Oleanolic Acid-amino Acids Derivatives: Design, Synthesis, and Hepatoprotective Evaluation In Vitro and In Vivo
Abstract
:1. Introduction
2. Results
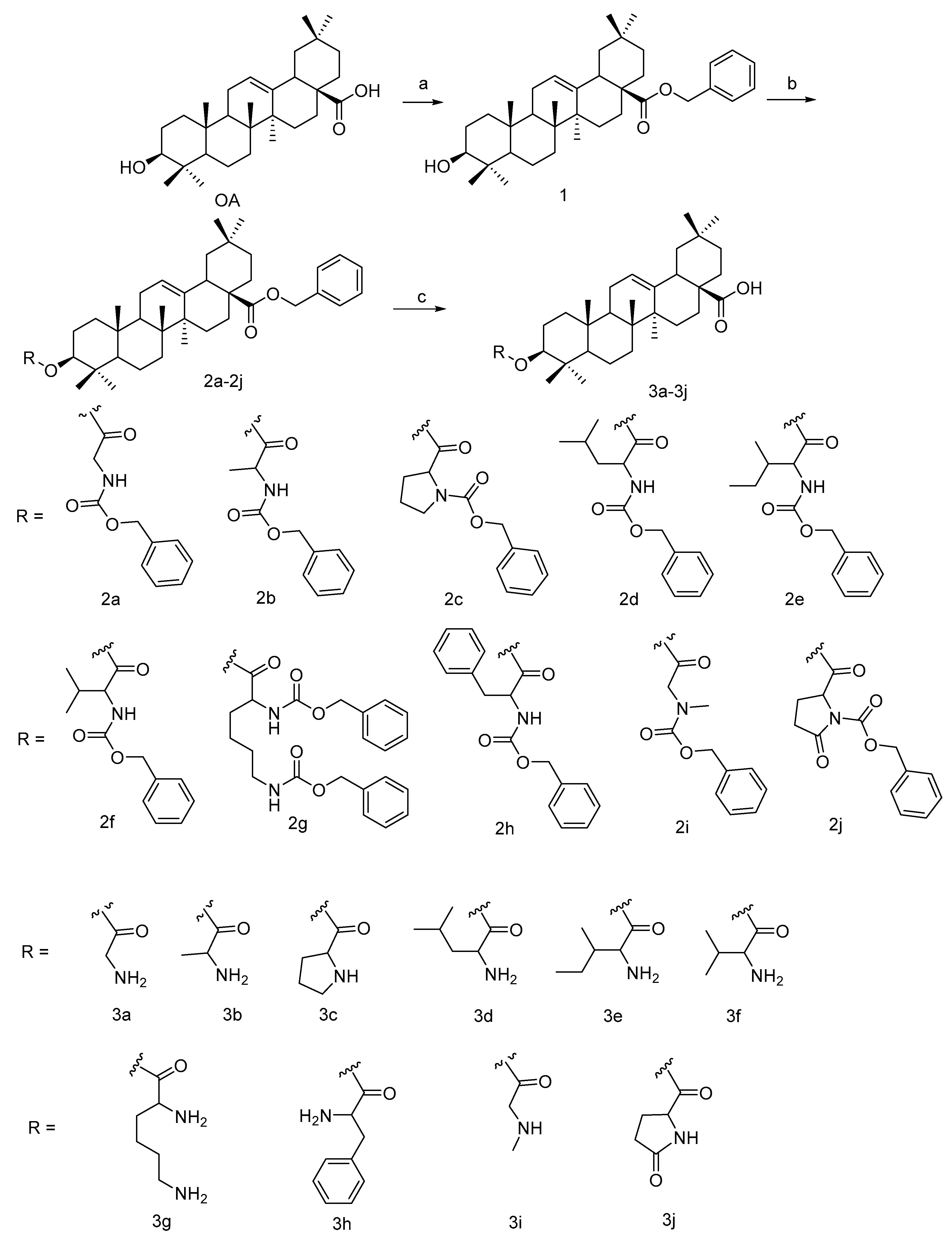
2.1. Chemical Synthesis
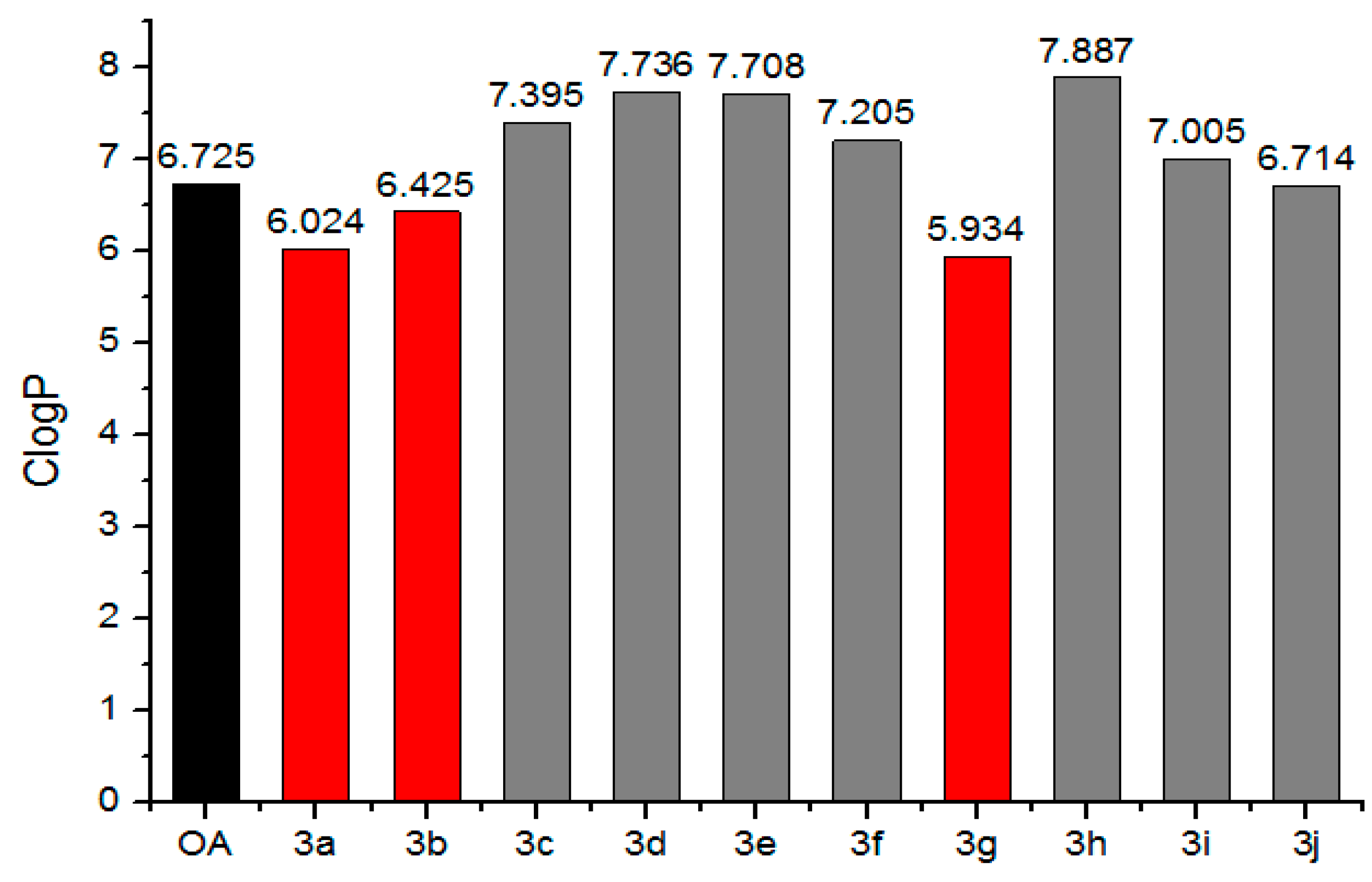
2.2. Simulation Online of ClogP
2.3. In Vitro Cytotoxicity
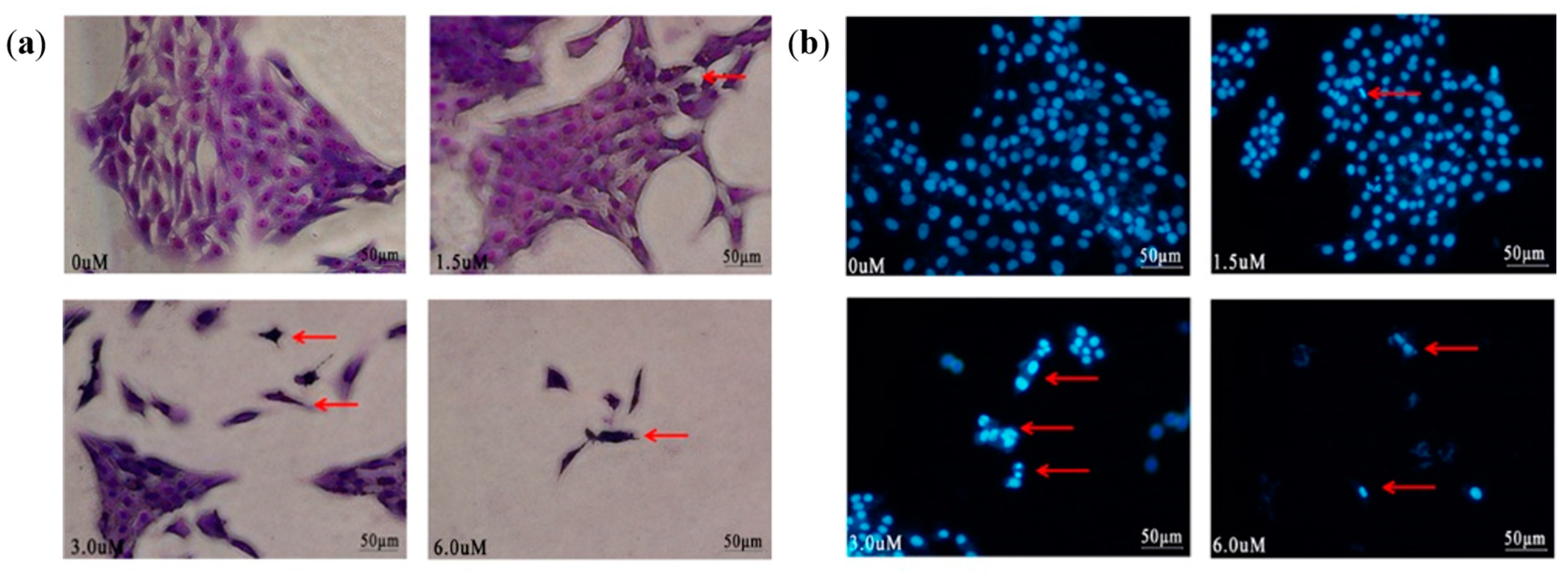
2.4. Morphological Changes
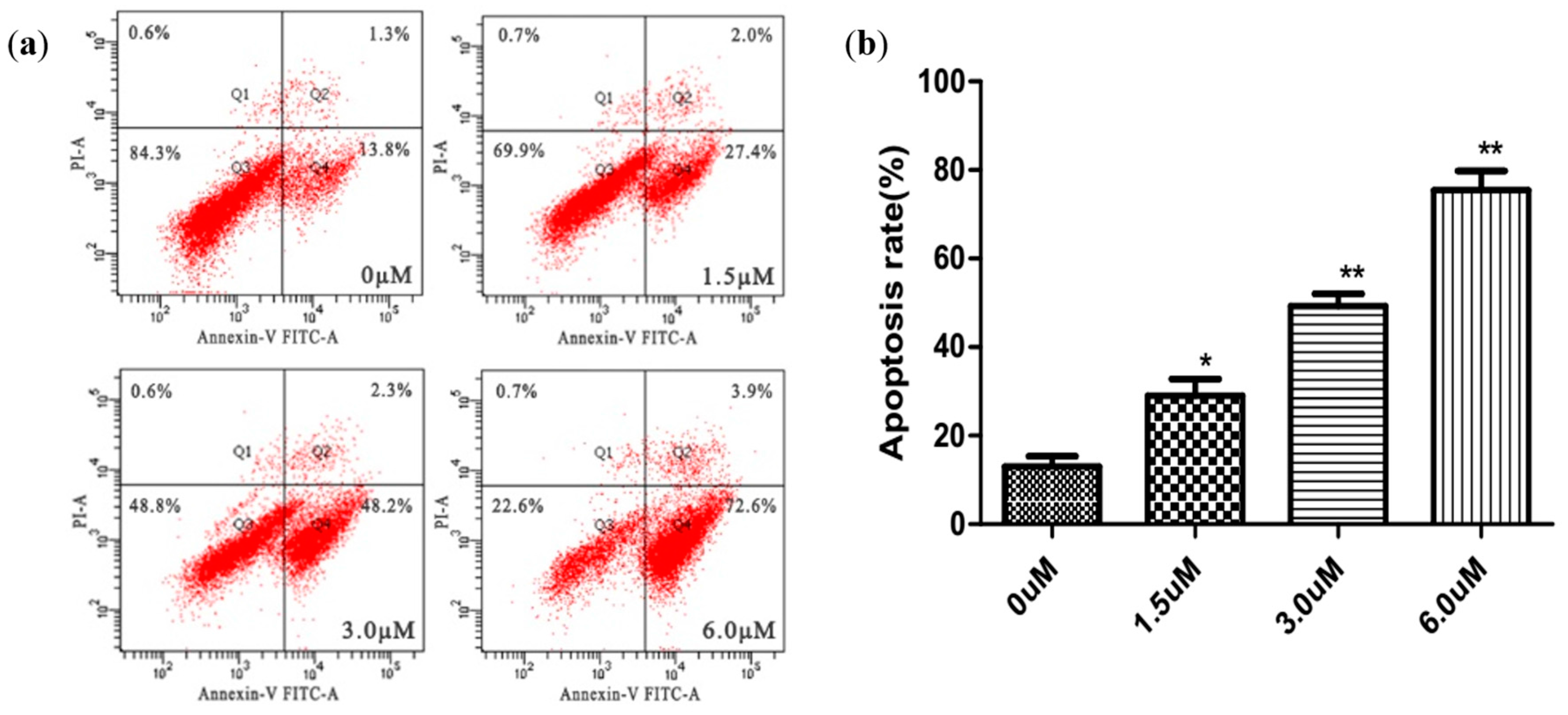
2.5. Apoptosis Analysis Using Annexin V-FITC/PI Staining
2.6. Measurement of Mitochondrial Membrane Potential (MMP)
2.7. Measurement of Intracellular Ca2+ Concentrations
2.8. In Vivo Activity on CCl4 Acute Liver Injury Mice Model
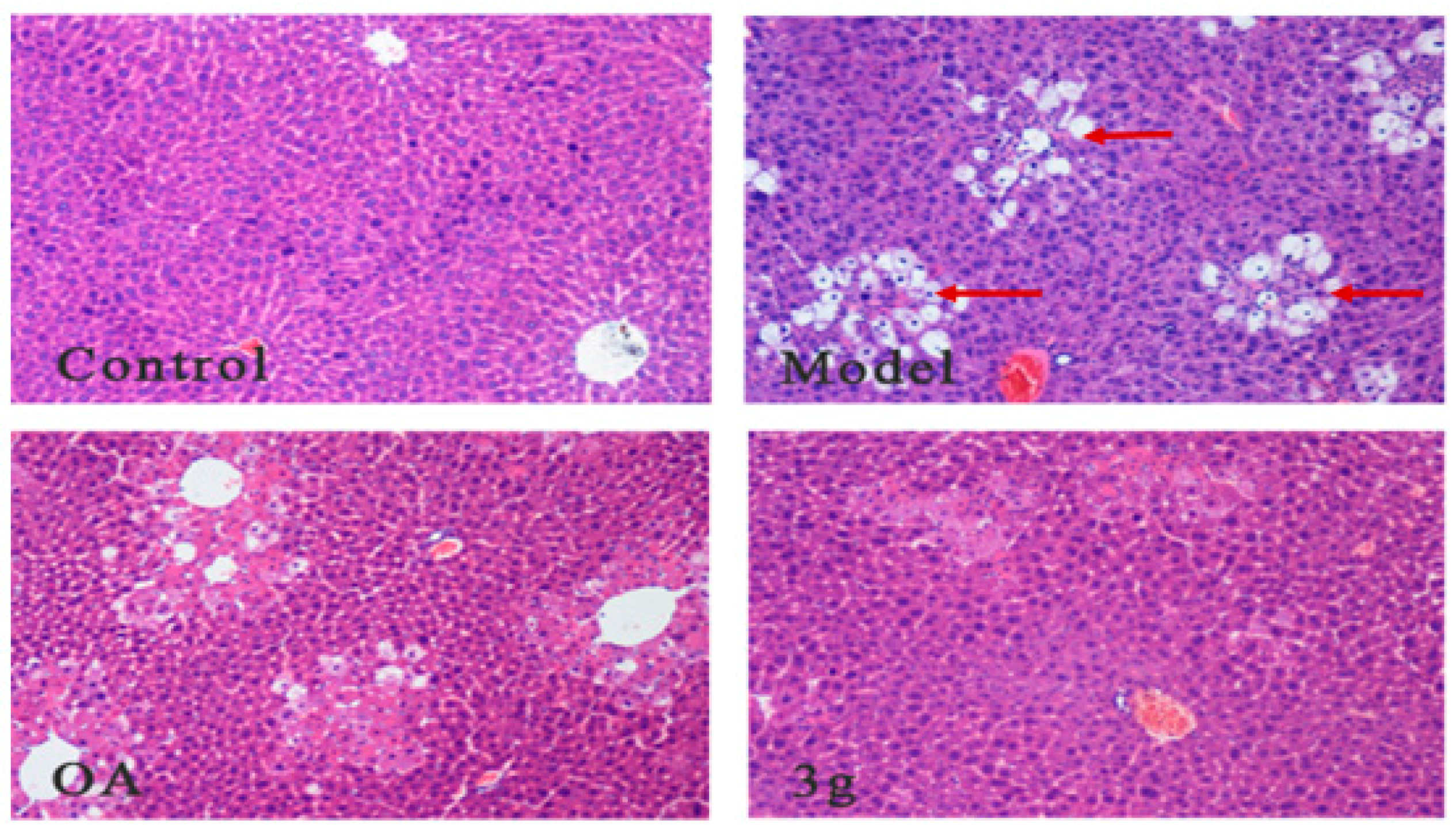
2.8.1. Morphological Observation by HE Staining
2.8.2. Determination of the Ability of ALT and AST in Serum
3. Discussion
4. Materials and Methods
4.1. General Synthesis of Compounds 3a–3j
4.2. Cell Viability Assay
4.3. Giemsa Staining Assay
4.4. DAPI Staining
4.5. Annexin V-FITC/PI Staining Assay
4.6. Mitochondrial Membrane Potential Assay
4.7. Intracellular Free Ca2+ Levels Assay
4.8. In Vivo Activity on CCl4 Acute Liver Injury Mice Model
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bataller, R.; Brenner, D.A. Liver fibrosis. J. Clin. Investig. 2005, 115, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, K.; Murata, M.; Yoshida, K.; Sekimoto, G.; Uemura, Y.; Sakaida, N.; Kaibori, M.; Kamiyama, Y.; Nishizawa, M.; Fujisawa, J.; et al. Chronic inflammation associated with hepatitis C virus infection perturbs hepatic transforming growth factor β signaling, promoting cirrhosis and hepatocellular carcinoma. Hepatology 2007, 46, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Friedman, S.L. Mechanisms of disease: Mechanisms of hepatic fibrosis and therapeutic implications. Nat. Clin. Pract. Gastroenterol. Hepatol. 2004, 1, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Gabele, E.; Brenner, D.A.; Rippe, R.A. Liver fibrosis: Signals leading to the amplification of the fibrogenic hepatic stellate cell. Front. Biosci. 2003, 8, 69–77. [Google Scholar]
- Izumi, H.; Torigoe, T.; Ishiguchi, H.; Uramoto, H.; Yoshida, Y.; Tanabe, M.; Ise, T.; Murakami, T.; Yoshida, T.; Nomoto, M.; et al. Cellular pH regulators: Potentially promising molecular targets for cancer chemotherapy. Cancer Treat. Rev. 2003, 29, 541–549. [Google Scholar] [CrossRef]
- Song, J.; Ge, Z.; Yang, X.; Luo, Q.; Wang, C.; You, H.; Ge, T.; Deng, Y.; Lin, H.; Cui, Y.; et al. Hepatic stellate cells activated by acidic tumor microenvironment promote the metastasis of hepatocellular carcinoma via osteopontin. Cancer Lett. 2015, 356, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Lingwal, P.; Bhaat, G.K.; Kothiyal, P. Hepatic Stellate Cells as a Target for the treatment of liver fibrosis. Int. J. Pharm. Res. Rev. 2015, 4, 32–37. [Google Scholar]
- Liu, J. Pharmacology of oleanolic acid and ursolic acid. J. Ethnopharmacol. 1995, 49, 57–68. [Google Scholar] [CrossRef]
- Liu, J. Oleanolic acid and ursolic acid: Research perspectives. J. Ethnopharmacol. 2005, 100, 92–94. [Google Scholar] [CrossRef] [PubMed]
- Kinjo, J.; Okawa, M.; Udayama, M.; Shono, Y.; Hirakawa, T.; Shii, Y.; Nohara, T. Hepatoprotective and hepatotoxic actions of oleanolic acid-type triterpenoidal glucuronides on rat primary hepatocyte cultures. Chem. Pharm. Bull. 1999, 47, 290–292. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.F.; Liu, J.; Wu, K.C.; Klaassen, C.D. Protection against phalloidin-induced liver injury by oleanolic acid involves Nrf2 activation and suppression of Oatp1b2. Toxicol. Lett. 2015, 232, 326–332. [Google Scholar] [CrossRef] [PubMed]
- China Pharmacopoeia Committee. Chinese Pharmacopoeia; China Medical Science Press: Beijing, China, 2010. [Google Scholar]
- Zheng, X.; Zheng, Q.; Bo, S.; Zeng, X.; Wei, Y.; Lei, S.; Xiao, X.; Xiao, J.; Wang, Z. Advances in research on hepatoprotective activity and synthesis of oleanolic acid derivatives. J. Appl. Biopharm. Pharmacokinet. 2015, 3, 27–33. [Google Scholar] [CrossRef]
- Shanmugam, M.K.; Dai, X.; Kumar, A.P.; Tan, B.K.H.; Sethi, G.; Bishayee, A. Oleanolic acid and its synthetic derivatives for the prevention and therapy of cancer: Preclinical and clinical evidence. Cancer Lett. 2014, 346, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Sun, W.; Peng, W.; Yu, R.; Li, G.; Jiang, T. Pharmacokinetics in vitro and in vivo of two novel prodrugs of oleanolic acid in rats and its hepatoprotective effects against liver injury induced by CCl4. Mol. Pharm. 2016, 13, 1699–1710. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Zhang, C.; Li, B.; Xu, X.; Liang, M.; Gu, S.; Chu, F.; Xu, B.; Ren, J.; Wang, P.; et al. A Series of oleanolic acid derivatives as anti-hepatitis B virus agents: Design, synthesis, and in vitro and in vivo biological evaluation. Molecules 2016, 21, 402. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Zheng, W.; Lin, S.; Guo, F.; Zhu, Y.; Wei, Y.; Liu, X.; Jin, L.; Li, Y. Identification of an Oleanane-type Triterpene Hedragonic Acid as a Novel Farnesoid X Receptor Ligand with Liver Protective Effects and Anti-inflammatory Activity. Mol. Pharmacol. 2017, 93, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Bi, S.; Chu, F.; Wang, M.; Li, B.; Mao, P.; Zhang, H.; Wang, P.; Guo, W.; Xu, L.; Ren, L.; et al. Ligustrazine-Oleanolic Acid Glycine Derivative, G-TOA, Selectively Inhibited the Proliferation and Induced Apoptosis of Activated HSC-T6 Cells. Molecules 2016, 21, 1599. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Han, Y.; Zhang, Y.; Yan, W.; Xu, B.; Chu, F.; Xie, T.; Jia, M.; Yan, M.; Zhao, R.; et al. A New Oleanolic Acid Derivative against CCl4-Induced Hepatic Fibrosis in Rats. Int. J. Mol. Sci. 2017, 18, 553. [Google Scholar] [CrossRef] [PubMed]
- Ayeleso, T.B.; Matumba, M.G.; Mukwevho, E. Oleanolic Acid and Its Derivatives: Biological Activities and Therapeutic Potential in Chronic Diseases. Molecules 2017, 22, 1915. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, Z.; Zhang, L.; Zhang, Y.; Mao, L.; Jiang, H. Synthesis, insecticidal activity and inhibition on topoisomerase I of 20 (S)-t-Boc-amino acid derivatives of camptothecin. Pestic. Biochem. Physiol. 2017, 139, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Cai, E.; Guo, S.; Yang, L.; Han, M.; Xia, J.; Zhao, Y. Synthesis and antitumour activity of arctigenin amino acid ester derivatives against H22 hepatocellular carcinoma. Nat. Prod. Res. 2017, 32, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Yan, W.Q.; Xu, X.; Wu, G.; Zhang, C.; Han, Y.; Chu, F.; Zhao, R.; Wang, P.; Lei, H. Combination of amino acid/dipeptide with ligustrazine-betulinic acid as antitumor agents. Eur. J. Med. Chem. 2017, 130, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Cai, E.; Yang, L.; Jia, C.; Zhang, W.; Zhao, Y.; Li, W.; Song, X.; Zheng, M. The synthesis and evaluation of arctigenin amino acid ester derivatives. Chem. Pharm. Bull. 2016, 64, 1466–1473. [Google Scholar] [CrossRef] [PubMed]
- Chu, F.; Xu, X.; Li, G.; Gu, S.; Xu, K.; Gong, Y.; Xu, B.; Wang, M.; Zhang, H.; Zhang, Y.; et al. Amino acid derivatives of ligustrazine-oleanolic acid as new cytotoxic agents. Molecules 2014, 19, 18215–18231. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Wang, M.; Gou, S.; Liu, X.; Zhang, H.; Cao, F. Combination of amino acid/dipeptide with nitric oxide donating oleanolic acid derivatives as PepT1 targeting antitumor prodrugs. J. Med. Chem. 2014, 57, 1116–1120. [Google Scholar] [CrossRef] [PubMed]
- Kondratenko, R.M.; Baltina, L.A.; Vasil’eva, E.V.; Nasyrov, K.M.; Kireeva, R.M.; Baschenko, N.Z.; Fridman, S.M.; Baltina, L.A., Jr.; Tolstikov, G.A. Synthesis and immunomodulating activity of new amino acid derivatives of glycyrrhizic acid and its methyl ester. Russ. J. Bioorg. Chem. 2004, 30, 148–153. [Google Scholar] [CrossRef]
- Wang, P.; She, G.; Yang, Y.; Li, Q.; Zhang, H.; Liu, J.; Cao, Y.; Xu, X.; Lei, H. Synthesis and biological evaluation of new ligustrazine derivatives as anti-tumor agents. Molecules 2012, 17, 4972–4985. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yan, W.; Li, B.; Xu, B.; Gong, Y.; Chu, F.; Zhang, Y.; Yao, Q.; Wang, P.; Lei, H. A new ligustrazine derivative-selective cytotoxicity by suppression of NF-κB/p65 and COX-2 expression on human hepatoma cells. Part 3. Int. J. Mol. Sci. 2015, 16, 16401–16413. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Chu, F.; Zhang, Y.; Wang, X.; Li, Q.; Liu, W.; Xu, X.; Xing, Y.; Chen, J.; Wang, P.; et al. A series of new ligustrazine-triterpenes derivatives as anti-tumor agents: Design, synthesis, and biological evaluation. Int. J. Mol. Sci. 2015, 16, 21035–21055. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Xu, X.; Chu, F.; Wang, M.; Wang, P.; Li, G.; Song, J.; Zhang, Y.; Lei, H. Synthesis and biological evaluation of T-OA analogues as the cytotoxic agents. Res. Chem. Intermed. 2015, 41, 6257–6269. [Google Scholar] [CrossRef]
- Meng, G.R.; Li, J.J.; Wang, G.L.; Dong, M.J.; Zhang, Q. Synthesis and cytotoxic activities of the amino Acid-conjugates of 10-hydroxycamptothecin. Chin. J. Org. Chem. 2014, 34, 155–160. [Google Scholar] [CrossRef]
- Ramachandran, P.; Iredale, J.P. Liver fibrosis: A bidirectional model of fibrogenesis and resolution. Int. J. Med. 2012, 105, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Popov, Y.; Schuppan, D. Targeting liver fibrosis: Strategies for development and validation of antifibrotic therapies. Hepatology 2009, 50, 1294–1306. [Google Scholar] [CrossRef] [PubMed]
- Gatenby, R.A.; Gillies, R.J. Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer 2004, 4, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Ozawa, S.; Miyamoto, C.; Maehata, Y.; Suzuki, A.; Maeda, T.; Baba, Y. Acidic extracellular microenvironment and cancer. Cancer Cell Int. 2013, 13, 89. [Google Scholar] [CrossRef] [PubMed]
- Getachew, Y.; Cusimano, F.A.; Gopal, P.; Reisman, S.A.; Shay, J.W. The synthetic triterpenoid RTA 405 (CDDO-EA) halts progression of liver fibrosis and reduces hepatocellular carcinoma size resulting in increased survival in an experimental model of chronic liver injury. Toxicol. Sci. 2015, 149, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.M.; Wu, X.X.; Sun, Y.; Kong, X.W.; Zhang, Y.H.; Xu, Q. A novel synthetic oleanolic acid derivative (CPU-II 2) attenuates liver fibrosis in mice through regulating the function of hepatic stellate cells. J. Biomed. Sci. 2008, 15, 251–259. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |



| Compounds | IC50 (μM) | |||
|---|---|---|---|---|
| HSCs | HepG2 | Bel-7402 | L02 | |
| OA | >50 a | >50 | >50 | >50 |
| 3a | 13.58 ± 1.12 | 11.50 ± 0.95 | 12.09 ± 1.32 | >25 b |
| 3b | 9.92 ± 1.05 | 12.17 ± 1.48 | >25 | >50 |
| 3c | 15.55 ± 2.31 | 11.05 ± 0.63 | 10.49 ± 1.49 | 10.08 ± 0.56 |
| 3d | >25 | >25 | >25 | >25 |
| 3e | 24.53 ± 1.99 | 18.00 ± 1.78 | 18.66 ± 1.63 | >25 |
| 3f | 19.61 ± 3.20 | 18.64 ± 2.65 | >25 | 15.22 ± 1.20 |
| 3g | 3.46 ± 1.16 | 3.67 ± 0.61 | 4.70 ± 0.75 | 12.31 ± 2.34 |
| 3h | 14.76 ± 0.80 | 11.49 ± 0.94 | 8.50 ± 1.65 | >50 |
| 3i | 7.30 ± 1.58 | >25 | 11.83 ± 0.83 | >50 |
| 3j | >25 | 23.90 ± 2.65 | >25 | >50 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, F.; Zhang, W.; Guo, W.; Wang, Z.; Yang, Y.; Zhang, X.; Fang, K.; Yan, M.; Wang, P.; Lei, H. Oleanolic Acid-amino Acids Derivatives: Design, Synthesis, and Hepatoprotective Evaluation In Vitro and In Vivo. Molecules 2018, 23, 322. https://doi.org/10.3390/molecules23020322
Chu F, Zhang W, Guo W, Wang Z, Yang Y, Zhang X, Fang K, Yan M, Wang P, Lei H. Oleanolic Acid-amino Acids Derivatives: Design, Synthesis, and Hepatoprotective Evaluation In Vitro and In Vivo. Molecules. 2018; 23(2):322. https://doi.org/10.3390/molecules23020322
Chicago/Turabian StyleChu, Fuhao, Wenxi Zhang, Wenbo Guo, Zhaoyi Wang, Yuqin Yang, Xinyu Zhang, Kang Fang, Mengmeng Yan, Penglong Wang, and Haimin Lei. 2018. "Oleanolic Acid-amino Acids Derivatives: Design, Synthesis, and Hepatoprotective Evaluation In Vitro and In Vivo" Molecules 23, no. 2: 322. https://doi.org/10.3390/molecules23020322





