Design, Synthesis and Biological Evaluation of 2-(2-Amino-5(6)-nitro-1H-benzimidazol-1-yl)-N-arylacetamides as Antiprotozoal Agents
Abstract
:1. Introduction
2. Results and Discussion
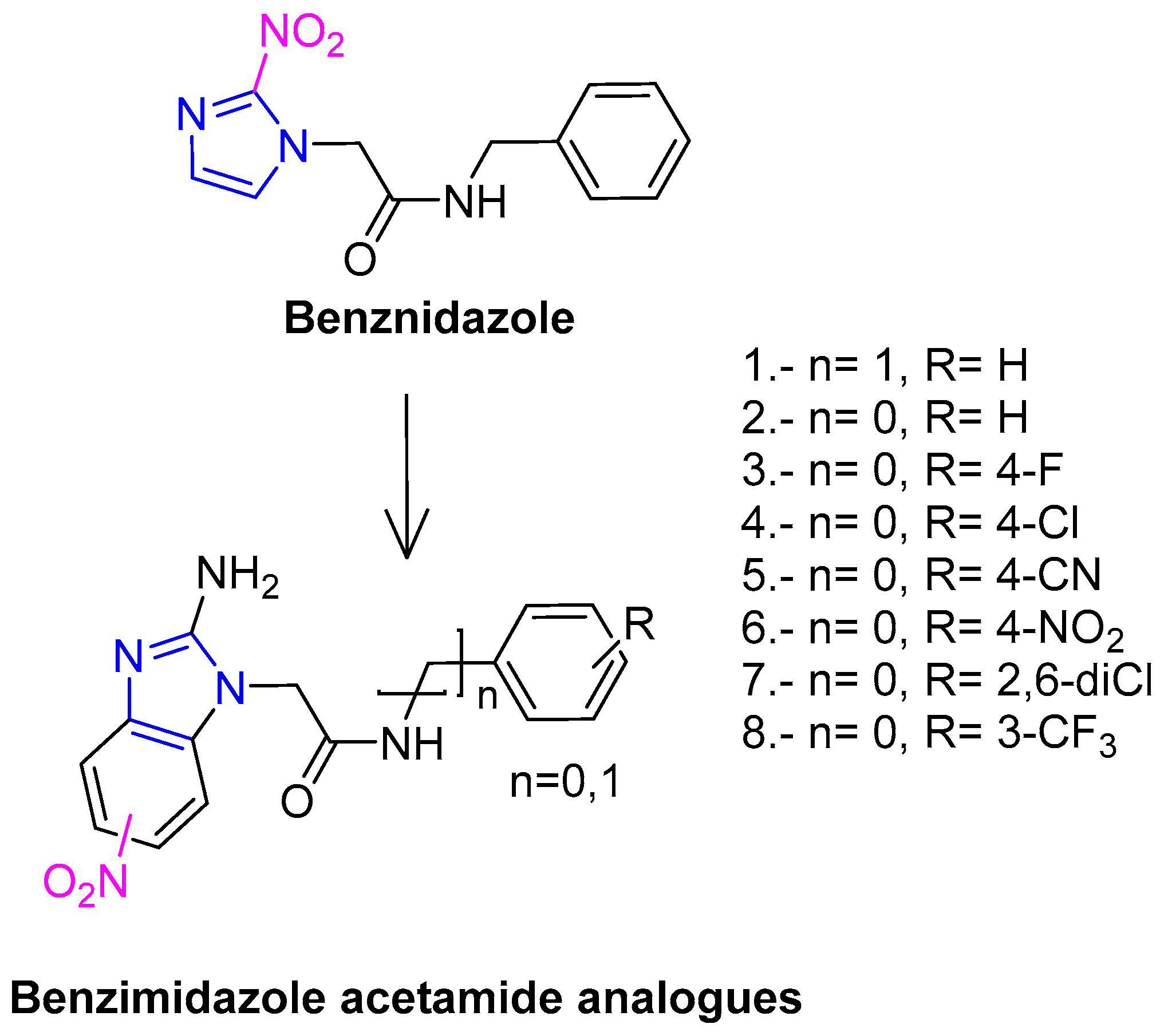
2.1. Drug Design of Benznidazole Derivatives
2.2. Evaluation in Silico (PASS and ACDTox/Suite)
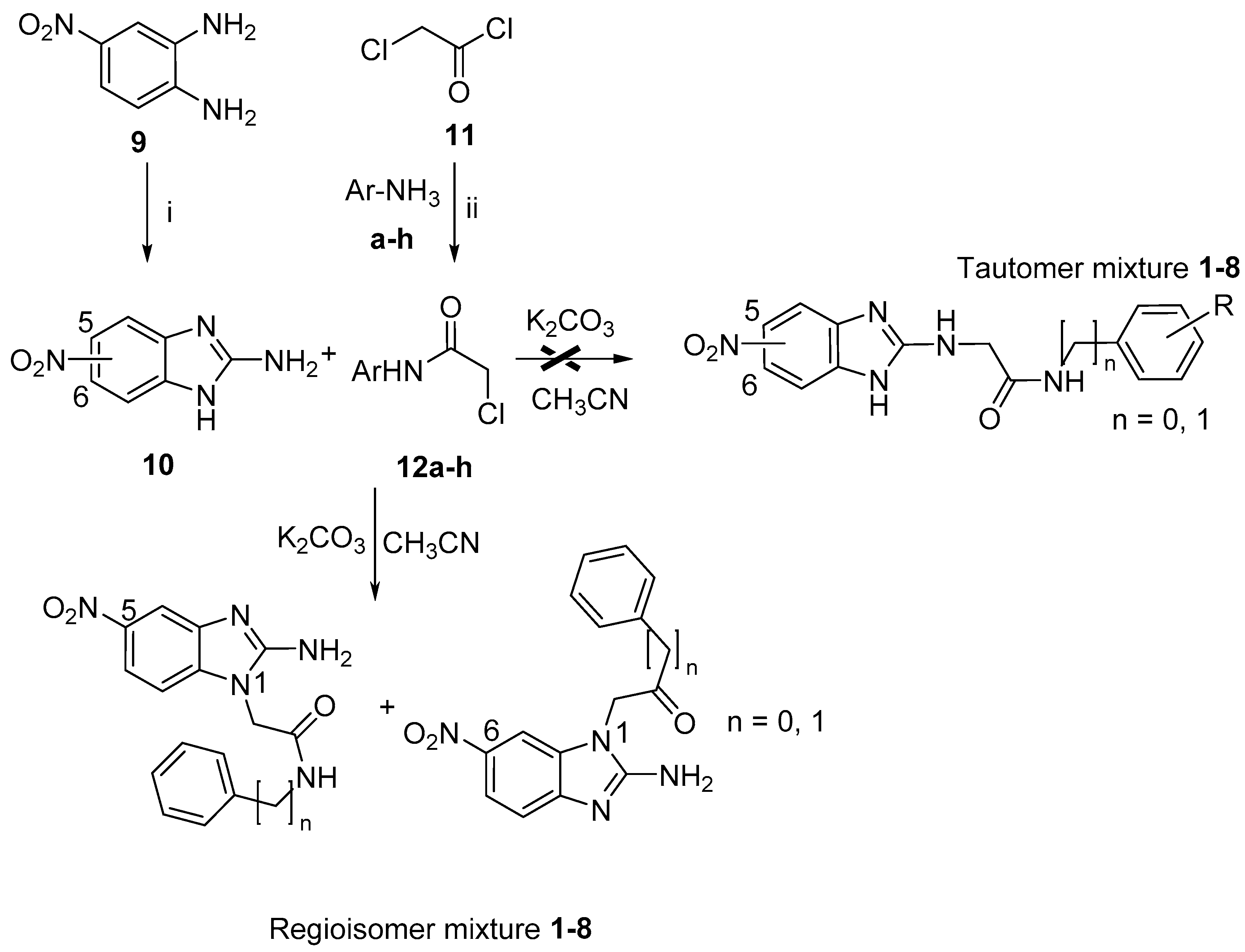
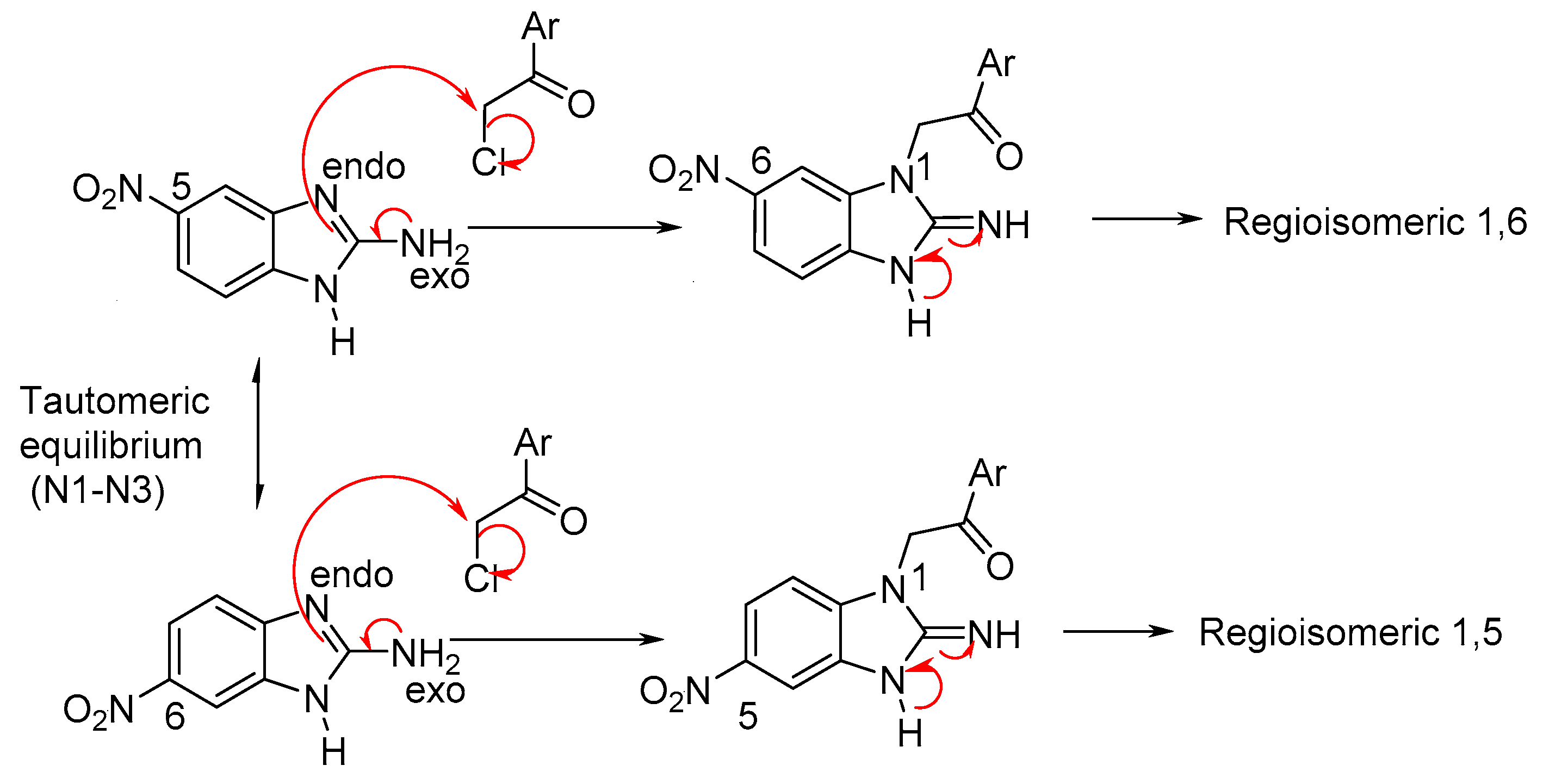
2.3. Chemical Synthesis and Characterization
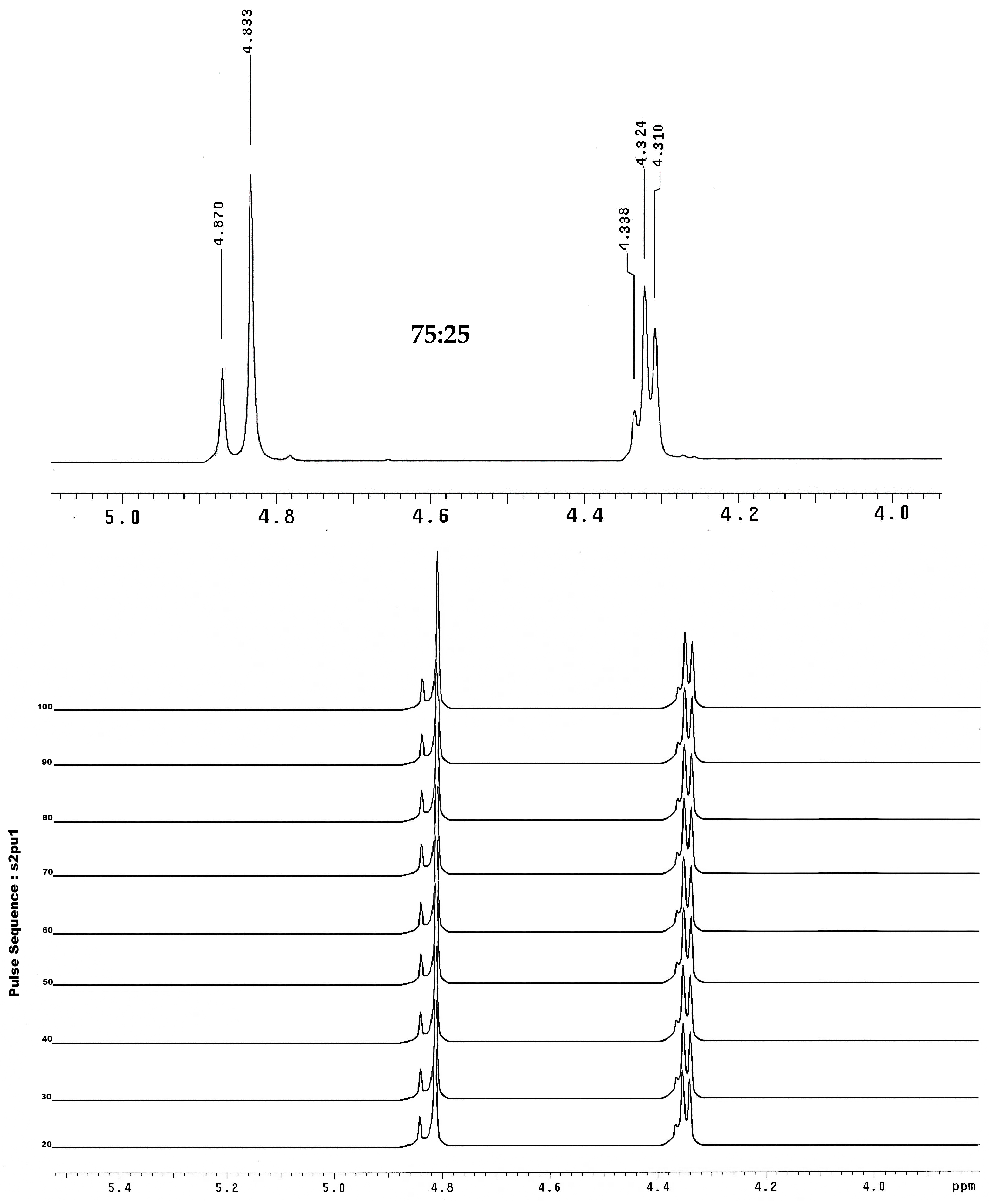
2.4. NMR Prediction
2.5. In Vitro Antiprotozoal Assays
- Physical properties known to be compatible with desirable pharmacokinetics (low molecular weight, favorable Clog P, favorable hydrogen bond donating and accepting capabilities);
- Potency and efficacy, with IC50 values at the low micromolar level;
- Simple synthetic access and thus low production costs;
- Bioisosteric groups improving the likelihood of reasonable solubility.
4. Materials and Methods
4.1. Chemistry
4.2. General Method of Synthesis of 2-(2-Amino-5(6)-nitro-1H-benzimidazol-1-yl)-N-Arylacetamides 1–8
4.3. Biological Assays
4.4. Computational Chemistry
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gupta, C.M.; Thiyagarajan, S.; Sahasrabuddhe, A.A. Unconventional actins and actin-binding proteins in human protozoan parasites. Int. J. Parasitol. 2015, 45, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Boiani, M.; Boiani, L.; Denicola, A.; Torres De Ortiz, S.; Serna, E.; Vera de Bilbao, N.; Sanabria, L.; Yaluff, G.; Rojas de Arias, A.; Vega, C.; et al. 2H-Benzimidazole 1,3-Dioxide Derivatives: A New Family of Water-Soluble Anti-Trypanosomatid Agents. J. Med. Chem. 2006, 49, 3215–3224. [Google Scholar] [CrossRef] [PubMed]
- Bloom, B.E. Recent successes and future predictions on drug repurposing for rare diseases. Expert Opin. Orphan Drugs 2016, 4, 1–4. [Google Scholar] [CrossRef]
- Abdelmohsen, U.R.; Balasubramanian, S.; Oelschlaeger, T.A.; Grkovic, T.; Pham, N.B.; Quinn, R.J.; Hentschel, U. Potential of marine natural products against drug-resistant fungal, viral, and parasitic infections. Lancet Infect. Dis. 2016, 17, e30–e41. [Google Scholar] [CrossRef]
- Jarrad, A.M.; Debnath, A.; Miyamoto, Y.; Hansford, K.A.; Pelingon, R.; Butler, M.S.; Bains, T.; Karoli, T.; Blaskovich, M.A.T.; Eckmann, L.; et al. Nitroimidazole carboxamides as antiparasitic agents targeting Giardia lamblia, Entamoeba histolytica and Trichomonas vaginalis. Eur. J. Med. Chem. 2016, 120, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Ibezim, A.; Nwodo, N.J.; Nnaji, N.J.N.; Ujam, O.T.; Olubiyi, O.O.; Mba, C.J. In silico investigation of morpholines as novel class of trypanosomal triosephosphate isomerase inhibitors. Med. Chem. Res. 2017, 26, 180–189. [Google Scholar] [CrossRef]
- Navarrete-Vázquez, G.; Rojano-Vilchis, M.D.M.; Yépez-Mulia, L.; Meléndez, V.; Gerena, L.; Hernández-Campos, A.; Castillo, R.; Hernández-Luis, F. Synthesis and antiprotozoal activity of some 2-(trifluoromethyl)-1H-benzimidazole bioisosteres. Eur. J. Med. Chem. 2006, 41, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Boiani, M.; González, M. Imidazole and Benzimidazole Derivatives as Chemotherapeutic Agents. Mini-Rev. Med. Chem. 2005, 5, 409–424. [Google Scholar] [CrossRef] [PubMed]
- Pradines, B.; Gallard, J.F.; Iorga, B.I.; Gueutin, C.; Loiseau, P.M.; Ponchel, G.; Bouchemal, K. Investigation of the complexation of albendazole with cyclodextrins for the design of new antiparasitic formulations. Carbohydr. Res. 2014, 398, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Molina, I.; Gómez i Prat, J.; Salvador, F.; Treviño, B.; Sulleiro, E.; Serre, N.; Pou, D.; Roure, S.; Cabezos, J.; Valerio, L.; et al. Randomized trial of posaconazole and benznidazole for chronic Chagas’ disease. N. Engl. J. Med. 2014, 370, 1899–1908. [Google Scholar] [CrossRef] [PubMed]
- Nava-Zuazo, C.; Chávez-Silva, F.; Moo-Puc, R.; Chan-Bacab, M.J.; Ortega-Morales, B.O.; Moreno-Díaz, H.; Díaz-Coutiño, D.; Hernández-Nuñez, E.; Navarrete-Vázquez, G. 2-Acylamino-5-nitro-1,3-thiazoles: Preparation and in vitro bioevaluation against four neglected protozoan parasites. Bioorg. Med. Chem. 2014, 22, 1626–1633. [Google Scholar] [CrossRef] [PubMed]
- Bernardino, A.M.R.; Gomes, A.O.; Charret, K.S.; Freitas, A.C.C.; Machado, G.M.C.; Canto-Cavalheiro, M.M.; Leon, L.L.; Amaral, V.F. Synthesis and leishmanicidal activities of 1-(4-X-phenyl)-N′-[(4-Y-phenyl)methylene]-1H-pyrazole-4-carbohydrazides. Eur. J. Med. Chem. 2006, 41, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Ashburn, T.T.; Thor, K.B. Drug repositioning: Identifying and developing new uses for existing drugs. Nat. Rev. Drug Discov. 2004, 3, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Raether, W.; Hänel, H. Nitroheterocyclic drugs with broad spectrum activity. Parasitol. Res. 2003, 90 (Suppl. S1), S19–S39. [Google Scholar] [PubMed]
- Shaukat, A.; Mirza, H.M.; Ansari, A.H.; Yasinzai, M.; Zaidi, S.Z.; Dilshad, S.; Ansari, F.L. Benzimidazole derivatives: Synthesis, leishmanicidal effectiveness, and molecular docking studies. Med. Chem. Res. 2013, 22, 3606–3620. [Google Scholar] [CrossRef]
- Bai, Y.-B.; Zhang, A.-L.; Tang, J.-J.; Gao, J.-M. Synthesis and antifungal activity of 2-chloromethyl-1H-benzimidazole derivatives against phytopathogenic fungi in vitro. J. Agric. Food Chem. 2013, 61, 2789–2795. [Google Scholar] [CrossRef] [PubMed]
- Sathaiah, G.; Ravi Kumar, A.; Chandra Shekhar, A.; Raju, K.; Shanthan Rao, P.; Narsaiah, B.; Raghuram Reddy, A.; Lakshmi, D.; Sridhar, B. Design and synthesis of positional isomers of 1-alkyl-2-trifluoromethyl-5 or 6-substituted benzimidazoles and their antimicrobial activity. Med. Chem. Res. 2013, 22, 1229–1237. [Google Scholar] [CrossRef]
- Tavman, A.; Cinarli, A.; Gürbüz, D.; Birteksöz, A.S. Synthesis, characterization and antimicrobial activity of 2-(5-H/Me/F/Cl/NO2–1H-benzimidazol-2-yl)-benzene-1,4-diols and some transition metal complexes. J. Iran. Chem. Soc. 2015, 29, 63–74. [Google Scholar] [CrossRef]
- Satyendra, R.V.; Vishnumurthy, K.A.; Vagdevi, H.M.; Shruthi, A. Synthesis, in vitro anthelmintic, and molecular docking studies of novel 5-nitro benzoxazole derivatives. Med. Chem. Res. 2011, 46, 3078–3084. [Google Scholar] [CrossRef]
- Shaker, Y.M.; Omar, M.A.; Mahmoud, K.; Elhallouty, S.M.; El-Senousy, W.M.; Ali, M.M.; Mahmoud, A.E.; Abdel-Halim, A.H.; Soliman, S.M.; El Diwani, H.I. Synthesis, in vitro and in vivo antitumor and antiviral activity of novel 1-substituted benzimidazole derivatives. J. Enzym. Inhib. Med. Chem. 2015, 30, 826–845. [Google Scholar] [CrossRef] [PubMed]
- Mavrova, A.T.; Yancheva, D.; Anastassova, N.; Anichina, K.; Zvezdanovic, J.; Djordjevic, A.; Markovic, D.; Smelcerovic, A. Synthesis, electronic properties, antioxidant and antibacterial activity of some new benzimidazoles. Bioorg. Med. Chem. 2015, 23, 6317–6326. [Google Scholar] [CrossRef] [PubMed]
- Gaba, M.; Singh, S.; Mohan, C. Benzimidazole: An emerging scaffold for analgesic and anti-inflammatory agents. Eur. J. Med. Chem. 2014, 76, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Da, Y.; Wu, D.; Zheng, H.; Zhu, L.; Wang, L.; Yan, Y.; Chen, Z. Design, synthesis and biological evaluation of new 5-nitro benzimidazole derivatives as AT1 antagonists with anti-hypertension activities. Bioorg. Med. Chem. 2014, 22, 2294–2302. [Google Scholar] [CrossRef] [PubMed]
- Bern, C.; Montgomery, S.P.; Herwaldt, B.L.; Rassi, A., Jr.; Marin-Neto, J.A.; Dantas, R.O.; Maguire, J.H.; Acquatella, H.; Morillo, C.; Kirchhoff, L.V.; et al. Evaluation and Treatment of Chagas Disease in the United States. A Systematic Review. Clin. Rev. 2007, 298, 2171–2181. [Google Scholar]
- Jannin, J.; Villa, L. An overview of Chagas disease treatment. Memórias do Instituto Oswaldo Cruz 2007, 102, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Núñez, E.; Tlahuext, H.; Moo-Puc, R.; Torres-Gómez, H.; Reyes-Martínez, R.; Cedillo-Rivera, R.; Nava-Zuazo, C.; Navarrete-Vazquez, G. Synthesis and in vitro trichomonicidal, giardicidal and amebicidal activity of N-acetamide(sulfonamide)-2-methyl-4-nitro-1H-imidazoles. Eur. J. Med. Chem. 2009, 44, 2975–2984. [Google Scholar] [CrossRef] [PubMed]
- Fuson, R.C. The Principle of Vinylogy. Chem. Rev. 1935, 16, 1–27. [Google Scholar] [CrossRef]
- Gaikwad, D.D.; Chapolikar, A.D.; Devkate, C.G.; Warad, K.D.; Tayade, A.P.; Pawar, R.P.; Domb, A.J. Synthesis of indazole motifs and their medicinal importance: An overview. Eur. J. Med. Chem. 2015, 90, 707–731. [Google Scholar] [CrossRef] [PubMed]
- Van de Waterbeemd, H.; Gifford, E. ADMET in silico modelling: Towards prediction paradise? Nat. Rev. Drug Discov. 2003, 2, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Bakhtyari, N.G.; Raitano, G.; Benfenati, E.; Martin, T.; Young, D. Comparison of in Silico Models for Prediction of Mutagenicity. J. Environ. Sci. Health Part C 2013, 31, 45–66. [Google Scholar] [CrossRef] [PubMed]
- Cronin, M.T.D. Computational Toxicology is Now Inseparable from Experimental Toxicology. J. Environ. Biol. 1999, 20, 1–4. [Google Scholar]
- Kulkarni, R.G.; Laufer, S.A.; Chandrashekhar, V.M.; Garlapati, A. Synthesis, p38 Kinase Inhibitory and Anti-inflammatory Activity of New Substituted Benzimidazole Derivatives. Med. Chem. 2013, 9, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Petrova, O.N.; Zamigajlo, L.L.; Ostras, K.S.; Shishkina, S.V.; Shishkin, O.V.; Borisov, A.V.; Musatov, V.I.; Shirobokova, M.G.; Lipson, V.V. Multicomponent Reaction of 2-aminobenzimidazole, Arylglyoxals, and 1,3-cyclohexanedione. Chem. Heterocycl. Compd. 2015, 51, 310–319. [Google Scholar] [CrossRef]
- Chen, Y.; Willis, M.C. An Aryne-Based Route to Substituted Benzoisothiazoles. Org. Lett. 2015, 17, 4786–4789. [Google Scholar] [CrossRef] [PubMed]
- Duckworth, D.M.; Lee-wong, S.; Slawin, A.M.Z.; Smith, E.H.; Williams, D.J. Co-cyclizations of nitrogen-containing acetylenes induced by a nickel triphenylphosphine complex to give aminoindane, isoindoline and isoindolinone derivatives. J. Chem. Soc. Perkin Trans. 1 1996, 1, 815–821. [Google Scholar] [CrossRef]
- Perchellet, E.M.; Wang, Y.; Lou, K.; Zhao, H.; Battina, S.K.; Hua, D.H.; Perchellet, J.P.H. Antitumor triptycene analogs directly interact with isolated mitochondria to rapidly trigger markers of permeability transition. Anticancer Res. 2007, 27, 3259–3271. [Google Scholar] [PubMed]
- Meng, X.; Li, X.; Chen, W.; Zhang, Y.; Wang, W.; Chen, J.; Song, J.; Feng, H.; Chen, B. Facile One-Pot Synthesis of N-Alkylated Benzimidazole and Benzotriazole from Carbonyl Compounds. J. Heterocycl. Chem. 2012, 49, 1458–1461. [Google Scholar]
- Wolinski, K.; Hinton, J.F.; Pulay, P. Efficient Implementation of the Gauge-Independent Atomic Orbital Method for NMR Chemical Shift Calculations. J. Am. Chem. Soc. 1990, 112, 8251. [Google Scholar] [CrossRef]
- Valiev, M.; Bylaska, E.J.; Govind, N.; Kowalski, K.; Straatsma, T.P.; Van Dam, H.J.J.; Wang, D.; Nieplocha, J.; Apra, E.; Windus, T.L.; et al. NWChem: A comprehensive and scalable open-source solution for large scale molecular simulations. Comput. Phys. Commun. 2010, 181, 1477–1489. [Google Scholar] [CrossRef]
- Stephens, P.J.; Devlin, F.J.; Chabalowski, C.F.; Frisch, M.J. Ab Initio Calculation of Vibrational Absorption and Circular Dichroism Spectra Using Density Functional Force Fields. J. Phys. Chem. 1994, 98, 11623–11627. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A. Drug-like properties and the causes of poor solubility and poor permeability. J. Pharmacol. Toxicol. Methods 2000, 44, 235–249. [Google Scholar] [CrossRef]
- Lipinski, C.A. Lead- and drug-like compounds: The rule-of-five revolution. Drug Discov. Today Technol. 2004, 1, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Lagunin, A.; Stepanchikova, A.; Filimonov, D.; Poroikov, V. PASS: Prediction of activity spectra for biologically active substances. Bioinformatics 2000, 16, 747–748. [Google Scholar] [CrossRef] [PubMed]
- Cedillo-Rivera, R.; Chávez, B.; González-Robles, A.; Tapia, A.; Yepez-mulia, L. In Vitro Effect of Nitazoxanide Against Entamoeba histolytica, Giardia intestinalis and Trichomonas vaginalis Trophozoites. J. Eukaryot. Microbiol. 2002, 49, 201–208. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of all compounds are available from the authors. |

| Compound | Antiparasitic Effect (Trichomonas vaginalis) | LD50 (mg/Kg) | ||||
|---|---|---|---|---|---|---|
| Mouse | Rat | |||||
| Pa | Pi | ip | p.o. | ip | po | |
| 1 | 0.313 | 0.023 | 300 | 1300 | 430 | 1200 |
| 2 | 0.319 | 0.020 | 460 | 1600 | 330 | 830 |
| 3 | 0.250 | 0.065 | 490 | 830 | 530 | 670 |
| 4 | 0.304 | 0.027 | 490 | 840 | 460 | 970 |
| 5 | 0.201 | 0.122 | 380 | 960 | 800 | 320 |
| 6 | 0.324 | 0.018 | 430 | 1200 | 1200 | 670 |
| 7 | 0.292 | 0.033 | 290 | 920 | 280 | 900 |
| 8 | 0.201 | 0.122 | 280 | 1100 | 440 | 750 |
| 10 | 0.333 | 0.015 | 150 | 1200 | 480 | 620 |
| Bnz | 0.705 | 0.002 | 1100 | 280 | 1300 | 1100 |
| Cmpd | m.p. (°C) | Reaction Time (h) | Yield (%) | MS FAB (+) | IC50 (μM) | |||
|---|---|---|---|---|---|---|---|---|
| Reigoisomers Mixture Ratio | T. vaginalis | G. lamblia | E. histolytica | |||||
| 1 | 279.0–282.0 | 5.0 | 90.1 | 326 | 75:25 | 11.34 | 11.26 | 18.43 |
| 2 | 285.0–287.8 | 5.0 | 80.1 | 297 | 60:40 | 26.76 | 10.44 | 46.16 |
| 3 | 313.0–315.0 | 7.0 | 72.0 | 330 | 70:30 | 15.61 | 16.47 | 26.14 |
| 4 | 294.0–296.0 | 12.0 | 92.1 | 345 | 69:31 | 14.66 | 15.26 | 28.28 |
| 5 | 286.0–289.0 | 8.0 | 81.5 | 337 | 57:43 | 11.16 | 14.45 | 21.07 |
| 6 | 309.0–312.0 | 8.0 | 79.8 | 357 | 59:41 | 7.09 | 16.52 | 13.23 |
| 7 | 303.0–306.0 | 18.0 | 61.4 | 381 | 55:45 | 5.92 | 9.20 | 34.01 |
| 8 | 282.0–284.0 | 18.0 | 60.7 | 380 | 61:39 | 5.62 | 3.84 | 45.60 |
| 10 | 192.0–194.0 | 5.0 | 94.1 | 179 | 50:50 * | 22.12 | 28.39 | 46.80 |
| Bnz | --- | --- | --- | --- | --- | 18.62 | 22.58 | 4.27 |

| Label | Experimental (δ, DMSO-d6) | In Silico (δ, DMSO-d6) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1H-NMR | 13C-NMR | 1H-NMR | 13C-NMR | |||||||||
| a | b | a | b | a | b | c | d | a | b | c | d | |
| 1 | --- | --- | --- | --- | --- | --- | 6.89 | 7.13 | --- | --- | --- | --- |
| 2 | --- | 158.05 | 159.58 | --- | --- | --- | --- | 163.58 | 166.01 | 161.77 | 158.55 | |
| 3 | --- | --- | --- | --- | --- | --- | --- | --- | --- | --- | --- | --- |
| 4 | 7.93 | 7.97 | 114.67 | 118.04 | 6.75 | 7.52 | 7.54 | 7.18 | 120.50 | 121.30 | 121.08 | 119.48 |
| 5 | 7.87 | --- | 140.14 | 138.70 | --- | 8.57 | 8.60 | --- | 152.59 | 148.66 | 149.48 | 152.78 |
| 6 | --- | 7.95 | 109.32 | 113.38 | 8.38 | --- | --- | 8.44 | 121.72 | 125.74 | 126.13 | 121.83 |
| 7 | 7.19 | 7.20 | 107.06 | 103.48 | 8.74 | 8.10 | 8.29 | 8.71 | 107.17 | 106.15 | 108.73 | 109.66 |
| 8 | --- | --- | 141.88 | 141.88 | --- | --- | --- | --- | 145.53 | 140.09 | 138.02 | 144.21 |
| 9 | --- | --- | 142.81 | 149.73 | --- | --- | --- | --- | 151.13 | 157.75 | 183.91 | 151.70 |
| 10 | 7.03 | 7.03 | --- | --- | 5.54 | 5.82 | 3.81 | 5.77 | --- | --- | --- | --- |
| 11 | 4.83 | 4.87 | 44.87 | 44.72 | 4.30 | 4.31 | 4.25 | 3.74 | 50.92 | 50.93 | 49.35 | 48.10 |
| 12 | --- | --- | 165.93 | 166.08 | --- | --- | --- | --- | 173.12 | 173.57 | 172.21 | 169.12 |
| 13 | 8.73 | 8.78 | --- | --- | 5.15 | 5.32 | 4.98 | 4.61 | --- | --- | --- | --- |
| 14 | 4.32 | 4.33 | 42.41 | 42.39 | 4.61 | 4.62 | 4.60 | 4.77 | 48.44 | 48.56 | 47.53 | 48.10 |
| 1′ | --- | --- | 138.77 | 134.31 | --- | --- | --- | --- | 145.57 | 144.75 | 147.96 | 147.03 |
| 2′ | 7.23–7.36 | 127.20 | 127.15 | 7.48 | 7.50 | 7.44 | 7.63 | 135.56 | 135.36 | 135.06 | 134.90 | |
| 3′ | 7.23–7.36 | 127.20 | 128.19 | 7.64 | 7.63 | 7.75 | 7.69 | 134.15 | 134.59 | 134.90 | 134.12 | |
| 4′ | 7.23–7.36 | 126.79 | 127.13 | 7.62 | 7.61 | 7.70 | 7.68 | 134.88 | 134.92 | 133.23 | 134.04 | |
| 5′ | 7.23–7.36 | 127.20 | 128.19 | 7.63 | 7.62 | 7.63 | 7.74 | 134.30 | 134.11 | 134.47 | 134.53 | |
| 6′ | 7.23–7.36 | 127.20 | 127.15 | 7.50 | 7.46 | 7.72 | 7.47 | 135.25 | 135.38 | 136.46 | 135.65 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Núñez, E.; Tlahuext, H.; Moo-Puc, R.; Moreno, D.; González-Díaz, M.O.; Vázquez, G.N. Design, Synthesis and Biological Evaluation of 2-(2-Amino-5(6)-nitro-1H-benzimidazol-1-yl)-N-arylacetamides as Antiprotozoal Agents. Molecules 2017, 22, 579. https://doi.org/10.3390/molecules22040579
Hernández-Núñez E, Tlahuext H, Moo-Puc R, Moreno D, González-Díaz MO, Vázquez GN. Design, Synthesis and Biological Evaluation of 2-(2-Amino-5(6)-nitro-1H-benzimidazol-1-yl)-N-arylacetamides as Antiprotozoal Agents. Molecules. 2017; 22(4):579. https://doi.org/10.3390/molecules22040579
Chicago/Turabian StyleHernández-Núñez, Emanuel, Hugo Tlahuext, Rosa Moo-Puc, Diego Moreno, María Ortencia González-Díaz, and Gabriel Navarrete Vázquez. 2017. "Design, Synthesis and Biological Evaluation of 2-(2-Amino-5(6)-nitro-1H-benzimidazol-1-yl)-N-arylacetamides as Antiprotozoal Agents" Molecules 22, no. 4: 579. https://doi.org/10.3390/molecules22040579







