3.1. VOC Analysis and Sniffing Test for C. indicum Linne
Table 1 shows the VOCs that were contained in the sample of
C. indicum Linné that was used in this study. We identified a total of 42 VOCs, with 32 hydrocarbons, four acids, three alcohols, two ketones, and one aldehyde. Individual VOCs included camphor (621.08 ± 18.51 μg/mL), germacrene D (117.43 ± 16.62 μg/mL), bornyl acetate (86.99 ± 17.21 μg/mL), 1,8-cineole (66.18 ± 8.59 μg/mL), and β-ocimene (60.77 ± 8.63 μg/mL).
In addition to measuring the concentrations of these VOCs, we also used a sniffing test to classify the VOCs that were associated with
C. indicum Linné by intensity. Of the 7 VOCs that could by identified by smell, 1,8-cineole gave a very strong sense of
C. indicum Linné, while camphor showed a low strength similarity to our sample of
C. indicum Linné. In general, the odor image of the
C. indicum Linné that was used in this study was considered to be mostly due to 1,8-cineole and camphor. In a study by Shin et al., 45 VOCs were reported to be present in
C. indicum Linné, while camphor and 1,8-cineol were reported to be the main VOCs [
12]. These results are consistent with the results of the current study. In the literature, 1,8-cineole is reported to be a VOC that is commonly found in the essential oils of
Eucalyptus globulus and
Laurus nobilis. This compound is used in ointments to treat coughs, muscular pain, and rheumatism [
30]. In a study by Miyazawa, 1,8-cineole was detected in the urine of people who had taken cold medicine, and 1,8-cineole was reported to be used as a major marker in urine [
31]. Meanwhile, camphor is known to be used commercially in moth repellent and antiseptic agents. Similar to 1,8-cineole, there are also reports of camphor being used to treat rheumatic pain and coughs [
30].
3.2. Effect of Inhalation of the VOCs in C. indicum Linné on Blood Pressure and EEG
We recorded the changes in heart rate and blood pressure in 10 adult male and female subjects after inhalation of VOCs using
C. indicum Linné essential oil (
Table 2). The heart rate decreased after VOC inhalation in eight out of 10 subjects, and this decrease was significant in five subjects (
p < 0.05). The systolic blood pressure decreased after VOC inhalation in nine out of 10 subjects, and this decrease was significant in seven subjects (
p < 0.05). In contrast, the diastolic blood pressure showed no consistent response pattern. However, there was a significant decrease in the diastolic blood pressure of four subjects (
p < 0.05). These results suggest that inhalation of the VOCs in
C. indicum Linné lowers systolic blood pressure and has a relaxing effect on heart rate.
Nasally inhaled VOCs are conveyed via olfactory receptors to the limbic system, where they cause an emotional, instinctive response; in turn, they stimulate the autonomic nervous system, modulating the heart rate, blood pressure, breathing, reproduction, memory, and stress response. Additionally, the VOCs that enter through the skin are transported in the blood stream and absorbed by the pulmonary alveoli, spreading to the whole body to take part in various chemical reactions [
24]. In a study by Jung et al., the majority of the 29 participants they tested showed a decrease in heart rate after inhaling the VOCs from ylang-ylang [
27]. Kikuchi et al. observed a decrease in the heart rate of participants following inhalation of the VOCs from rose oil and concluded that rose oil inhalation had a relaxing effect [
32]. Kovar et al. observed an increase in blood 1,8-cineole levels after rosemary oil inhalation in a mouse model and found that this increase correlated with locomotor stimulation [
33]. Sayorwan detected a decrease in the heart rate of humans following sweet almond oil inhalation, while rosemary oil inhalation was, conversely, associated with a temporary increase in heart rate [
34]. Based on these studies, we can deduce that inhalation of plant-derived essential oils does not induce a decrease in heart rate but that the actions of these oils are mediated by the compounds that they contain. It will be necessary to investigate the VOCs that cause a decrease in heart rate in further studies.
We used EEG to measure the brainwaves of participants before and after the inhalation of the VOCs from
C. indicum Linné; the results are displayed in
Table 3,
Table 4,
Table 5 and
Table 6, while the brain mapping is shown in
Figure 1,
Figure 2,
Figure 3 and
Figure 4. Different regions of the cortex fulfill different roles. EEG allows us to examine different regions of the brain where electrodes are attached. Fp1 and Fp2 were placed over the prefrontal cortex, which is involved in attention and logical reasoning; abnormalities in attention are highlighted by the responses in the area of Fp1, while reasoning and impulse control are associated with the responses in the area of Fp2. F3 and F4 were located over the frontal cortex, with each of these regions affecting the contralateral side of the body. These regions are involved in thought processes, including goal selection, decision making, social judgments, and initiation of behavior; in cases of hyperactivity or impairment in the frontal area function, defects are observed in attention, awareness, motor function, affect, and memory [
35]. T3 and T4 were located over the temporal cortex, which is responsible for receiving and processing auditory and olfactory information, and is also involved in linguistic understanding, stimulation, and control of emotion. Injury to the temporal area is known to manifest in auditory defects, aphasia, and impaired stimulus recognition [
36]. P3 and P4 were located over the parietal cortex, which is associated with sensory and motor function. The parietal cortex is involved in receiving and verifying sensory information and affects physical awareness, left–right discrimination, abstract thought, and interpretation of mathematical symbols; abnormalities in the parietal area are known to cause agnosia and impaired awareness of physical sensation [
37]. O1 and O2 were placed over the occipital cortex, which is responsible for visual function. Injury in this area is known to lead to impaired visual cognition. In addition, when the temporal lobe and occipital lobe are both injured, this can result in visual field defects or cortical blindness accompanied by object agnosia, achromatopsia, and visual agnosia [
38].
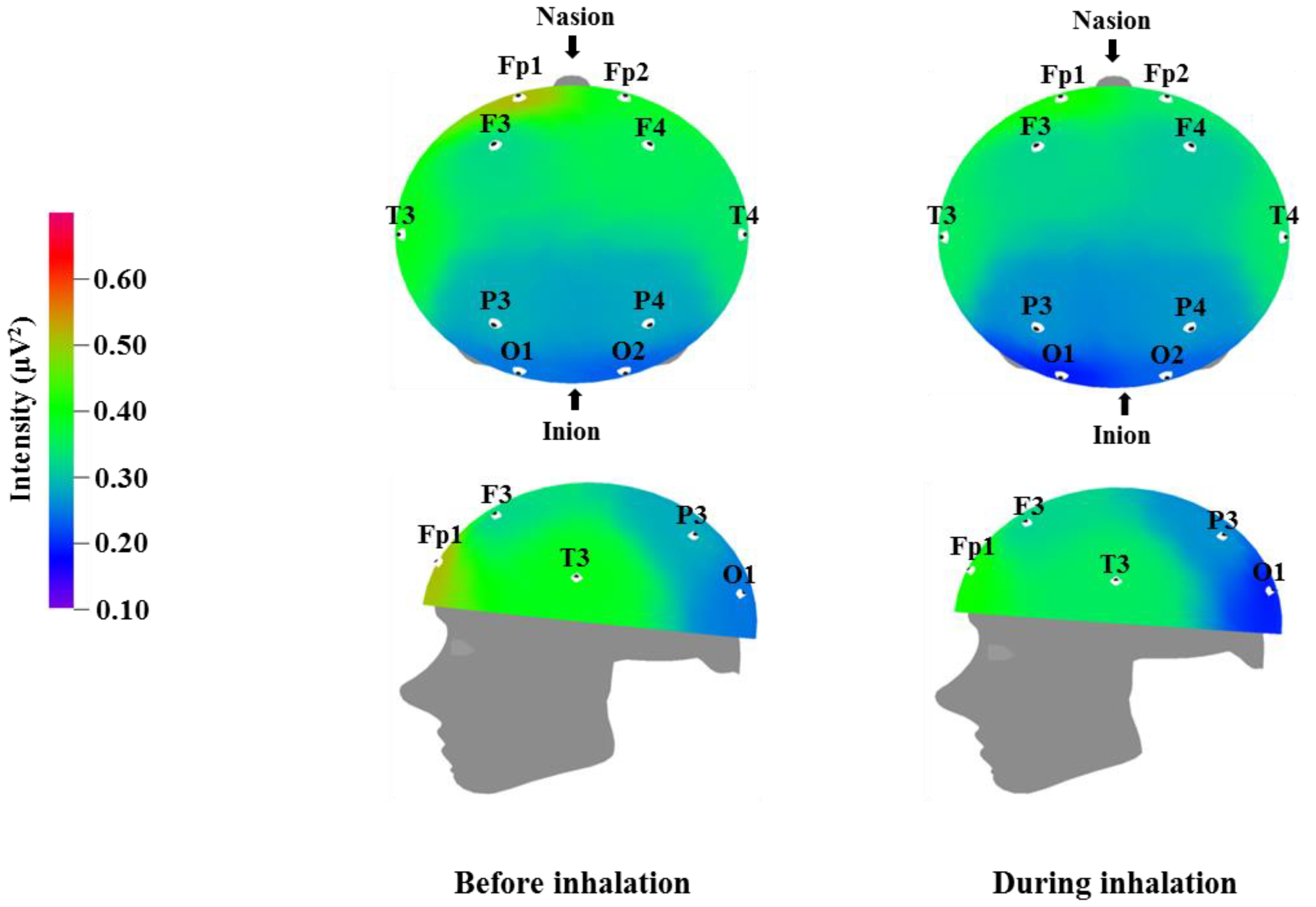
Table 3 and
Figure 1 show the recorded changes in the relative theta waves; an increase was observed at eight electrode sites: Fp2, F3, F4, T3, T4, P3, P4, and O2. Theta waves are in the 4–8 Hz frequency band. These waves are usually observed in adults during the early sleep stages, when the individual begins to feel drowsy, and are known to also increase when an individual is thinking of new ideas. Theta waves are associated with the hippocampus, which plays an important role in memory in humans. Better memory has been observed for events occurring at times of increased theta wave activity [
39]. Given that theta waves are detected during peaceful meditation, the overall increase in theta waves that was observed after essential oil inhalation may be related to mental and physical relaxation.
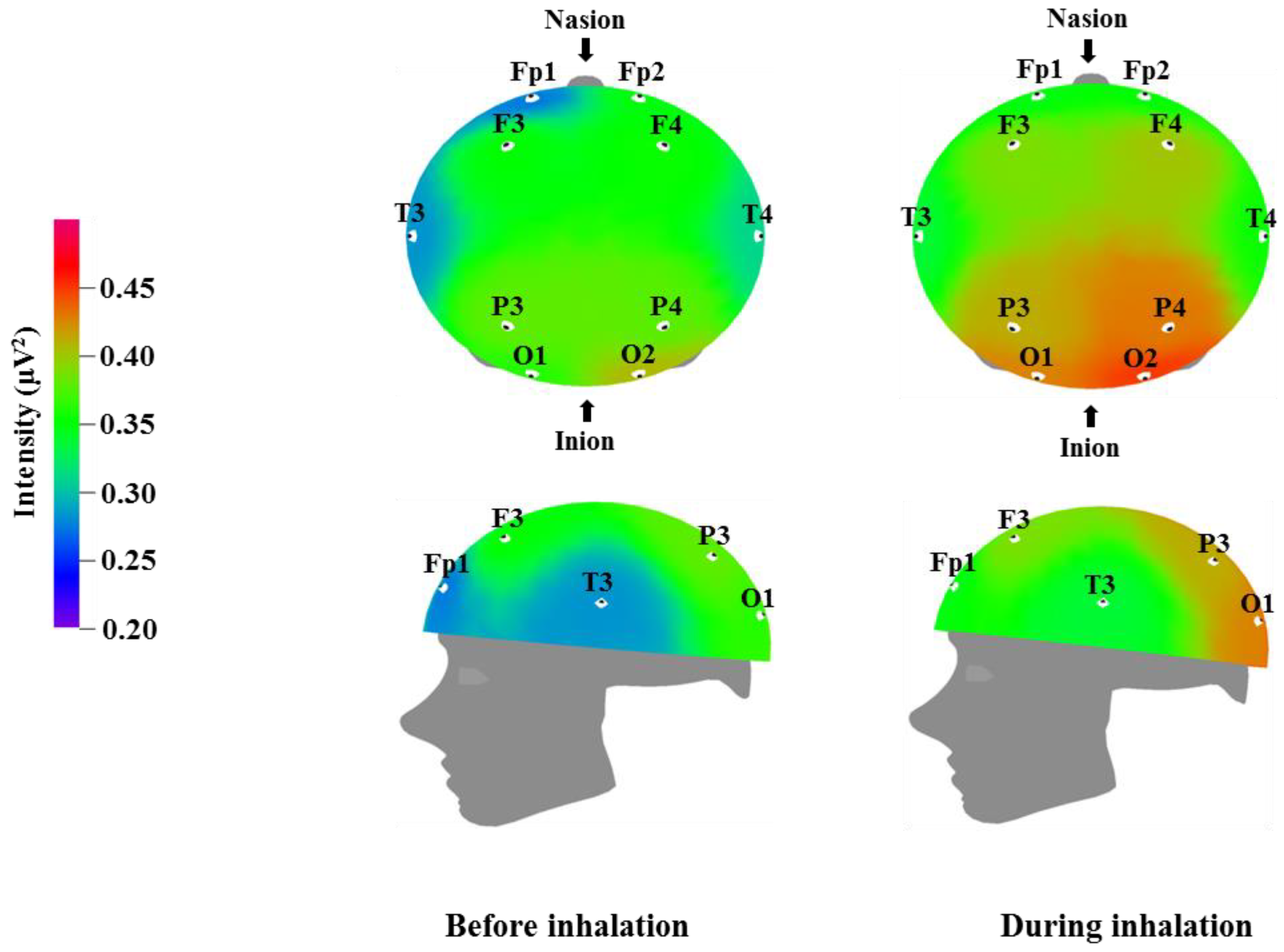
Table 4 and
Figure 2 show the recorded changes in the relative alpha waves; an increase was observed at nine electrode sites: Fp1, F3, F4, T3, T4, P3, P4, O1, and O2. Alpha waves are in the 8–12 Hz frequency band, and are commonly observed in a state of rest, such as meditation or peaceful relaxation. An increase in alpha wave activity indicates a decrease in activity in the corresponding cortical region. Alpha wave increases occur when the subject feels physical and mental relaxation and comfort and can be considered to be correlated with the reduced blood pressure and heart rate shown in
Table 2. Given that alpha wave activity typically becomes weaker during excited states or cognitive activity, the overall alpha wave response observed following essential oil inhalation is suggestive of mental relaxation. Similarly, Sugano reported increases in alpha wave activity following the inhalation of α-pinene, 1,8-cineole, lavender, sandalwood, musk, and eucalyptus [
40]. Lee et al. detected various brainwave changes following the inhalation of citrus, lavender, and floral flavors, with lavender in particular showing an increase in alpha wave activity in the occipital region [
41]. Lorig reported that pleasant odors promote increased alpha waves, whereas unpleasant odors cause a decrease in alpha waves [
42]. Pleasant odors were also found to result in different breathing patterns; specifically, pleasant odor inhalation was associated with deeper breathing. This change in breathing pattern could be one factor that contributes to the mental and physical relaxation that appears to be induced by essential oil inhalation. Our results are consistent with the results of previous studies, which have reported that 1,8-cineole and methyl jasmonate are factors that could cause increases in theta and alpha waves similar to those described in
Table 3 and
Table 4 [
43]. In particular, the effects of 1,8-cineole have been demonstrated in both human and animal studies [
33,
44].
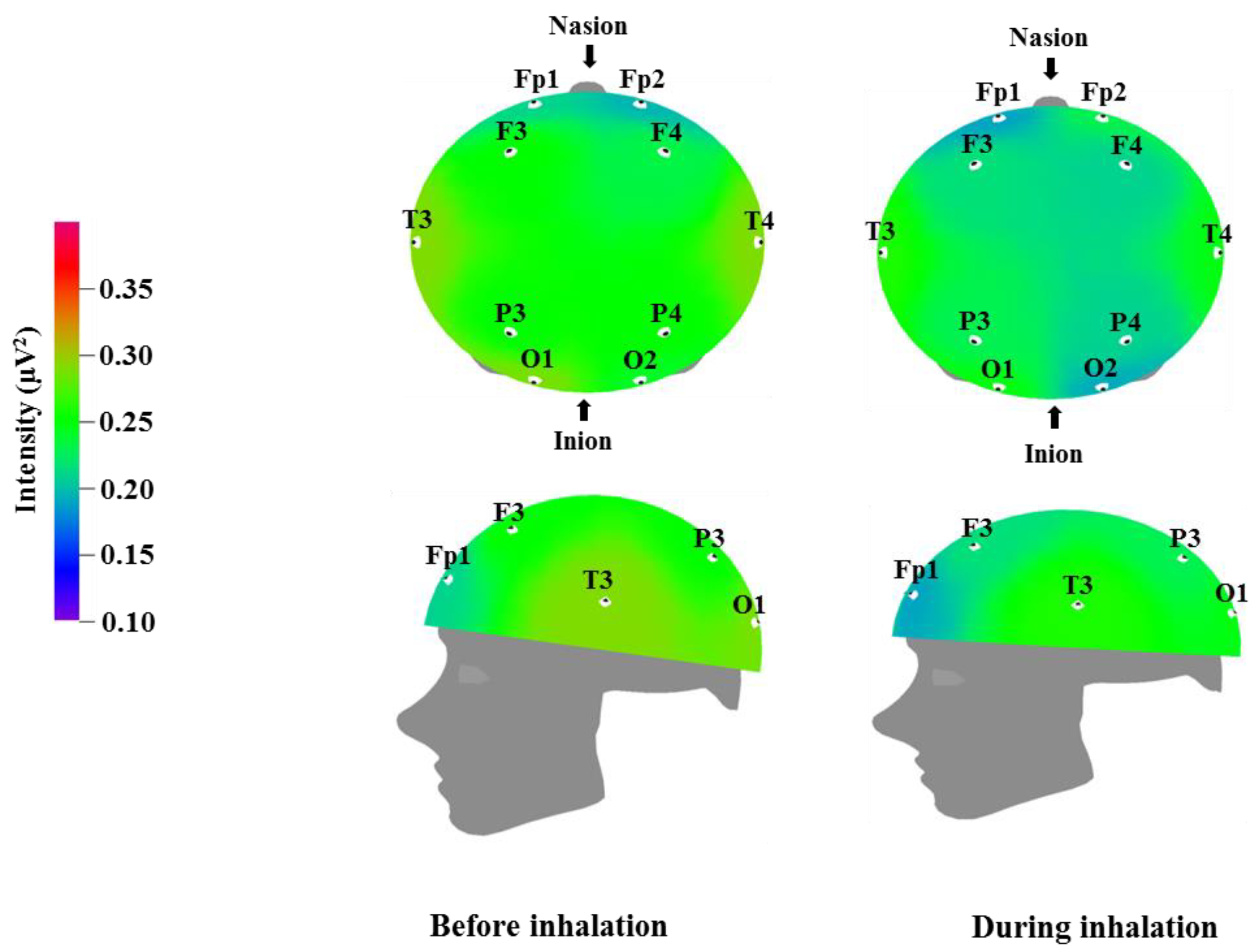
Table 5 and
Figure 3 show the recorded changes in the relative beta waves; a decreasing trend was observed at nine electrode sites, excluding Fp2: Fp1, F3, F4, T3, T4, P3, P4, O1, and O2. Beta waves are in the 12–30 Hz frequency band and can be categorized further into low beta waves and high beta waves. Low beta wave activity is known to increase awareness. Excessive high beta wave activity can be a major cause of anxiety in subjects, and highly anxious subjects who are overly cautious can show strong high beta wave activity [
39]. Recent research has demonstrated that high beta wave activity is associated with migraines [
45]. The results in
Table 3 and
Table 4 show a trend towards a negative correlation between increased theta and alpha wave activity, which is associated with relaxation and attention, and decreased beta wave activity, which is associated with anxiety and nervousness.
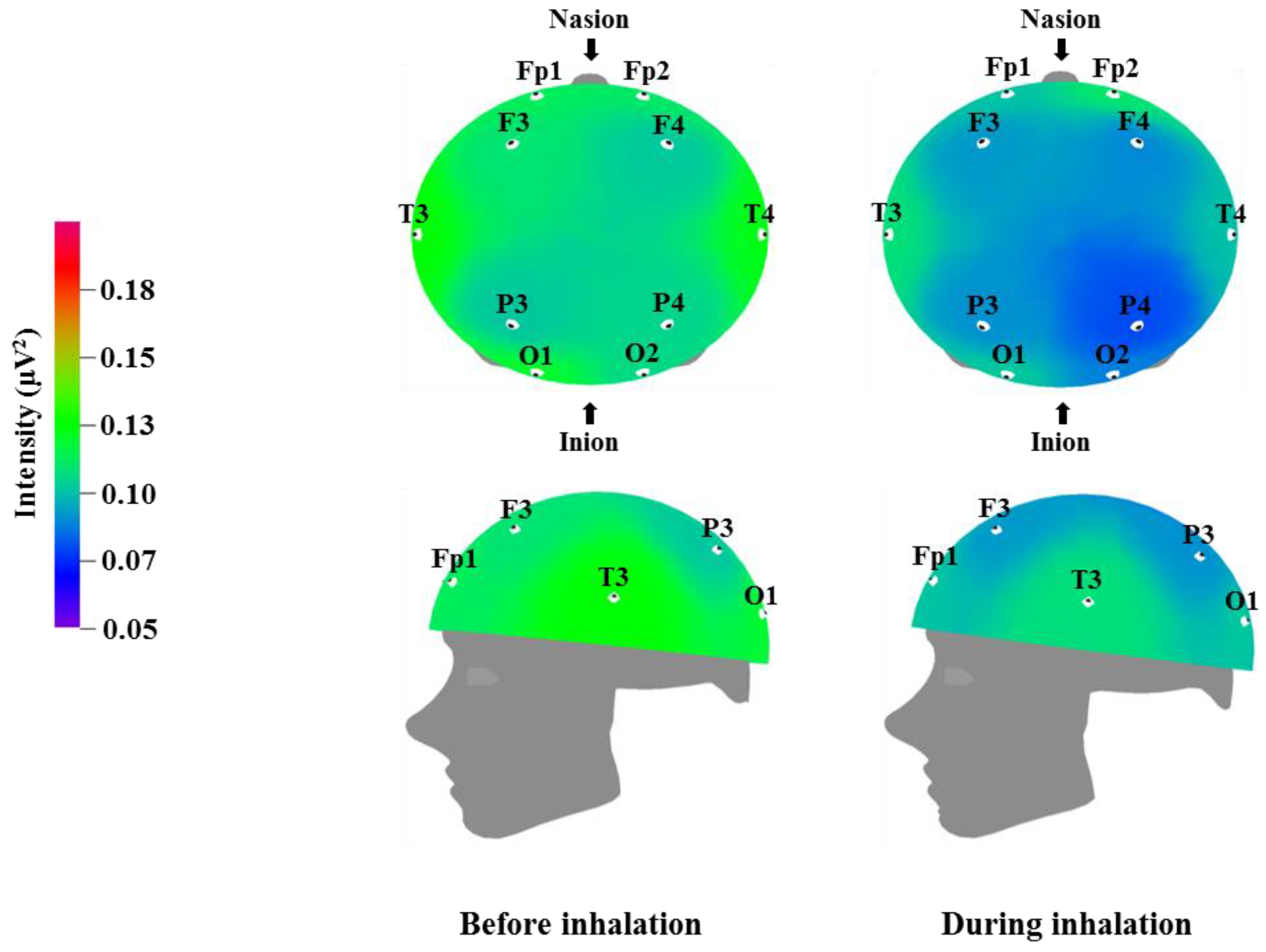
Table 6 and
Figure 4 show the recorded changes in the relative gamma waves; similar to the relative beta wave results, a decrease was observed in the gamma waves at nine electrode sites, excluding Fp2: Fp1, F3, F4, T3, T4, P3, P4, O1, and O2. Gamma waves are in the 31–50 Hz frequency band and represent the highest frequency band for brainwaves. Gamma waves usually appear during focused attention during complex problem solving or while using multiple cortical areas to think of an answer; thus, they are closely related to learning [
39]. However, excessive gamma wave activity can cause cortical fatigue. Similar to the relative beta wave activity shown in
Table 5 and
Figure 3, the decrease in gamma wave activity observed after inhalation of
C. indicum Linné essential oil is thought to be related to brain relaxation and the decrease in heart rate and blood pressure.