HPLC Analysis and Biochemical Characterization of LOX from Eschscholtzia californica Cham.
Abstract
:1. Introduction
2. Results and Discussion
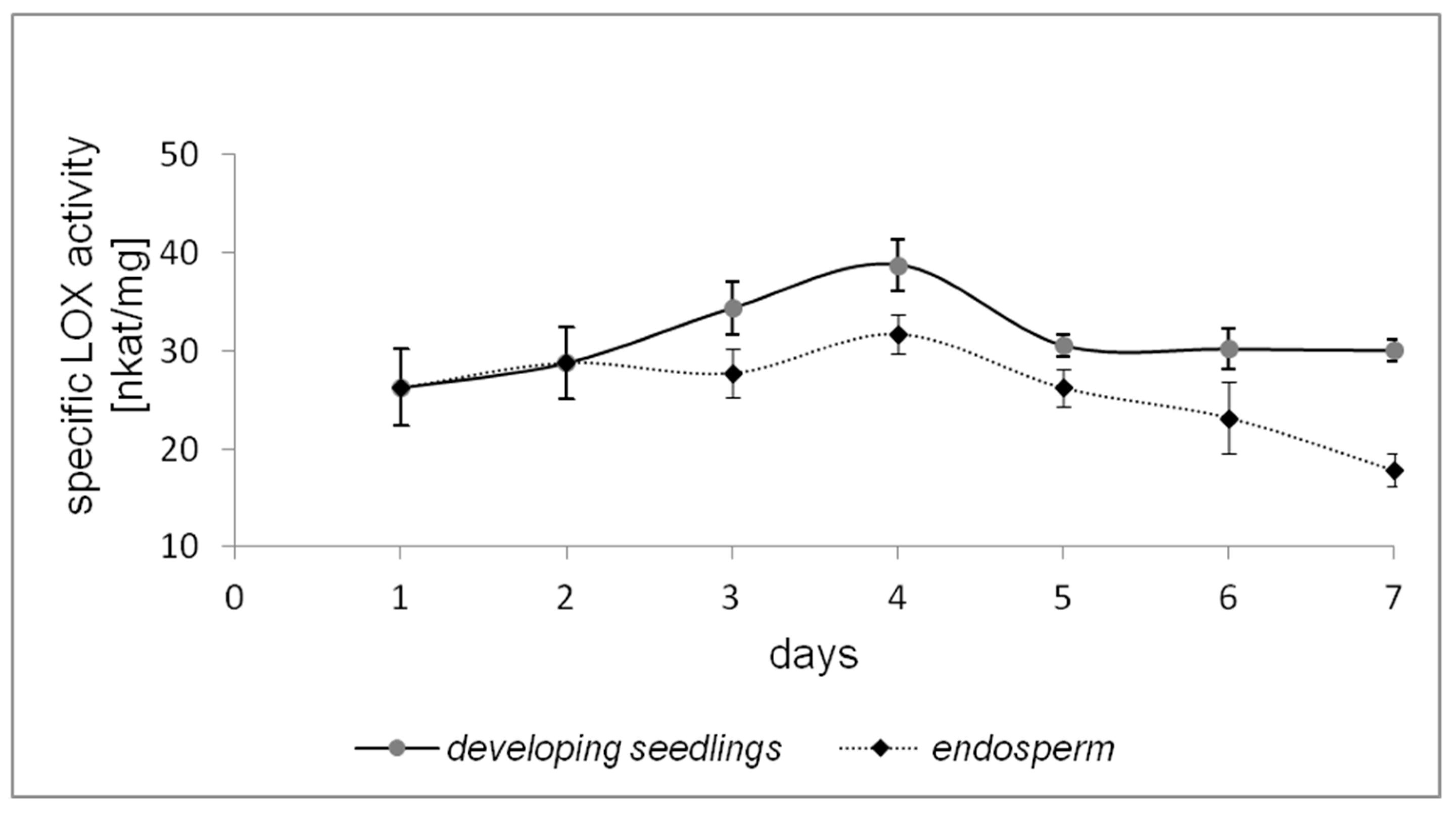
2.1. LOX Activity during Germination of California Poppy Seeds
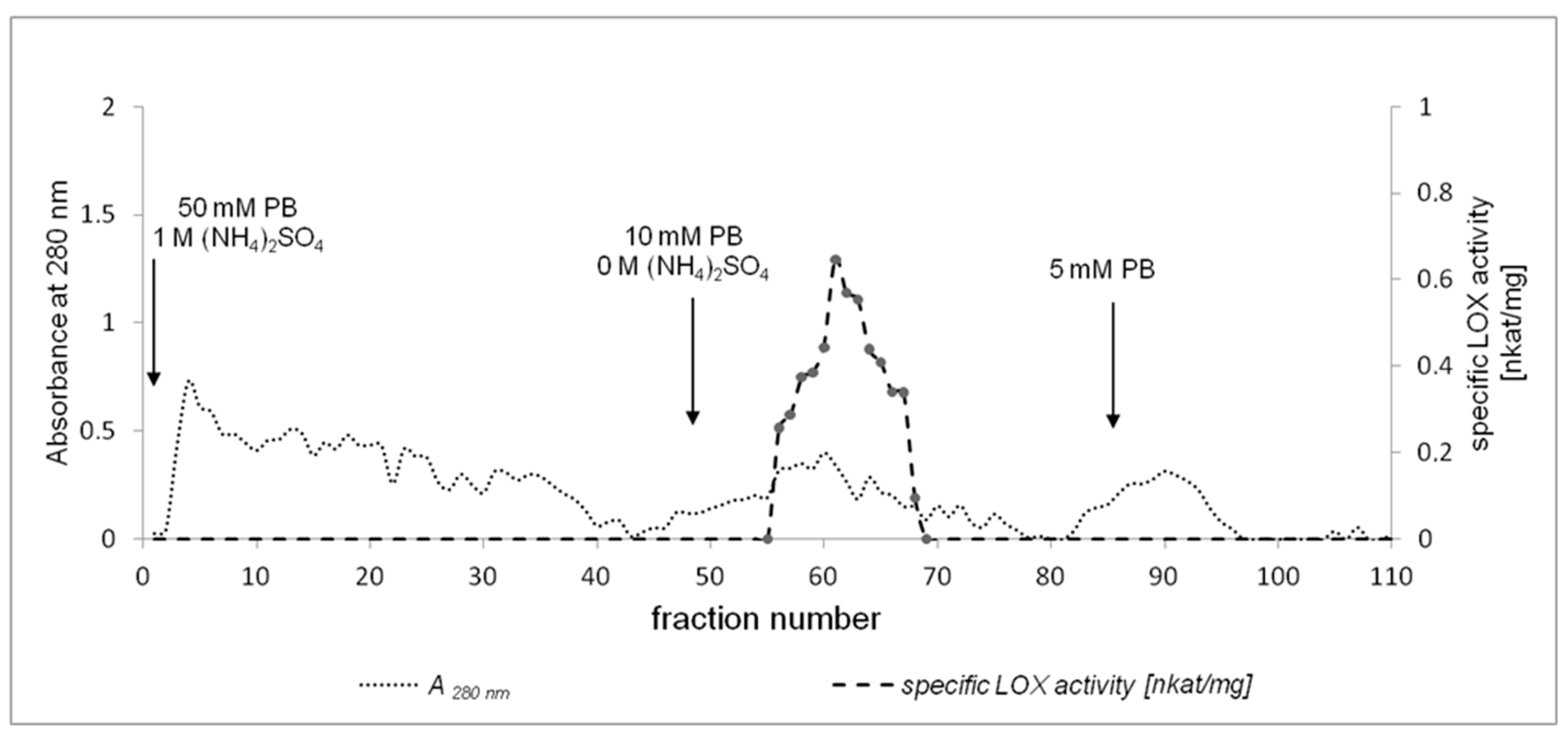
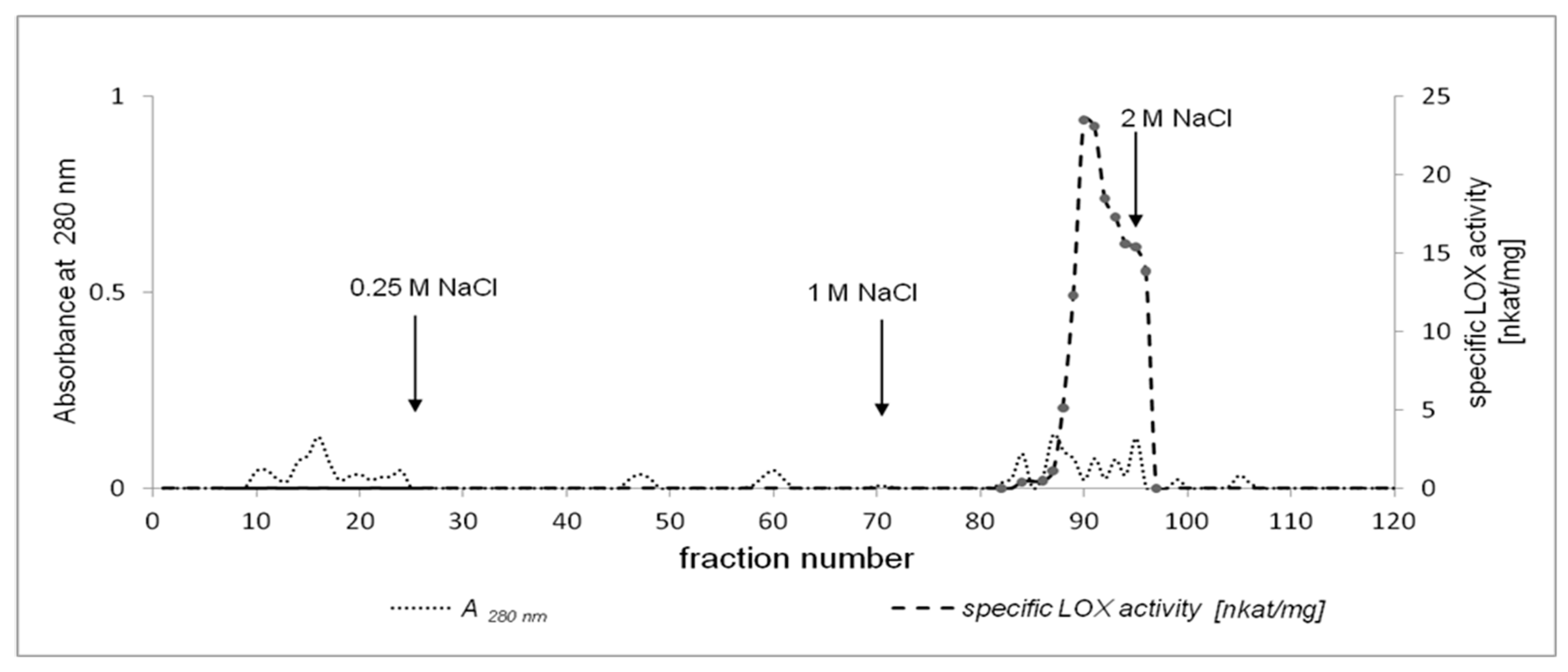
2.2. Purification of California Poppy LOX
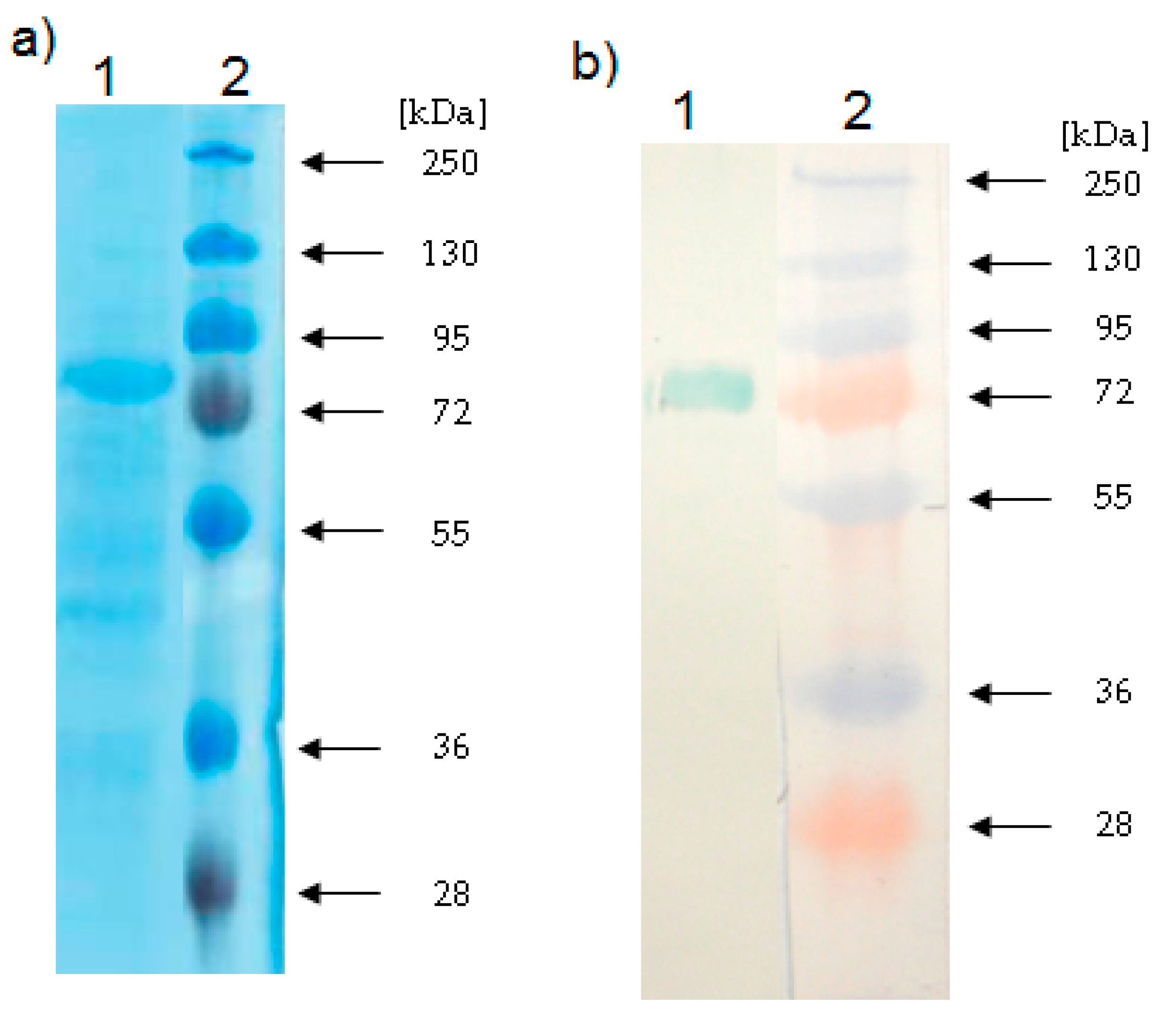
2.3. Determination of the Relative Molecular Mass of the LOX
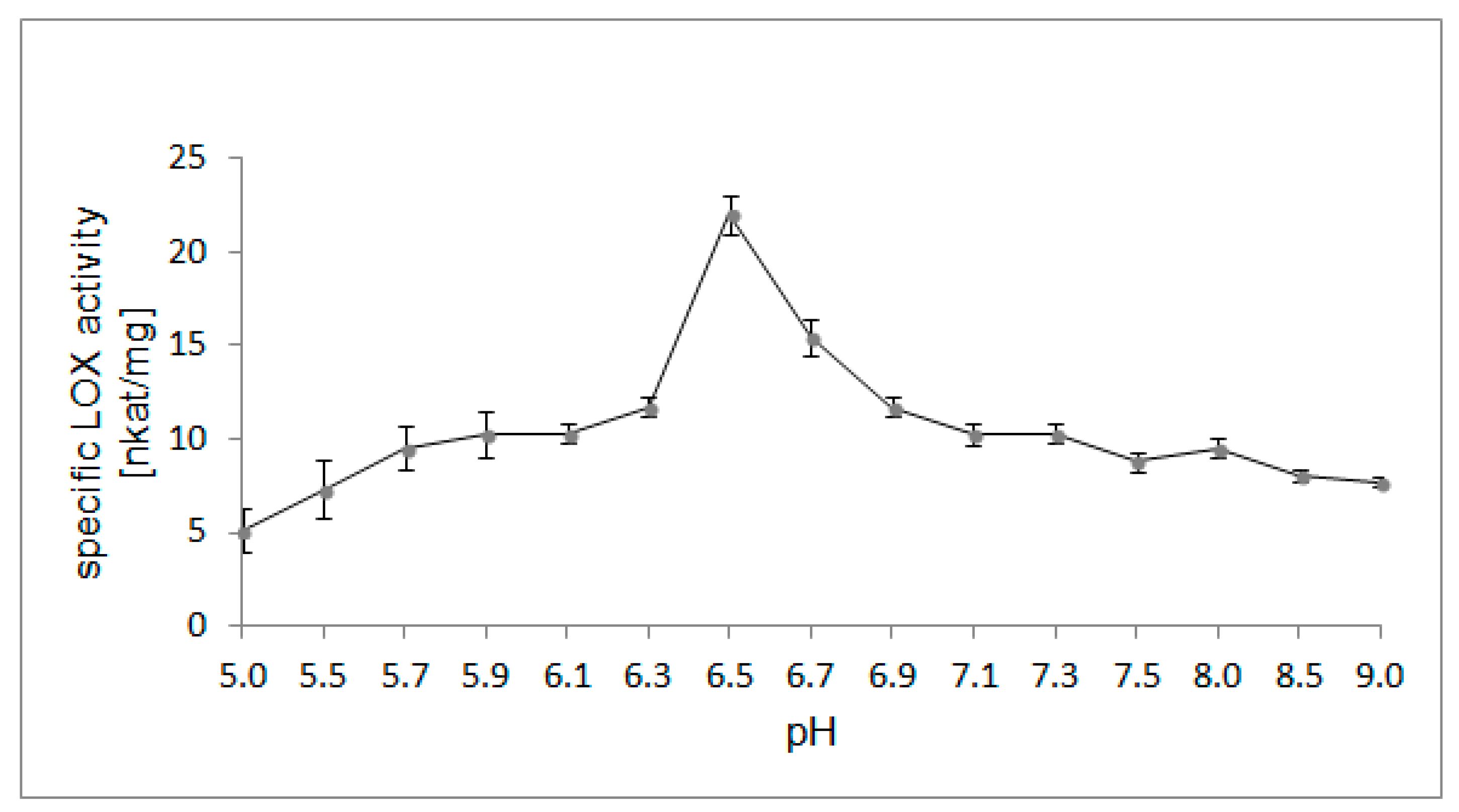
2.4. Optimal pH for LOX Activity
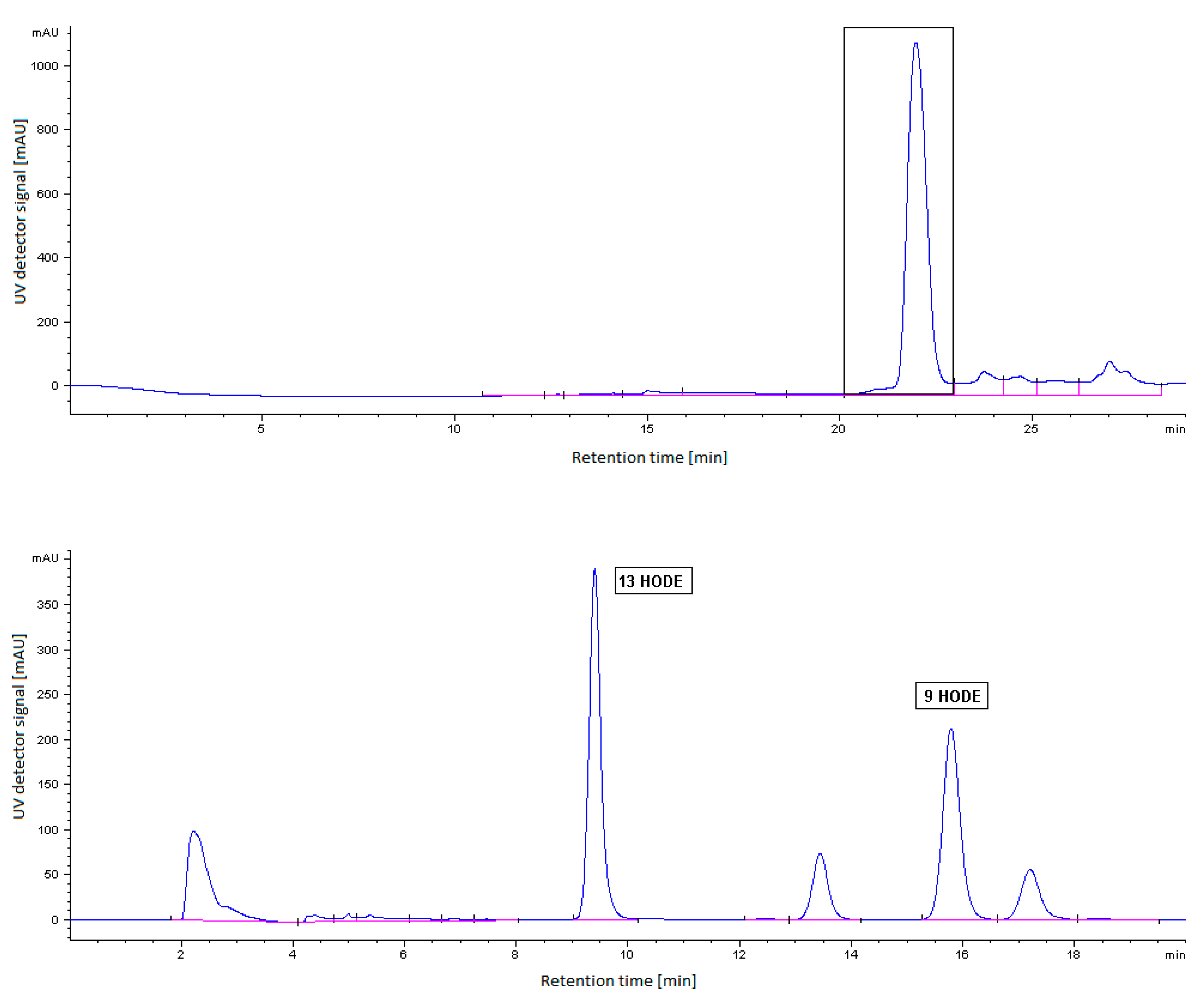
2.5. HPLC Analysis of LOX Reaction Products
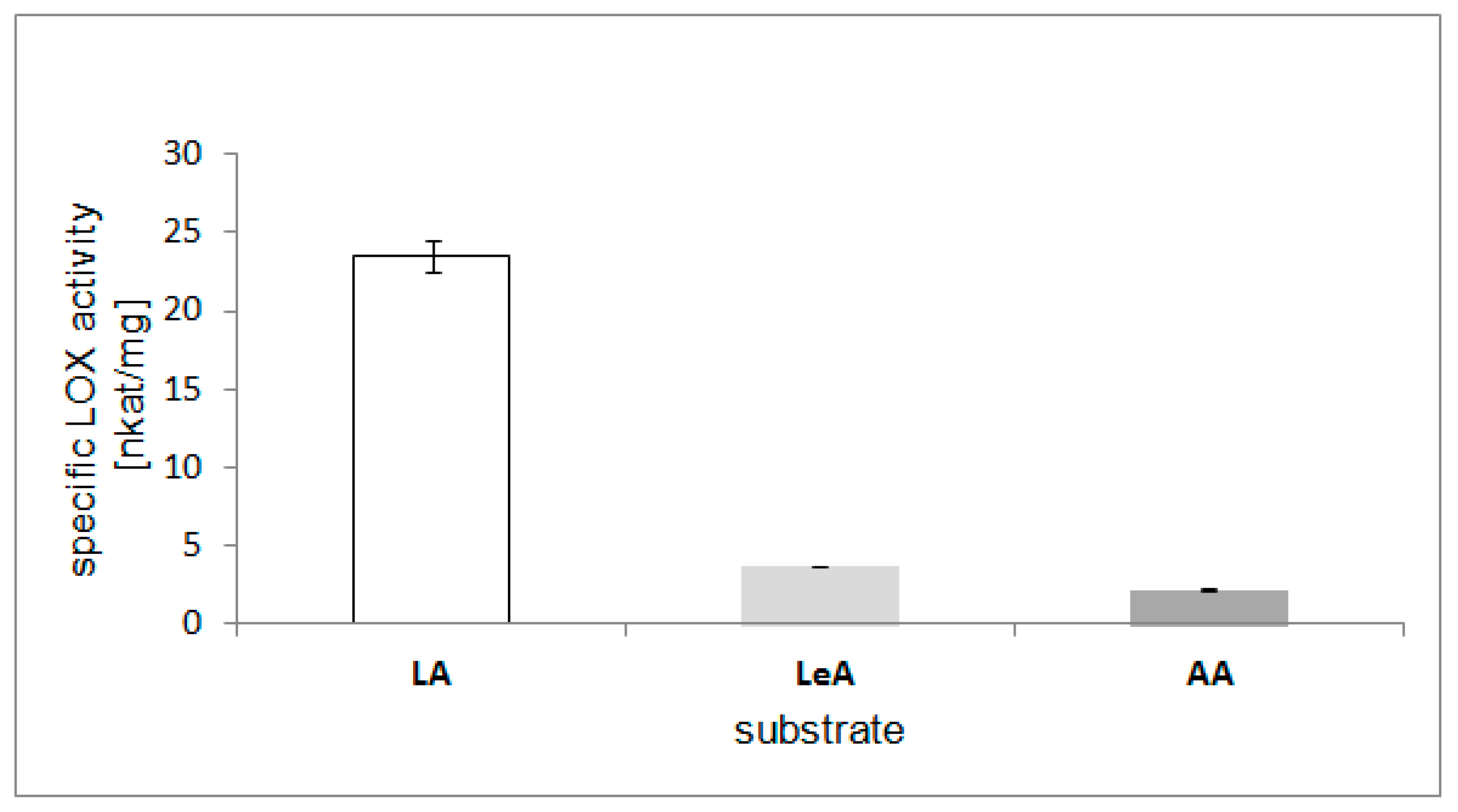
2.6. Substrate Specificity of the LOX
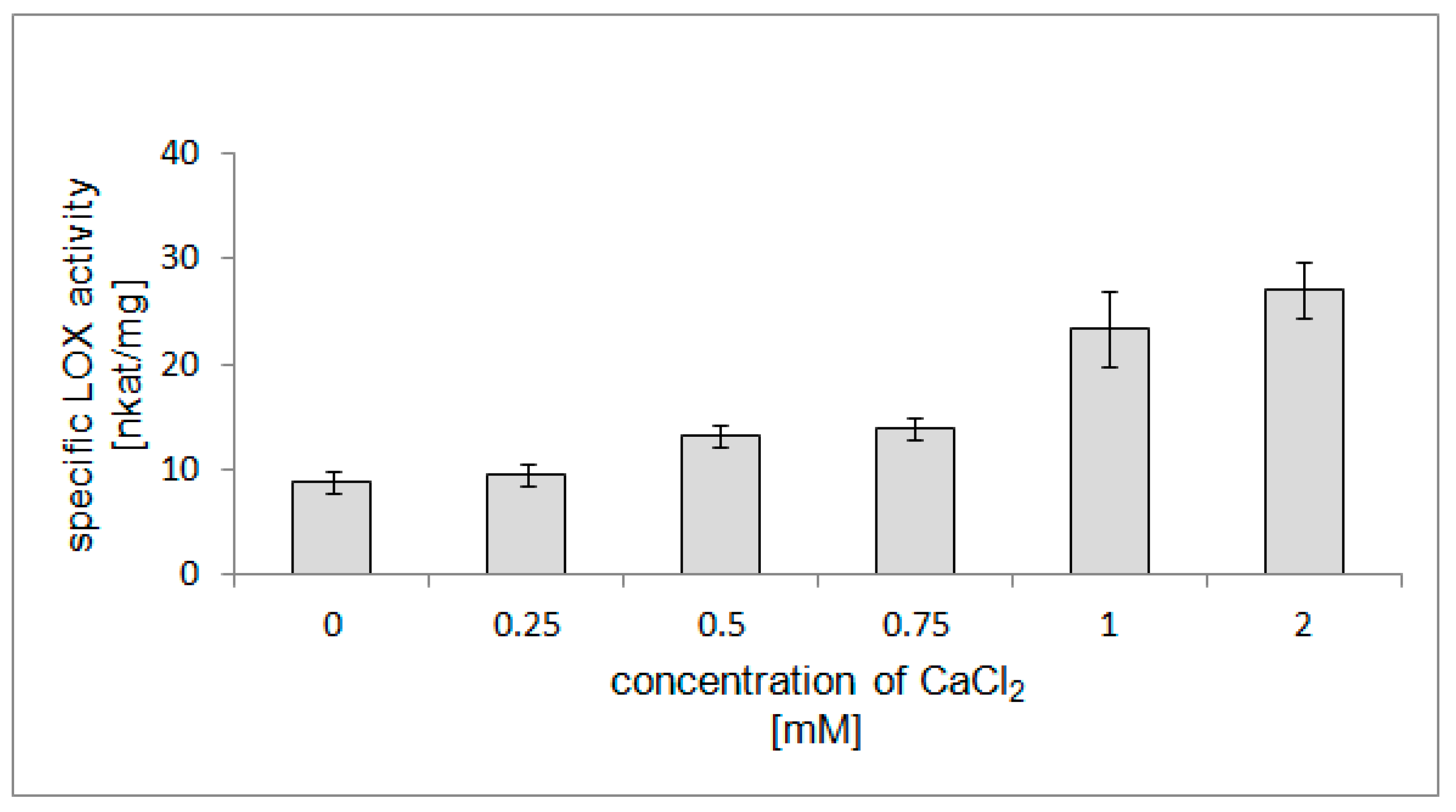
2.7. Stimulatory Effect of Ca2+ Ions on LOX Activity
2.8. Kinetic Paramaters
3. Materials and Methods
3.1. Plant Material
3.2. Preparation of Crude Extracts
3.3. Enzyme Purification
3.4. Lipoxygenase Activity Assay and Protein Determination
3.5. HPLC Analysis of LOX Positional Specificity
3.6. Effect of pH and Ca2+ Ions on LOX Activity
3.7. Kinetic Assay of Purified LOX
3.8. Substrate Specificity
3.9. Electrophoresis and Western Blotting
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fabre, N.; Claparols, C.; Richelme, S.; Angelin, M.-L.; Fourasté, I.; Moulis, C. Direct characterization of isoquinoline alkaloids in a crude plant extract by ion-pair liquid chromatography—Electrospray ionization tandem mass spectrometry: Example of Eschscholtzia californica. J. Chromarogr. A 2000, 904, 35–46. [Google Scholar] [CrossRef]
- Kollárová, R.; Obložinský, M.; Kováčiková, V.; Holková, I.; Balažová, A.; Pekárová, M.; Hoffman, P.; Bezáková, L. Lipoxygenase activity and sanguinarine production in cell suspension cultures of California poppy (Eschscholtzia californica CHAM.). Pharmazie 2014, 69, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Villegas, M.; Sommarin, M.; Brodelius, P.E. Effects of sodium orthovanadate on benzophenantridine alkaloid formation and distribution in cell suspension cultures of Eschscholtzia californica. Plant Physiol. Biochem. 2000, 38, 233–241. [Google Scholar] [CrossRef]
- Liavonchanka, A.; Feussner, I. Lipoxygenases: Occurrence, functions and catalysis. J. Plant Physiol. 2006, 163, 348–357. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, I.; Heydeck, D.; Hofheinz, K.; Roffeis, J.; O´Donnell, V.B.; Kuhn, H.; Walther, M. Molecular enzymology of lipoxygenases. Arch. Biochem. Biophys. 2010, 503, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Andreou, A.; Feussner, I. Lipoxygenases—Structure and reaction mechanism. Phytochemistry 2009, 70, 1504–1510. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Shen, W.; Liu, L.; Jiang, L.; Liu, Y.; Su, N.; Wan, J. A novel lipoxygenase gene from developing rice seeds confers dual position specificity and responds to wounding and insect attack. Plant Mol. Biol. 2008, 66, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Han, Y.; Woo, J.C.; Baudisch, B.; Klösgen, R.B.; Oh, S.; Han, J.; Han, O. Cellular localization of dual positional specific maize lipoxygenase-1 in transgenic rice and calcium-mediated membrane association. Plant Sci. 2011, 181, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Holková, I.; Bilka, F.; Rauová, D.; Bezáková, L. Purification and properties of lipoxygenase from opium poopy seedlings (Papaver somniferum L.). Turk. J. Biol. 2016, 40, 772–780. [Google Scholar] [CrossRef]
- Porta, H.; Rocha-Sosa, M. Plant lipoxygenases. Physiological and molecular features. Plant Physiol. 2002, 130, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Wasternack, C.; Hause, B. Jasmonates: Biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany. Ann. Bot. 2013, 111, 1021–1058. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Dahuja, A.; Kar, A.; Santha, I.M. In vitro kinetics of soybean lipoxygenase with combinatorial fatty substrates and its functional significance in off flavour development. Food Chem. 2014, 146, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Santino, A.; Iannacone, R.; Hughes, R.; Casey, R.; Mita, G. Cloning and characterization of an almond 9-lipoxygenase expressed early during seed development. Plant Sci. 2005, 168, 699–706. [Google Scholar] [CrossRef]
- Meyer, D.; Herrfurth, C.; Brodhun, F.; Feussner, I. Degradation of lipoxygenase-derived oxylipins by glyoxysomes from sunflower and cucumber cotyledons. BMC Plant Biol. 2013, 13, 177. [Google Scholar] [CrossRef] [PubMed]
- Feussner, I.; Wasternack, C. The lipoxygenase pathway. Annu. Rev. Plant Biol. 2002, 53, 275–297. [Google Scholar] [CrossRef] [PubMed]
- Mosblech, A.; Feussner, I.; Heilmann, I. Oxylipins: Structurally diverse metabolites from fatty acid oxidation. Plant Physiol. Biochem. 2009, 47, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Yadav, M.K.; Bhatla, S.C. Localization of lipoxygenase activity on the oil bodies and in protoplasts using a novel fluorescence imaging method. Plant Physiol. Biochem. 2011, 49, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Terp, N.; Göbel, C.; Brandt, A.; Feussner, I. Lipoxygenases during Brassica napus seed germination. Phytochemistry 2006, 67, 2030–2040. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.; Harwood, J.L. Characterisation of lipoxygenase isoforms from olive callus cultures. Phytochemistry 2008, 69, 2532–2538. [Google Scholar] [CrossRef] [PubMed]
- Vanko, M.; Rauová, D.; Bezáková, L.; Holková, I.; Bilka, F.; Cupáková, M. Biochemical properties of lipoxygenase from opium poppy chloroplasts. Biol. Plant. 2012, 56, 105–110. [Google Scholar] [CrossRef]
- Lorenzi, V.; Maury, J.; Casanova, J.; Berti, L. Purification, product characterization and kinetic properties of lipoxygenase from olive fruit (Olea europaea L.). Plant Physiol. Biochem. 2006, 44, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Clemente, A.; Olías, R.; Olías, J.M. Purification and characterization of broad bean lipoxygenase isoenzymes. J. Agric. Food Chem. 2000, 48, 1070–1075. [Google Scholar] [CrossRef] [PubMed]
- Kuo, J.M.; Hwang, A.; Yeh, D.B.; Pan, M.H.; Tsai, M.L.; Pan, B.S. Lipoxygenase from banana leaf: Purification and characterization of an enzyme that catalyzes linoleic acid oxygenation at the 9-position. J. Agric. Food Chem. 2006, 54, 3151–3156. [Google Scholar] [CrossRef] [PubMed]
- Coffa, G.; Schneider, C.; Brash, A.R. A comprehensive model of positional and stereo control in lipoxygenases. Biochem. Biophys. Res. Commin. 2005, 338, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Sicilia, F.; Mattei, B.; Cervone, F.; Bellincampi, D.; De Lorenzo, G. Characterization of a membrane-associated apoplastic lipoxygenase in Phaseolus vulgaris L. Biochim. Biophys. Acta 2005, 1748, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Chugh, L.K. Two isoforms of lipoxygenase from mature grains of pearl millet [Pennisetum glaucum (L.) R. Br.]: Purification and physic-chemico-kinetic characterization. J. Food Sci. Technol. 2017, 54, 1577. [Google Scholar] [CrossRef] [PubMed]
- Jacobo-Velázquez, D.A.; Hernández-Brenes, C.; Cisneros-Zevallos, L.; Benavides, J. Partial purification and enzymatic characterization of avocado (Persea Americana Mill, cv. Hass) lipoxygenase. Food Res. Int. 2010, 43, 1079–1085. [Google Scholar] [CrossRef]
- Bowsher, C.G.; Ferrie, B.J.; Ghosh, S.; Todd, J.; Thompson, J.E.; Rothstein, S.J. Purification and partial characterization of a membrane-associated lipoxygenase in tomato fruit. Plant Physiol. 1992, 100, 1802–1807. [Google Scholar] [CrossRef] [PubMed]
- Leone, A.; Melillo, M.T.; Bleve-Zacheo, T. Lipoxygenase in pea roots subjected to biotic stress. Plant Sci. 2001, 161, 703–717. [Google Scholar] [CrossRef]
- Sheu, S.C.; Chen, A.O. Lipoxygenase as blanching index for frozen vegetable soybeans. J. Food Sci. 1991, 56, 448–451. [Google Scholar] [CrossRef]
- Chedea, V.S.; Vicas, S.; Socaciu, C. Kinetics of soybean lipoxygenases are related to pH, substrate availability and extraction procedures. J. Foof Biochem. 2008, 32, 153–172. [Google Scholar] [CrossRef]
- Palmieri-Thiers, C.; Canaan, S.; Brunini, V.; Lorenzi, V.; Tomi, F.; Desseyn, J.L.; Garscha, U.; Oliw, E.H.; Berti, L.; Maury, J. A lipoxygenase with dual positional specifity is expressed in olives (Olea europaea L.) during ripening. Biochim. Biophys. Acta 2009, 1791, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.S.; Choi, E.; Kim, Y.; Cho, K.; Lee, A.; Shim, J.; Rakwal, R.; Agrawal, G.K.; Han, O. Dual positional specificity and expression of non-traditional lipoxygenase induced by wounding and methyl jasmonate in maize seedlings. Plant Mol. Biol. 2003, 52, 1203–1213. [Google Scholar] [CrossRef] [PubMed]
- Daglia, M.; Aceti, C.; Giorgetti, S.; Papetti, A.; Gazzani, G. Purification and characterization of soluble Cichorium intybus var. silvestre lipoxygenase. J. Agric. Food Chem. 2005, 53, 6448–6454. [Google Scholar] [CrossRef] [PubMed]
- López-Nicolás, J.M.; Pérez-Gilabert, M.; García-Carmona, F. Eggplant lipoxygenase (Solanum melongena): Product characterization and effect of physicochemical properties of linoleic acid on the enzymatic activity. J. Agric. Food Chem. 2001, 49, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Szymanowska, U.; Jakubczyk, A.; Baraniak, B.; Kur, A. Characterisation of lipoxygenase from pea seeds (Pisum sativum var Telephone L.). Food Chem. 2009, 116, 906–910. [Google Scholar] [CrossRef]
- Cho, K.; Han, J.; Rakwal, R.; Han, O. Calcium modulates membrane association, positional specificity and product distribution in dual positional specific maize lipoxygenase-1. Bioorg. Chem. 2015, 60, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Gökmen, V.; Bahçeci, S.; Acar, J. Characterization of crude lipoxygenase extract from green pea using a modified spectrophotometric method. J. Eur. Food Res. Technol. 2002, 215, 42–45. [Google Scholar] [CrossRef]
- Yilmaz, E. Kinetic studies with crude tomato lipoxygenase. Turk. J. Agric. For. 2001, 25, 291–296. [Google Scholar]
- Obložinský, M.; Schoeps, R.; Ulbrich-Hofmann, R.; Bezáková, L. Two uncommon phospholipase D isoenzymes from poppy seedlings (Papaver somniferum L.). Biochim. Biophys. Acta 2003, 1631, 153–159. [Google Scholar] [CrossRef]
- Chen, A.O.; Whitaker, J.R. Purification and characterization of a lipoxygenase from immature English peas. J. Agric. Food Chem. 1986, 34, 203–211. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utulizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Andreou, A.Z.; Vanko, M.; Bezáková, L.; Feussner, I. Properties of a mini 9R-lipoxygenase from Nostoc sp. PCC 7120 and its mutant forms. Phytochemistry 2008, 69, 1832–1837. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, R.J.; Prvan, T. Current statistical methods for estimating the Km and Vmax of Michaelis-Menten kinetics. Biochem. Educ. 1996, 24, 196–206. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc. Natl. Acad. Sci. USA 1979, 76, 4350–4354. [Google Scholar] [CrossRef] [PubMed]
- Holková, I.; Bezáková, L.; Bilka, F.; Balažová, A.; Vanko, M.; Blanáriková, V. Involvement of lipoxygenase in elicitor-stimulated sanguinarine accumulation in Papaver somniferum suspension cultures. Plant Physiol. Biochem. 2010, 48, 887–892. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Sample of purified LOX is available from the authors. |
| Purification Step | Proteins (mg/mL) | Activity (nkat/mL) | Specific Activity (nkat/mg) | Purification Fold |
|---|---|---|---|---|
| Crude extract | 0.78 | 0.27 | 0.34 | 1 |
| Phenyl-Sepharose CL-4B | 1.07 | 0.48 | 0.49 | 1.4 |
| Q-Sepharose | 0.04 | 0.90 | 23.47 | 69 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kollárová, R.; Holková, I.; Rauová, D.; Bálintová, B.; Mikuš, P.; Obložinský, M. HPLC Analysis and Biochemical Characterization of LOX from Eschscholtzia californica Cham. Molecules 2017, 22, 1899. https://doi.org/10.3390/molecules22111899
Kollárová R, Holková I, Rauová D, Bálintová B, Mikuš P, Obložinský M. HPLC Analysis and Biochemical Characterization of LOX from Eschscholtzia californica Cham. Molecules. 2017; 22(11):1899. https://doi.org/10.3390/molecules22111899
Chicago/Turabian StyleKollárová, Renáta, Ivana Holková, Drahomíra Rauová, Barbora Bálintová, Peter Mikuš, and Marek Obložinský. 2017. "HPLC Analysis and Biochemical Characterization of LOX from Eschscholtzia californica Cham." Molecules 22, no. 11: 1899. https://doi.org/10.3390/molecules22111899





