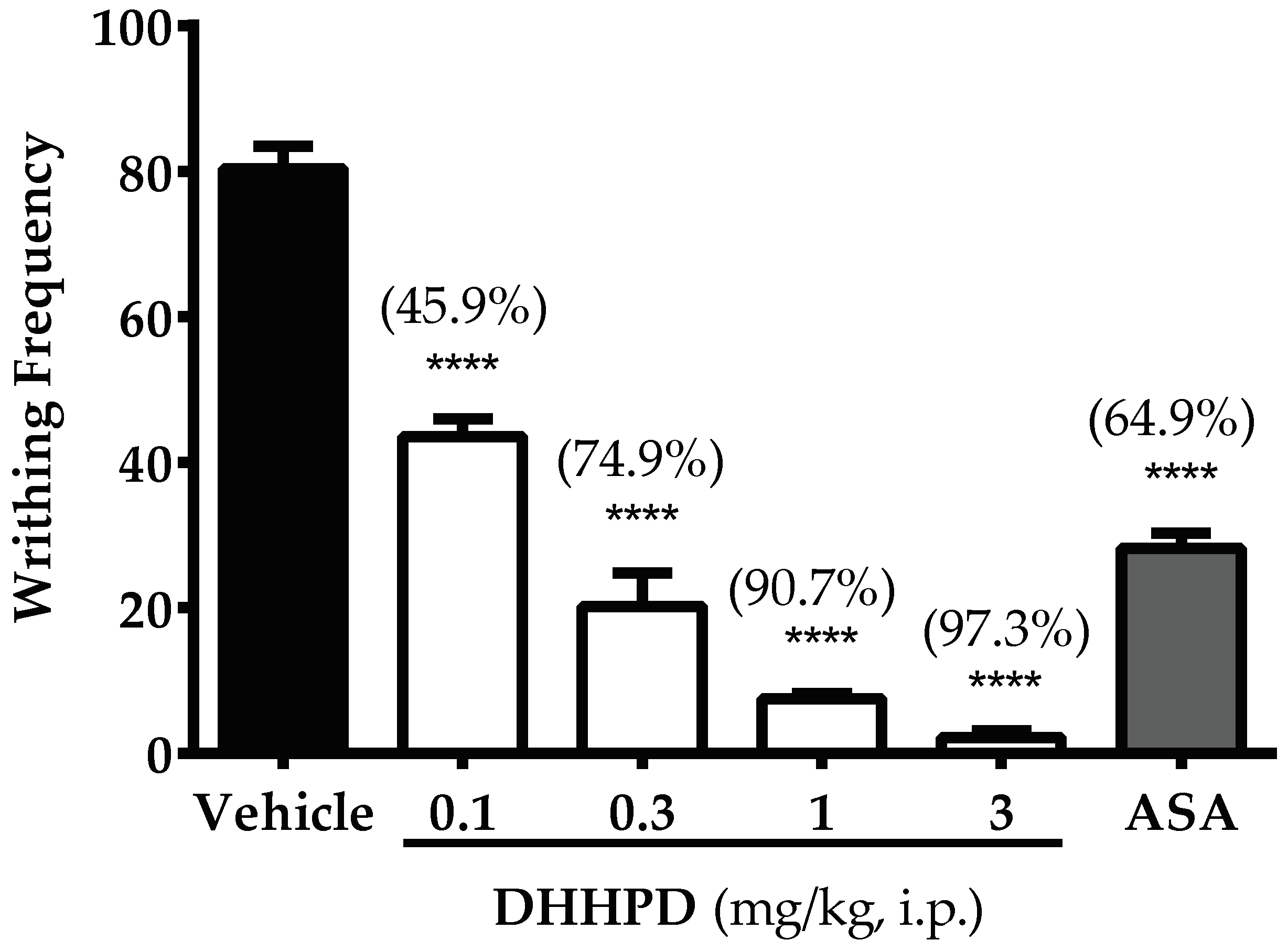
2.2.1. Acetic Acid-Induced Writhing Test
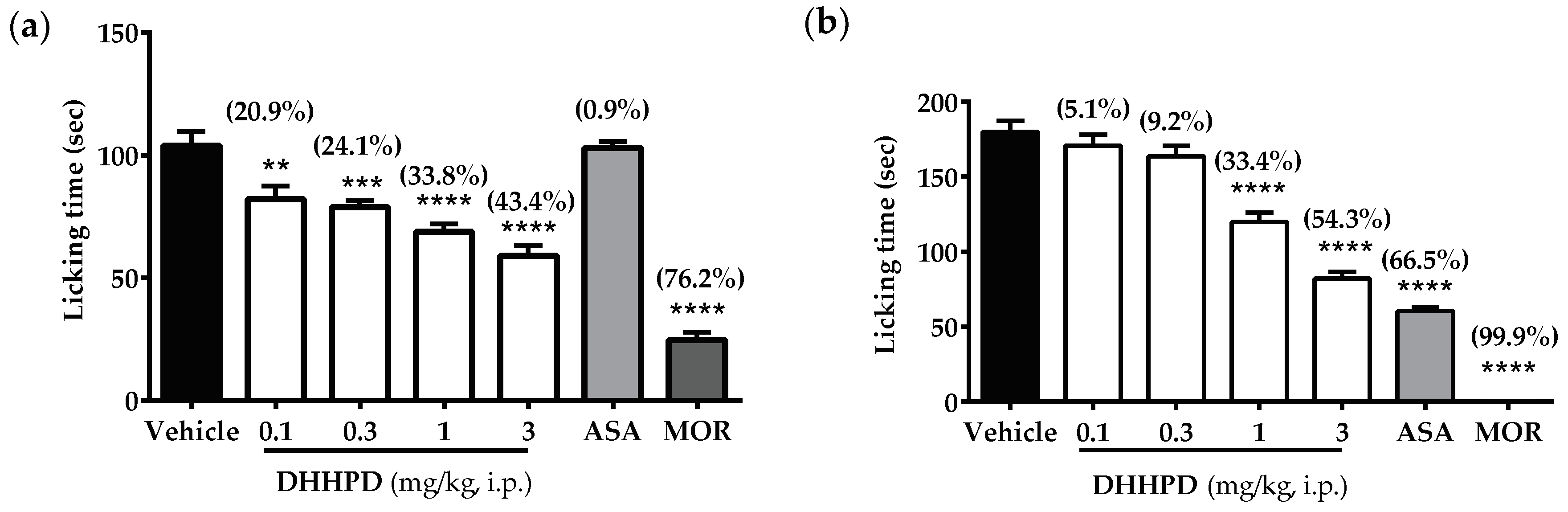
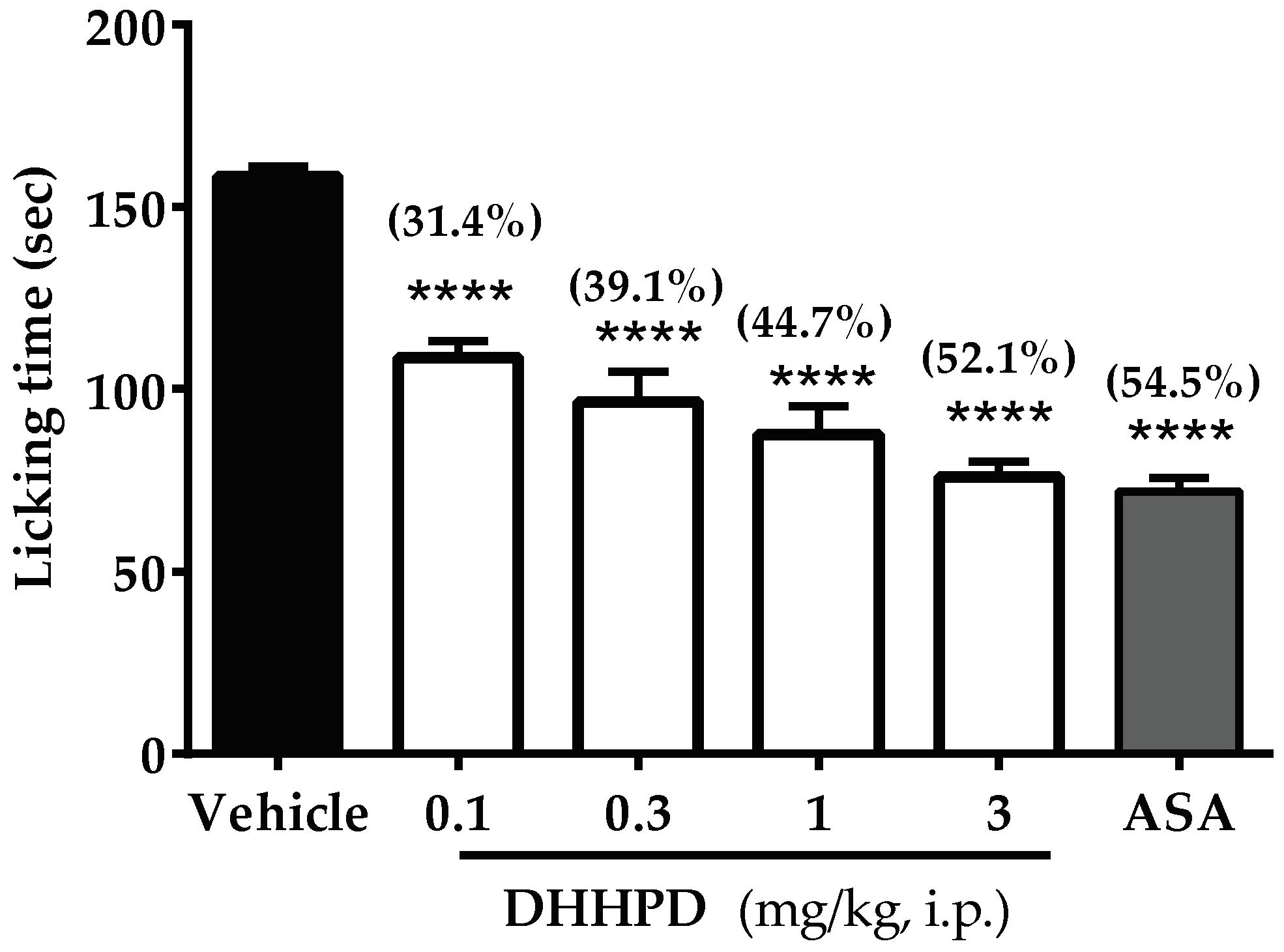
2.2.2. Formalin-Induced Paw Licking Test
2.2.3. Hot-Plate Test
2.2.4. Involvement of the Opioidergic System
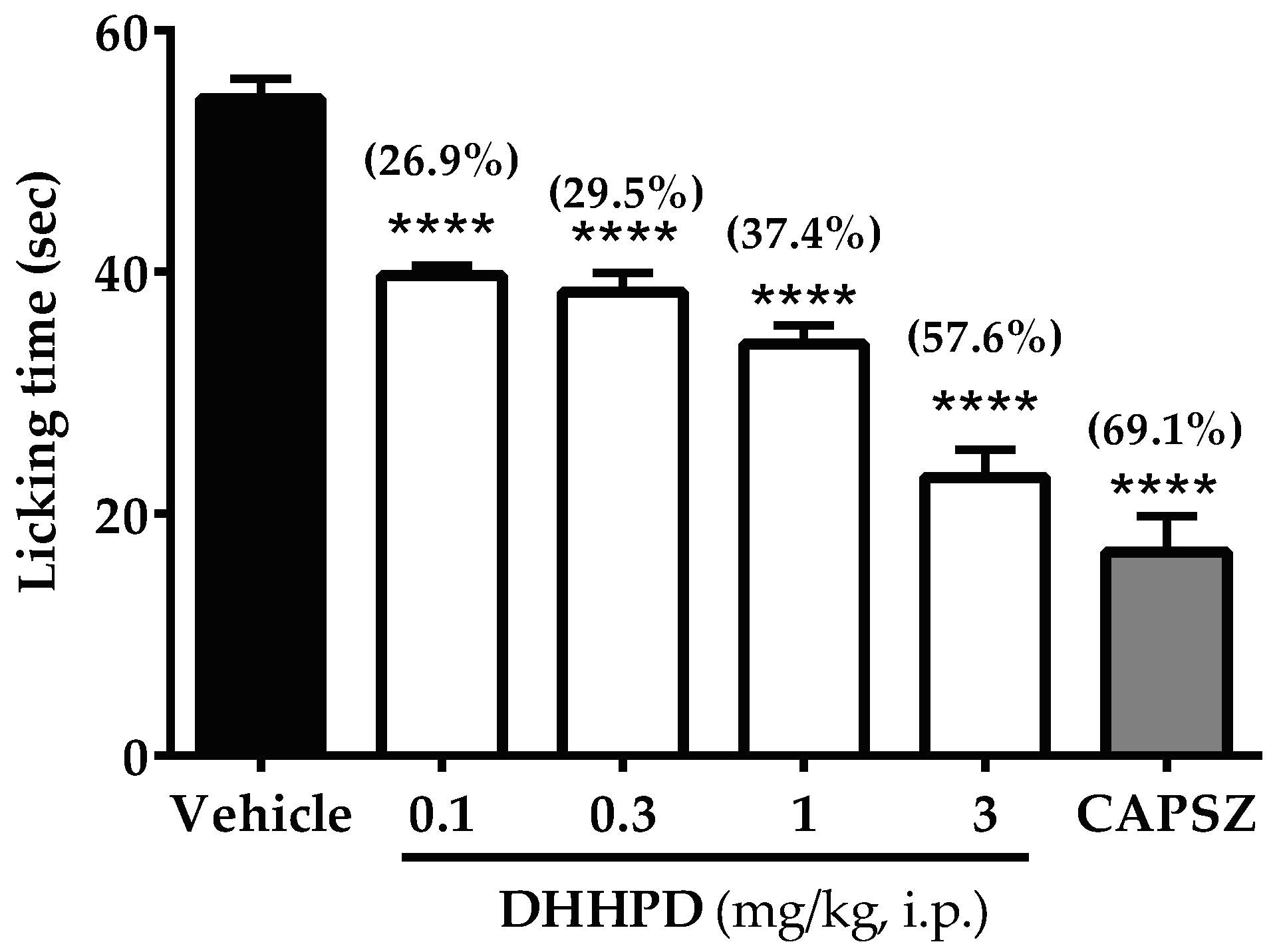
2.2.5. Capsaicin-Induced Paw Licking Test
2.2.6. Glutamate-Induced Paw Licking Test
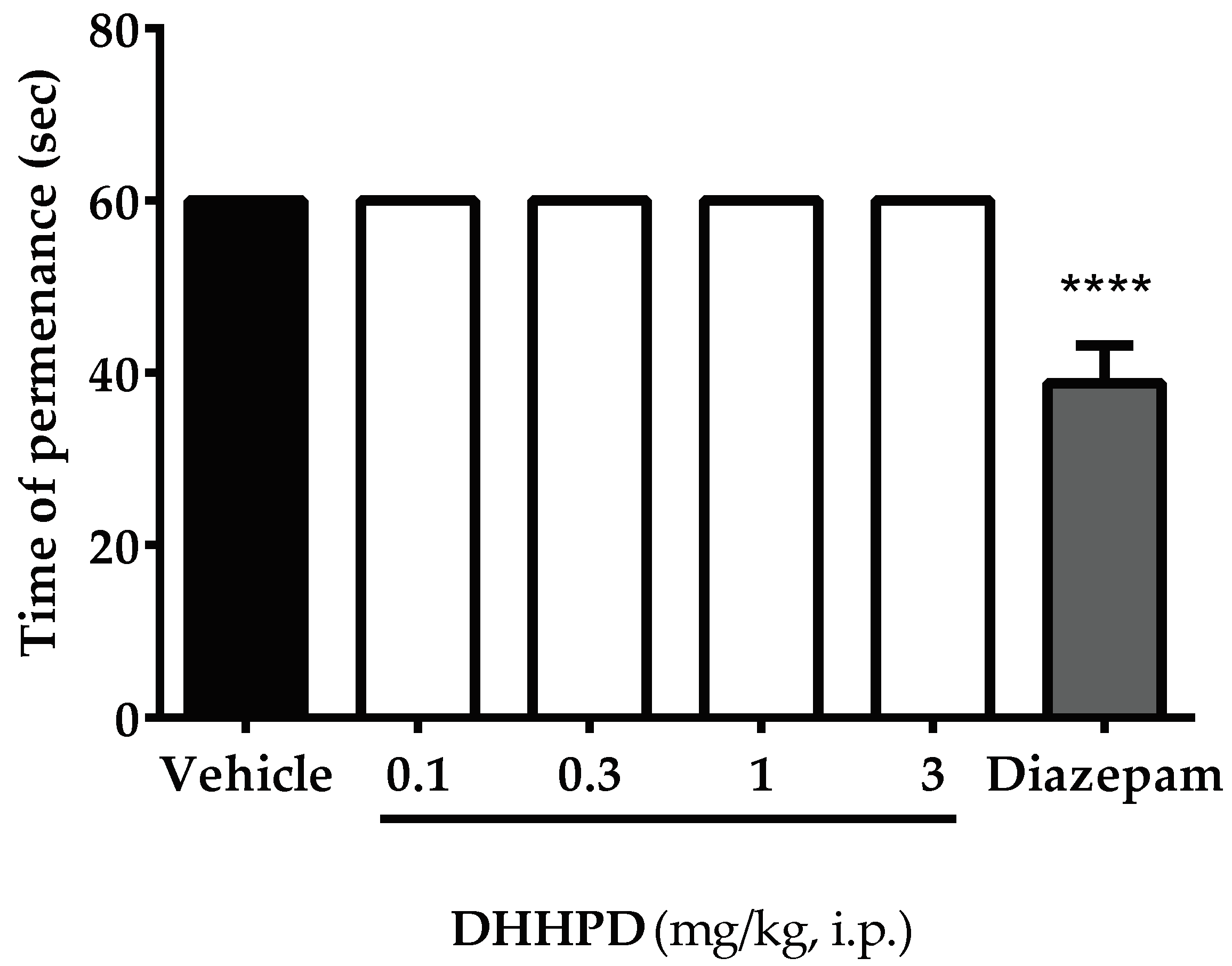
2.3. Motor Performance Test
Rotarod Test
3. Discussion
4. Materials and Methods
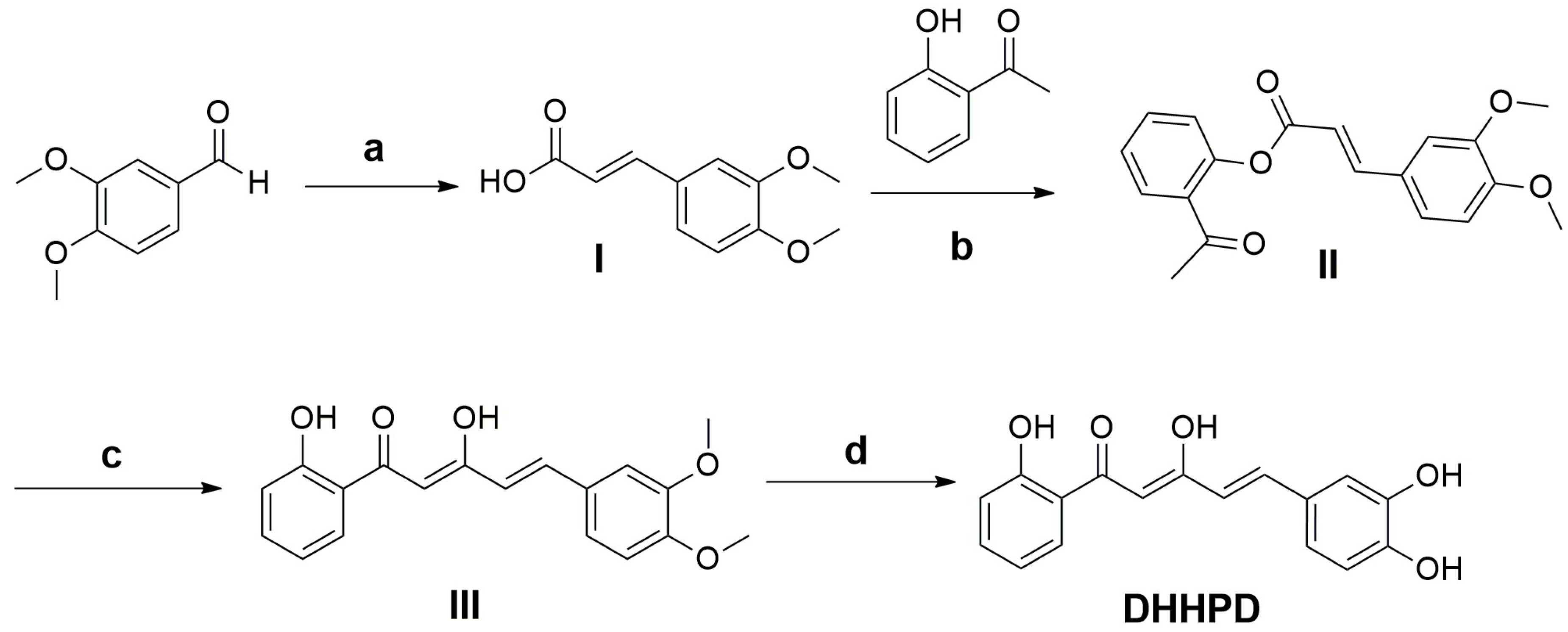
4.1. Synthesis of DHHPD
4.2. Chemicals and Drugs
4.3. Animals
4.4. Acute Toxicity Test
4.5. Antinociceptive Studies
4.5.1. Acetic Acid-Induced Writhing Test
4.5.2. Formalin-Induced Paw Licking Test
4.5.3. Hot-Plate Test
4.5.4. Involvement of the Opioidergic System
4.5.5. Capsaicin-Induced Paw Licking Test
4.5.6. Glutamate-Induced Paw Licking Test
4.6. Motor Performance Test
Rotarod Test
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, Y.; Wang, C.; Wang, L.; Parks, G.S.; Zhang, X.; Guo, Z.; Ke, Y.; Li, K.; Kim, M.K.; Vo, B.; et al. A novel analgesic isolated from a traditional Chinese medicine. Curr. Biol. 2014, 24, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Basbaum, A.I.; Bautista, D.M.; Scherrer, G.; Julius, D. Cellular and molecular mechanisms of pain. Cell 2010, 139, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Simmons, D.L. What makes a good anti-inflammatory drug target? Drug Discov. Today 2006, 11, 210–219. [Google Scholar] [CrossRef]
- North, R.A. Drug receptors and the inhibition of nerve cells. Br. J. Pharmacol. 1989, 98, 13–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Honey; Thareja, S.; Kumar, M.; Sinha, V.R. Self-organizing molecular field analysis of NSAIDs: Assessment of pharmacokinetic and physicochemical properties using 3D-QSPkR approach. Eur. J. Med. Chem. 2012, 53, 76–82. [Google Scholar] [CrossRef]
- Bavry, A.A.; Khaliq, A.; Gong, Y.; Handberg, E.M.; Cooper-DeHoff, R.M.; Pepine, C.J. Harmful Effects of NSAIDs among Patients with Hypertension and Coronary Artery Disease. Am. J. Med. 2011, 124, 614–620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deghrigue, M.; Festa, C.; Ghribi, L.; D’Auria, M.V.; De Marino, S.; Ben Jannet, H.; Bouraoui, A. Anti-inflammatory and analgesic activities with gastroprotective effect of semi–purified fractions and isolation of pure compounds from Mediterranean gorgonian Eunicella singularis. Asian Pac. J. Trop. Med. 2015, 8, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Bertam, G.K. Basic and Clinical Pharmacology, 7th ed.; Appleton & Lange: New York, NY, USA, 1998; pp. 496–513. ISBN 0838505651. [Google Scholar]
- Akil, H.; Owens, C.; Gutstein, H.; Taylor, L.; Curran, E.; Watson, S. Endogenous opioids: Overview and current issues. Drug Alcohol. Depend. 1998, 51, 127–140. [Google Scholar] [CrossRef]
- Terman, G.W.; Shavit, Y.; Lewis, J.W.; Cannon, J.T.; Liebeskind, J.C. Intrinsic mechanisms of pain inhibition: Activation by stress. Science 1984, 226, 1270–1277. [Google Scholar] [CrossRef] [PubMed]
- Porreca, F.; Ossipov, M.H. Nausea and vomiting side effects with opioid analgesics during treatment of chronic pain: Mechanisms, implications, and management options. Pain Med. 2009, 10, 654–662. [Google Scholar] [CrossRef] [PubMed]
- Benyamin, R.; Trescot, A.; Datta, S.; Buenaventura, R.; Adlaka, R.; Sehgal, N.; Glaser, S.E.; Vallejo, R. Opioid complications and side effects. Pain Phys. 2008, 11, S105–S120. [Google Scholar]
- McNicol, E.; Horowicz-Mehler, N.; Fisk, R.; Bennett, K.; Gialeli-Goudas, M.; Chew, P.; Lau, J.; Carr, D. Management of opioid side effects in cancer-related and chronic noncancer pain: A systematic review. J. Pain 2003, 4, 231–256. [Google Scholar] [CrossRef]
- Neal, M.J. Medical Pharmacology at a Glance, 3rd ed.; Blackwell Science Inc.: Hoboken, NJ, USA, 1997; pp. 64–65. [Google Scholar]
- Shipton, E.A. Development of narcotic addiction, tolerance and crosstolerance. S. Afr. Med. J. 1984, 66, 163. [Google Scholar] [PubMed]
- Max, M.B.; Payne, R.; Edwards, W.T.; Sunshine, A.; Inturrisi, C.E. Principles of Analgesic Use in the Treatment of Acute Pain and Cancer Pain, 4th ed.; American Pain Society: Glenview, IL, USA, 1999. [Google Scholar]
- Portenoy, R.K.; Payne, R. Acute and chronic pain. In Comprehensive Textbook of Substance Abuse, 3rd ed.; Lowinson, J.H., Ruiz, P., Millman, R., Eds.; Williams & Wilkins: Baltimore, MD, USA, 1997. [Google Scholar]
- Chapman, C.R.; Hill, H.F. Prolonged morphine self-administration and addiction liability. Evaluation of two theories in a bone marrow transplant unit. Cancer 1989, 63, 1636–1644. [Google Scholar] [CrossRef]
- Newman, R.G. The need to redefine “addiction”. N. Engl. J. Med. 1983, 308, 1096–1098. [Google Scholar] [CrossRef] [PubMed]
- Yob, N.J.; Jofrry, S.M.; Affandi, M.M.R.M.M.; Teh, L.K.; Salleh, M.Z.; Zakaria, Z.A. Zingiber zerumbet (L.) Smith: A Review of Its Ethnomedicinal, Chemical, and Pharmacological Uses. Evid. Based Complement. Altern. Med. 2011, 2011, 12. [Google Scholar] [CrossRef] [PubMed]
- Suloon, J.; Sulaiman, M.R.; Azlina, N.; Abu Bakar, N.A.; Makhtar, N.A.; Ismail, N.I.; Lee, M.T.; Kamaldin, M.N.; Mohamad, A.S.; Lajis, N.; et al. Antinociceptive Activity of Curcuma longa Essential Oil. UMTAS 2011, 2011, 555–560. [Google Scholar]
- Ammon, H.P.T.; Anazodo, M.I.; Safayhi, H.; Dhawan, B.N.; Srimal, R.C. Curcumin: A potent inhibitor of leukotriene B4 formation in rat peritoneal polymorphonuclear neutrophils (PMNL). Planta Med. 1992, 58, 226. [Google Scholar] [CrossRef] [PubMed]
- Azuine, M.A.; Bhide, S.V. Chemopreventive effect of turmeric against stomach and skin tumors induced by chemical carcinogens in Swiss mice. Nutr. Cancer 1992, 17, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Ammon, H.; Wahl, M. Pharmacology of Curcuma longa. Planta Med. 1991, 57, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kuttan, R.; Bhanumathy, P.; Nirmala, K.; George, M.C. Potential anticancer activity of turmeric (Curcuma longa). Cancer Lett. 1985, 29, 197–202. [Google Scholar] [CrossRef]
- Anand, P.; Thomas, S.G.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Sung, B.; Tharakan, S.T.; Misra, K.; Priyadarsini, I.K.; Rajasekharan, K.N.; et al. Biological activities of curcumin and its analogues (Congeners) made by man and Mother Nature. Biochem. Pharmacol. 2008, 76, 1590–1611. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.; Shao, L.; Wang, Y.; Zhao, C.; Chu, Y.; Xiao, J.; Zhao, Y.; Li, X.; Yang, S. Exploration and synthesis of curcumin analogues with improved structural stability both in vitro and in vivo as cytotoxic agents. Bioorg. Med. Chem. 2009, 17, 2623–2631. [Google Scholar] [CrossRef] [PubMed]
- Leong, S.W.; Mohd Faudzi, S.M.; Abas, F.; Mohd Aluwi, M.F.F.; Rullah, K.; Wai, L.K.; Abdul Bahari, M.N.; Ahmad, S.; Tham, C.L.; Shaari, K.; et al. Synthesis and sar study of diarylpentanoid analogues as new anti-inflammatory agents. Molecules 2014, 19, 16058–16081. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Farida, F.H.; Syahida, A.; Abas, F.; Shaari, K.; Israf, D.A.; Lajis, N.H. Synthesis and biological evaluation of curcumin-like diarylpentanoid analogues for anti-inflammatory, antioxidant and anti-tyrosinase activities. Eur. J. Med. Chem. 2009, 44, 3195–3200. [Google Scholar] [CrossRef] [PubMed]
- Ahmad Wani, T.; Kumar, D.; Prasad, R.; Verma, P.K.; Sardar, K.K.; Tandan, S.K.; Kumar, D. Analgesic activity of the ethanolic extract of Shorea robusta resin in experimental animals. Indian J. Pharmacol. 2012, 44, 493–499. [Google Scholar] [CrossRef]
- Couto, V.M.; Vilela, F.C.; Dias, D.F.; Santos, M.H.; Soncini, R.; Nascimento, C.G.O.; Giusti-paiva, A. Antinociceptive effect of extract of Emilia sonchifolia in mice. J. Ethnopharmacol. 2011, 134, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Woode, E.; Amoh-Barimah, A.K.; Abotsi, W.K.M.; Ainooson, G.K.; Owusu, G. Analgesic effects of stem bark extracts of Trichilia monadelpha (Thonn.) JJ De Wilde. Indian J. Pharmacol. 2012, 44, 765–773. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, D.P. Analgesic-like activity of essential oils constituents. Molecules 2011, 16, 2233–2252. [Google Scholar] [CrossRef] [PubMed]
- Farouk, L.; Laroubi, A.; Aboufatima, R.; Benharref, A.; Chait, A. Evaluation of the analgesic effect of alkaloid extract of Peganum harmala L.: Possible mechanisms involved. J. Ethnopharmacol. 2008, 115, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.; Fernandez, M.; Alvarez, A.; Saenz, M. Antinociceptive and anti-inflammatory effect of the aqueous extract from leaves of Pimenta racemosa var. ozua (Mirtaceae). J. Ethnopharmacol. 2004, 91, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, M.R.; Hussain, M.K.; Zakaria, Z.A.; Somchit, M.N.; Moin, S.; Mohamad, A.S.; Israf, D.A. Evaluation of the antinociceptive activity of Ficus deltoidea aqueous extract. Fitoterapia 2008, 79, 557–561. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Trujano, M.; Pena, E.; Martinez, A.; Moreno, J.; Guevara-Fefer, P.; Deciga-Campos, M.; Lopez-Moreno, F. Evaluation of the antinociceptive effect of Rosmarinus officinalis L. using three different experimental models in rodents. J. Ethnopharmacol. 2007, 111, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Hossain, C.F.; Al-Amin, M.; Rahman, K.M.M.; Sarker, A.; Alam, M.M.; Chowdhury, M.H.; Khan, S.N.; Sultana, G.N.N. Analgesic principle from Curcuma amada. J. Ethnopharmacol. 2015, 163, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Moniruzzaman, M.; Imam, M.Z. Evaluation of antinociceptive effect of methanolic extract of leaves of Crataeva nurvala Buch.-Ham. BMC Complement. Altern. Med. 2014, 14, 354. [Google Scholar] [CrossRef] [PubMed]
- Ming-Tatt, L.; Khalivulla, S.I.; Akhtar, M.N.; Mohamad, A.S.; Perimal, E.K.; Khalid, M.H.; Akira, A.; Lajis, N.; Ahmad Israf, D.; Sulaiman, M.R. Antinociceptive Activity of a Synthetic Curcuminoid Analogue, 2,6-bis-(4-hydroxy-3-methoxybenzylidene)cyclohexanone, on Nociception-induced Models in Mice. Basic Clin. Pharmacol. Toxicol. 2012, 110, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Bastos, G.N.T.; Santos, A.R.S.; Ferreira, V.M.M.; Costa, A.M.R.; Bispo, C.I.; Silveira, A.J.A.; Do Nascimento, J.L.M. Antinociceptive effect of the aqueous extract obtained from roots of Physalis angulata L. on mice. J. Ethnopharmacol. 2006, 103, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Ismail, N.I.; Ming-Tatt, L.; Lajis, N.; Akhtar, M.N.; Akira, A.; Perimal, E.K.; Israf, D.A.; Sulaiman, M.R. Antinociceptive Effect of 3-(2,3-Dimethoxyphenyl)-1-(5-methylfuran-2-yl)prop-2-en-1-one in Mice Models of Induced Nociception. Molecules 2016, 21, 1077. [Google Scholar] [CrossRef] [PubMed]
- Hajhashemi, V.; Ghannadi, A.; Pezeshkian, S.K. Antinociceptive and anti-inflammatory effects of Satureja hortensis L. extracts and essential oil. J. Ethnopharmacol. 2002, 82, 83–87. [Google Scholar] [CrossRef]
- Viana, A.F.; Heckler, A.P.; Fenner, R.; Rates, S.M.K. Antinociceptive activity of Hypericum caprifoliatum and Hypericum polyanthemum (Guttiferae). Braz. J. Med. Biol. Res. 2003, 36, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Hitner, H.; Nagle, B. Basic Pharmacology, 4th ed.; Glencoe/McGraw-Hill Education: New York, NY, USA, 2002; pp. 219–254. ISBN 0028046544. [Google Scholar]
- Khatun, A.; Imam, M.; Rana, M. Antinociceptive effect of methanol extract of leaves of Persicaria hydropiper in mice. BMC Complement. Altern. Med. 2015, 15, 63. [Google Scholar] [CrossRef] [PubMed]
- Fattori, V.; Hohmann, M.; Rossaneis, A.; Pinho-Ribeiro, F.; Verri, W. Capsaicin: Current Understanding of Its Mechanisms and Therapy of Pain and Other Pre-Clinical and Clinical Uses. Molecules 2016, 21, 844. [Google Scholar] [CrossRef] [PubMed]
- Lebovitz, E.E.; Keller, J.M.; Kominsky, H.; Kaszas, K.; Maric, D.; Iadarola, M.J. Positive Allosteric Modulation of TRPV1 as a Novel Analgesic Mechanism. Mol. Pain 2012, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Gosu, V.; Basith, S.; Hong, S.; Choi, S. Polymodal Transient Receptor Potential Vanilloid Type 1 Nocisensor: Structure, Modulators, and Therapeutic Applications. Adv. Protein Chem. Struct. Biol. 2016, 104, 81–125. [Google Scholar] [PubMed]
- Chizh, B.A.; O’Donnell, M.B.; Napolitano, A.; Wang, J.; Brooke, A.C.; Aylott, M.C.; Bullman, J.N.; Gray, E.J.; Lai, R.Y.; Williams, P.M.; et al. The effects of the TRPV1 antagonist SB-705498 on TRPV1 receptor-mediated activity and inflammatory hyperalgesia in humans. Pain 2007, 132, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Bleakman, D.; Alt, A.; Nisenbaum, E.S. Glutamate receptors and pain. Semin. Cell Dev. Biol. 2006, 17, 592–604. [Google Scholar] [CrossRef] [PubMed]
- Nucci-Martins, C.; Martins, D.F.; Nascimento, L.F.; Venzke, D.; Oliveira, A.S.; Frederico, M.J.S.; Silva, F.R.M.B.; Brighente, I.M.C.; Pizzolatti, M.G.; Santos, A.R.S. Ameliorative potential of standardized fruit extract of Pterodon pubescens Benth on neuropathic pain in mice: Evidence for the mechanisms of action. J. Ethnopharmacol. 2015, 175, 273–286. [Google Scholar] [CrossRef] [PubMed]
- Osikowicz, M.; Mika, J.; Przewlocka, B. The glutamatergic system as a target for neuropathic pain relief. Exp. Physiol. 2013, 98, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Beirith, A.; Santos, A.R.S.; Calixto, J.B. Mechanisms underlying the nociception and paw oedema caused by injection of glutamate into the mouse paw. Brain Res. 2002, 924, 219–228. [Google Scholar] [CrossRef]
- Beirith, A.; Santos, A.R.S.; Rodrigues, A.L.S.; Creczynski-Pasa, T.B.; Calixto, J.B. Spinal and supraspinal antinociceptive action of dipyrone in formalin, capsaicin and glutamate tests. Study of the mechanism of action. Eur. J. Pharmacol. 1998, 345, 233–245. [Google Scholar] [CrossRef]
- Azmi, F.; Leong, S.W.; Abas, F.; Ming, O.H.; Perimal, E.K.; Akira, A.; Ahmad Israf, D.; Sulaiman, M.R. Antinociceptive effect of 2-benzoyl-6-(3,4-dihydroxybenzylidene)cyclohexen-1-ol on nociception induced models in mice. J. Pharmacol. Toxicol. Investig. 2016, 2, 1–9. [Google Scholar]
- Sulaiman, M.R.; Perimal, E.K.; Zakaria, Z.A.; Mokhtar, F.; Akhtar, M.N.; Lajis, N.H.; Israf, D.A. Preliminary analysis of the antinociceptive activity of zerumbone. Fitoterapia 2009, 80, 230–232. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, A.S.; Akhtar, M.N.; Zakaria, Z.A.; Perimal, E.K.; Khalid, S.; Mohd, P.A.; Khalid, M.H.; Israf, D.A.; Lajis, N.H.; Sulaiman, M.R. Antinociceptive activity of a synthetic chalcone, flavokawin B on chemical and thermal models of nociception in mice. Eur. J. Pharmacol. 2010, 647, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Guilhon, C.C.; Rahayu, I.; Wahab, A.; Boylan, F.; Fernandes, P.D. Central Antinociceptive and Mechanism of Action of Pereskia bleo Kunth Leaves Crude Extract, Fractions, and Isolated Compounds. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.M.; Mohamad, A.S.; Makhtar, N.; Khalid, M.H.; Khalid, S.; Perimal, E.K.; Mastuki, S.N.; Zakaria, Z.A.; Lajis, N.; Ahmad Israf, D.; et al. Antinociceptive activity of methanolic extract of Acmella uliginosa (Sw.) Cass. J. Ethnopharmacol. 2011, 133, 227–233. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compound, DHHPD 5-(3,4-dihydroxyphenyl)-3-hydroxy-1-(2-hydroxyphenyl)penta-2,4-dien-1-one, are available from F.A., S.W.L. and M.R.S. |
| Interval Following Treatment (min) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | 0 | 30 | 60 | 90 | 120 | 150 | 180 | 210 | 240 | |
| Dose (mg/kg) | Latency Time (s) | |||||||||
| Vehicle (10 mL/kg) | - | 6.16 ± 0.17 | 6.83 ± 0.30 | 6.67 ± 0.33 | 6.67 ± 0.21 | 6.83 ± 0.31 | 6.67 ± 0.21 | 6.83 ± 0.31 | 6.33 ± 0.21 | 6.30 ± 0.03 |
| DHHPD | 0.1 | 7.08 ± 0.35 | 7.94 ± 1.11 | 8.11 ± 0.56 | 9.58 ± 1.31 ** | 8.79 ± 0.45 | 7.29 ± 0.54 | 7.08 ± 0.36 | 6.57 ± 0.59 | 8.17 ± 0.84 |
| 0.3 | 7.34 ± 0.34 | 8.30 ± 0.93 | 9.28 ± 0.34 * | 10.01 ± 0.72 ** | 9.62 ± 0.80 * | 8.83 ± 0.38 | 7.92 ± 0.44 | 8.21 ± 0.48 | 8.51 ± 0.63 | |
| 1 | 6.52 ± 0.23 | 7.58 ± 1.10 | 11.80 ± 1.22 **** | 10.14 ± 1.34 *** | 9.67 ± 0.42 * | 10.60 ± 0.50 **** | 10.80 ± 1.14 **** | 7.59 ± 0.48 | 8.30 ± 0.90 | |
| 3 | 7.14 ± 0.39 | 9.51 ± 0.43 * | 10.93 ± 0.78 **** | 11.55 ± 1.06 **** | 12.34 ± 1.07 **** | 10.23 ± 0.45 *** | 8.84 ± 0.53 | 9.41 ± 0.61 ** | 7.68 ± 0.34 | |
| Morphine | 5 | 7.50 ± 0.34 | 18.33 ± 0.67 **** | 17.00 ± 0.82 **** | 16.33 ± 0.42 **** | 16.33 ± 0.99 **** | 15.33 ± 0.62 **** | 15.17 ± 0.40 **** | 14.83 ± 0.40 **** | 13.70 ± 0.05 |
| Naloxone + DHHPD | 5 + 3 | 7.13 ± 0.22 | 08.56 ± 0.52 | 9.79 ± 1.05 | 9.01 ± 0.89 | 13.76 ± 1.03 | 9.95 ± 0.43 | 8.25 ± 0.71 | 7.30 ± 0.53 | 7.88 ± 0.35 |
| Naloxone + Morphine | 5 + 5 | 6.33 ± 0.21 | 6.67 ± 0.33 # | 6.67 ± 0.33 # | 6.50 ± 0.22 # | 7.00 ± 0.37 # | 6.50 ± 0.22 # | 6.83 ± 0.17 # | 6.50 ± 0.22 # | 6.36 ± 0.01 # |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamarudin, N.; Hisamuddin, N.; Ong, H.M.; Ahmad Azmi, A.F.; Leong, S.W.; Abas, F.; Sulaiman, M.R.; Shaik Mossadeq, W.M. Analgesic Effect of 5-(3,4-Dihydroxyphenyl)-3-hydroxy-1-(2-hydroxyphenyl)penta-2,4-dien-1-one in Experimental Animal Models of Nociception. Molecules 2018, 23, 2099. https://doi.org/10.3390/molecules23092099
Kamarudin N, Hisamuddin N, Ong HM, Ahmad Azmi AF, Leong SW, Abas F, Sulaiman MR, Shaik Mossadeq WM. Analgesic Effect of 5-(3,4-Dihydroxyphenyl)-3-hydroxy-1-(2-hydroxyphenyl)penta-2,4-dien-1-one in Experimental Animal Models of Nociception. Molecules. 2018; 23(9):2099. https://doi.org/10.3390/molecules23092099
Chicago/Turabian StyleKamarudin, Nadhirah, Nadia Hisamuddin, Hui Ming Ong, Ahmad Farhan Ahmad Azmi, Sze Wei Leong, Faridah Abas, Mohd Roslan Sulaiman, and Wan Mastura Shaik Mossadeq. 2018. "Analgesic Effect of 5-(3,4-Dihydroxyphenyl)-3-hydroxy-1-(2-hydroxyphenyl)penta-2,4-dien-1-one in Experimental Animal Models of Nociception" Molecules 23, no. 9: 2099. https://doi.org/10.3390/molecules23092099