Probing Gas Adsorption in Zeolites by Variable-Temperature IR Spectroscopy: An Overview of Current Research
Abstract
:1. Introduction
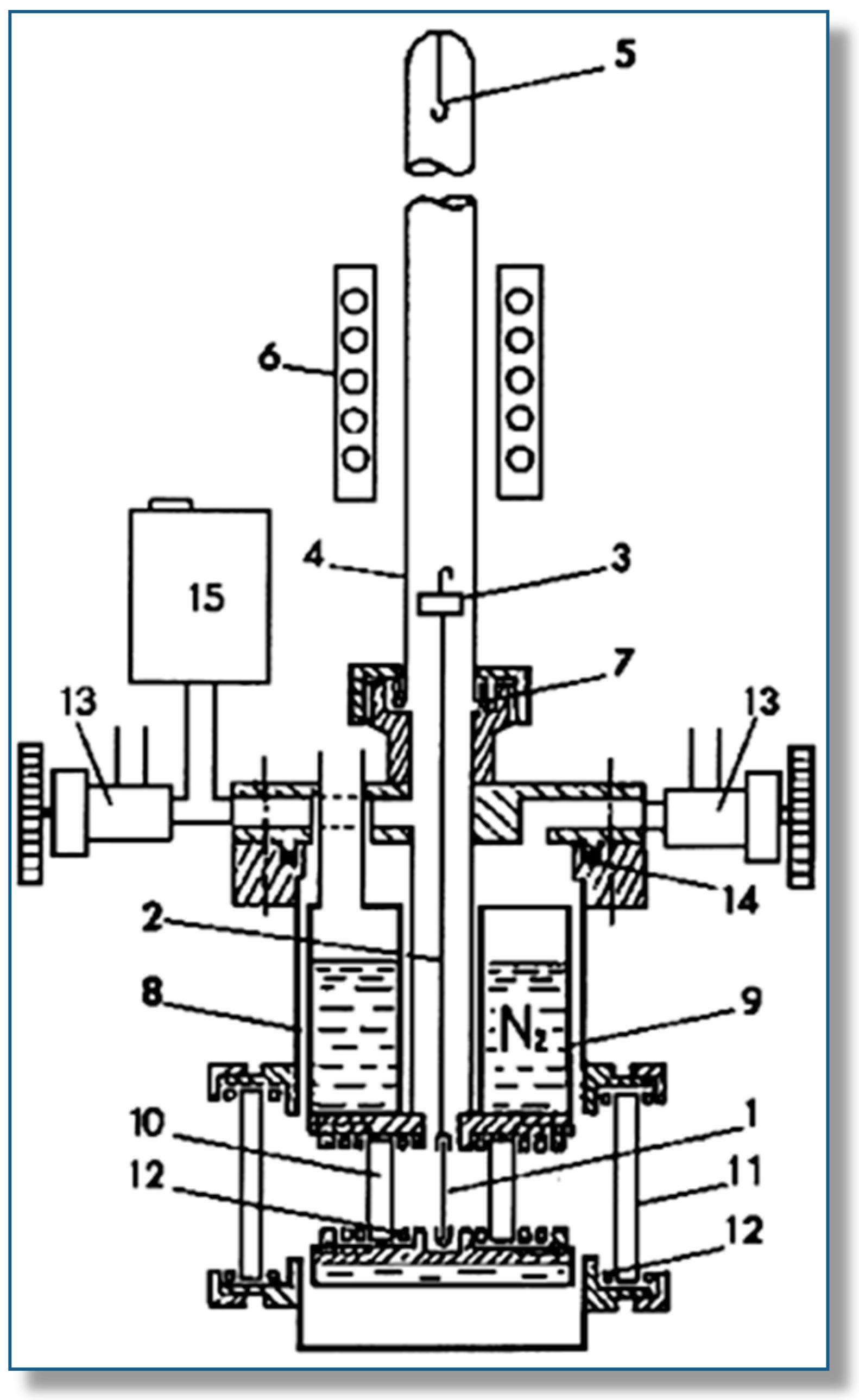
2. Outline of the VTIR Method
3. Selected Case Studies
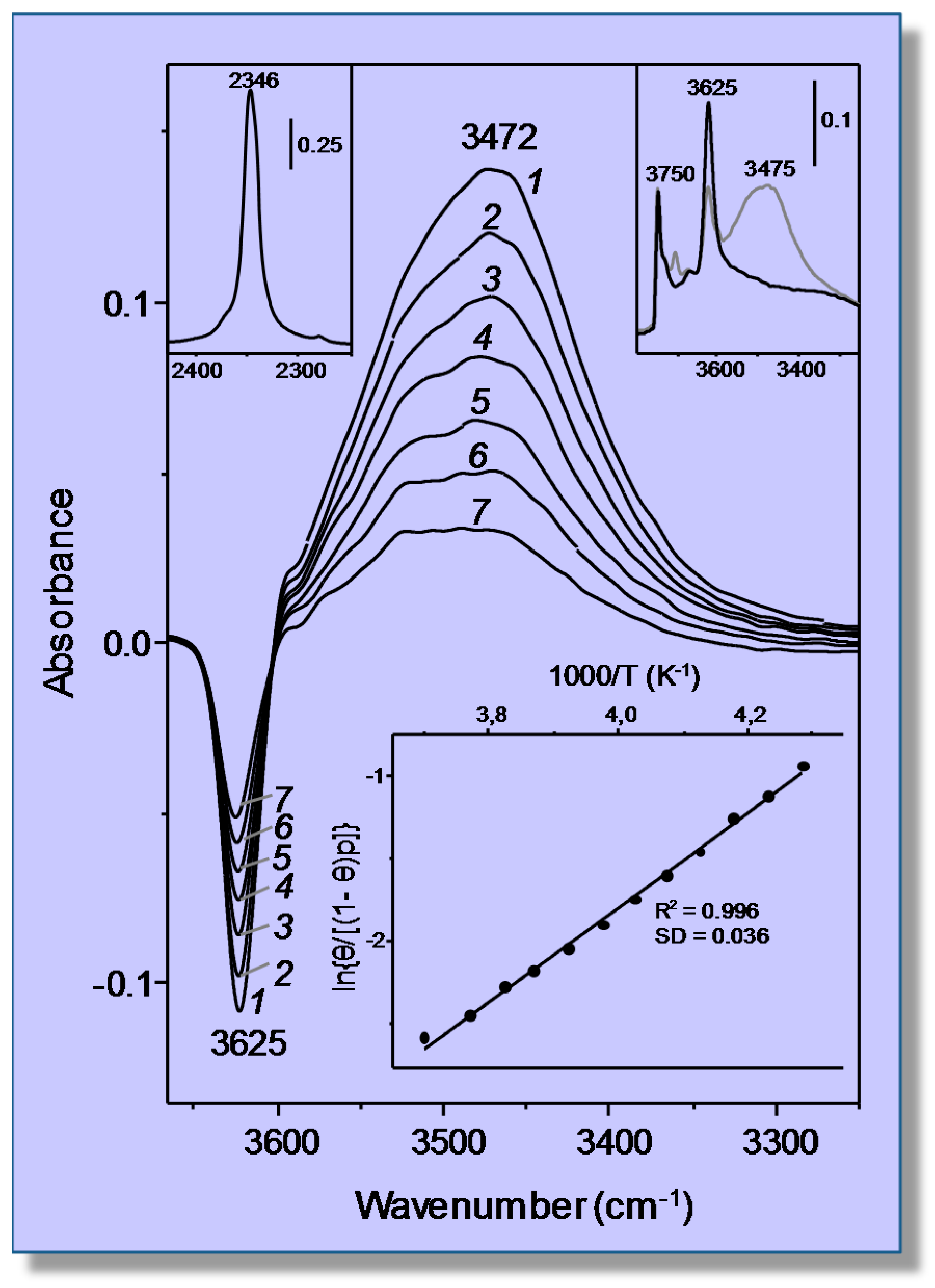
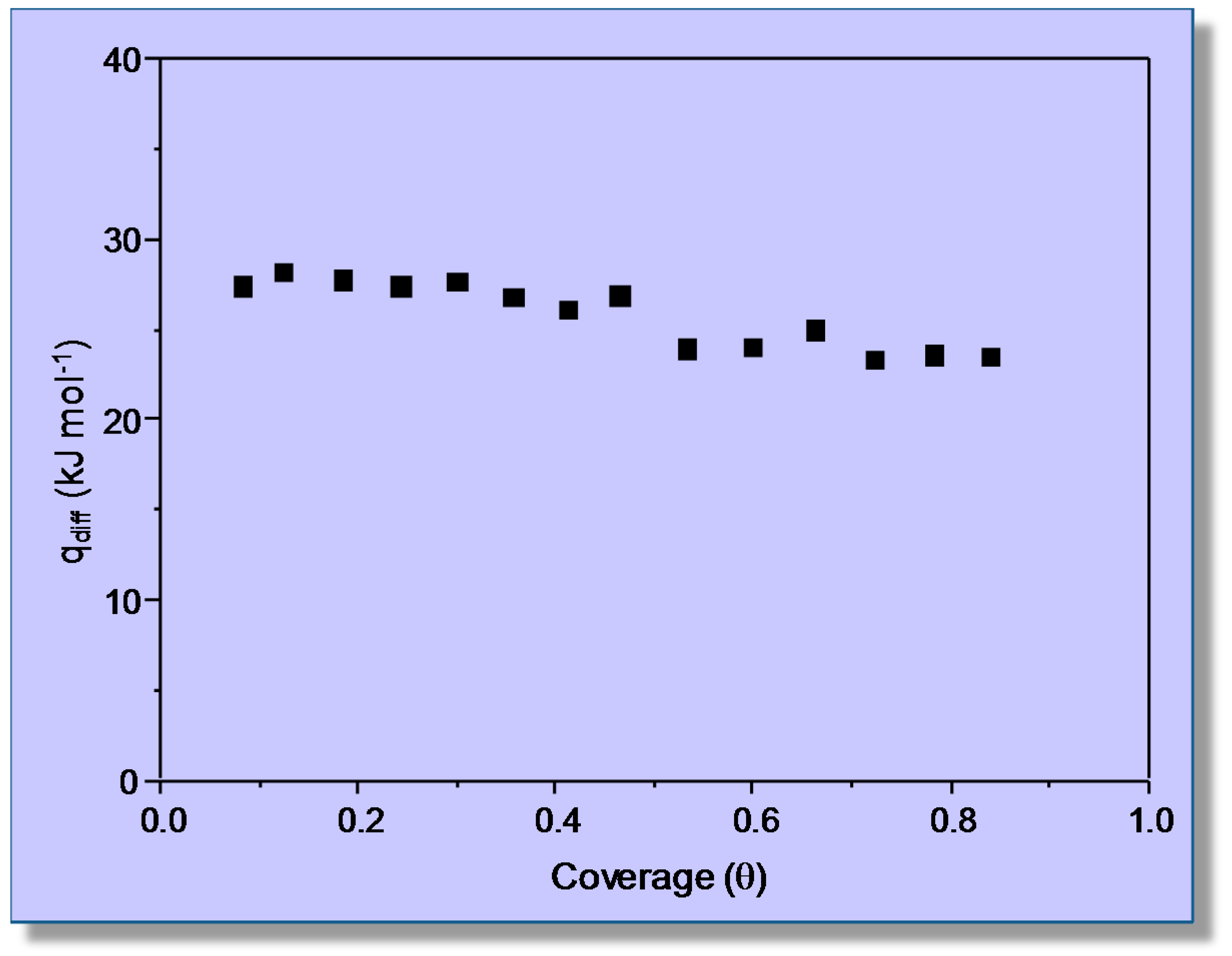
3.1. Carbon Dioxide Adsorption in H-MCM-22
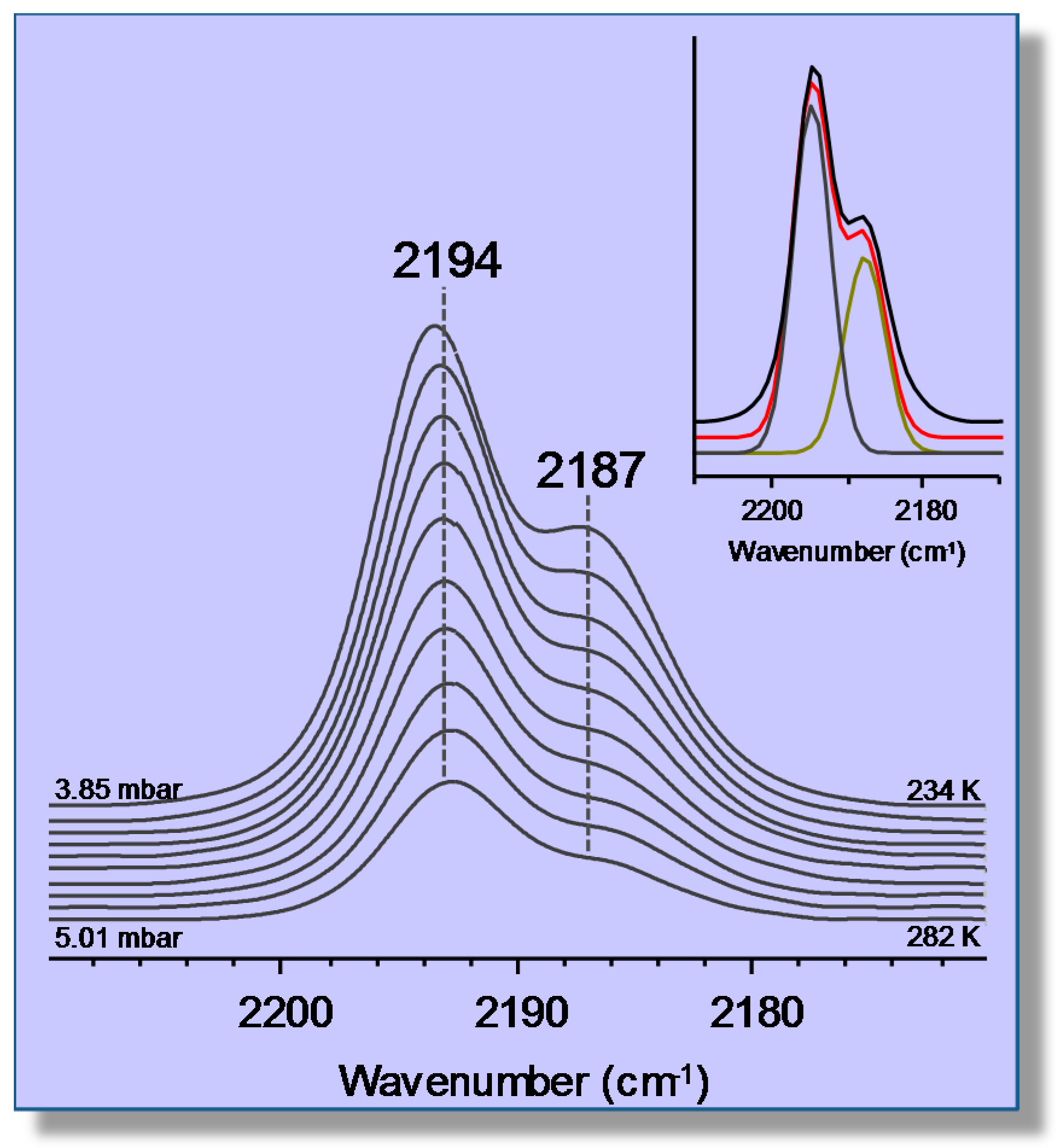
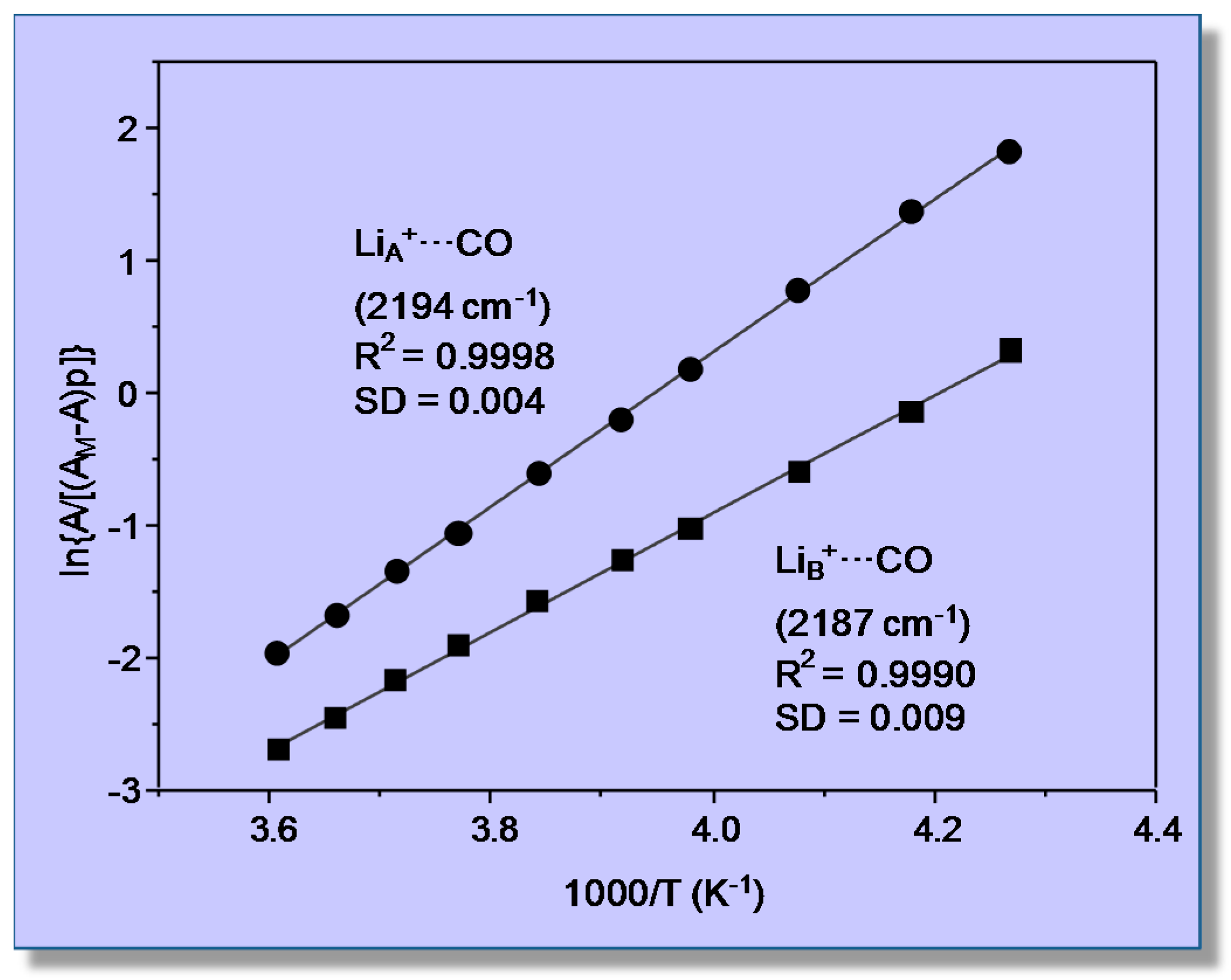
3.2. Carbon Monoxide Adsorption in the Alkaline Zeolite Li-ZSM-5
4. Adsorption Sites Involving More Than One Cation
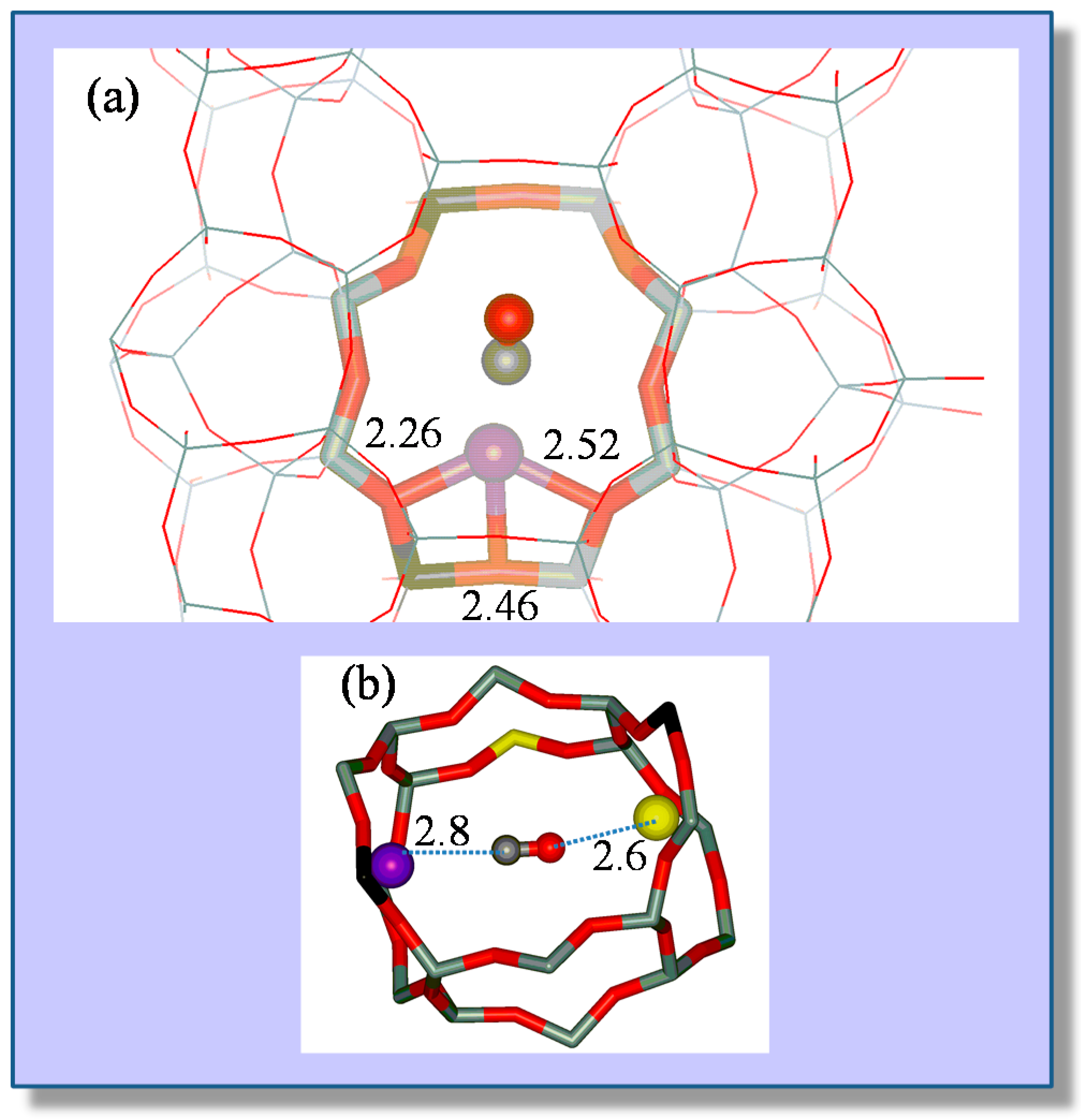
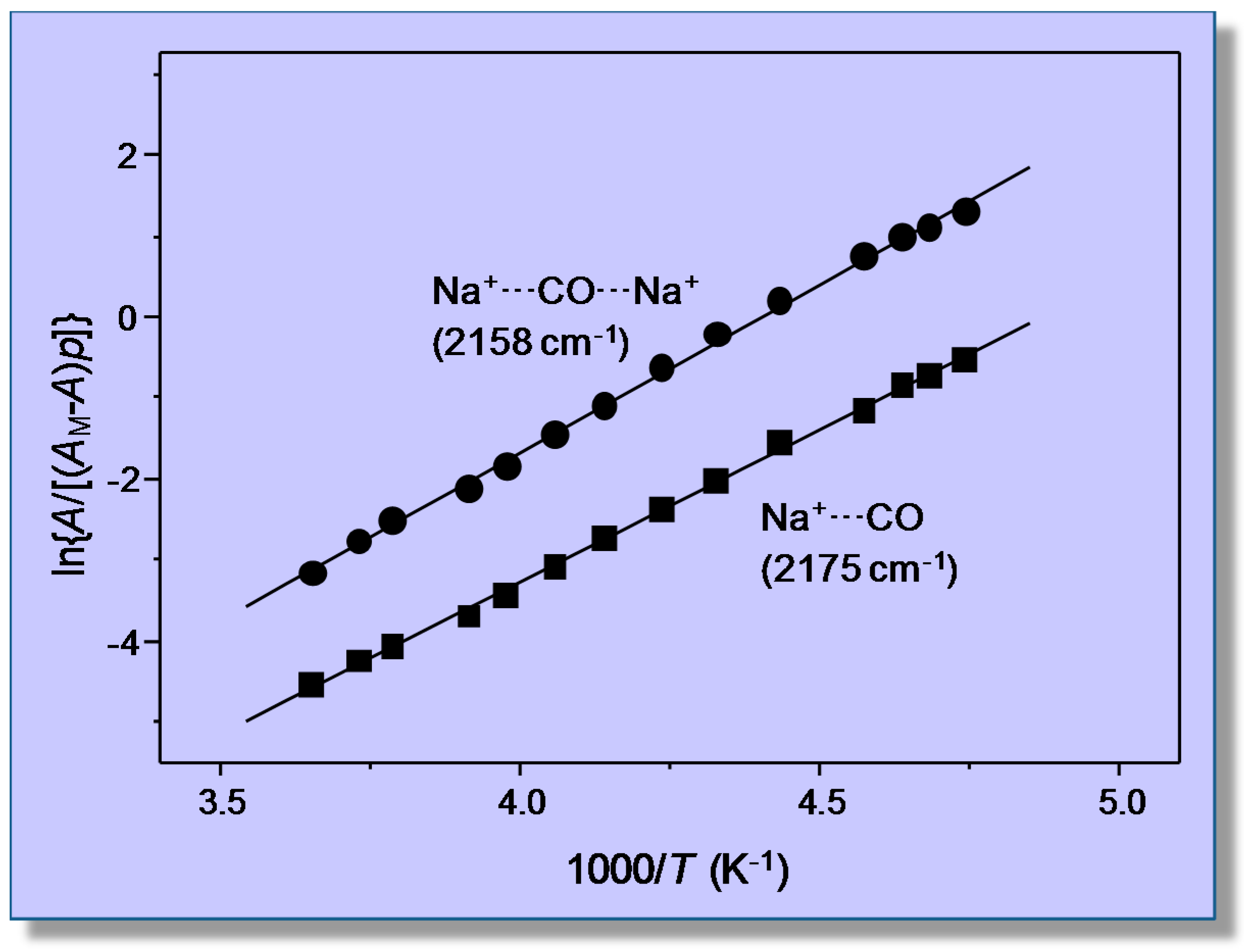
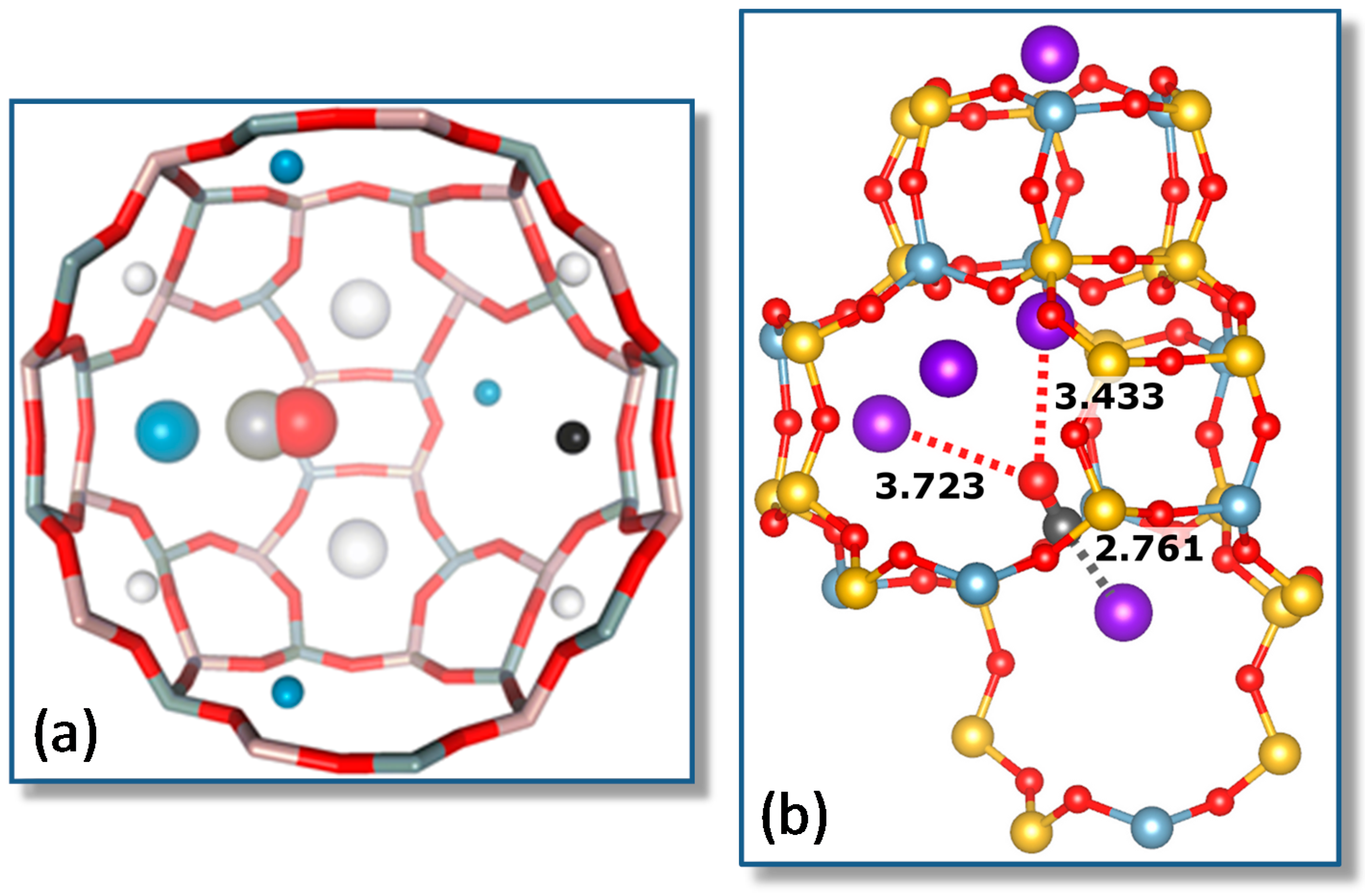
4.1. Carbon Monoxide Adsorption on Na-FER
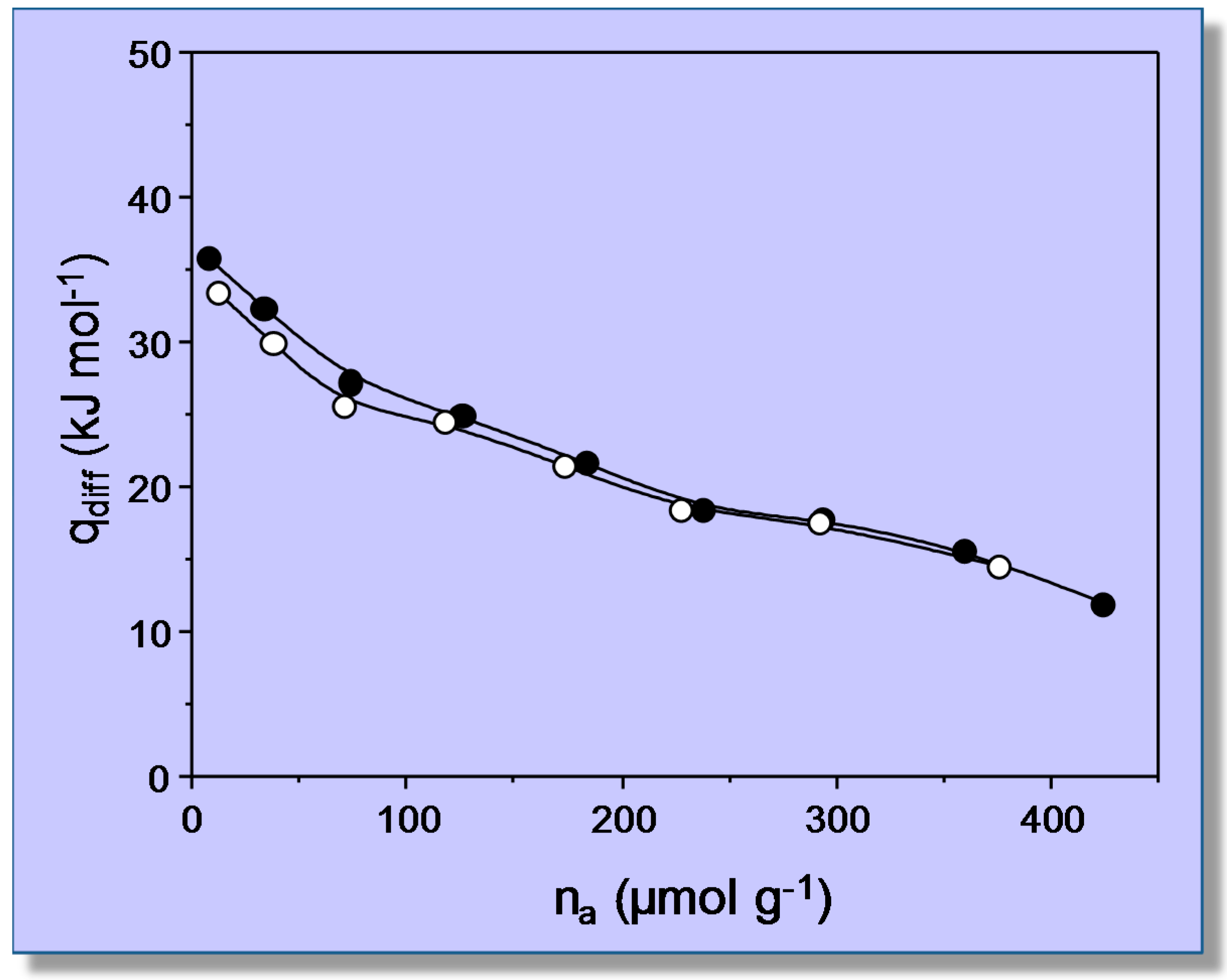
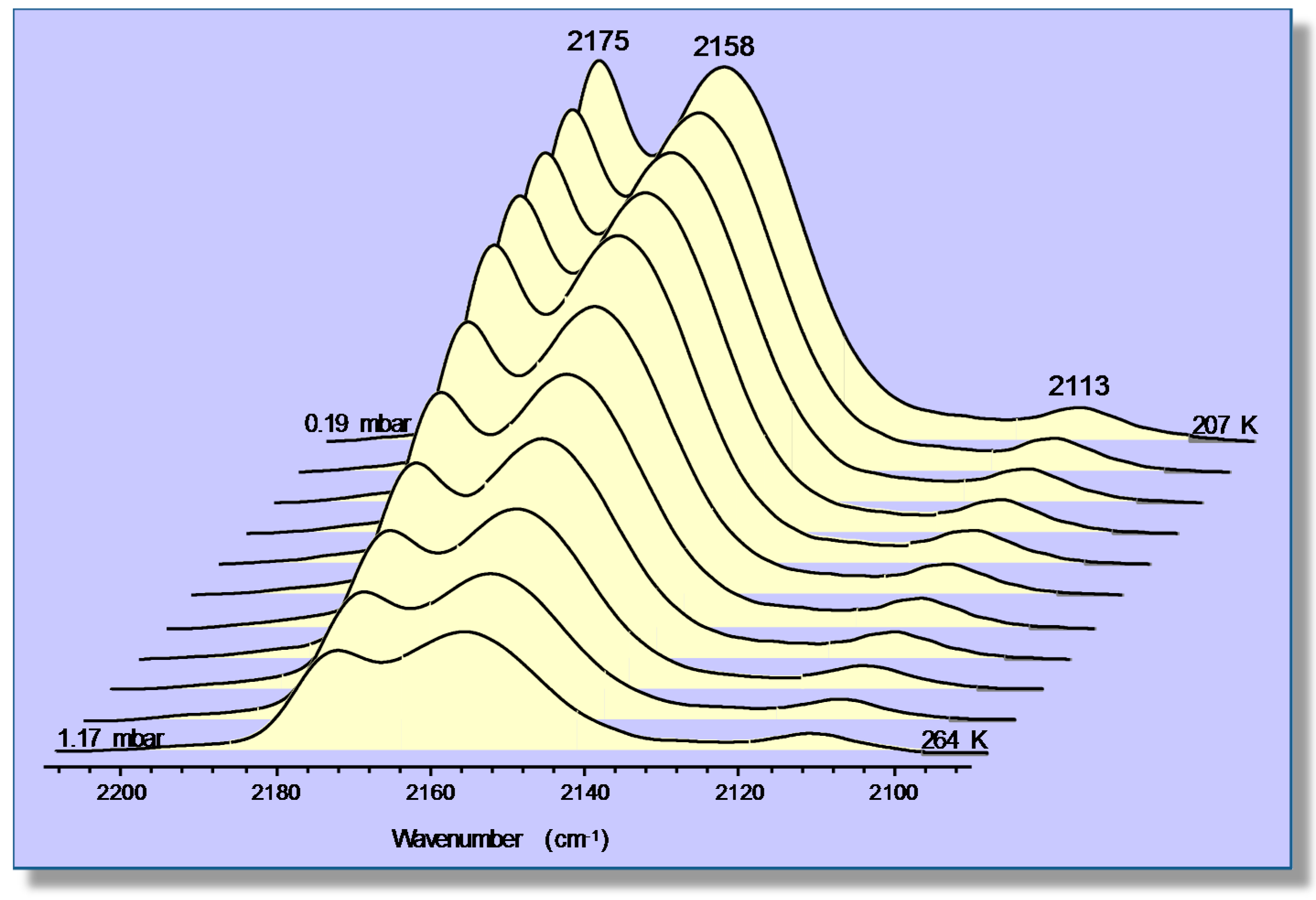
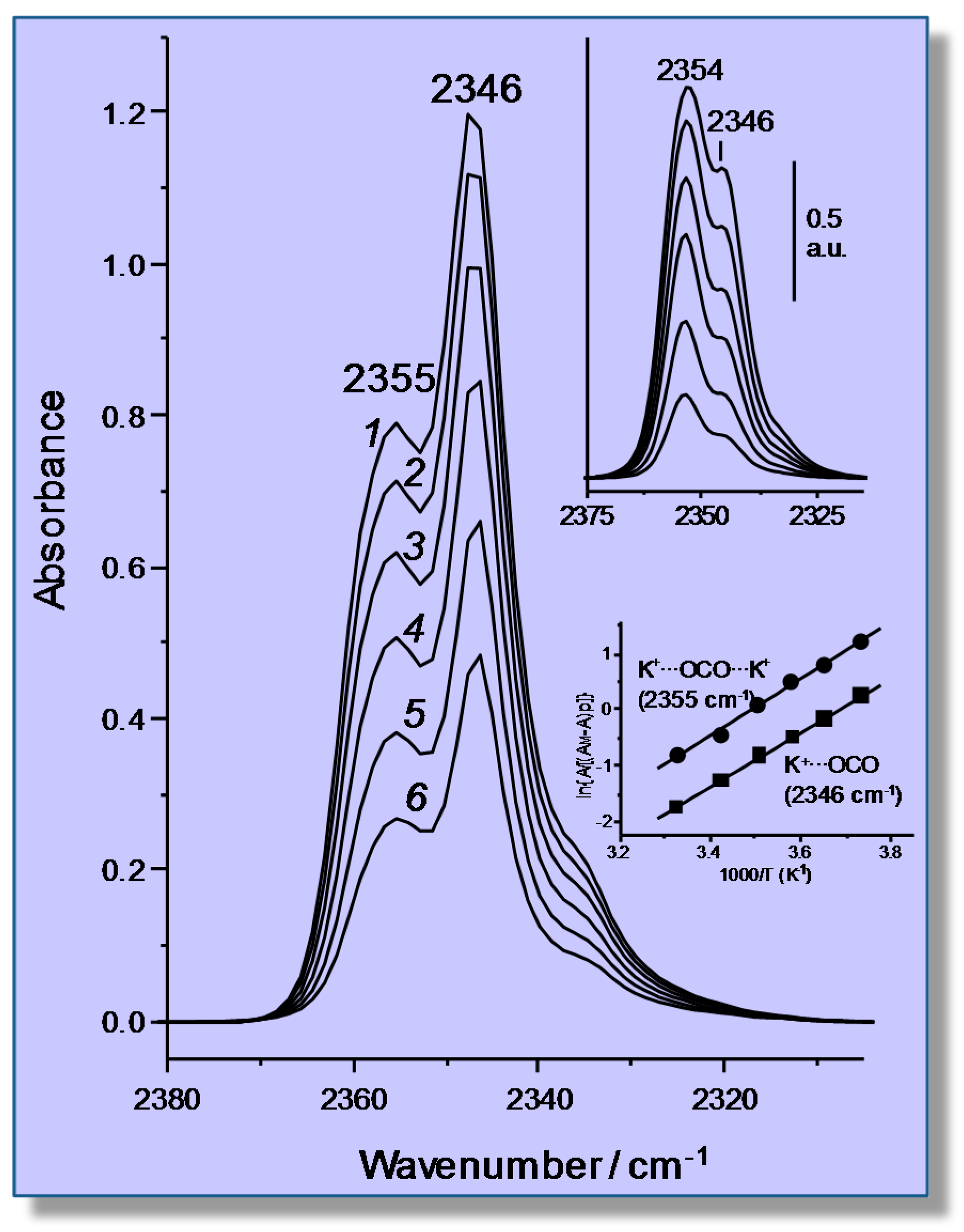
4.2. Carbon Dioxide Adsorption in K-FER
5. Summary and Conclusions
Author Contributions
Conflicts of Interest
References
- Sumida, K.; Rogow, D.L.; Mason, J.A.; McDonald, T.M.; Bloch, E.D.; Herm, Z.R.; Bae, T.H.; Long, J.R. Carbon dioxide capture in metal-organic frameworks. Chem. Rev. 2012, 112, 724–781. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.A.; Luo, J.Z.; Zhong, Z.Y.; Borgna, A. CO2 capture by solid adsorbents and their applications: Current status and new trends. Energy Environ. Sci. 2011, 4, 42–55. [Google Scholar] [CrossRef]
- Yazaydin, A.O.; Snurr, R.Q.; Park, T.H.; Koh, K.; Liu, J.; LeVan, M.D.; Benin, A.I.; Jakubeczak, P.; Lanuza, M.; Galloway, D.B.; et al. Screening metal-organic frameworks for carbon dioxide capture from flue gas using a combined experimental and modeling approach. J. Am. Chem. Soc. 2009, 131, 18198–18199. [Google Scholar] [CrossRef] [PubMed]
- Dietzel, P.D.C.; Johnsen, R.E.; Fjellvaj, H.; Bordiga, S.; Groppo, E.; Chavan, S.; Blom, R. Adsorption properties and structure of CO2 adsorbed on open coordination sites of metal-organic framework Ni2(dhtp) from gas adsorption, IR spectroscopy and X-ray diffraction. Chem. Commun. 2008, 7, 5125–5127. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Alvarez, P.; Calero, S. Highly selective zeolite topologies for flue gas separation. Chem. Eur. J. 2016, 22, 18705–18708. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.A.; Sumida, K.; Herm, Z.R.; Krishna, R.; Long, J.R. Evaluating metal-organic frameworks for post-combustion carbon dioxide capture via temperature swing adsorption. Energy Environ. Sci. 2011, 4, 3030–3040. [Google Scholar] [CrossRef]
- Huck, J.M.; Li-Chian, L.; Berger, A.H.; Shahrak, M.N.; Martin, R.L.; Bhown, A.S.; Haranczyk, M.; Reuter, K.; Smit, B. Evaluating different classes of porous materials for carbon capture. Energy Environ. Sci. 2014, 7, 4132–4146. [Google Scholar] [CrossRef]
- Bae, T.H.; Hudson, M.R.; Mason, J.A.; Queen, W.L.; Dutton, J.J.; Sumida, K.; Micklash, K.J.; Kaye, S.S.; Brown, C.M.; Long, J.R. Evaluation of cation-exchanged zeolite adsorbents for post-combustion carbon dioxide capture. Energy Environ. Sci. 2013, 6, 128–138. [Google Scholar] [CrossRef]
- Fischer, M.; Bell, R.G. Influence of zeolite topology on CO2/N2 separation behavior: Force-field simulations using a DFT-derived charge model. J. Phys. Chem. C 2012, 116, 26449–26463. [Google Scholar] [CrossRef]
- Grajciar, L.; Cejka, J.; Zukal, A.; Arean, C.O.; Palomino, G.T.; Nachtigall, P. Controlling the adsorption enthalpy of CO2 in zeolites by framework topology and composition. ChemSusChem 2012, 5, 2011–2022. [Google Scholar] [CrossRef] [PubMed]
- Thang, H.V.; Grajciar, L.; Nachtigall, P.; Bludsky, O.; Arean, C.O.; Frydova, E.; Bulanek, R. Adsorption of CO2 in FAU zeolites: Effect of zeolite composition. Catal. Today 2014, 227, 50–56. [Google Scholar] [CrossRef]
- Pham, T.D.; Hudson, M.R.; Brown, C.M.; Lobo, R.F. On the structure-property relationships of cation-exchanged ZK-5 zeolites for CO2 adsorption. ChemSusChem 2017, 10, 946–957. [Google Scholar] [CrossRef] [PubMed]
- Rui, J.; Liu, F.; Wang, R.; Lu, Y.; Yang, X. Adsorptive desulfurization of model gasoline by using different Zn sources exchanged NaY zeolites. Molecules 2017, 22, 305. [Google Scholar] [CrossRef] [PubMed]
- Getman, R.B.; Bae, Y.S.; Wilmer, C.E.; Snurr, R.Q. Review and analysis of molecular simulations of methane, hydrogen and acetylene storage in metal-organic frameworks. Chem. Rev. 2012, 112, 703–723. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.A.; Veenstra, M.; Long, J.R. Evaluating metal-organic frameworks for natural gas storage. Chem. Sci. 2014, 5, 32–51. [Google Scholar] [CrossRef]
- Chen, B.; Xiang, S.; Qian, G. Metal-organic frameworks with functional pores for recognition of small molecules. Acc. Chem. Res. 2010, 43, 1115–1124. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Yang, S.; Blake, A.J.; Schröder, M. Studies on metal-organic frameworks with isophthalate linkers for hydrogen storage. Acc. Chem. Res. 2013, 47, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Simon, C.M.; Kim, J.; Lin, L.C.; Martin, R.L.; Haranczyk, M.; Smit, B. Optimizing nanoporous materials for gas storage. Phys. Chem. Chem. Phys. 2014, 16, 5499–5513. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, J.Y.; Yeh, Y.H.; Gorte, R.J.; Rangarajan, S.; Mavrikakis, M. An adsorption study of CH4 on ZSM-5, MOR, and ZSM-12 zeolites. J. Phys. Chem. C 2015, 119, 28970–28979. [Google Scholar] [CrossRef]
- Kumar, K.V.; Preus, K.; Titirici, M.M.; Rodríguez-Reinoso, F. Nanoporous materials for onboard storage of natural gas. Chem. Rev. 2017, 117, 1796−1825. [Google Scholar] [CrossRef] [PubMed]
- Vogt, E.T.C.; Weckhuysen, B.M. Fluid catalytic cracking: Recent developments on the grand old lady of zeolite catalysis. Chem. Soc. Rev. 2015, 44, 7342–7370. [Google Scholar] [CrossRef] [PubMed]
- Olsbye, U.; Bjørgen, M.; Svelle, S.; Lillerud, K.P.; Kolbe, S. Mechanistic insight into the methanol-to-hydrocarbons reaction. Catal. Today 2005, 106, 108–111. [Google Scholar] [CrossRef]
- Tian, P.; Wei, Y.; Ye, M.; Liu, Z. Methanol to olefins MTO.: From fundamentals to commercialization. ACS Catal. 2015, 5, 1922–1938. [Google Scholar] [CrossRef]
- Losch, P.; Pinar, A.B.; Willinger, M.G.; Soukup, K.; Chavan, S.; Vincent, B.; Pale, P.; Louis, B. H-ZSM-5 zeolite model crystals: Structure-diffusion-activity relationship in methanol-to-olefins catalysis. J. Catal. 2017, 345, 11–23. [Google Scholar] [CrossRef]
- Corma, A. State of the art and future challenges of zeolites as catalysts. J. Catal. 2003, 216, 298–312. [Google Scholar] [CrossRef]
- Eimer, G.; Girola, P.; Tomas, L.; Pirella, L.B.; Anunziata, O.A. Catalytic activity of MEL zeolites modified with metallic couples for the conversion of ethane. Molecules 2000, 5, 560–561. [Google Scholar] [CrossRef]
- Okomura, K.; Tomiyama, T.; Moriyama, S.; Nakamici, A.; Niwa, M. Enhancement in the catalytic activity of Pd/USY in the Heck reaction induced by H2 bubbling. Molecules 2011, 16, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Jwa, E.; Lee, S.B.; Lee, H.W.; Mok, Y.S. Plasma-assisted catalytic methanation of CO and CO2 over Ni-zeolite catalysts. Fuel Process. Technol. 2013, 108, 89–93. [Google Scholar] [CrossRef]
- Westermann, A.; Azambre, B.; Bacariza, M.C.; Graça, I.; Ribeiro, M.F.; Lopes, J.M.; Henriques, C. Insight into CO2 methanation mechanism over NiUSY zeolites: An operando IR study. Appl. Catal. B 2015, 174, 120–125. [Google Scholar] [CrossRef]
- Garrone, E.; Arean, C.O. Variable temperature infrared spectroscopy: A convenient tool for studying the thermodynamics of weak solid-gas interactions. Chem. Soc. Rev. 2005, 34, 846–857. [Google Scholar] [CrossRef] [PubMed]
- Bond, G.C.; Keane, M.A.; Kral, H.; Lercher, J.A. Compensation phenomena in heterogeneous catalysis. Catal. Rev. Sci. Eng. 2000, 42, 223–283. [Google Scholar] [CrossRef]
- Arean, C.O.; Palomino, G.T.; Zecchina, A.; Spoto, G.; Bordiga, S.; Roy, P. Cation-carbon stretching vibration of adducts formed upon CO adsorption on alkaline zeolites. Phys. Chem. Chem. Phys. 1999, 1, 4139–4140. [Google Scholar] [CrossRef]
- Arean, C.O.; Manoilova, O.V.; Tsyganenko, A.A.; Palomino, G.T.; Mentruit, M.P.; Geobaldo, F.; Garrone, E. Thermodynamics of hydrogen bonding between CO and the supercage Brønsted acid sites of the H-Y zeolite—studies from variable temperature IR spectrometry. Eur. J. Inorg. Chem. 2001, 1739–1743. [Google Scholar]
- Tsyganenko, A.A.; Storozhev, P.Y.; Arean, C.O. Infraed spectroscopic studies on the binding isomerism of adsorbed molecules. Kinet. Catal. 2004, 45, 530–540. [Google Scholar] [CrossRef]
- Arean, C.O.; Delgado, M.R.; Bulánek, R.; Frolich, K. Combined microcalorimetric and IR spectroscopic study on carbon dioxide adsorption in H-MCM-22. Appl. Surf. Sci. 2014, 316, 532–536. [Google Scholar] [CrossRef]
- Pulido, A.; Delgado, M.R.; Bludsky, O.; Rubes, M.; Nachtigall, P.; Arean, C.O. Combined DFT/CC and IR spectroscopic studies on carbon dioxide adsorption on the zeolite H-FER. Energy Environ. Sci. 2009, 2, 1187–1195. [Google Scholar] [CrossRef]
- Arean, C.O.; Delgado, M.R.; Nachtigall, P.; Thang, H.V.; Rubes, M.; Bulánek, R.; Chlubná-Eliásova, P. Measuring the Brønsted acid strength of zeolites—Does it correlate with the O-H frequency shift probed by a weak base? Phys. Chem. Chem. Phys. 2014, 16, 10129–10141. [Google Scholar] [CrossRef] [PubMed]
- Onida, B.; Geobaldo, F.; Testa, F.; Crea, F.; Garrone, E. FTIR investigation of the interaction at 77 K of diatomic molecular probes on MCM-22 zeolite. Micropor. Mesopor. Mater. 1999, 30, 119–127. [Google Scholar] [CrossRef]
- Bonelli, B.; Garrone, E.; Fubini, B.; Onida, B.; Delgado, M.R.; Arean, C.O. Two distinguishable lithium sites in the zeolite Li-ZSM-5 as revealed by adsorption of CO: An infrared spectroscopic and thermodynamic characterization. Phys. Chem. Chem. Phys. 2003, 5, 2900–2905. [Google Scholar] [CrossRef]
- Arean, C.O.; Manoilova, O.V.; Delgado, M.R.; Tsyganenko, A.A.; Garrone, E. Formation of several types of coordination complexes upon CO adsorption on the zeolite Li-ZSM-5. Phys. Chem. Chem. Phys. 2001, 3, 4187–4188. [Google Scholar] [CrossRef]
- Nachtigall, P.; Frolich, K.; Drobná, H.; Bludsky, O.; Nachtigallova, D.; Bulanek, R. FTIR study of CO interactions with Li+ ions in micro- and mesoporous matrices: Coordination and localization of Li+ ions. J. Phys. Chem. C 2007, 111, 11353–11362. [Google Scholar] [CrossRef]
- Sillar, K.; Burk, P. Adsorption of carbon monoxide on Li-ZSM-5: Theoretical study of complexation of Li+ cation with two CO molecules. Phys. Chem. Chem. Phys. 2007, 9, 824–827. [Google Scholar] [CrossRef] [PubMed]
- Thang, H.V.; Nachtigall, P.; Grajciar, L. The Lewis acidity of three- and two-dimensional zeolites: The effect of framework topology. Catal. Today. 2017, in press. [Google Scholar] [CrossRef]
- Nachtigall, P.; Delgado, M.R.; Nachtigallova, D.; Arean, C.O. The nature of cationic adsorption sites in alkaline zeolites—single, dual and multiple cation sites. Phys. Chem. Chem. Phys. 2012, 14, 1552–1569. [Google Scholar] [CrossRef] [PubMed]
- Garrone, E.; Bulanek, R.; Frolich, K.; Arean, C.O.; Delgado, M.R.; Palomino, G.T.; Nachtigallova, D.; Nachtigall, P. Single and dual cation sites in zeolites: Theoretical calculations and FTIR spectroscopic studies on CO adsorption in K-FER. J. Phys. Chem. B 2006, 110, 22542–22550. [Google Scholar] [CrossRef] [PubMed]
- Zecchina, A.; Arean, C.O. Diatomic molecular probes for mid-IR studies of zeolites. Chem. Soc. Rev. 1996, 25, 187–197. [Google Scholar] [CrossRef]
- Garrone, E.; Fubini, B.; Bonelli, B.; Onida, B.; Arean, C.O. Thermodynamics of CO adsorption on the zeolite Na-ZSM-5: A combined microcalorimetric and FTIR spectroscopic study. Phys. Chem. Chem. Phys. 1999, 1, 513–518. [Google Scholar] [CrossRef]
- Arean, C.O. Zeolites and intrazeolite chemistry: Insights from infrared spectroscopy. Comments Inorg. Chem. 2000, 22, 241–273. [Google Scholar] [CrossRef]
- Hadjiivanov, K.I.; Vayssilov, G.N. Characterization of oxide surfaces and zeolites by carbon monoxide as an IR probe molecule. Adv. Catal. 2002, 47, 307–511. [Google Scholar]
- Scarano, D.; Bertarione, S.; Spoto, G.; Zecchina, A.; Arean, C.O. FTIR spectroscopy of hydrogen, carbon monoxide and methane adsorbed and co-adsorbed on zinc oxide. Thin Solid Films 2001, 400, 50–55. [Google Scholar] [CrossRef]
- Lamberti, C.; Zecchina, A.; Groppo, E.; Bordiga, S. Probing the surfaces of heterogeneous catalysts by in situ IR spectroscopy. Chem. Soc. Rev. 2010, 39, 4951–5001. [Google Scholar] [CrossRef] [PubMed]
- Katoh, M.; Yamazaki, T.; Ozawa, S. IR spectroscopic study of adsorption of binary gases over ion-exchanged ZSM-5 zeolites. J. Colloid Interface Sci. 1998, 203, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Manoilova, O.V.; Mentruit, M.P.; Palomino, G.T.; Tsyganenko, A.A.; Arean, C.O. Variable-temperature infrared spectrometry of carbon monoxide adsorbed on the zeolite K-ZSM-5. Vib. Spectrosc. 2001, 26, 107–111. [Google Scholar] [CrossRef]
- Salla, I.; Montanari, T.; Salagre, P.; Cesteros, Y.; Busca, G. A reexamination of the adsorption of CO and nitriles on alkali-metal mordenites: Characterization of multiple interactions. Phys. Chem. Chem. Phys. 2005, 7, 2526–2532. [Google Scholar] [CrossRef] [PubMed]
- Nachtigall, P.; Delgado, M.R.; Frolich, K.; Bulanek, R.; Palomino, G.T.; Bauça, C.L.; Arean, C.O. Periodic density functional and FTIR spectroscopic studies on CO adsorption on the zeolite Na-FER. Microporous Mesoporous Mater. 2007, 106, 162–173. [Google Scholar] [CrossRef]
- Arean, C.O.; Tsyganenko, A.A.; Platero, E.E.; Garrone, E.; Zecchina, A. Two coordination modes of CO in zeolites: A temperature dependent equilibrium. Angew. Chem. Int. Ed. 1998, 37, 3161–3163. [Google Scholar] [CrossRef]
- Arean, C.O.; Palomino, G.T.; Tsyganenko, A.A.; Garrone, E. Quantum chemical and FTIR spectroscopic studies on the linkage isomerism of carbon monoxide in alkali-metal-exchanged zeolites: A review of current research. Int. J. Mol. Sci. 2002, 3, 764–776. [Google Scholar] [CrossRef]
- Arean, C.O.; Delgado, M.R.; Frolich, K.; Bulánek, R.; Pulido, A.; Bibiloni, G.F.; Nachtigall, P. Computational and Fourier Transform infrared spectroscopic studies on carbon monoxide adsorption on the zeolites Na-ZSM-5 and K-ZSM-5: Evidence of dual-cation sites. J. Phys. Chem. C 2008, 112, 4658–4666. [Google Scholar] [CrossRef]
- Arean, C.O.; Delgado, M.R.; Bauça, C.L.; Vrbka, L.; Nachtigall, P. Carbon monoxide adsorption on low-silica zeolites: From single to dual and to multiple cation sites. Phys. Chem. Chem. Phys. 2007, 9, 4657–4661. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.; Delgado, M.R.; Arean, C.O.; Duran, C.O. CO adsorption complexes in zeolites: How does the inclusion of dispersion interactions affect predictions made from DFT calculations? The case of Na-CHA. Theor. Chem. Acc. 2015, 134, 91. [Google Scholar] [CrossRef]
- Arean, C.O.; Delgado, M.R.; Bibiloni, G.F.; Bludsky, O.; Nachtigall, P. Variable-temperature IR spectroscopic and theoretical studies on CO2 adsorbed in zeolite K-FER. ChemPhysChem 2011, 12, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Pulido, A.; Nachtigall, P.; Zukal, A.; Dominguez, I.; Cejka, J. Adsorption of CO2 on sodium-exchanged ferrierites: The bridged CO2 complexes formed between two extraframework cations. J. Phys. Chem. C 2009, 113, 2928–2935. [Google Scholar] [CrossRef]
- Itadani, A.; Torigoe, H.; Yumura, T.; Ohkubo, T.; Kobayashi, H.; Kuroda, Y. Dual-copper catalytic site formed in CuMFI zeolite makes effective activation of ethane possible even at room temperature. J. Phys. Chem. C 2012, 116, 10680–10691. [Google Scholar] [CrossRef]
- Itadani, A.; Sogawa, Y.; Oda, A.; Torigoe, H.; Ohkubo, T.; Kuroda, Y. Further evidence for the existence of a dual-Cu+ site in MFI working as the efficient site for C2H6 adsorption at room temperature. Langmuir 2013, 29, 9727–9733. [Google Scholar] [CrossRef] [PubMed]
- Hakemian, A.S.; Rosenweig, A.C. The biochemistry of methane oxidation. Annu. Rev. Biochem. 2007, 76, 223–241. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, R.; Smith, S.M.; Rawat, S.; Yatsunyk, L.A.; Stemmler, T.L.; Rosenzweig, A.C. Oxidation of methane by a biological dicopper center. Nature 2010, 465, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, R.; Proshlyakov, Y.; Lipscom, J.D.; Proshlyakov, D.A. Structure of the key species in the enzymatic oxidation of methane to methanol. Nature 2015, 518, 431–4343. [Google Scholar] [CrossRef] [PubMed]
- Smeets, P.J.; Groothaert, M.H.; Schoonheydt, R.A. Cu based zeolites: A UV-Vis study of the active site in the selective methane oxidation at low temperatures. Catal. Today 2005, 110, 303–309. [Google Scholar] [CrossRef]
- Beznis, N.V.; Weckhuysen, B.M.; Bitter, J.H. Cu-ZSM-5 zeolites for the formation of methanol from methane and oxygen: Probing the active sites and spectator species. Catal. Lett. 2010, 138, 14–22. [Google Scholar] [CrossRef]
- Sheppard, T.; Hamill, C.D.; Goguet, A.; Rooney, D.W.; Thompson, J.M. A low temperature, isothermal gas-phase system for conversion of methane to methanol over Cu-ZSM-5. Chem. Commun. 2014, 50, 11053–11055. [Google Scholar] [CrossRef] [PubMed]
- Wulfers, M.J.; Teketel, S.; Ipek, B.; Lobo, R.F. Conversion of methane to methanol on copper-containing small-pore zeolites and zeotypes. Chem. Commun. 2015, 51, 4447–4450. [Google Scholar] [CrossRef] [PubMed]
- Woertink, J.S.; Smeets, P.J.; Groothaert, M.H.; Vance, M.A.; Sels, B.F.; Schoonheydt, R.A.; Solomon, E.I. A [Cu2O](2+) core in Cu-ZSM-5, the active site in the oxidation of methane to methanol. Proc. Natl. Acad. Sci. USA 2009, 106, 18908–18913. [Google Scholar] [CrossRef] [PubMed]
- Arvidsson, A.A.; Zhdanov, V.P.; Carlsson, P.A.; Grönbeck, H.; Hellman, A. Metal dimer sites in ZSM-5 zeolite for methane-to-methanol conversion from first-principles kinetic modelling: Is the [Cu-O-Cu]2+ motif relevant for Ni, Co, Fe, Ag, and Au? Catal. Sci. Technol. 2017, 7, 1470–1477. [Google Scholar] [CrossRef]
- Recchia, S.; Dossi, C.; Fusi, A.; Psaro, R.; Ugo, R.; Moretti, G. Irreversible dinitrogen adsorption on Cu-ZSM-5 catalysts and in situ IR identification of the NO decomposition sites. Chem. Commun. 1997, 19, 1909–1910. [Google Scholar] [CrossRef]
- Moretti, G.; Ferraris, G.; Fierro, G.; Lo Jacono, M.; Morpurgo, S.; Faticanti, M. Dimeric Cu(I) species in Cu-ZSM-5 catalysts: The active sites for the NO decomposition. J. Catal. 2005, 232, 476–487. [Google Scholar] [CrossRef]
- Morpurgo, S.; Moretti, G.; Bossa, M. A computational study on N2 adsorption in Cu-ZSM-5. Phys. Chem. Chem. Phys. 2007, 9, 417–424. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garrone, E.; Delgado, M.R.; Bonelli, B.; Arean, C.O. Probing Gas Adsorption in Zeolites by Variable-Temperature IR Spectroscopy: An Overview of Current Research. Molecules 2017, 22, 1557. https://doi.org/10.3390/molecules22091557
Garrone E, Delgado MR, Bonelli B, Arean CO. Probing Gas Adsorption in Zeolites by Variable-Temperature IR Spectroscopy: An Overview of Current Research. Molecules. 2017; 22(9):1557. https://doi.org/10.3390/molecules22091557
Chicago/Turabian StyleGarrone, Edoardo, Montserrat R. Delgado, Barbara Bonelli, and Carlos O. Arean. 2017. "Probing Gas Adsorption in Zeolites by Variable-Temperature IR Spectroscopy: An Overview of Current Research" Molecules 22, no. 9: 1557. https://doi.org/10.3390/molecules22091557




