Five New Limonoids from Peels of Satsuma Orange (Citrus reticulata)
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental
3.1. General Experimental Procedure
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Cell Culture
3.5. Cytotoxicity Assay
3.6. Inhibitory Assay of NO Production
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Okada, M.; Mitsuhashi, H. (Eds.) Newly Revised Illustrated Medicinal Plants of the World; Hokuryukan Co., Ltd.: Tokyo, Japan, 2002; p. 249. [Google Scholar]
- Sawabe, A.; Morita, M.; Kiso, T.; Kishine, H.; Ohtsubo, Y.; Minematsu, T.; Matsubara, Y.; Okamoto, T. Studies on physiologically active substances in citrus fruit peel. Part 23. Isolation and characterization of new limonoid glycosides from Citrus unshiu peels. Carbohydr. Res. 1999, 315, 142–147. [Google Scholar] [CrossRef]
- Baba, S.; Ueno, Y.; Kikuchi, T.; Tanaka, R.; Fujimori, K. A Limonoid Kihadanin B from Immature Citrus unshiu Peels Suppresses Adipogenesis through Repression of the Akt-FOXO1-PPARγ Axis in Adipocytes. J. Agric. Food Chem. 2016, 64, 9607–9615. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Lee, K.T.; Eun, J.S.; Zee, O.P.; Lim, J.P.; Eum, S.S.; Kim, S.H.; Shin, T.Y. Anti-allergic components from the peels of Citrus unshiu. Arch. Pharm. Res. 1999, 22, 642–645. [Google Scholar] [CrossRef] [PubMed]
- Sawabe, A.; Matsubara, Y.; Iizuka, Y.; Okamoto, K. Studies on physiologically active substances in citrus fruit peel. Part XIV. Structures and hypotensive effect of flavonoid glycosides in young Citrus unshiu peelings. Yukagaku 1989, 38, 53–59. [Google Scholar]
- Yang, X.; Kang, S.-M.; Jeon, B.-T.; Kim, Y.-D.; Ha, J.-H.; Kim, Y.-T.; Jeon, Y.-J. Isolation and identification of an antioxidant flavonoid compound from citrus-processing by-product. J. Sci. Food Agric. 2011, 91, 1925–1927. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.J.; Nam, J.-W.; Yoon, U.J.; Han, A.-R.; Seo, E.-K. Identification of three new flavonoids from the peels of Citrus unshiu. Helv. Chim. Acta 2012, 95, 240–245. [Google Scholar] [CrossRef]
- Seo, K.-H.; Lee, D.-Y.; Nam, T.-G.; Kim, D.-O.; Lee, D.-G.; Kim, E.-K.; Kang, H.-C.; Song, M.-C.; Baek, N.-I. New tocopherol analogue with radical-scavenging activity from the peels of Citrus unshiu Marcovich. J. Korean Soc. Appl. Biol. Chem. 2013, 56, 747–750. [Google Scholar] [CrossRef]
- Seo, K.H.; Lee, D.Y.; Lee, D.S.; Kang, H.C.; Kim, H.S.; Kim, Y.C.; Baek, N.I. Hepatoprotective and neuroprotective tocopherol analogs isolated from the peels of Citrus unshiu Marcovich. Nat. Prod. Res. 2015, 29, 571–573. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, K.; Kobayashi, M.; Arihara, S. Flower fragrance precursors from flower buds of Citrus unshiu MARCOV. Nat. Med. 1996, 50, 176–178. [Google Scholar]
- Eom, H.J.; Lee, D.; Lee, S.; Noh, H.J.; Hyun, J.W.; Yi, P.H.; Kang, K.S.; Kim, K.H. Flavonoids and a Limonoid from the Fruits of Citrus unshiu and Their Biological Activity. J. Agric. Food Chem. 2016, 64, 7171–7178. [Google Scholar] [CrossRef] [PubMed]
- Noh, H.J.; Hwang, D.; Lee, E.S.; Hyun, J.W.; Yi, P.H.; Kim, G.S.; Lee, S.E.; Pang, C.; Park, Y.J.; Chung, K.H.; et al. Anti-inflammatory activity of a new cyclic peptide, citrusin XI, isolated from the fruits of Citrus unshiu. J. Ethnopharmacol. 2015, 163, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.J.; Ahn, H.M.; Kim, S.S.; Yun, P.Y.; Jeon, G.L.; Ko, Y.H.; Riu, K.Z. Composition and antimicrobial activities of essential oils in the peel of citrus fruits. J. Appl. Biol. Chem. 2007, 50, 148–154. [Google Scholar]
- Nakatani, M.; Takao, H.; Iwashita, T.; Naoki, H.; Hase, T. The structure of graucin A, a new bitter limonoid from Evodia grauca Miq. (Rutaceae). Bull. Chem. Soc. Jpn. 1987, 60, 2503–2507. [Google Scholar] [CrossRef]
- Min, Y.D.; Kwon, H.C.; Yang, M.C.; Lee, K.H.; Choi, S.U.; Lee, K.R. Isolation of limonoids and alkaloids from Phellodendron amurense and their multidrug resistance (MDR) reversal activity. Arch. Pharm. Res. 2007, 30, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Kondo, Y.; Suzuki, H.; Nozoe, S. Two γ-hydroxybutenolides from the bark of Phellodendron amurense and photooxidation of limonoids. Yakugaku Zasshi 1985, 105, 742–746. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Morita, H.; Takeya, K.; Itokawa, H.; Fukaya, H. Limonoids from Citrus nippokoreana. Nat. Med. 1999, 53, 255–258. [Google Scholar]
- Yang, X.-W.; Zhang, H.; Hu, J. Isolation and structure characterization of evolimorutanin from the unripe fruits of Evodia rutaecarpa var. bodinieri. Fenxi Huaxue 2008, 36, 219–222. [Google Scholar]
- Garcez, F.R.; Garcez, W.S.; Tsutsumi, M.T.; Roque, N.F. Limonoids from Trichilia elegans ssp. elegans. Phytochemistry 1997, 45, 141–148. [Google Scholar] [CrossRef]
- Liu, J. Two new limonoids from Polygonum orientale L. Indian J. Chem. Sect. B Org. Chem. Incl. Med. Chem. 2001, 40, 644–646. [Google Scholar]
- Khalil, A.T.; Maatooq, G.T.; El Sayed, K.A. Limonoids from Citrus reticulata. Z. Naturforsch. C J. Biosci. 2003, 58, 165–170. [Google Scholar] [CrossRef]
- Bennett, R.D.; Hasegawa, S. Limonoids of calamondin seeds. Tetrahedron 1981, 37, 17–24. [Google Scholar] [CrossRef]
- Nakagawa, H.; Takaishi, Y.; Tanaka, N.; Tsuchiya, K.; Shibata, H.; Higuti, T. Chemical Constituents from the Peels of Citrus sudachi. J. Nat. Prod. 2006, 69, 1177–1179. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, C.; Dong, B.; Ma, X.; Hou, L.; Cao, X.; Wang, C. Anti-inflammatory activity and mechanism of surfactin in lipopolysaccharide-activated macrophages. Inflammation 2015, 38, 756–764. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Muroga, Y.; Jinno, M.; Kajimoto, T.; Usami, Y.; Numata, A.; Tanaka, R. New class azaphilone produced by a marine fish-derived Chaetomium globosum. The stereochemistry and biological activities. Bioorg. Med. Chem. 2011, 19, 4106–4113. [Google Scholar] [CrossRef] [PubMed]
- Matsui, Y.; Kikuchi, T.; Inoue, T.; Muraoka, O.; Yamada, T.; Tanaka, R. Carapanolides J-L from the seeds of Carapa guianensis (Andiroba) and their effects on LPS-activated NO production. Molecules 2014, 19, 17130–17140. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
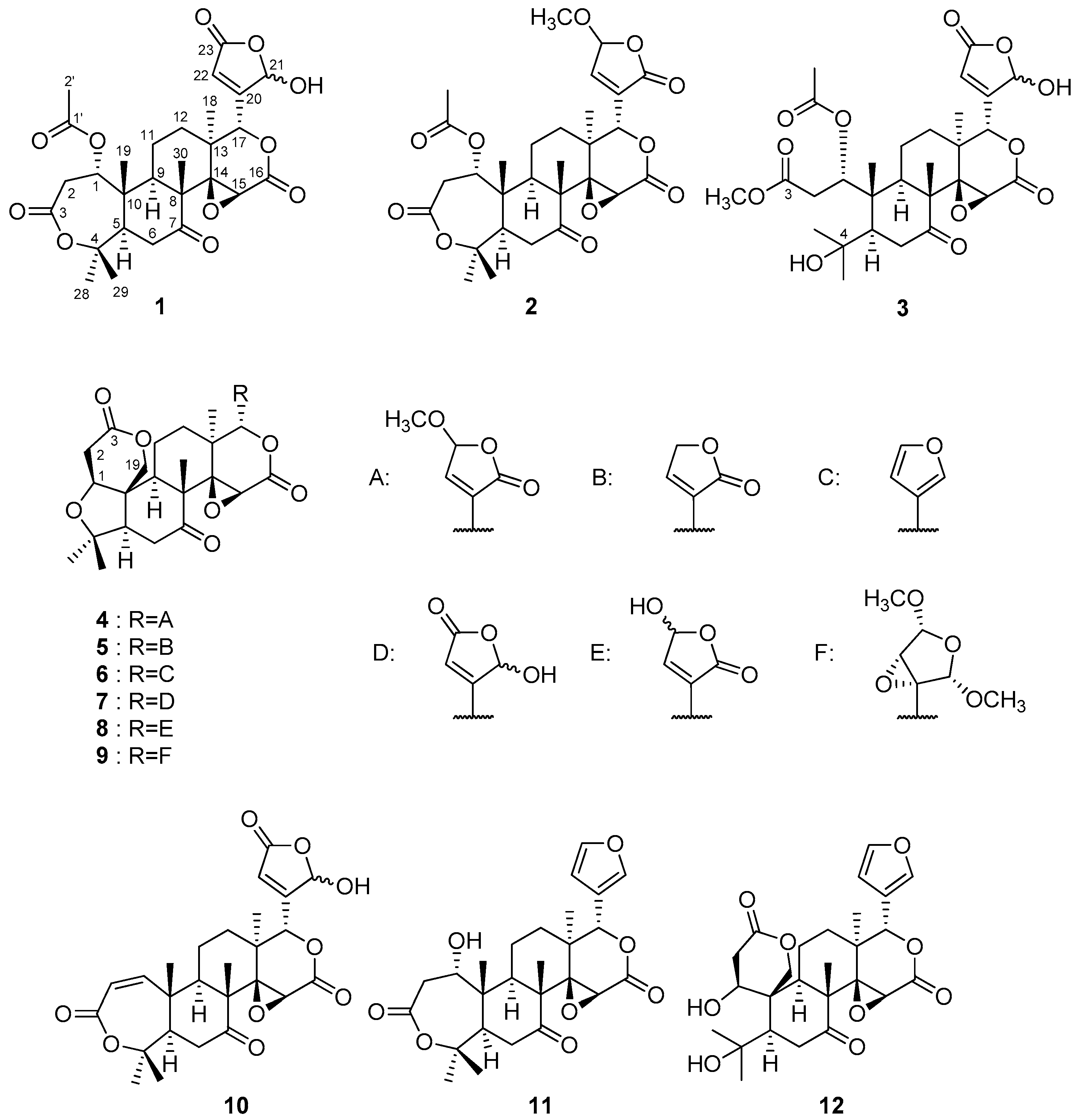
 ) and 1H-1H COSY (
) and 1H-1H COSY (  ) (A) and NOE (
) (A) and NOE (  ) (B) correlations of compound 1.
) (B) correlations of compound 1.
 ) of compound 3.
) of compound 3.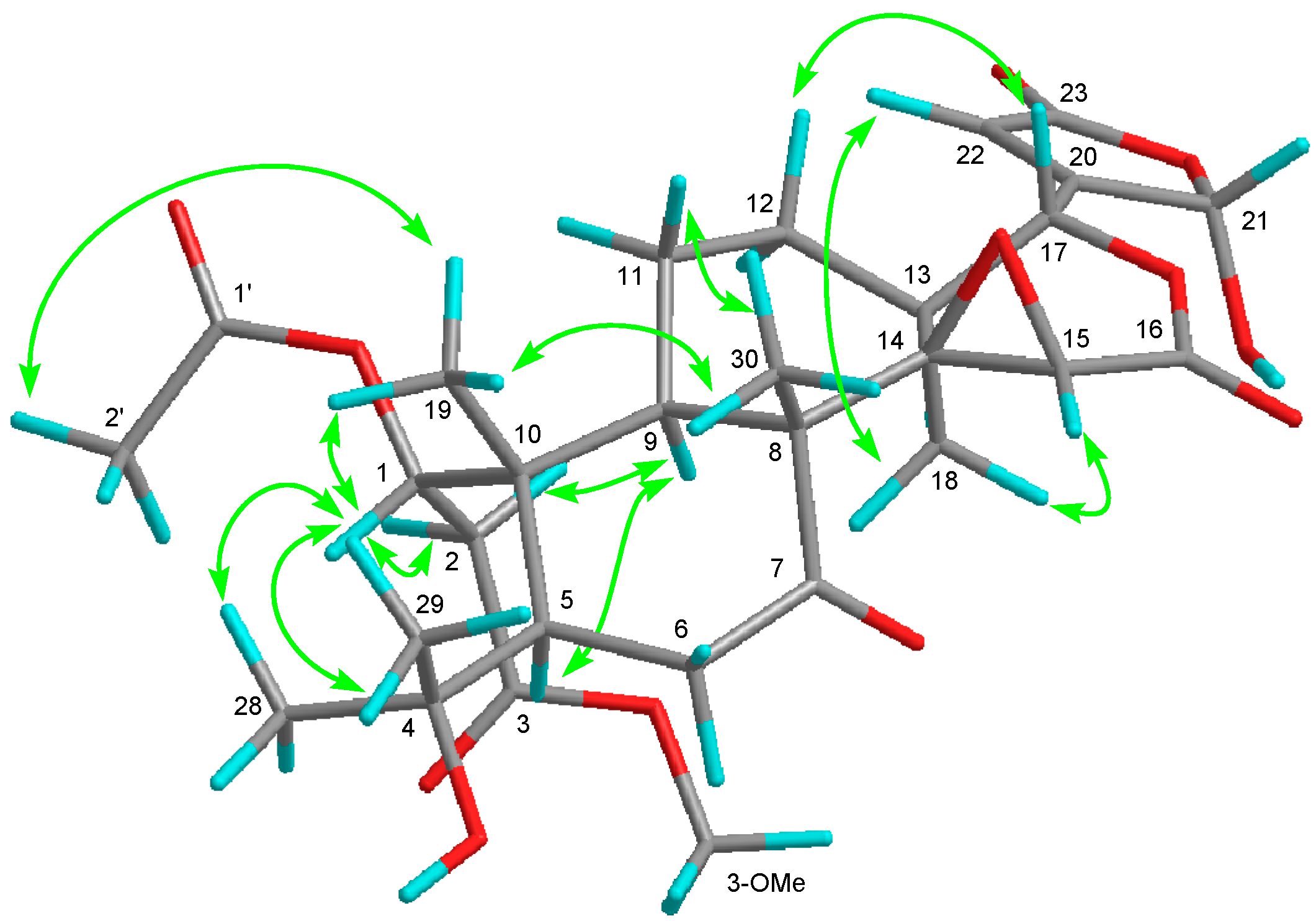
 ) and 1H-1H COSY (
) and 1H-1H COSY (  ) (A) and NOE (
) (A) and NOE (  ) (B) correlations of compound 4.
) (B) correlations of compound 4.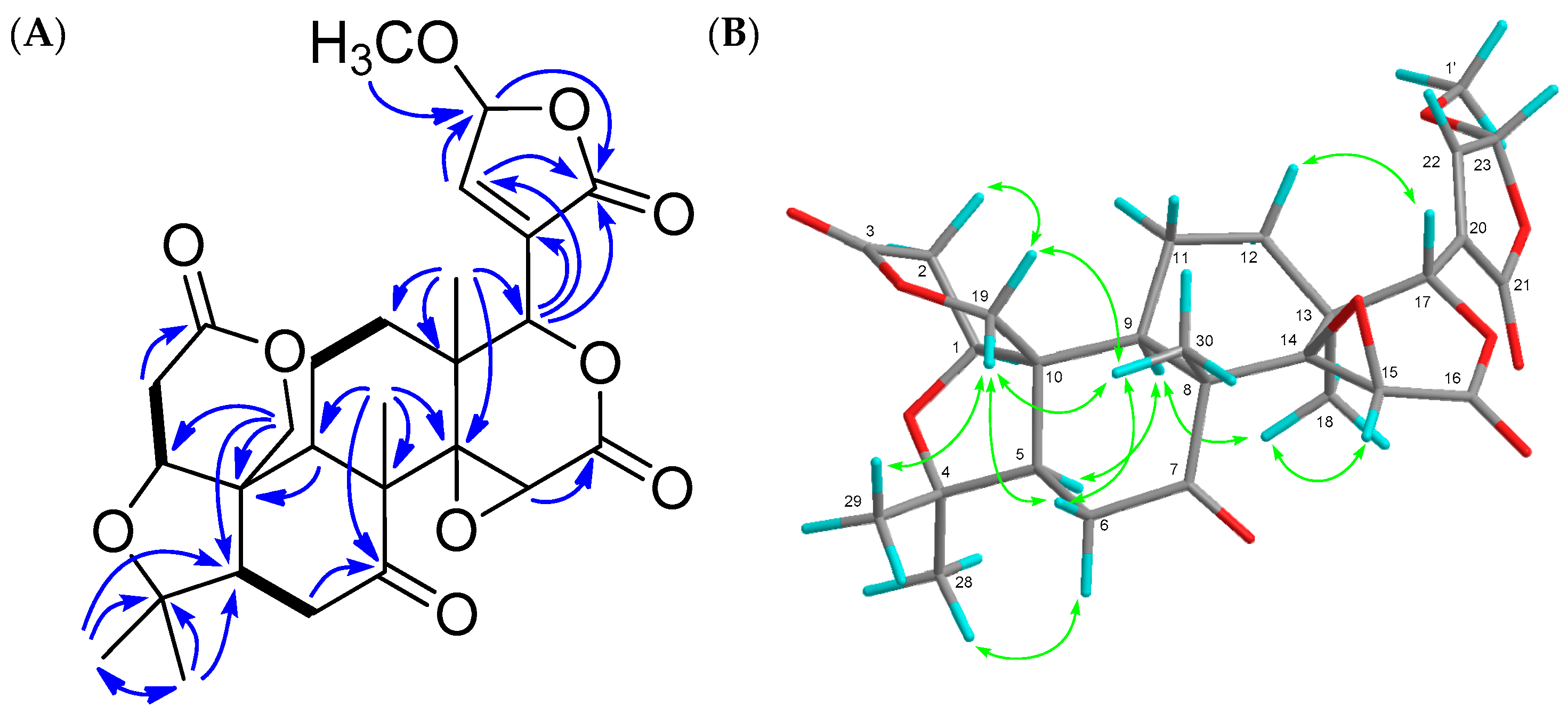
| Position | 1 | 2 | 3 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| δH | Mult. (J in Hz) | δC | δH | Mult. (J in Hz) | δC | δH | Mult. (J in Hz) | δC | ||||
| 1β | 5.05 | brd (6.4) | 70.7 | d | 4.97 | d (7.1) | 72.0 | d | 6.54 | brs | 76.3 | d |
| 2 | α 3.11 | dd (6.4, 15.8) | 35.3 | t | α 2.98 | dd (7.1, 15.8) | 36.0 | t | A 2.35 | m | 35.4 | t |
| β 3.24 | brd (15.8) | β 3.50 | dd (1.2, 15.8) | B 2.82 | m | |||||||
| 3 | 169.1 | s | 169.5 | s | 172.0 | s | ||||||
| 4 | 84.4 | s | 84.7 | s | 74.1 | s | ||||||
| 5 | 2.62 | dd (3.9, 15.8) | 51.1 | d | 2.69 | dd (3.5, 14.7) | 51.7 | d | 2.01 | m | 53.0 | |
| 6α | α 2.60 | dd (3.9, 15.8) | 38.6 | t | α 2.51 | dd (3.5, 14.7) | 39.6 | t | α 2.46 | dd (5.3, 14.9) | 38.9 | t |
| 6β | β 2.78 | t (15.8) | β 3.10 | t (14.7) | β 2.80 | t (14.9) | ||||||
| 7 | 206.5 | s | 208.3 | s | 210.0 | s | ||||||
| 8 | 53.1 | s | 53.2 | s | 52.5 | s | ||||||
| 9 | 2.44 | brd (11.2) | 44.3 | d | 2.61 | dd (2.4, 11.8) | 44.8 | d | 2.14 | brd (11.4) | 44.4 | d |
| 10 | 44.1 | s | 45.1 | s | 46.1 | s | ||||||
| 11 | α 1.70 | m | 17.3 | t | α 1.63 | m | 17.44 | t | α 2.49 | m | 19.0 | t |
| β 1.66 | m | β 1.73 | m | β 1.72 | m | |||||||
| 12 | α 1.29 | m | 32.0 | t | α 1.16 | m | 30.6 | t | α 1.61 | m | 31.6 | t |
| β 2.02 | dd (7.9, 13.0) | β 2.10 | m | β 2.01 | m | |||||||
| 13 | 37.7 | s | 39.0 | s | 37.5 | s | ||||||
| 14 | 65.0 | s | 66.8 | s | 65.0 | s | ||||||
| 15 | 3.70 | s | 52.6 | d | 3.95 | m | 54.5 | d | 3.64 | s | 52.5 | d |
| 16 | 165.5 | s | 166.9 | 166.3 | s | |||||||
| 17 | 5.34 | brd (1.7) | 78.1 | d | 5.32 | t (1.1) | 76.2 | d | 5.33 | d (1.5) | 78.6 | d |
| 18 | 1.11 | s | 21.4 | q | 1.20 | s | 20.4 | q | 1.10 | s | 21.4 | q |
| 19 | 1.34 | s | 16.64 | q | 1.47 | m | 16.1 | q | 1.31 | s | 16.7 | q |
| 20 | 162.5 | s | 134.0 | s | 163.6 | s | ||||||
| 21 | 5.99 | brs | 97.4 | d | 169.80 | s | 6.00 | brs | 98.2 | d | ||
| 22 | 6.30 | dd (0.9, 1.7) | 123.2 | d | 7.48 | t (1.1) | 150.9 | d | 6.29 | brs | 122.7 | d |
| 23 | 168.9 | s | 6.02 | t (1.1) | 103.5 | d | 169.8 | s | ||||
| 28 | 1.48 | s | 33.4 | q | 1.39 | s | 33.9 | q | 1.35 | s | 33.6 | q |
| 29 | 1.56 | s | 23.4 | q | 1.64 | s | 23.3 | q | 1.34 | s | 27.5 | q |
| 30 | 1.16 | s | 16.66 | q | 1.28 | s | 17.49 | q | 1.14 | s | 16.3 | q |
| 1′ | 169.6 | s | 2.00 | s | 20.7 | q | 170.8 | s | ||||
| 2′ | 2.10 | s | 20.9 | q | 169.82 | s | 2.08 | s | 21.1 | q | ||
| 3-OCH3 | 3.67 | s | 52.3 | q | ||||||||
| 23-OCH3 | 3.45 | s | 56.9 | q |
| Position | 4 | 5 | ||||||
|---|---|---|---|---|---|---|---|---|
| δH | Mult. (J in Hz) | δC | δH | Mult. (J in Hz) | δC | |||
| 1β | 4.03 | brd (4.2) | 79.2 | d | 4.27 | brd (4.1) | 80.1 | d |
| 2α | 2.66 | dd (1.7, 16.8) | 35.6 | t | 2.87 | dd (1.5, 16.7) | 36.5 | t |
| 2β | 2.98 | dd (4.2, 16.8) | 2.73 | dd (4.1, 16.7) | ||||
| 3 | 168.9 | s | 170.0 | s | ||||
| 4 | 80.3 | s | 80.7 | s | ||||
| 5 | 2.22 | m | 60.4 | d | 2.60 | dd (3.6, 15.2) | 59.8 | d |
| 6α | 2.47 | dd (3.5, 14.6) | 36.3 | t | 2.40 | dd (3.6, 15.2) | 37.1 | t |
| 6β | 2.84 | dd (14.6, 15.8) | 3.16 | t (15.2) | ||||
| 7 | 206.1 | s | 208.2 | s | ||||
| 8 | 51.1 | s | 51.7 | s | ||||
| 9 | 2.50 | dd (3.3, 12.7) | 48.0 | d | 2.83 | m | 48.2 | d |
| 10 | 45.8 | s | 46.7 | s | ||||
| 11α | 1.77 | (2H) | 18.5 | t | 1.96 | m | 18.8 | t |
| 11β | 2.07 | m | ||||||
| 12α | 1.40 | ddd (7.3, 9.1, 14.4) | 28.7 | t | 1.44 | m | 29.0 | t |
| 12β | 2.24 | m | 2.06 | m | ||||
| 13 | 38.6 | s | 39.9 | s | ||||
| 14 | 65.7 | s | 67.6 | s | ||||
| 15 | 4.12 | s | 53.8 | d | 4.19 | s | 55.2 | d |
| 16 | 166.0 | s | 167.3 | s | ||||
| 17 | 5.43 | t (1.5) | 75.2 | d | 5.35 | d (1.2) | 76.6 | d |
| 18 | 1.18 | s | 20.0 | q | 1.27 | s | 19.7 | q |
| 19α | 4.46 | d (13.2) | 65.1 | t | 4.65 | d (13.5) | 65.7 | t |
| 19β | 4.74 | d (13.2) | 4.97 | d (13.5) | ||||
| 20 | 133.8 | s | 129.6 | s | ||||
| 21 | 168.8 | s | 173.1 | s | ||||
| 22 | 7.25 | t (1.5) | 149.1 | d | 7.85 | dd (1.7, 2.9) | 154.4 | d |
| 23 | 5.77 | t (1.5) | 102.5 | d | 4.99 | dd (1.7, 3.5) | 71.9 | t |
| 28 | 1.29 | s | 30.2 | q | 1.13 | s | 21.8 | q |
| 29 | 1.18 | s | 21.2 | q | 1.24 | s | 30.3 | q |
| 30 | 1.09 | s | 17.8 | q | 1.18 | s | 18.2 | q |
| 23-OCH3 | 3.60 | s | 57.8 | q |
| Inhibitory Ratio of NO % (Cell Viability %) a,b | |||||
|---|---|---|---|---|---|
| Compound | 1 μM | 3 μM | 10 μM | 30 μM | IC50 (μM) |
| 1 | 109.9 ± 2.8 | 110.9 ± 1.7 | 104.8 ± 2.2 | 106.6 ± 2.5 | >30 |
| (99.1 ± 1.3) | (102.5 ± 1.8) | (103.7 ± 0.7) | (99.3 ± 1.3) | ||
| 2 | 95.2 ± 1.6 | 89.8 ± 2.1 * | 80.4 ± 2.1 ** | 39.3 ± 0.5 ** | 25.4 |
| (101.3 ± 1.5) | (98.7 ± 1.4) | (99.1 ± 0.3) | (94.5 ± 0.4) | ||
| 3 | 99.4 ± 0.8 | 99.4 ± 2.3 | 94.1 ± 1.9 | 80.3 ± 1.2 ** | >30 |
| (96.8 ± 0.2) | (94.5 ± 0.3) | (93.2 ± 0.4) | (93.4 ± 0.6) | ||
| 4 | 96.2 ± 7.9 | 94.0 ± 2.7 | 100.8 ± 1.2 | 95.2 ± 3.4 | >30 |
| (102.6 ± 0.4) | (97.0 ± 0.3) | (95.1 ± 0.2) | (91.7 ± 0.5) | ||
| 5 | 99.5 ± 5.6 | 100.6 ± 4.7 | 93.8 ± 1.6 | 83.2 ± 5.6* | >30 |
| (97.9 ± 1.9) | (97.9 ± 0.2) | (99.5 ± 0.5) | (96.6 ± 0.2) | ||
| 6 | 102.3 ± 4.4 | 93.4 ± 5.7 | 90.9 ± 3.1 | 86.0 ± 5.5 | >30 |
| (100.1 ± 0.3) | (95.6 ± 0.5) | (97.2 ± 0.9) | (99.3 ± 0.9) | ||
| 7 | 99.8 ± 1.4 | 99.0 ± 3.0 | 96.8 ± 2.5 | 93.9 ± 4.0 | >30 |
| (102.3 ± 1.5) | (106.3 ± 2.2) | (104.2 ± 1.4) | (100.2 ± 1.4) | ||
| 8 | 97.2 ± 1.1 | 94.7 ± 2.4 | 90.9 ± 1.6 ** | 76.0 ± 2.2 ** | >30 |
| (98.1 ± 2.4) | (97.6 ± 0.7) | (92.0 ± 0.5) | (89.3 ± 0.5) | ||
| 9 | 94.3 ± 4.3 | 85.2 ± 6.4 | 91.3 ± 2.8 | 89.9 ± 3.2 | >30 |
| (101.4 ± 0.7) | (97.7 ± 0.6) | (101.3 ± 1.2) | (97.6 ± 2.1) | ||
| 10 | 95.0 ± 4.2 | 96.8 ± 1.3 | 97.7 ± 2.2 | 86.6 ± 1.6 | >30 |
| (104.7 ± 2.3) | (101.8 ± 1.0) | (98.9 ± 0.5) | (98.6 ± 1.8) | ||
| 11 | 103.6 ± 3.1 | 93.2 ± 3.2 | 84.7 ± 3.2 * | 78.1 ± 3.4 ** | >30 |
| (95.9 ± 0.1) | (100.1 ± 0.4) | (99.3 ± 1.5) | (100.4 ± 0.6) | ||
| 12 | 106.6 ± 4.1 | 92.6 ± 3.2 | 88.4 ± 5.5 | 73.9 ± 3.4 ** | >30 |
| (100.7 ± 0.3) | (98.1 ± 1.0) | (98.3 ± 1.6) | (98.3 ± 1.2) | ||
| l-NMMA c | 93.3 ± 2.2 | 91.4 ± 0.8 | 68.9 ± 4.5 ** | 43.1 ± 1.1 ** | 23.9 |
| (101.5 ± 0.9) | (101.9 ± 0.4) | (98.5 ± 0.9) | (109.4 ± 0.5) | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kikuchi, T.; Ueno, Y.; Hamada, Y.; Furukawa, C.; Fujimoto, T.; Yamada, T.; Tanaka, R. Five New Limonoids from Peels of Satsuma Orange (Citrus reticulata). Molecules 2017, 22, 907. https://doi.org/10.3390/molecules22060907
Kikuchi T, Ueno Y, Hamada Y, Furukawa C, Fujimoto T, Yamada T, Tanaka R. Five New Limonoids from Peels of Satsuma Orange (Citrus reticulata). Molecules. 2017; 22(6):907. https://doi.org/10.3390/molecules22060907
Chicago/Turabian StyleKikuchi, Takashi, Yasuaki Ueno, Yoshino Hamada, Chika Furukawa, Takako Fujimoto, Takeshi Yamada, and Reiko Tanaka. 2017. "Five New Limonoids from Peels of Satsuma Orange (Citrus reticulata)" Molecules 22, no. 6: 907. https://doi.org/10.3390/molecules22060907






