Medicinal Plants Based Products Tested on Pathogens Isolated from Mastitis Milk
Abstract
:1. Introduction
2. Results
2.1. Determination of Phenolic Compounds Content
2.2. Antioxidant Activity Assays
2.3. Determination of Mineral Content
2.4. Antibacterial Activity
3. Discussion
4. Materials and Methods
4.1. Plant Material and Alcoholic Extracts
4.2. Bacterial Strains
4.3. Chemicals
4.4. Polyphenolic and Flavones Contents of the Extracts
4.5. Radical Scavenging Activity Assay (DPPH)
4.6. Determination of Mineral Content by Atomic Absorption Spectroscopy
4.7. Antibacterial Activity Determination
4.8. Plant Biological Products
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bradley, A.J. Bovine mastitis: An evolving disease. Vet. J. 2002, 164, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Viguier, C.; Arora, S.; Gilmartin, N.; Welbeck, K.; O’Kennedy, R. Mastitis detection: Current trends and future perspectives. Trends Biotechnol. 2009, 27, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Wellenberg, G.J.; Van der Poel, W.H.; Van Oirschot, J.T. Viral infections and bovine mastitis: A review. Vet. Microbiol. 2002, 88, 27–45. [Google Scholar] [CrossRef]
- Barkema, H.W.; Schukken, Y.H.; Zadoks, R.N. Invited Review: The Role of Cow, Pathogen and Treatment Regimen in the Therapeutic Success of Bovine Staphylococcus aureus Mastitis. J. Dairy Sci. 2006, 89, 1877–1895. [Google Scholar] [CrossRef]
- White, D.G.; McDermott, P.F. Emergence and transfer of antibiotic resistance. J. Dairy Sci. 2001, 84, E151–E155. [Google Scholar] [CrossRef]
- Rios, J.L.; Recio, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol. 2005, 100, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Brisibe, E.A.; Umoren, U.E.; Owai, P.U.; Brisibe, F. Dietary inclusion of dried Artemisia annua leaves for management of coccidiosis and growth enhancement in chickens. Afr. J. Biotechnol. 2008, 7, 4083–4093. [Google Scholar] [CrossRef]
- Upadhyay, A.; Upadhyaya, I.; Kollanoor-Johny, A.; Venkitanarayanan, K. Combating pathogenic microorganisms using plant-derived antimicrobials: A minireview of the mechanistic basis. BioMed Res. Int. 2014. [Google Scholar] [CrossRef] [PubMed]
- Boldootar, D.; El-Seedi, H.R.; Findakly, M.; Jabri, S.; Javzan, B.; Choidash, B.; Goransson, U.; Hellman, B. Antigenotoxic and antioxidant effects of the Mongolian medicinal plant Leptopyrum fumarioides (L): An in vitro study. J. Ethnopharmacol. 2014, 155, 559–606. [Google Scholar] [CrossRef]
- Dorman, H.J.D.; Deans, S.G. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Min, B.R.; Pinchak, W.E.; Merkel, R.; Walker, S.; Tomita, G.; Anderson, R.C. Comparative antimicrobial activity of tannin extracts from perennial plants on mastitis pathogens. Sci. Res. Essays 2008, 3, 66–73. [Google Scholar]
- Gopinath, S.M.; Suneetha, T.B.; Mruganka, V.D. Chemical prophylaxis and antibacterial activity of methanolic and aqueous extracts of some medicinal plants against bovine mastitis. Int. J. Adv. Biol. Res. 2011, 1, 93–95. [Google Scholar]
- Taemchuay, D.; Rukkwamsuk, T.; Sakpuaram, T.; Ruangwises, N. Antibacterial activity of crude extracts of Centella asiatica against Staphylococcus aureus in bovine mastitis. J. Kasetsart Vet. 2009, 19, 119–128. [Google Scholar]
- Voravuthikunchai, S.; Kitpipit, L. Antibacterial activity of crude extracts of Thai medicinal plants against clinical isolates of methicillin-resistant Staphylococcus aureus. Songklanakarin J. Sci. Technol. 2005, 27 (Suppl. 2), 525–534. [Google Scholar]
- Nath, G.; Suneetha, T.B.; Deshpande, M. Preliminary analysis of two medicinal plants against causative organism of bovine mastitis. Phytomedicine 2011, 3, 333–337. [Google Scholar]
- Dhanabalan, R. In vitro phytochemical screening and antibacterial activity of aqueous and methanolic leaf extracts of Tridax procumbens against bovine mastitis isolated Staphylococcus aureus. Ethnobot. Leaflets 2008, 12, 1090–1095. [Google Scholar]
- Stefan, M.; Zamfirache, M.; Padurariu, C.; Trută, E.; Gostin, I. The composition and antibacterial activity of essential oils in three Ocimum species growing in Romania. Open Life Sci. 2013, 8, 600–608. [Google Scholar] [CrossRef]
- Petrişor, A.I.; Ianoş, I.; Tălângă, C. Land cover and use changes focused on the urbanization processes in Romania. Environ. Eng. Manag. J. 2010, 9, 765–771. [Google Scholar]
- Pavel, M.; Ristić, M.; Stević, T. Essential oils of Thymus pulegioides and Thymus glabrescens from Romania: Chemical composition and antimicrobial activity. J. Serbian Chem. Soc. 2010, 75, 27–34. [Google Scholar] [CrossRef]
- Jianu, C.; Misca, C.; Pop, G.; Rusu, L.C.; Ardelean, L.; Gruia, A.T. Chemical composition and antimicrobial activity of essential oils obtained from dill (Anethum graveolens L.) grown in western Romania. Rev. Chim. 2012, 63, 641–645. [Google Scholar]
- Soare, L.C.; Ferdes, M.; Stefanov, S.; Denkova, Z.; Nicolova, R.; Denev, P.; Bejan, C.; Paunescu, A. Antioxidant activity, polyphenols content and antimicrobial activity of several native pteridophytes of Romania. Not. Bot. Hort. Agrobot. Cluj Napoca 2012, 40, 53–57. [Google Scholar]
- Benedec, D.; Vlase, L.; Oniga, I.; Mot, A.C.; Damian, G.; Hanganu, D.; Duma, M.; Silaghi-Dumitrescu, R. Polyphenolic composition, antioxidant and antibacterial activities for two Romanian subspecies of Achillea distans Waldst. et Kit. ex Willd. Molecules 2013, 18, 8725–8739. [Google Scholar] [CrossRef] [PubMed]
- Jianu, C.; Pop, G.; Gruia, A.T.; Horhat, F.G. Chemical composition and antimicrobial activity of essential oils of lavender (Lavandula angustifolia) and lavandin (Lavandula x intermedia) grown in Western Romania. Int. J. Agric. Biol. 2013, 15, 772–776. [Google Scholar]
- Mocan, A.; Crișan, G.; Vlase, L.; Crișan, O.; Vodnar, D.C.; Raita, O.; Gheldiu, A.M.; Toiu, A.; Oprea, R.; Tilea, I. Comparative studies on polyphenolic composition, antioxidant and antimicrobial activities of Schisandra chinensis leaves and fruits. Molecules 2014, 19, 15162–15179. [Google Scholar] [CrossRef] [PubMed]
- Moldovan, R.I.; Oprean, R.; Benedec, D.; Hanganu, D.; Duma, M.; Oniga, I.; Vlase, L. LC-MS analysis, antioxidant and antimicrobial activities for five species of Mentha cultivated in Romania. Dig. J. Nanomater. Biostruct. 2014, 9, 559–566. [Google Scholar]
- Vlase, L.; Benedec, D.; Hanganu, D.; Damian, G.; Csillag, I.; Sevastre, B.; Moţ, A.C.; Silaghi-Dumitrescu, R.; Tilea, I. Evaluation of antioxidant and antimicrobial activities and phenolic profile for Hyssopus officinalis, Ocimum basilicum and Teucrium chamaedrys. Molecules 2014, 19, 5490–5507. [Google Scholar] [CrossRef] [PubMed]
- Bobis, O.; Dezmirean, D.S.; Tomos, L.; Chirila, F.; Marghitas, L. Influence of phytochemical profile on antibacterial activity of different medicinal plants against gram-positive and gram-negative bacteria. Appl. Biochem. Microbiol. 2015, 51, 113–118. [Google Scholar] [CrossRef]
- Zajmi, A.; Hashim, N.M.; Noordin, M.; Khalifa, S.A.; Ramli, F.; Ali, H.M.; El-Seedi, H.R. Ultrastructural study on the antibacterial activity of Artonin E versus streptomycin against Staphylococcus aureus strains. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Duda, S.C.; Marghitas, L.A.; Dezmirean, D.S.; Duda, M.; Margaoan, R.; Bobis, O. Changes in major bioactive compounds with antioxidant activity of Agastache foeniculum, Lavandula angustifolia, Melissa officinalis and Nepeta cataria: Effect of harvest time and plant species. Ind. Crops Prod. 2015, 77, 499–507. [Google Scholar] [CrossRef]
- Fit, I.N.; Rapuntean, G.; Rapuntean, S.; Chirila, F.; Nadas, G.C. Antibacterial effect of essential vegetal extracts on Staphylococcus aureus compared to antibiotics. Not. Bot. Hort. Agrobot. Cluj Napoca 2009, 37, 117–123. [Google Scholar]
- Bubulica, M.V.; Anghel, I.; Grumezescu, A.M.; Saviuc, C.; Anghel, G.A.; Chifiriuc, M.C.; Gheorghe, I.; Lazăr, V.; Popescu, A. In vitro evaluation of bactericidal and antibiofilm activity of Lonicera tatarica and Viburnum opulus plant extracts on Staphylococcus strains. Farmacia 2012, 60, 80–94. [Google Scholar]
- Gogoasa, I.; Jurca, V.; Alda, L.M.; Velciov, A.; Rada, M.; Alda, S.; Sirbulescu, C.; Bordean, D.M.; Gergen, I. Mineral Content of Some Medicinal Herbs. J. Hort. For. Biotechnol. 2013, 17, 65–67. [Google Scholar]
- Paşca, C.; Marghitas, L.A.; Dezmirean, D.S.; Bobiș, O.; Bonta, V.; Mihăiescu, T.; Chirilă, F.; Fit, N. Mineral Profile Evolution of Some Medicinal Plants with Antibacterial Effects. Bull. Universit. Agric. Sci. Vet. Med. Cluj-Napoca 2016, 73, 223–229. [Google Scholar] [CrossRef]
- Newman, D.J. Natural products as leads to potential drugs: An old process or the new hope for drug discovery? J. Med. Chem. 2008, 51, 2589–2599. [Google Scholar] [CrossRef] [PubMed]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [PubMed]
- Mayer, M.; Vogl, C.R.; Amorena, M.; Hamburger, M.; Walkenhorst, M. Treatment of organic livestock with medicinal plants: A systematic review of European ethnoveterinary research. Forsch. Komplementärmed. 2014, 21, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Proestos, C.; Lytoudi, K.; Mavromelanidou, O.K.; Zoumpoulakis, P.; Sinanoglou, V.J. Antioxidant Capacity of Selected Plant Extracts and Their Essential Oils. Antioxidants 2013, 2, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Maslennikov, P.V.; Chupakhina, G.N.; Skrypnik, L.N. The content of phenolic compounds in medicinal plants of a botanical garden (Kaliningrad oblast), Plant Physiology. Biol. Bull. Russ. Acad. Sci. 2014, 41, 133. [Google Scholar] [CrossRef]
- Mihaylova, D.; Georgieva, L.; Pavlov, A. In vitro antioxidant activity and phenolic composition of Nepeta cataria L. extracts. Int. J. Agric. Sci. Technol. 2013, 1, 74–79. [Google Scholar]
- Stagos, D.; Portesis, N.; Spanou, C.; Mossialos, D.; Aligiannis, N.; Chaita, E.; Panagoulis, C.; Reri, E.; Skaltsounis, L.; Tsatsakis, A.M.; et al. Correlation of total polyphenolic content with antioxidant and antibacterial activity of 24 extracts from Greek domestic Lamiaceae species. Food Chem. Toxicol. 2012, 50, 4115–4124. [Google Scholar] [CrossRef] [PubMed]
- Sordillo, L.M.; Shafer-Weaver, K.; Derosa, D. Immunobiology of the Mammary Gland. J. Dairy Sci. 1997, 80, 1851–1865. [Google Scholar] [CrossRef]
- Miao, B.H.; Han, X.G.; Zhang, W.H. The ameliorative effect of silicon on soybean seedlings grown in potassium-deficient medium. Ann. Bot. 2010, 105, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Idris, S.; Iyaka, Y.A.; Ndamitso, M.M.; Paiko, Y.B. Nutritional Composition of the Leaves and Stems of Ocimum Gratissimum. J. Emerg. Trends Eng. Appl. Sci. 2011, 2, 801–805. [Google Scholar]
- Shukla, V.; Joshi, G.P.; Rawat, M.S.M. Lichens as a potential natural source of bioactive compounds: A review. Phytochem. Rev. 2010, 9, 303–314. [Google Scholar] [CrossRef]
- Kosanić, M.; Manojlović, N.; Janković, S.; Stanojković, T.; Ranković, B. Evernia prunastri and Pseudoevernia furfuraceae lichens and their major metabolites as antioxidant, antimicrobial and anticancer agents. Food Chem. Toxicol. 2013, 53, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Halama, P.; Haluwin, C.V. Antifungal activity of lichen extracts and lichenic acids. BioControl 2004, 49, 95–107. [Google Scholar] [CrossRef]
- Aslan, A.; Güllüce, M.; Sökmen, M.; Adιgüzel, A.; Sahin, F.; Özkan, H. Antioxidant and Antimicrobial Properties of the Lichens Cladonia foliacea, Dermatocarpon miniatum, Everinia divaricata, Evernia prunastri, and Neofuscella pulla. Pharm. Biol. 2006, 44, 247–252. [Google Scholar] [CrossRef]
- Mitrović, T.; Stamenković, S.; Cvetković, V.; Tošić, S.; Stanković, M.; Radojević, I.; Stefanović, O.; Čomić, L.; Đačić, D.; Ćurčić, M.; Marković, S. Antioxidant, antimicrobial and antiproliferative activities of five lichen species. Int. J. Mol. Sci. 2011, 12, 5428–5448. [Google Scholar] [CrossRef] [PubMed]
- Juteau, F.; Jerkovic, I.; Masotti, V.; Milos, M.; Mastelic, J.; Bessiere, J.M.; Viano, J. Composition and antimicrobial activity of the essential oil of Artemisia absinthium from Croatia and France. Planta Med. 2003, 69, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Bartha, S.G.; Quave, C.L.; Balogh, L.; Papp, N. Ethnoveterinary practices of Covasna County, Transylvania, Romania. J. Ethnobiol. Ethnomed. 2015, 11, 35–57. [Google Scholar] [CrossRef] [PubMed]
- Erdogrul, Ö.T. Antibacterial activities of some plant extracts used in folk medicine. Pharm. Biol. 2002, 40, 269–273. [Google Scholar] [CrossRef]
- Kordali, S.; Kotan, R.; Mavi, A.; Cakir, A.; Ala, A.; Yildirim, A. Determination of the chemical composition and antioxidant activity of the essential oil of Artemisia dracunculus and of the antifungal and antibacterial activities of Turkish Artemisia absinthium, A. dracunculus, Artemisia santonicum, and Artemisia spicigera essential oils. J. Agric. Food Chem. 2005, 53, 9452–9458. [Google Scholar] [CrossRef] [PubMed]
- Lopes-Lutz, D.; Alviano, D.S.; Alviano, C.S.; Kolodziejczyk, P.P. Screening of chemical composition, antimicrobial and antioxidant activities of Artemisia essential oils. Phytochemistry 2008, 69, 1732–1738. [Google Scholar] [CrossRef] [PubMed]
- Diaz, M.A.; Rossi, C.C.; Mendonça, V.R.; Silva, D.M.; Ribon, A.D.O.; Aguilar, A.P.; Muñoz, G.D. Screening of medicinal plants for antibacterial activities on Staphylococcus aureus strains isolated from bovine mastitis. Rev. Bras. Farmacogn. 2010, 20, 724–728. [Google Scholar] [CrossRef]
- Lis-Balchin, M.; Deans, S.G. Bioactivity of selected plant essential oils against Listeria monocytogenes. J. Appl. Microbiol. 1997, 82, 759–762. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Rota, C.; Carraminana, J.J.; Burillo, J.; Herrera, A. In vitro antimicrobial activity of essential oils from aromatic plants against selected foodborne pathogens. J. Food Prot. 2004, 67, 1252–1256. [Google Scholar] [CrossRef] [PubMed]
- Soković, M.; Glamočlija, J.; Marin, P.D.; Brkić, D.; van Griensven, L.J. Antibacterial effects of the essential oils of commonly consumed medicinal herbs using an in vitro model. Molecules 2010, 15, 7532–7546. [Google Scholar] [CrossRef] [PubMed]
- Blazekovic, B.; Stanic, G.; Pepeljnjak, S.; Vladimir-Knezevic, S. In vitro antibacterial and antifungal activity of Lavandula x intermedia Emeric ex Loisel. ‘Budrovka’. Molecules 2011, 16, 4241–4253. [Google Scholar] [CrossRef]
- Pașca, C.; Mărghitaș, L.A.; Dezmirean, D.; Bobiș, O.; Bonta, V.; Mărgăoan, R.; Chirilă, F.; Fit, N. The Assessment of the Antibacterial Activity of some Plant Extracts on Normal and Pathogenic Microflora from Milk. Sci. Pap. Anim. Sci. Biotechnol. 2015, 48, 166–172. [Google Scholar]
- Cavanagh, H.M.A.; Wilkinson, J.M. Biological activities of lavender essential oil. Phytother. Res. 2002, 16, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Folin, O.; Ciocâlteu, V. On tyrosine and tryptophane determination in proteins. J. Biol.Chem. 1927, 73, 627. [Google Scholar]
- Pașca, C.; Mărghitaș, L.A.; Bobiș, O.; Dezmirean, D.; Mărgăoan, R.; Mureșan, C. Total content of polyphenols and antioxidant activity of different melliferous plants. Bull. Anim. Sci. Biotechnol. 2016, 73, 59–65. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Kim, D.O.; Chun, O.K.; Kim, Y.J.; Moon, H.Y.; Lee, C.Y. Quantification of polyphenolics and their antioxidant capacity in fresh plums. J. Agric. Food Chem. 2003, 51, 6509–6515. [Google Scholar] [CrossRef] [PubMed]
- Dowd, L.E. Spectrophotometric determination of quercetin. Anal. Chem. 1959, 31, 1184–1187. [Google Scholar] [CrossRef]
- Farrukh, M.A. Atomic Absorption Spectroscopy; InTech Europe: Rijeka, Croația, 2012; pp. 1–12. ISBN 978-953-307-817-5. [Google Scholar]
- Berghof Products+Instruments GMBH. User Manual, SpeedwaveTM mws-2 Microwave Digestion System with Built-in, Non-Contact Temperature Measurement; Berghof Products+Instruments GMBH: Eningen, Germany, 2004; Volume 5, pp. 23–29. [Google Scholar]
- Finger, D.; Filho, I.K.; Torres, Y.R.; Quinaia, S.P. Propolis as an Indicator of Environmental Contamination by Metals. Bull. Environ. Contam. Toxicol. 2014, 92, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Quinn, P.; Carter, M.E.; Markey, B.; Carter, G.R. Clinical Veterinary Microbiology; Wolfe: London, UK, 1994; ISBN 9780723432371. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
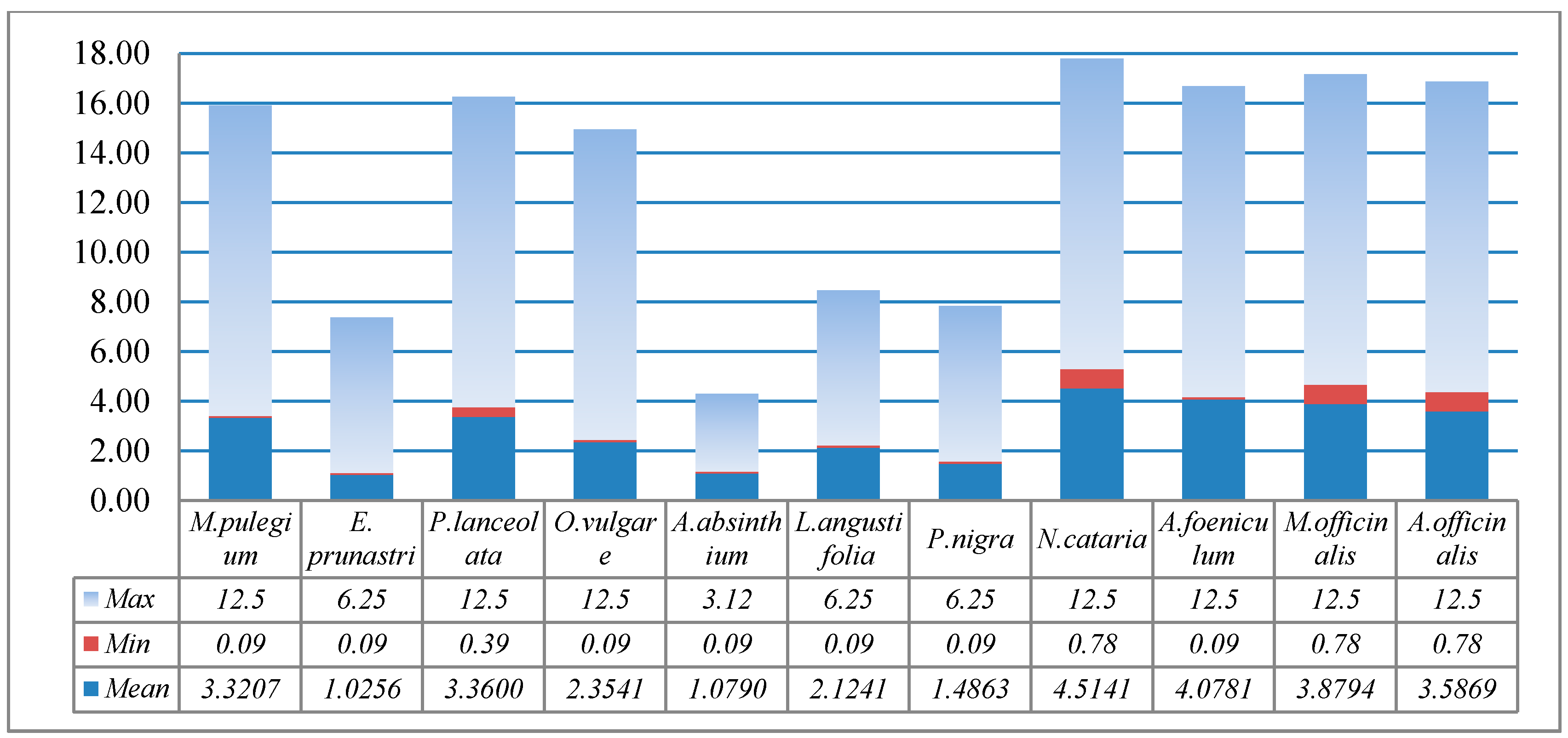
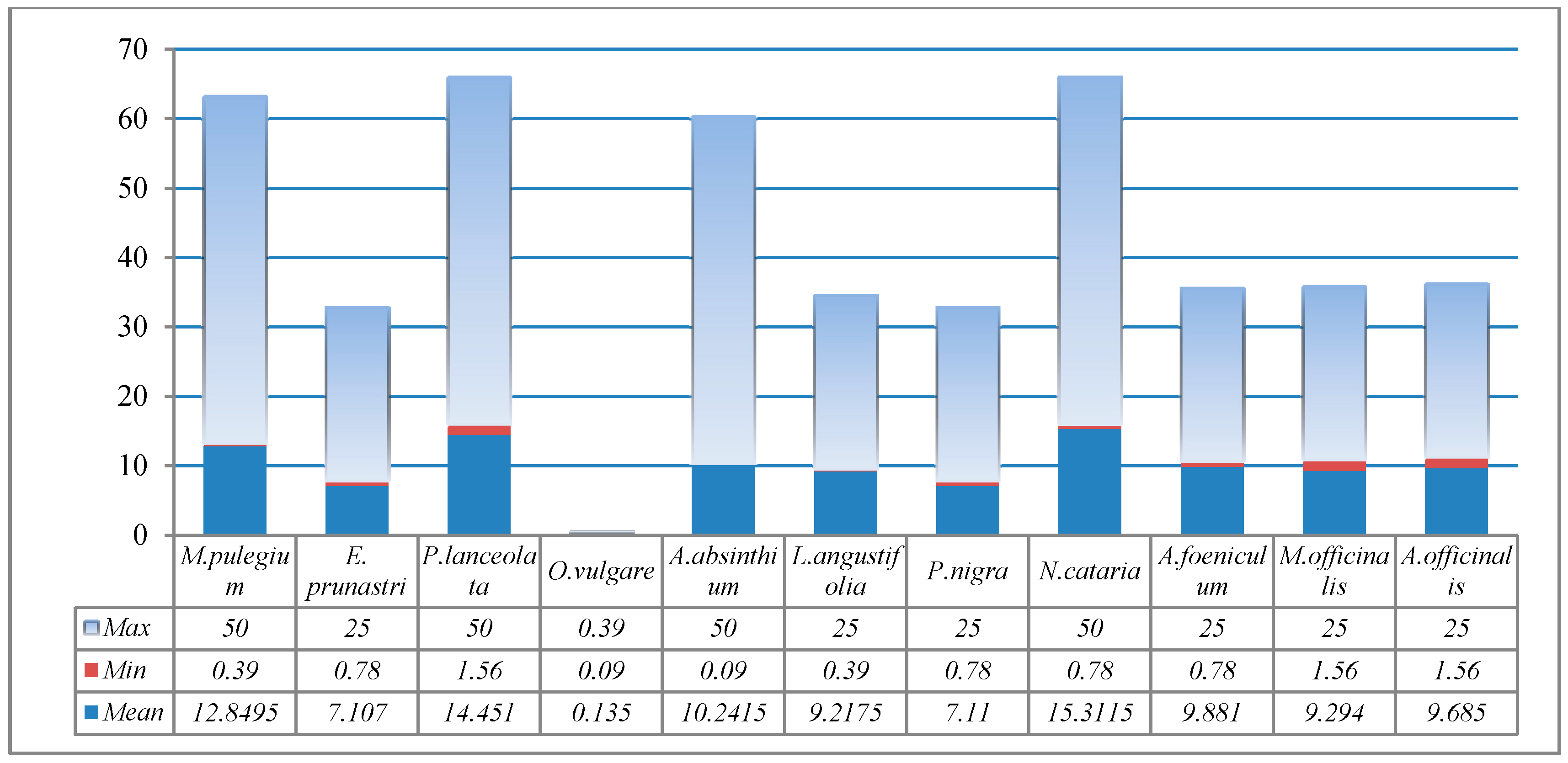
| Plant Extracts | Polyphenols (mg·GAE/g DW) | Flavones (mg·QE/g DW) | Inhibition Percentage (%) | RSA (Mmoli Trolox/g) |
|---|---|---|---|---|
| Mp | 30.2 ± 0.84 | 150.6 ± 1.51 | 58.60 ± 1.32 | 1.24 ± 0.03 |
| Pn | 2.4 ± 0.45 | 20.8 ± 0.95 | 70.01 ± 1.13 | 1.08 ± 0.03 |
| Ao | 3.3 ± 0.61 | 16.5 ± 0.67 | 74.95 ± 0.90 | 1.82 ± 0.03 |
| Pl | 6.1 ± 0.76 | 31.0 ± 0.55 | 77.66 ± 1.43 | 1.56 ± 0.04 |
| Ov | 14.3 ± 0.77 | 32.1 ± 0.56 | 79.88 ± 1.95 | 1.82 ± 0.05 |
| Ep | 14.2 ± 0.56 | 74.1 ± 1.21 | 70.40 ± 0.98 | 1.65 ± 0.02 |
| La | 15.7 ± 0.91 | 34.6 ± 0.44 | 77.76 ± 1.02 | 1.51 ± 0.04 |
| Nc | 18.1 ± 0.68 | 4.8 ± 0.22 | 77.92 ± 1.95 | 1.52 ± 0.03 |
| Mo | 26.5 ± 0.87 | 14.8 ± 0.34 | 74.19 ± 1.85 | 1.72 ± 0.02 |
| Aa | 79.2 ± 1.36 | 9.1 ± 0.24 | 81.65 ± 1.82 | 1.69 ± 0.03 |
| Af | 45.5 ± 1.02 | 89.0 ± 1.01 | 80.79 ± 1.91 | 1.75 ± 0.03 |
| Sample | Ca (µg/kg) | Mg (µg/kg) | Fe (µg/kg) | Na (µg/kg) | K (µg/kg) | Cr (µg/kg) | Cd (µg/kg) | Ni (µg/kg) | Mn (µg/kg) | Cu (µg/kg) | Se (µg/kg) | Pb (µg/kg) | Zn (µg/kg) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mp | 4630.15 ± 17.59 | 504.82 ± 1.29 | 1717.64 ± 17.52 | 0 | 632.73 ± 3.95 | 88.64 ± 4.26 | 42.18 ± 5.31 | 35.75 ± 0.61 | 84.83 ± 1.56 | 121.14 ± 1.35 | 181.02 ± 4.57 | 3.68 ± 0.23 | 51.17 ± 0.17 |
| Pn | 6749.50 ± 39.27 | 539.84 ± 1.41 | 1478.07 ± 14.10 | 12.38 ± 0.24 | 641.17 ± 6.88 | 77.65 ± 1.83 | 41.29 ± 7.31 | 19.26 ± 0.32 | 245.18 ± 4.37 | 118.52 ± 7.52 | 185.59 ± 8.22 | 2.18 ± 0.27 | 67.54 ± 0.17 |
| Ao | 6963.69 ± 17.65 | 535.86 ± 2.72 | 303.99 ± 4.78 | 0 | 644.24 ± 9.42 | 117.67 ± 3.30 | 90.44 ± 2.72 | 11.04 ± 0.41 | 262.63 ± 4.85 | 177.42 ± 9.48 | 107.88 ± 14.16 | 0.68 ± 0.2 | 64.76 ± 0.53 |
| Pl | 7098.04 ± 12.95 | 535.42 ± 2.05 | 56.21 ± 8.28 | 165.45 ± 11.18 | 630.78 ± 9.00 | 32.79 ± 1.14 | 36.06 ± 9.61 | 12.86 ± 0.01 | 199.29 ± 1.63 | 46.81 ± 5.24 | 190.16 ± 0.35 | 5.88 ± 0.78 | 29.63 ± 0.22 |
| Ov | 5071.45 ± 10.39 | 501.47 ± 2.65 | 0 | 1445.66 ± 8.31 | 552.18 ± 1.17 | 37.94 ± 1.29 | 45.66 ± 4.31 | 9.48 ± 0.49 | 244.90 ± 4.82 | 146.85 ± 8.64 | 40.10 ± 9.63 | 4.50 ± 0.49 | 25.87 ± 0.35 |
| Ep | 10,287.02 ± 17.32 | 418.76 ± 1.71 | 1653.08 ± 11.44 | 103.99 ± 5.88 | 378.45 ± 12.59 | 123.85 ± 5.66 | 13.99 ± 3.88 | 15.38 ± 0.2 | 237.38 ± 2.64 | 259.36 ± 1.24 | 517.28 ± 11.29 | 0 | 36.22 ± 0.13 |
| La | 2027.21 ± 12.35 | 1420.07 ± 10.71 | 323.13 ± 7.89 | 0 | 23,443.88 ± 16.52 | 0 | 0 | 11.05 ± 0.98 | 14.12 ± 1.76 | 13.44 ± 1.26 | 85.6 ± 9. 02 | 0 | 20.83 ± 1.11 |
| Nc | 4836.56 ± 11.13 | 2113.91 ± 11.23 | 73.63 ± 3.45 | 0 | 20,013.34 ± 15.78 | 0 | 0 | 9.09 ± 0.55 | 20.76 ± 1.85 | 10.09 ± 0.35 | 35.55 ± 1.01 | 0 | 26.13 ± 0.96 |
| Mo | 4064.49 ± 8.32 | 2357.07 ± 11.63 | 69.10 ± 5.67 | 0 | 18,911.22 ± 16.35 | 0 | 0 | 8.96 ± 0.63 | 33.42 ± 1.79 | 10.30 ± 0.29 | 21.22 ± 0.87 | 0 | 24.10 ± 1.34 |
| Aa | 9005.82 ± 5.88 | 867.72 ± 11.77 | 97.25 ± 9.15 | 0 | 20,991.05 ± 14.47 | 0 | 0 | 11.70 ± 1.21 | 46.54 ± 1.39 | 120.78 ± 2.56 | 212.34 ± 4.06 | 0 | 35.63 ± 0.86 |
| Af | 9505.12 ± 19.58 | 491.85 ± 3.53 | 3.87 ± 0.65 | 0 | 19,984.98 ± 14.82 | 0 | 0 | 1.10 ± 1.12 | 63.75 ± 1.75 | 79.44 ± 1.59 | 246.62 ± 1.65 | 0 | 26.78 ± 0.35 |
| Species | Mp | Nc | Mo | Af | La | Ov | Ao | Pl | Aa | Pn | Ep | C | O | C | G | P | F | E | A |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A. hydrophila caviae | 6.0 | 11.0 | 11.0 | 6.0 | 8.0 | 8.5 | 7.0 | 8.0 | 13.0 | 7.0 | 14.0 | 0.0 | 0.0 | 20.9 | 8.5 | 0.0 | 19.7 | 19 | 0.0 |
| B. cereus | 8.0 | 8.0 | 0.0 | 7.5 | 10.0 | 12.0 | 10.0 | 6.0 | 20.0 | 8.0 | 13.0 | 8.0 | 12.7 | 0.0 | 7.6 | 0.0 | 17.1 | 13.1 | 0.0 |
| E. intermedius | 8.0 | 10.5 | 10.0 | 9.0 | 13.0 | 8.0 | 9.0 | 11.0 | 7.0 | 9.5 | 13.0 | 8.5 | 10.7 | 6.7 | 13.3 | 0.0 | 0.0 | 20.2 | 11.3 |
| E. coli | 7.0 | 10 | 9.0 | 9.0 | 12.0 | 8.0 | 6.0 | 0.0 | 7.0 | 7.0 | 22.0 | 0.0 | 14.4 | 0.0 | 7.5 | 0.0 | 12.3 | 18.3 | 7.7 |
| E. coli | 10.0 | 8.5 | 12.0 | 9.0 | 12.5 | 7.0 | 12.0 | 13.0 | 6.0 | 9.0 | 9.0 | 8.0 | 20.6 | 8.5 | 14.5 | 0.0 | 22.7 | 19.6 | 0.0 |
| K. sedentarius | 8.0 | 10.0 | 10.0 | 7.0 | 7.5 | 7.5 | 7.0 | 8.0 | 14.5 | 12.0 | 19.0 | 6.0 | 22.2 | 16.1 | 15.5 | 21.8 | 17.1 | 19.2 | 13.9 |
| L. lactis | 8.0 | 7.5 | 7.0 | 10.0 | 7.5 | 8.0 | 9.0 | 7.0 | 10.0 | 6.0 | 10.0 | 8.0 | 19.6 | 12.2 | 14.1 | 16.2 | 14.7 | 13.8 | 19.9 |
| L. lactis | 8.0 | 10.0 | 8.0 | 8.5 | 10.0 | 11.0 | 8.0 | 8.5 | 12.0 | 12.0 | 21.0 | 8.0 | 21.6 | 10.9 | 14.7 | 21.7 | 20.6 | 18.2 | 18.2 |
| S. liquefaciens | 10.0 | 11.0 | 11.0 | 9.0 | 10.0 | 10.0 | 16.0 | 13.0 | 11.0 | 10.0 | 10.0 | 6.0 | 0.0 | 18.9 | 9.5 | 0.0 | 17.8 | 11.0 | 0.0 |
| S. liquefaciens | 7.0 | 9.0 | 9.0 | 7.0 | 8.0 | 8.0 | 7.0 | 7.0 | 0.0 | 11.0 | 10.0 | 8.0 | 0.0 | 17.4 | 9.9 | 0.0 | 18.3 | 18.8 | 0.0 |
| S. aureus | 6.0 | 8.0 | 11.0 | 8.5 | 10.0 | 9.0 | 7.0 | 9.0 | 23.0 | 9.0 | 8.0 | 8.5 | 17.8 | 13.3 | 9.5 | 25.1 | 15.3 | 17.4 | 23.5 |
| S. chromogenes | 7.5 | 8.0 | 9.0 | 8.0 | 10.0 | 11.0 | 8.5 | 8.5 | 12.0 | 9.5 | 12.0 | 8.0 | 21.4 | 11.6 | 16.6 | 20.9 | 18.1 | 22.2 | 14.4 |
| S. hyicus | 8.5 | 10.0 | 11.0 | 9.0 | 10.0 | 12.0 | 8.5 | 11.5 | 9.5 | 11.5 | 9.0 | 9.0 | 17.5 | 11.7 | 12.6 | 21.7 | 21.9 | 21.5 | 20.6 |
| S. intermedius | 11.5 | 11.0 | 9.5 | 8.5 | 9.0 | 8.5 | 10.0 | 8.0 | 10.0 | 12.0 | 9.5 | 8.0 | 17.1 | 24.1 | 8.7 | 0.0 | 19.2 | 18.7 | 0.0 |
| S. intermedius | 8.5 | 8.5 | 9.5 | 10.0 | 10.0 | 0.0 | 11.0 | 10.0 | 10.5 | 10.0 | 20.0 | 8.0 | 16.7 | 9.5 | 12.4 | 21.5 | 19.5 | 20.8 | 19.5 |
| S. intermedius | 8.5 | 10.0 | 11.0 | 11.5 | 10.0 | 11.5 | 7.0 | 8.5 | 9.0 | 9.5 | 11.5 | 8.5 | 15.9 | 14.1 | 5.8 | 0.0 | 17.4 | 14.7 | 0.0 |
| S. intermedius | 9.0 | 11.0 | 10.0 | 10.0 | 11.0 | 9.0 | 8.0 | 11.5 | 8.0 | 9.5 | 12.5 | 8.0 | 17.2 | 10.4 | 12.4 | 22.8 | 18.1 | 21.2 | 19.6 |
| S. xylosus | 8.0 | 12.5 | 12.5 | 7.0 | 11.5 | 7.5 | 16.0 | 12.0 | 20.0 | 14.0 | 8.0 | 8.5 | 23.7 | 14.9 | 15.9 | 6.2 | 18.9 | 18.1 | 10.3 |
| S. xylosus | 9.0 | 9.0 | 11.0 | 6.0 | 9.0 | 18.5 | 10.0 | 7.5 | 14.0 | 13.0 | 12.0 | 8.5 | 23.8 | 14.1 | 15.4 | 22.1 | 15.9 | 19.9 | 19.2 |
| S. xylosus | 10.0 | 8.5 | 11.5 | 9.0 | 10.0 | 8.0 | 10.0 | 8.0 | 7.0 | 9.0 | 12.0 | 8.5 | 15.1 | 18.5 | 10.1 | 0.0 | 21.5 | 21.4 | 0.0 |
| S. xylosus | 13.0 | 11.5 | 12.0 | 10.0 | 11.0 | 11.0 | 10.0 | 10.0 | 8.5 | 13.5 | 12.0 | 10.0 | 19.3 | 17.4 | 6.8 | 0.0 | 18.8 | 17.4 | 10.9 |
| S. xylosus | 13.0 | 8.5 | 9.0 | 9.5 | 10.5 | 8.0 | 13.5 | 9.0 | 12.0 | 9.0 | 10.0 | 10.0 | 23.2 | 11.1 | 11.4 | 7.08 | 0.0 | 13.1 | 11.1 |
| S. xylosus | 0.0 | 10.0 | 8.0 | 7.0 | 11.0 | 10.0 | 8.0 | 6.0 | 20.0 | 8.0 | 12.0 | 7.0 | 13.3 | 6.7 | 18.4 | 13.2 | 15.3 | 20.5 | 19.4 |
| S. xylosus | 7.0 | 11.0 | 10.0 | 8.0 | 12.0 | 6.0 | 9.0 | 9.0 | 14.5 | 9.0 | 12.0 | 8.0 | 13.6 | 5.76 | 17.61 | 9.6 | 26.1 | 18.9 | 16.5 |
| V. fluvialis | 6.0 | 8.0 | 10.0 | 6.0 | 11.0 | 12.0 | 12.0 | 10.0 | 8.0 | 9.0 | 14.0 | 8.5 | 0.0 | 17.6 | 9.2 | 0.0 | 18.6 | 18.9 | 0.0 |
| V. fluvialis | 10.0 | 8.0 | 7.0 | 9.0 | 9.0 | 10.5 | 11.0 | 9.0 | 10.0 | 10.0 | 11.0 | 7.0 | 9.9 | 27.7 | 14.8 | 0.0 | 25.4 | 25.7 | 0.0 |
| V. fluvialis | 11.0 | 10.0 | 12.0 | 8.0 | 12.0 | 12.0 | 13.0 | 8.0 | 17.0 | 9.0 | 12.0 | 8.0 | 0.0 | 20.1 | 10.9 | 0.0 | 10.3 | 19.2 | 0.0 |
| V. fluvialis | 10.0 | 8.0 | 7.5 | 7.0 | 10.0 | 12.0 | 8.0 | 7.0 | 20.5 | 13.0 | 11.0 | 7.0 | 0.0 | 20.9 | 6.2 | 0.0 | 9.9 | 10.8 | 0.0 |
| V. fluvialis | 7.0 | 9.5 | 10.0 | 7.0 | 11.0 | 8.0 | 12.0 | 11.0 | 19.5 | 9.0 | 8.0 | 7.0 | 0.0 | 20.1 | 7.2 | 0.0 | 6.2 | 16.2 | 0.0 |
| V. fluvialis | 11.0 | 10.0 | 8.5 | 7.0 | 9.0 | 8.0 | 10.0 | 9.0 | 0.0 | 7.0 | 9.0 | 0.0 | 0.0 | 19.2 | 9.2 | 0.0 | 18.2 | 16.3 | 9.1 |
| V. fluvialis | 12.5 | 10.5 | 10.0 | 8.0 | 10.0 | 10.5 | 10.0 | 13.0 | 24.0 | 11.0 | 12.5 | 9.5 | 26.7 | 20.1 | 22.3 | 30.3 | 32.1 | 28.1 | 23.1 |
| Y. ruckeri | 8.5 | 10 | 8.5 | 8.0 | 10.0 | 0.0 | 8.0 | 7.5 | 7.0 | 11.0 | 12.0 | 7.5 | 16.5 | 14.2 | 6.3 | 0.0 | 19.5 | 19.7 | 0.0 |
| Mean ± SD | 8.6 ± 2.5 | 9.6 ± 1.3 | 9.5 ± 2.3 | 8.3 ± 1.34 | 10.2 ± 1.4 | 9.1 ± 3.4 | 9.7 ± 2.5 | 8. 9 ± 2.5 | 12.0 ± 6.3 | 9.9 ± 1.9 | 12.2 ± 3.6 | 7.3 ± 2.5 | 12.7 ± 8.9 | 14.2 ± 6.4 | 11.7 ± 4.1 | 8.1 ± 10.4 | 17.1 ± 6.6 | 18.5 ± 3.7 | 8.9 ± 8.8 |
| Species | R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 | O | C | G | P | F | E | A |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A. hydrophila caviae | 7.3 | 6.9 | 7.5 | 6.1 | 5.7 | 8.1 | 9.7 | 8.3 | 0 | 20.9 | 8.5 | 0.0 | 19.7 | 19.0 | 0.0 |
| B. cereus | 20.5 | 19.1 | 20.6 | 21.7 | 11.2 | 19.9 | 22.5 | 17.5 | 12.7 | 0.0 | 7.6 | 0.0 | 17.1 | 13.1 | 0.0 |
| E. intermedius | 8.6 | 7.1 | 10.8 | 13.1 | 7.6 | 10.2 | 11.3 | 12.7 | 10.7 | 6.7 | 13.3 | 0.0 | 0 | 20.2 | 11.3 |
| E. coli | 10.1 | 5.9 | 9.3 | 12.6 | 13.7 | 7.5 | 13.2 | 9.9 | 14.4 | 0.0 | 7.5 | 0.0 | 12.3 | 18.3 | 7.7 |
| E. coli | 11.3 | 6.3 | 9.1 | 11.9 | 12.9 | 7.9 | 12.9 | 10.2 | 20.6 | 8.5 | 14.5 | 0.0 | 22.7 | 19.6 | 0.0 |
| K. sedentarius | 20.1 | 21.5 | 25.1 | 25.3 | 20.2 | 21.9 | 27.3 | 20.3 | 22.2 | 16.1 | 15.5 | 21.8 | 17.1 | 19.2 | 13.9 |
| L. lactis | 16.9 | 22.2 | 22.3 | 25.7 | 18.8 | 16.44 | 18.3 | 19.2 | 19.6 | 12.2 | 14.1 | 16.2 | 14.7 | 13.8 | 19.9 |
| L. lactis | 17.1 | 21.8 | 21.5 | 23.9 | 21.2 | 18.2 | 23.7 | 22.1 | 21.6 | 10.9 | 14.7 | 21.7 | 20.6 | 18.2 | 18.2 |
| S. liquefaciens | 5.8 | 8.5 | 12.3 | 10.8 | 0.0 | 12.1 | 9.1 | 10.1 | 0.0 | 18.9 | 9.5 | 0.0 | 17.8 | 11 | 0.0 |
| S. liquefaciens | 8.9 | 7.1 | 10.9 | 13.4 | 7.9 | 10.4 | 11.2 | 12.1 | 0.0 | 17.4 | 9.9 | 0.0 | 18.3 | 18.8 | 0.0 |
| S. aureus | 17.1 | 17.5 | 13.9 | 17.8 | 14.9 | 13.9 | 14.2 | 16.1 | 17.8 | 13.3 | 9.5 | 25.1 | 15.3 | 17.4 | 23.5 |
| S. chromogenes | 13.9 | 17.1 | 21.5 | 17.2 | 17.1 | 15.4 | 18.6 | 12.3 | 21.4 | 11.6 | 16.6 | 20.9 | 18.1 | 22.2 | 14.4 |
| S. hyicus | 14.6 | 15.9 | 16.3 | 24.3 | 17.1 | 18.6 | 19.3 | 18.7 | 17.5 | 11.7 | 12.6 | 21.7 | 21.9 | 21.5 | 20.6 |
| S. intermedius | 7.3 | 10.9 | 15.2 | 10.4 | 6.7 | 14.1 | 14.4 | 12.3 | 17.1 | 24.1 | 8.7 | 0.0 | 19.2 | 18.7 | 0.0 |
| S. intermedius | 21.2 | 21.7 | 24.5 | 22.9 | 20.9 | 22.3 | 21.8 | 22.9 | 16.7 | 9.5 | 12.4 | 21.5 | 19.5 | 20.8 | 19.5 |
| S. intermedius | 18.5 | 19.6 | 20.4 | 22.2 | 16.67 | 13.3 | 20.6 | 18.9 | 15.9 | 14.1 | 5.8 | 0.0 | 17.4 | 14.7 | 0.0 |
| S. intermedius | 19.7 | 20.3 | 22.3 | 22.5 | 18.7 | 17.1 | 21.5 | 19.9 | 17.2 | 10.4 | 12.4 | 22.8 | 18.1 | 21.2 | 19.6 |
| S. xylosus | 16.7 | 18.1 | 18.1 | 23.4 | 23.2 | 14.2 | 14.2 | 14.1 | 23.7 | 14.9 | 15.9 | 6.2 | 18.9 | 18.1 | 10.3 |
| S. xylosus | 16.9 | 18.2 | 17.9 | 23.5 | 23.4 | 14.5 | 14.6 | 14.3 | 23.8 | 14.1 | 15.4 | 22.1 | 15.9 | 19.9 | 19.2 |
| S. xylosus | 16.5 | 17.9 | 18.2 | 23.3 | 23.1 | 14.1 | 14.2 | 14.1 | 15.1 | 18.5 | 10.1 | 0.0 | 21.5 | 21.4 | 0.0 |
| S. xylosus | 17.9 | 16.4 | 19.7 | 21.7 | 13.9 | 22.1 | 21.3 | 20.7 | 19.3 | 17.4 | 6.8 | 0.0 | 18.8 | 17.4 | 10.9 |
| S. xylosus | 18.2 | 20.8 | 21.5 | 24.1 | 17.7 | 17.9 | 21.8 | 20.4 | 23.2 | 11.1 | 11.4 | 7.08 | 0.0 | 13.1 | 11.1 |
| S. xylosus | 18.1 | 16.5 | 19.8 | 21.9 | 13.9 | 22.2 | 21.5 | 20.9 | 13.3 | 6.7 | 18.4 | 13.2 | 15.3 | 20.5 | 19.4 |
| S. xylosus | 18.1 | 20.1 | 21.2 | 24.1 | 17.7 | 17.8 | 21.6 | 20.7 | 13.6 | 5.76 | 17.61 | 9.6 | 26.1 | 18.9 | 16.5 |
| V. fluvialis | 5.51 | 5.8 | 6.3 | 5.9 | 6.3 | 9.4 | 11.1 | 11.9 | 0.0 | 17.6 | 9.2 | 0.0 | 18.6 | 18.9 | 0.0 |
| V. fluvialis | 5.6 | 7.8 | 8.1 | 5.5 | 7.8 | 8.1 | 11.2 | 10.7 | 9.9 | 27.7 | 14.8 | 0.0 | 25.4 | 25.7 | 0.0 |
| V. fluvialis | 5.5 | 9.2 | 9.4 | 0.0 | 9.2 | 0.0 | 11.3 | 9.6 | 0.0 | 20.1 | 10.9 | 0.0 | 10.3 | 19.2 | 0.0 |
| V. fluvialis | 15.4 | 12.6 | 14.3 | 18.1 | 12.7 | 12.8 | 17.9 | 12.8 | 0.0 | 20.9 | 6.2 | 0.0 | 9.9 | 10.8 | 0.0 |
| V. fluvialis | 13.2 | 15.1 | 17.9 | 10.8 | 9.3 | 18.3 | 19.3 | 13.5 | 0.0 | 20.1 | 7.2 | 0.0 | 6.2 | 16.2 | 0.0 |
| V. fluvialis | 10.6 | 5.9 | 9.1 | 12.3 | 13.9 | 7.8 | 13.4 | 9.8 | 0.0 | 19.2 | 9.2 | 0.0 | 18.2 | 16.3 | 9.1 |
| V. fluvialis | 20.5 | 19.4 | 20.9 | 21.5 | 11.7 | 19.8 | 22.9 | 17.5 | 26.7 | 20.1 | 22.3 | 30.3 | 32.1 | 28.1 | 23.1 |
| Y. ruckeri | 7.8 | 10.6 | 15.2 | 9.9 | 8.0 | 14.1 | 15.1 | 10.9 | 16.5 | 14.2 | 6.3 | 0.0 | 19.5 | 19.7 | 0.0 |
| Mean ± SD | 13.9 ± 5.2 | 14.5 ± 5.8 | 16.3 ± 5.5 | 17.1 ± 7.1 | 13.8 ± 5.9 | 14.4 ± 5.3 | 16.9 ± 4.9 | 15.2 ± 4.4 | 12.7 ± 8.9 | 14.2 ± 6.4 | 11.7 ± 4.1 | 8.1 ± 10.4 | 17.1 ± 6.6 | 18.5 ± 3.7 | 8.9 ± 8.8 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pașca, C.; Mărghitaș, L.; Dezmirean, D.; Bobiș, O.; Bonta, V.; Chirilă, F.; Matei, I.; Fiț, N. Medicinal Plants Based Products Tested on Pathogens Isolated from Mastitis Milk. Molecules 2017, 22, 1473. https://doi.org/10.3390/molecules22091473
Pașca C, Mărghitaș L, Dezmirean D, Bobiș O, Bonta V, Chirilă F, Matei I, Fiț N. Medicinal Plants Based Products Tested on Pathogens Isolated from Mastitis Milk. Molecules. 2017; 22(9):1473. https://doi.org/10.3390/molecules22091473
Chicago/Turabian StylePașca, Claudia, Liviu Mărghitaș, Daniel Dezmirean, Otilia Bobiș, Victorița Bonta, Flore Chirilă, Ioana Matei, and Nicodim Fiț. 2017. "Medicinal Plants Based Products Tested on Pathogens Isolated from Mastitis Milk" Molecules 22, no. 9: 1473. https://doi.org/10.3390/molecules22091473








