Simultaneous Determination of Seven Phenolic Acids in Rat Plasma Using UHPLC-ESI-MS/MS after Oral Administration of Echinacea purpurea Extract
Abstract
:1. Introduction
2. Results
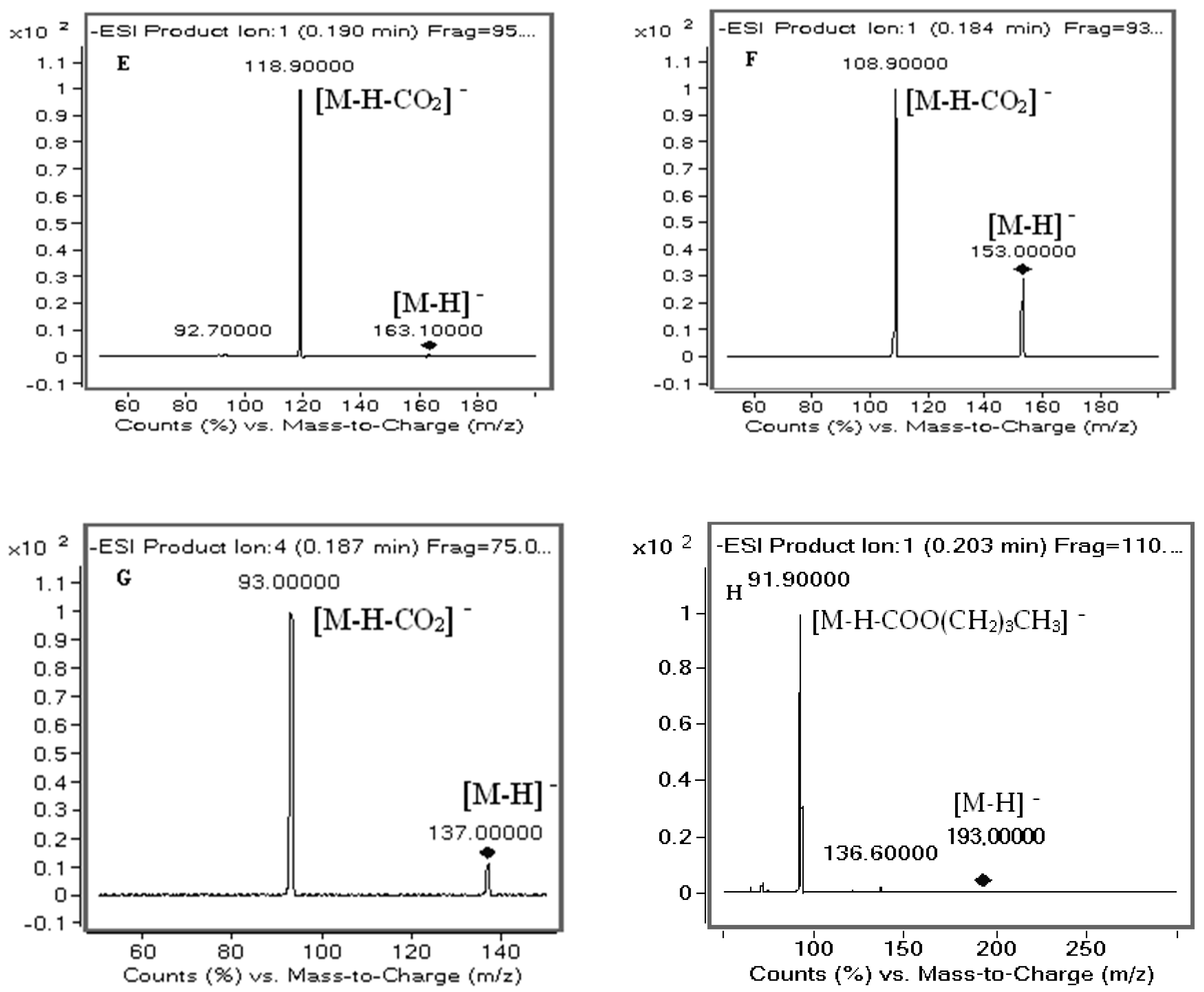
2.1. UHPLC-MS/MS Optimization
2.2. Method Validation
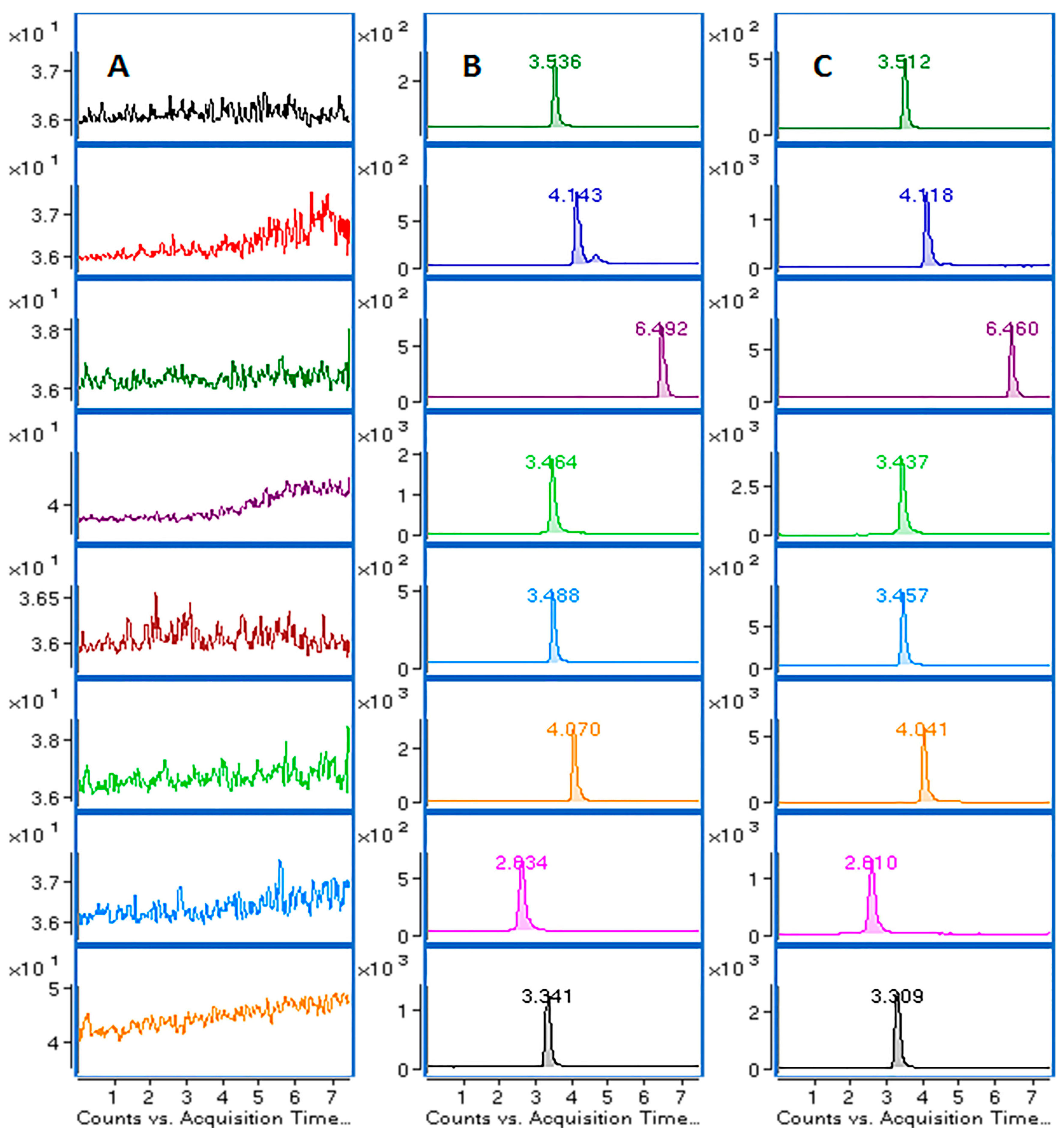
2.2.1. Selectivity
2.2.2. Linearity and Sensitivity
2.2.3. Accuracy and Precision
2.2.4. Extraction Recovery and Matrix Effect
2.2.5. Stability
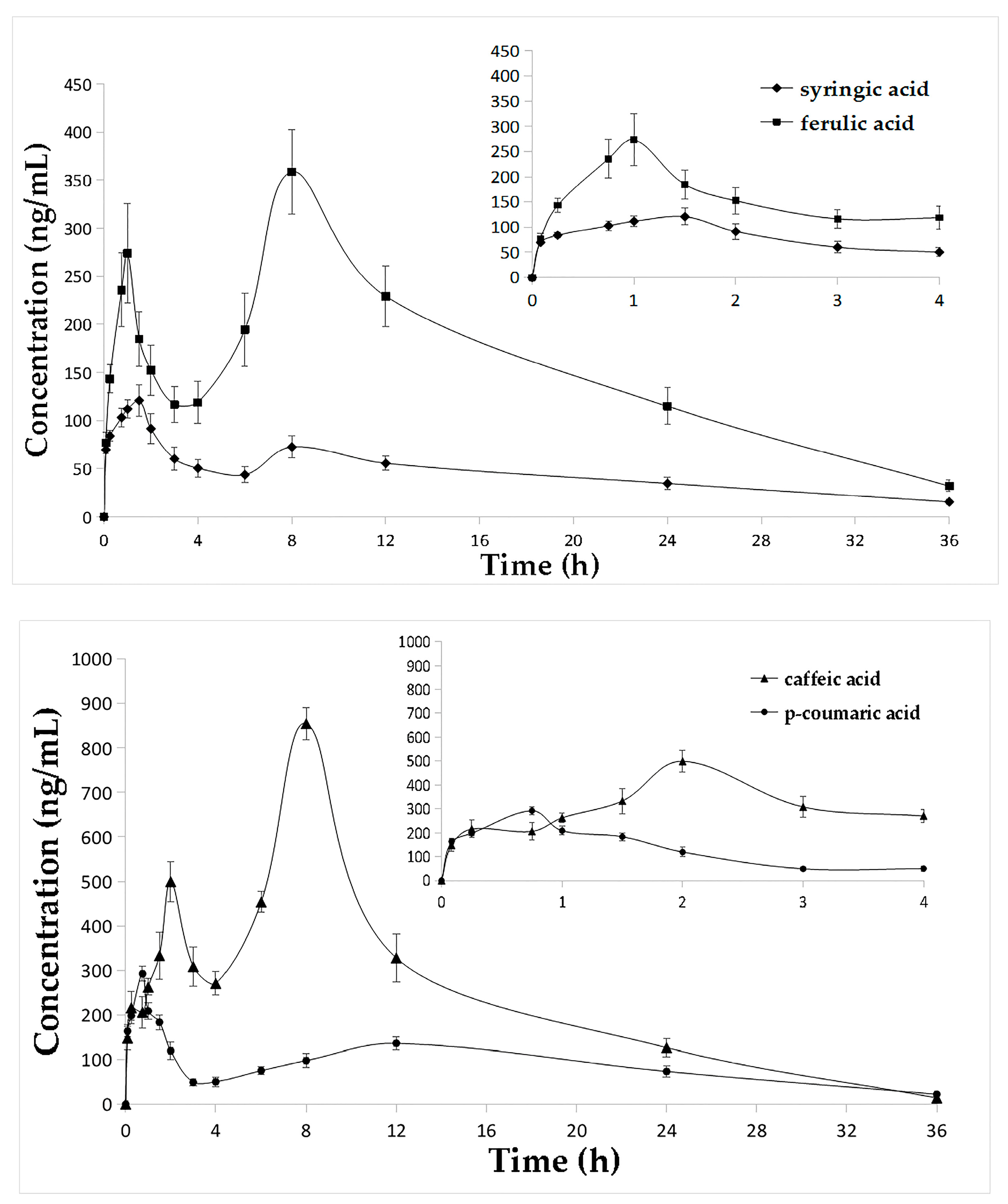
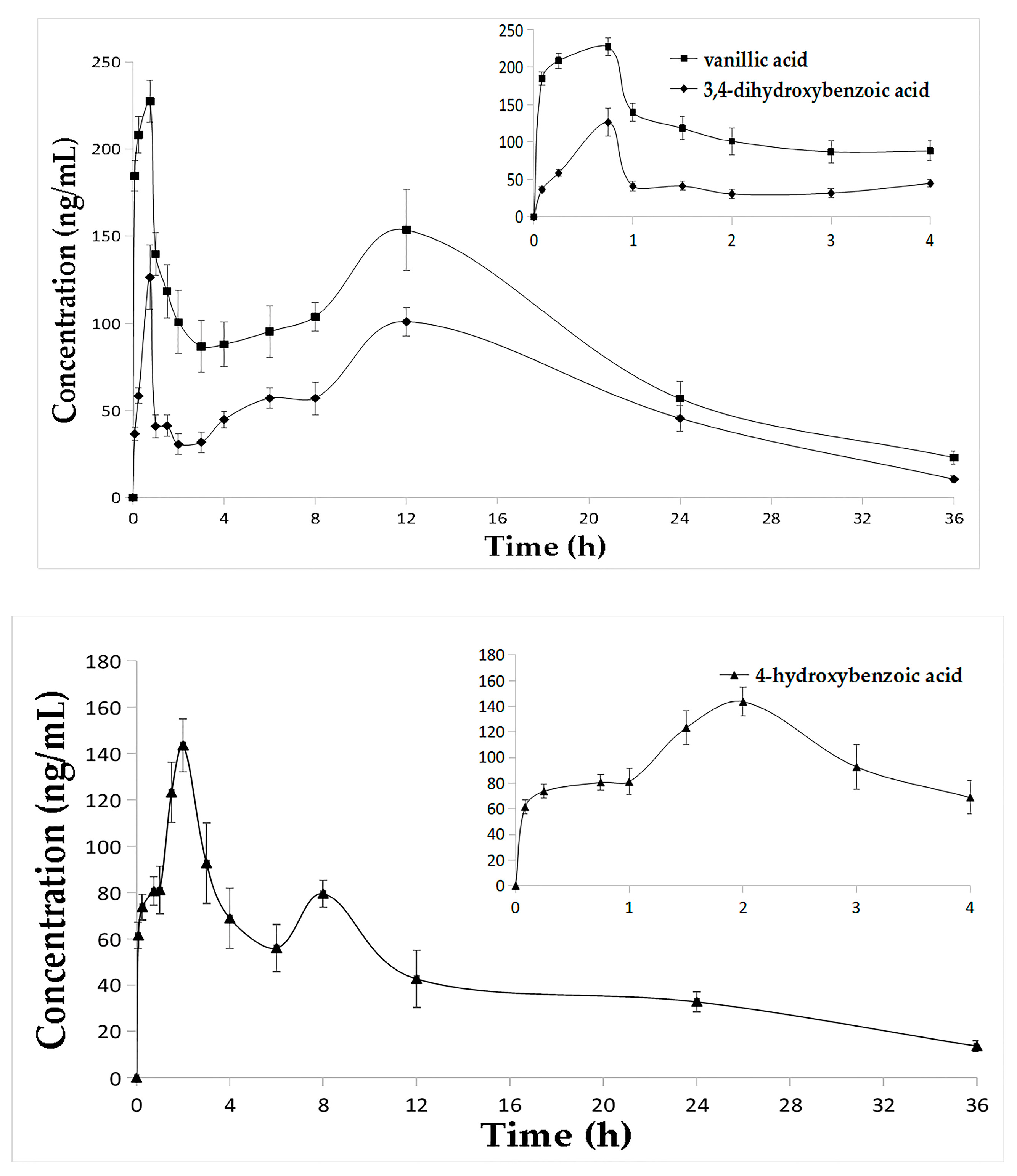
2.3. Pharmacokinetic Studies
3. Discussion
3.1. The Content of Phenolic Acids in Echinacea purpurea
3.2. Selection of Extraction Method
3.3. Selection of IS
3.4. Pharmacokinetic Studies
4. Materials and Methods
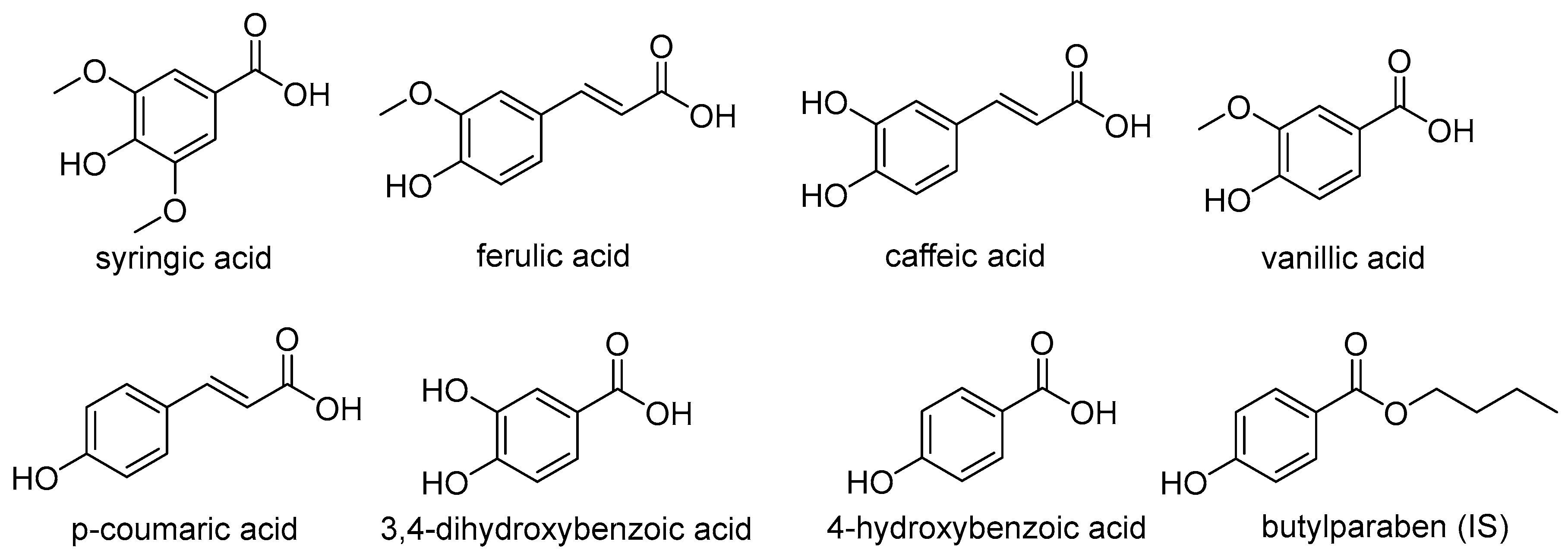
4.1. Materials and Reagents
4.2. Preparation of E. purpurea
4.3. Preparation of Standard Solutions and Quality Control Samples
4.4. Animal Experiments and Plasma Sample Extraction Procedure
4.5. UHPLC-ESI-MS/MS Conditions

4.6. Method Validations
4.6.1. Selectivity
4.6.2. Linearity and Sensitivity
4.6.3. Accuracy and Precision
4.6.4. Extraction Recovery and Matrix Effect
4.6.5. Stability
4.7. Pharmacokinetic Studies
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Han, L.N.; Zhou, F.Q. Research progress of introduced Echinacea purpurea. J. Archtcm 2012, 30, 1799–1802. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, K.; Wu, L.J. Advances in studies on Echinacea Moench. Chin. Tradit. Herb. Drugs 2001, 32, 852–855. [Google Scholar]
- Šutovská, M.; Capek, P.; Kazimierová, I.; Pappová, L.; Jošková, M.; Matulová, M.; Fraňová, S.; Pawlaczyk, I.; Gancarz, R. Echinacea complex—Chemical view and anti-asthmatic profile. J. Ethnopharmacol. 2015, 175, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.M. Echinacea purpurea: A proprietary extract of Echinacea purpurea in shown to be safe and effective in the prevention of the common cold. Holist. Nurs. Pract. 2016, 30, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.M.; Choi, K.M.; Lee, Y.S.; Kim, W.; Shin, K.O.; Oh, S.; Jung, J.C.; Lee, M.K.; Lee, Y.M.; Hong, J.T.; et al. Echinacea purpurea root extract enhances the adipocyte differentiation of 3T3-L1 cells. Arch. Pharm. Res. 2016, 37, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Barrett, B. Medicinal properties of Echinacea: A critical review. Phytomedicine 2003, 10, 66–86. [Google Scholar] [CrossRef] [PubMed]
- Barnes, J.; Anderson, L.A.; Gibbons, S.; Phillipson, J.D. Echinacea species (Echinacea angustifolia (DC.) Hell., Echinacea pallida (Nutt.) Nutt., Echinacea purpurea (L.) Moench): A review of their chemistry, pharmacology and clinical properties. J. Pharm. Pharmacol. 2005, 57, 929–954. [Google Scholar] [CrossRef] [PubMed]
- Awortwe, C.; Manda, V.K.; Avonto, C.; Khan, S.I.; Khan, I.A.; Walker, L.A.; Bouic, P.J.; Rosenkranz, B. Echinacea purpurea up-regulates CYP1A2, CYP3A4 and MDR1 gene expression by activation of pregnane X receptor pathway. Xenobiotica 2015, 45, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Goey, A.K.; Meijerman, I.; Rosing, H.; Burgers, J.A.; Mergui-Roelvink, M.; Keessen, M.; Marchetti, S.; Beijnen, J.H.; Schellens, J.H. The effect of Echinacea purpurea on the pharmacokinetics of docetaxel. Br. J. Clin. Pharmacol. 2013, 76, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Abdelmonem, M.; Kassem, S.H.; Gabr, H.; Shaheen, A.A.; Aboushousha, T. Avemar and Echinacea extracts enhance mobilization and homing of CD34(+) stem cells in rats with acute myocardial infarction. Stem Cell Res. Ther. 2015, 6, 172. [Google Scholar] [CrossRef] [PubMed]
- Woelkart, K.; Xu, W.; Pei, Y.; Makriyannis, A.; Picone, R.P.; Bauer, R. The endocannabinoid system as a target for alkamides from Echinacea angustifolia roots. Planta Med. 2005, 71, 701–705. [Google Scholar] [CrossRef] [PubMed]
- Hohmann, J.; Rédei, D.; Forgo, P.; Szabó, P.; Freund, T.F.; Haller, J.; Bojnik, E.; Benyhe, S. Alkamides and a neolignan from purpurea roots and the interaction of alkamides with G-protein-coupled cannabinoid receptors. Phytochemistry 2011, 72, 1848–1853. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.; Senol, F.S.; Gülpinar, A.R.; Kartal, M.; Sekeroglu, N.; Deveci, M.; Kan, Y.; Sener, B. Acetylcholinesterase inhibitory and antioxidant properties of Cyclotrichium niveum, Thymus praecox subsp. caucasicus var. caucasicus, Echinacea purpurea and E. pallida. Food Chem. Toxicol. 2009, 47, 1304–1310. [Google Scholar] [CrossRef] [PubMed]
- Kurkin, V.A.; Dubishchev, A.V.; Ezhkov, V.N.; Titova, I.N.; Avdeeva, E.V. Antidepressant activity of some phytopharmaceuticals and phenylpropanoids. Pharm. Chem. J. 2006, 40, 614–619. [Google Scholar] [CrossRef]
- Chen, Q.L.; Wang, L.; Feng, F. Chemical constituents from the aerial part of Echinacea purpurea. J. Chin. Med. Mater. 2013, 36, 739–743. [Google Scholar] [CrossRef]
- Sandasi, M.; Vermaak, I.; Chen, W.; Vijoen, A.M. Hyperspectral imaging and chemometric modeling of Echinacea—A novel approach in the quality control of herbal medicines. Molecules 2014, 19, 13104–13121. [Google Scholar] [CrossRef] [PubMed]
- Pomponio, R.; Gotti, R.; Hudaib, M.; Cavrini, V. Analysis of phenolic acids by micellar electrokinetic chromatography: Application to Echinacea purpurea plant extracts. J. Chromatogr. A 2002, 945, 239–247. [Google Scholar] [CrossRef]
- Luo, X.B.; Chen, B.; Yao, S.Z.; Zeng, J.G. Simultaneous analysis of caffeic acid derivatives and alkamides in roots and extracts of Echinacea purpurea by high-performance liquid chromatography photodiode array detection–electrospray Mass Spectrometry. J. Chromatogr. A 2003, 986, 73–81. [Google Scholar] [CrossRef]
- Hu, C.; Kitts, D.D. Studies on the antioxidant activity of Echinacea root extract. J. Agric. Food Chem. 2000, 48, 1466–1472. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Wang, Y.; Ding, H.; Liu, Z.H. On antioxidation activity of extracts of Echinacea purpurea. J. Hunan Agric. Univ. 2009, 35, 151–154. [Google Scholar] [CrossRef]
- Li, Z.Y.; Zhang, J.; Cheng, F.; Gui, B.Z.; Liu, J.H. Research progress of antioxidation and its mechanism in Echinacea. JTCVM 2016, 4, 72–73. [Google Scholar] [CrossRef]
- Chiou, S.Y.; Sung, J.M.; Huang, P.W.; Lin, S.D. Antioxidant, Antidiabetic, and Anti hypertensive Properties of Echinacea purpurea Flower Extract and Caffeic Acid Derivatives Using In Vitro Models. J. Med. Food 2017, 20, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Scagel, C.F. Chicoric acid: Chemistry, distribution, and production. Front. Chem. 2013, 1, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.N.; Chan, M.; Paley, L.; Betz, J.M. Determination of major phenolic compounds in Echinacea spp. Raw materials and finished products by high-performance liquid chromatography with ultraviolet detection: Single laboratory validation matrix extension. J. AOAC Int. 2011, 94, 1400–1410. [Google Scholar] [CrossRef] [PubMed]
- Ramasahayam, S.; Baraka, H.N.; Abdel Bar, F.M.; Abuasal, B.S.; Widrlechner, M.P.; Sayed, K.A.; Meyer, S.A. Effects of Chemically Characterized Fractions from Aerial Parts of Echinacea purpurea and E. angustifolia on Myelopoiesis in Rats. Planta Med. 2011, 77, 1883–1889. [Google Scholar] [CrossRef] [PubMed]
- Han, L.N.; Sun, J.Y.; Guo, Q.M. Dynamic determination of active ingredients in introduced Echinacea purpurea. WST 2014, 16, 1858–1860. [Google Scholar] [CrossRef]
- Gan, C.L.; Liu, L.; Du, Y.; Wang, L.Q.; Gao, M.J.; Wu, L.J.; Yang, C.J. Simutaneous determination and pharmacokinetic study of four phenolic acids in rat plasma by ultra-high performance liquid chromatography with tandem mass spectrometry after oral administration of Echinacea purpurea extract. J. Sep. Sci. 2016, 39, 1628–1637. [Google Scholar] [CrossRef] [PubMed]
- Plusquellec, Y.; Efthymiopoulos, C.; Duthil, P.; Houin, G. A pharmacokinetic model for multiple sites discontinuous gastrointestinal absorption. Med. Eng. Phys. 1999, 21, 525–532. [Google Scholar] [CrossRef]
- Issar, M.; Singh, S.K.; Mishra, B.; Gupta, R.C. Pharmacokinetics, n-situ absorption and protein binding studies of a new neuroleptic agent centbutindole in rats. Eur. J. Pharm. Sci. 2003, 19, 105–113. [Google Scholar] [CrossRef]
- Niu, X.F.; Wang, H.Y.; Zhang, J.J. The immune inhibitory effect of Echinacea to the regulation of immune function in mice. Chin. J. Vet. Med. 2015, 2, 50–52. [Google Scholar]
- U.S. Food and Drug Administration. Available online: http://www.fda.gov/downloads/Drugs/ GuidanceComplianceRegulatoryInformation/Guidances/UCM368107.pdf (accessed on 1 September 2013).
Sample Availability: Samples of the compounds syringic acid, ferulic acid, caffeic acid, vanillic acid, p-coumaric acid, 3,4-dihydroxybenzoic acid, 4-hydroxybenzoic acid and butylparaben are available from the authors. |
| Compounds | Spiked Conc (ng/mL) | Measured Conc (ng/mL) | Accuracy (%) | Intra-Day Precision (%) | Inter-Day Precision (%) |
|---|---|---|---|---|---|
| Syringic acid | 1.050 | 1.08 ± 0.13 | 2.66 | 6.28 | 12.61 |
| 5.250 | 5.14 ± 0.34 | −2.12 | 6.99 | 3.89 | |
| 65.63 | 64.14 ± 4.30 | −1.39 | 7.05 | 1.37 | |
| 840.0 | 802.1 ± 44.09 | −4.51 | 5.64 | 4.28 | |
| Ferulic acid | 0.832 | 0.85 ± 0.10 | 2.52 | 12.60 | 8.23 |
| 4.160 | 4.00 ± 0.43 | −3.80 | 11.03 | 8.98 | |
| 52.00 | 53.41 ± 3.47 | 2.71 | 4.82 | 13.60 | |
| 665.6 | 608.4 ± 36.12 | −8.59 | 4.42 | 12.39 | |
| Caffeic acid | 0.880 | 0.86 ± 0.09 | −2.04 | 9.98 | 13.93 |
| 4.400 | 4.71 ± 0.31 | 7.09 | 7.03 | 2.16 | |
| 55.00 | 55.55 ± 3.74 | 1.01 | 7.10 | 2.84 | |
| 704.0 | 701.2 ± 29.50 | −0.39 | 4.26 | 3.75 | |
| Vanillic acid | 0.326 | 0.36 ± 0.03 | 10.93 | 9.04 | 13.26 |
| 1.632 | 1.62 ± 0.16 | −0.59 | 9.32 | 13.00 | |
| 20.40 | 20.72 ± 1.77 | 1.57 | 8.53 | 8.72 | |
| 261.1 | 255.9 ± 14.17 | −1.45 | 4.82 | 9.30 | |
| p-Coumaric acid | 0.844 | 0.94 ± 0.10 | 11.17 | 11.10 | 10.48 |
| 4.220 | 3.97 ± 0.39 | −5.97 | 10.22 | 5.47 | |
| 52.75 | 51.93 ± 5.04 | −1.55 | 9.81 | 8.81 | |
| 675.2 | 632.7 ± 50.98 | −6.30 | 8.33 | 5.65 | |
| 3,4-Dihydroxy benzoic acid | 0.808 | 0.78 ± 0.06 | −3.27 | 7.11 | 10.18 |
| 4.040 | 4.03 ± 0.42 | −0.33 | 10.77 | 6.11 | |
| 50.50 | 53.82 ± 5.71 | 6.57 | 10.71 | 9.81 | |
| 646.4 | 663.5 ± 45.89 | 2.46 | 6.95 | 6.67 | |
| 4-Hydroxy benzoic acid | 0.856 | 0.84 ± 0.12 | −1.52 | 14.74 | 9.14 |
| 4.280 | 4.25 ± 0.47 | −0.76 | 11.41 | 7.89 | |
| 53.50 | 53.60 ± 5.80 | 0.18 | 11.35 | 5.38 | |
| 684.8 | 683.2 ± 66.79 | −0.23 | 10.11 | 6.71 |
| Compounds | Spiked Conc (ng/mL) | Matrix Effect | Extraction Recovery | ||
|---|---|---|---|---|---|
| Mean (%) | RSD (%) | Mean (%) | RSD (%) | ||
| Syringic acid | 5.250 | 86.06 | 14.39 | 89.96 | 6.18 |
| 65.63 | 84.84 | 5.71 | 92.25 | 10.45 | |
| 840.0 | 81.17 | 12.72 | 90.45 | 4.72 | |
| Ferulic acid | 4.160 | 82.87 | 13.68 | 94.50 | 7.65 |
| 52.00 | 91.19 | 12.57 | 87.47 | 4.02 | |
| 665.6 | 100.2 | 10.92 | 95.79 | 13.19 | |
| Caffeic acid | 4.400 | 94.75 | 9.79 | 81.43 | 12.43 |
| 55.00 | 97.88 | 11.04 | 95.03 | 11.56 | |
| 704.0 | 102.9 | 12.46 | 96.17 | 11.26 | |
| Vanillic acid | 1.632 | 89.37 | 12.80 | 82.27 | 12.52 |
| 20.40 | 100.0 | 11.00 | 89.38 | 11.82 | |
| 261.1 | 83.94 | 14.43 | 95.85 | 11.83 | |
| p-Coumaric acid | 4.220 | 101.7 | 10.93 | 98.08 | 14.44 |
| 52.75 | 101.3 | 11.40 | 96.40 | 12.72 | |
| 675.2 | 97.20 | 11.21 | 94.63 | 11.68 | |
| 3,4-Dihydroxybenzoic acid | 4.040 | 99.45 | 14.28 | 87.88 | 12.85 |
| 50.50 | 96.16 | 9.03 | 84.62 | 14.87 | |
| 646.4 | 88.73 | 10.49 | 89.98 | 11.82 | |
| 4-Hydroxybenzoic acid | 4.280 | 102.4 | 12.80 | 80.28 | 10.51 |
| 53.50 | 88.88 | 11.00 | 85.06 | 6.74 | |
| 684.8 | 93.22 | 9.72 | 90.83 | 6.25 | |
| IS | 212.0 | 95.74 | 8.95 | 86.55 | 7.63 |
| Compounds | Spiked Conc (ng/mL) | Stability (RE%) | |||
|---|---|---|---|---|---|
| Freeze-Thaw | Short-Term | Long-Term | Post-Preparative | ||
| Syringic acid | 5.250 | −3.75 | −1.95 | −0.66 | −10.74 |
| 65.63 | 4.48 | −1.50 | −0.79 | −7.64 | |
| 840.0 | −2.61 | −5.73 | −1.30 | −6.08 | |
| Ferulic acid | 4.160 | −7.10 | −4.21 | 7.93 | −0.89 |
| 52.00 | −3.80 | 4.61 | 6.80 | 4.84 | |
| 665.6 | −13.12 | −8.77 | −1.89 | −4.91 | |
| Caffeic acid | 4.400 | 7.79 | 7.45 | 6.01 | 10.35 |
| 55.00 | 2.18 | −0.16 | 1.00 | 3.89 | |
| 704.0 | 6.15 | 6.12 | −1.54 | 9.83 | |
| Vanillic acid | 1.632 | −6.56 | 2.24 | 2.91 | 9.11 |
| 20.40 | 5.74 | −0.67 | −0.37 | −6.34 | |
| 261.1 | 5.28 | 1.59 | −0.32 | −8.03 | |
| p-Coumaric acid | 4.220 | −7.57 | −3.59 | −6.75 | 8.52 |
| 52.75 | −0.82 | 1.57 | −5.40 | 11.50 | |
| 675.2 | −8.78 | −4.81 | −5.30 | 7.12 | |
| 3,4-Dihydroxy benzoic acid | 4.040 | −0.11 | 2.03 | −2.93 | 9.29 |
| 50.50 | 6.57 | 10.18 | 2.30 | −5.47 | |
| 646.4 | −0.52 | 4.80 | 3.65 | −4.01 | |
| 4-Hydroxybenzoic acid | 4.280 | 10.03 | 10.62 | −3.31 | −6.42 |
| 53.50 | 2.63 | −0.46 | 0.21 | 10.47 | |
| 684.8 | −1.01 | −2.50 | 2.81 | 7.71 | |
| Compounds | Cmax (ng/mL) | Tmax (h) | T1/2 (h) | AUC0→t (ng h/mL) | AUC0→∞ (ng h/mL) |
|---|---|---|---|---|---|
| Syringic acid | 122.50 ± 16.43 | 1.54 ± 0.14 | 13.21 ± 1.66 | 1645.22 ± 132.79 | 1946.57 ± 164.14 |
| Ferulic acid | 358.68 ± 43.84 | 8.00 ± 0 | 8.48 ± 0.71 | 5620.34 ± 453.15 | 6015.42 ± 497.85 |
| Caffeic acid | 854.61 ± 35.88 | 8.00 ± 0 | 5.31 ± 0.50 | 9217.13 ± 452.54 | 9326.39 ± 441.68 |
| Vanillic acid | 227.56 ± 11.94 | 0.75 ± 0 | 8.89 ± 1.12 | 3136.01 ± 263.85 | 3435.67 ± 233.61 |
| p-Coumaric acid | 292.51 ± 16.38 | 0.75 ± 0 | 9.32 ± 1.29 | 3132.51 ± 156.23 | 3439.38 ± 217.96 |
| 3,4-Dihydroxy benzoic acid | 126.64 ± 18.07 | 0.77 ± 0.07 | 7.45 ± 0.87 | 1934.80 ± 123.40 | 2051.96 ± 115.85 |
| 4-Hydroxy benzoic acid | 144.71 ± 11.62 | 1.96 ± 0.14 | 14.91 ± 5.37 | 1643.96 ± 121.54 | 1947.84 ± 175.74 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, Y.; Wang, Z.; Wang, L.; Gao, M.; Wang, L.; Gan, C.; Yang, C. Simultaneous Determination of Seven Phenolic Acids in Rat Plasma Using UHPLC-ESI-MS/MS after Oral Administration of Echinacea purpurea Extract. Molecules 2017, 22, 1494. https://doi.org/10.3390/molecules22091494
Du Y, Wang Z, Wang L, Gao M, Wang L, Gan C, Yang C. Simultaneous Determination of Seven Phenolic Acids in Rat Plasma Using UHPLC-ESI-MS/MS after Oral Administration of Echinacea purpurea Extract. Molecules. 2017; 22(9):1494. https://doi.org/10.3390/molecules22091494
Chicago/Turabian StyleDu, Yan, Zhibin Wang, Libo Wang, Mingjie Gao, Liqian Wang, Chunli Gan, and Chunjuan Yang. 2017. "Simultaneous Determination of Seven Phenolic Acids in Rat Plasma Using UHPLC-ESI-MS/MS after Oral Administration of Echinacea purpurea Extract" Molecules 22, no. 9: 1494. https://doi.org/10.3390/molecules22091494





