Adaptive Responses to Oxidative Stress in the Filamentous Fungal Shiraia bambusicola
Abstract
:1. Introduction
2. Results
2.1. Effect of H2O2 on Hypocrellin Production and Growth
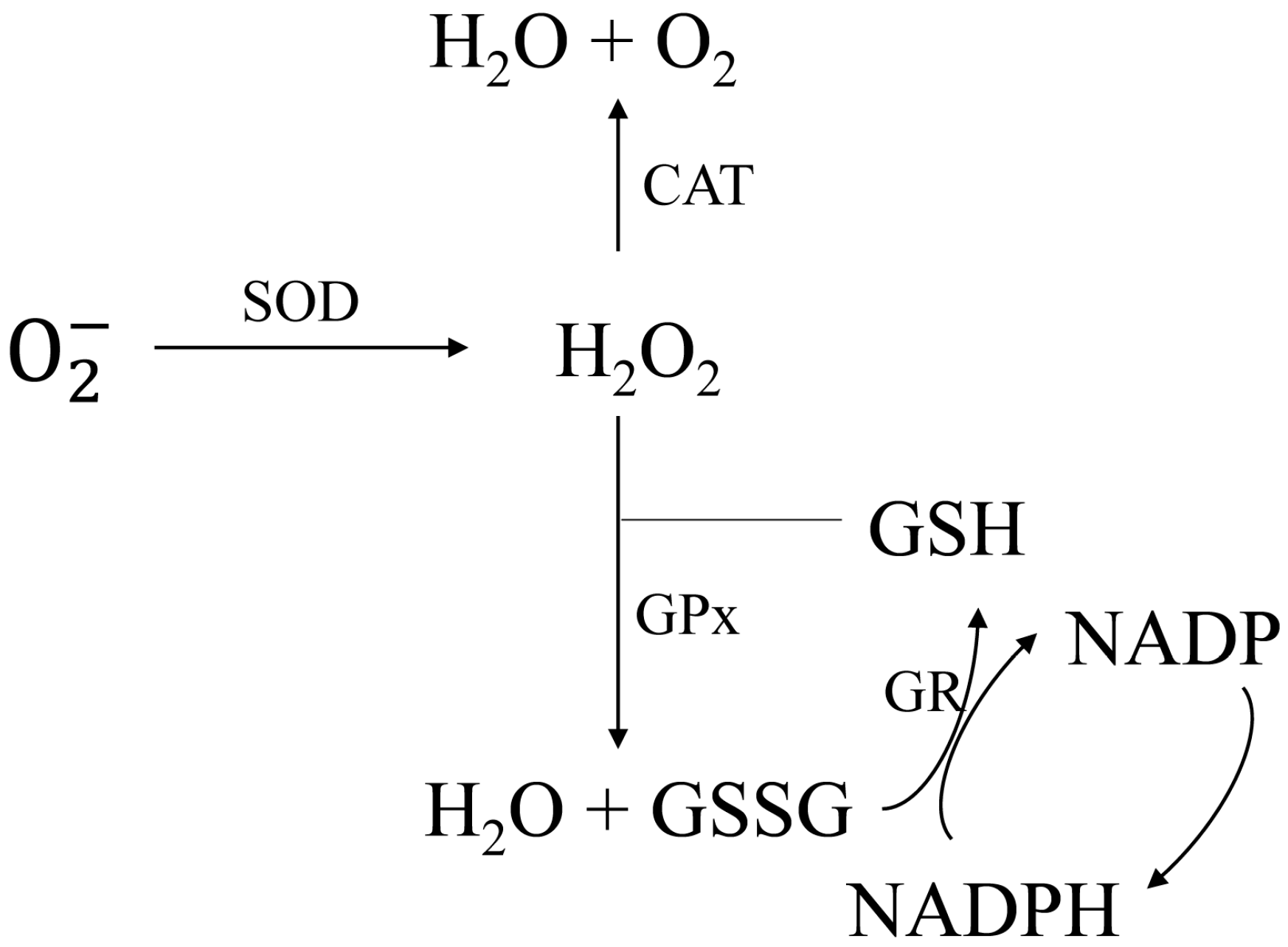
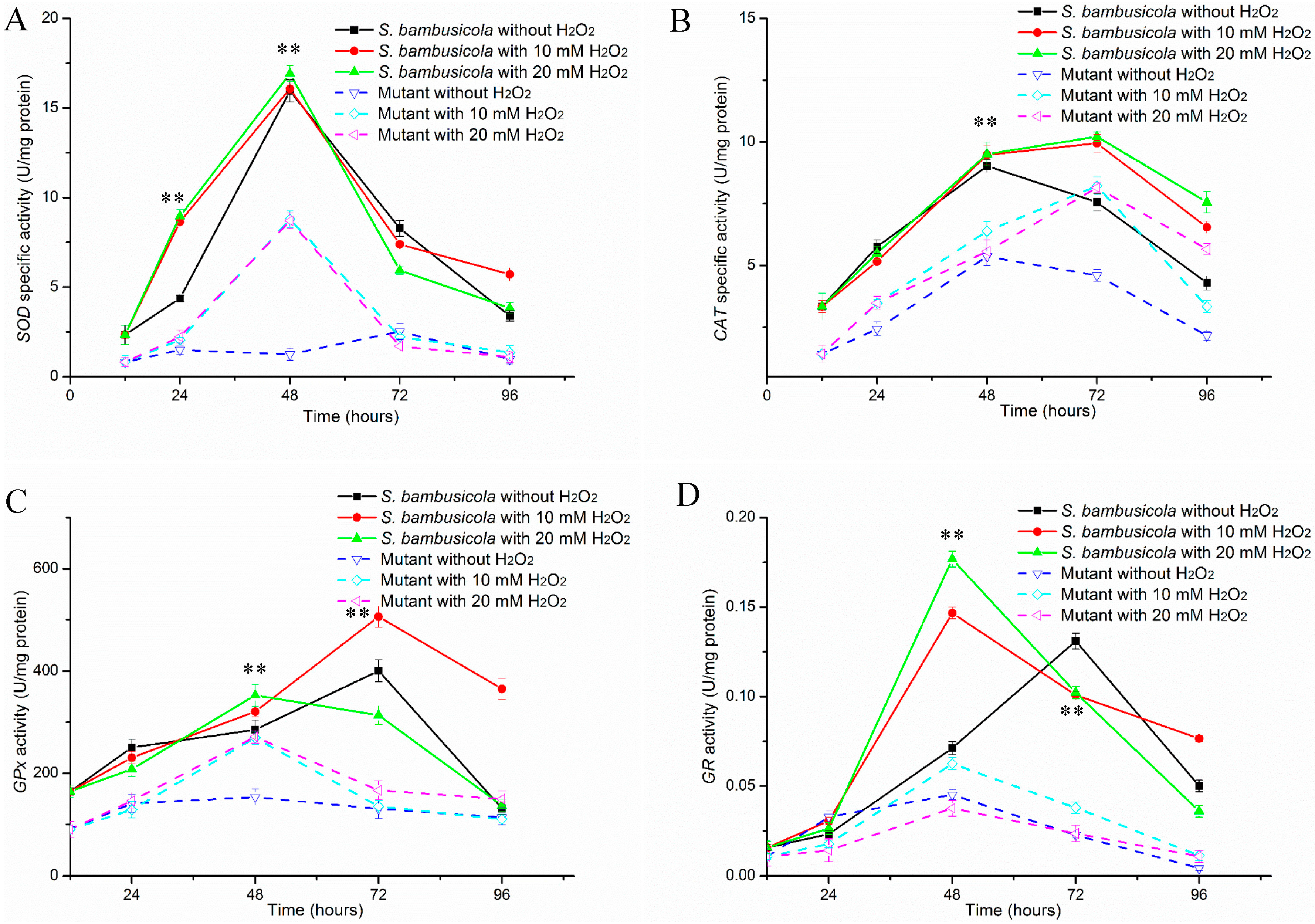
2.2. Antioxidant Enzyme Activity Analysis
2.2.1. Specific Activities of SOD and CAT
2.2.2. Determinations of GPx and GR Activities
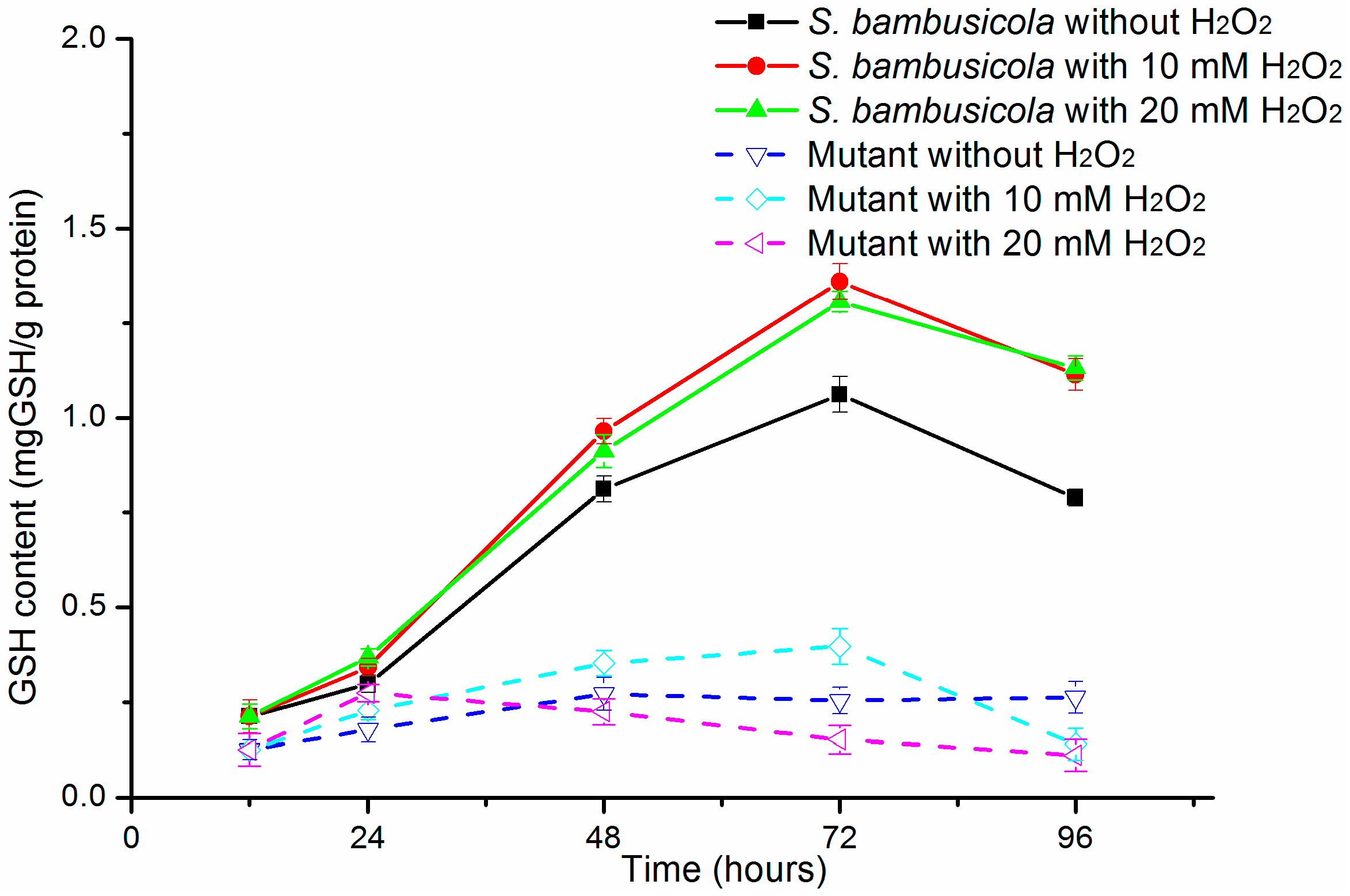
2.3. GSH Content
3. Discussion
4. Experimental Section
4.1. Strain and Cultivation
4.2. Biochemical Assays
5. Statistical Analysis
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cheng, T.F.; Jia, X.M.; Ma, X.H.; Lin, H.P.; Zhao, Y.H. Phylogenetic study on Shiraia bambusicola by rDNA sequence analyses. J. Basic Microbiol. 2004, 44, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Hino, I.; Katumoto, K. Icones Fungorum Bambusicolorum Japonicorum, Yamagutiensi; The Fuji Bamboo Garden: Gotenba, Japan, 1961; p. 335. [Google Scholar]
- Cai, Y.; Liang, X.H.; Liao, X.R.; Ding, Y.R.; Sun, J.; Li, X.H. High-Yield Hypocrellin A Production in Solid-State Fermentation by Shiraia sp. SUPER-H168. Appl. Biochem. Biotechnol. 2010, 160, 2275–2286. [Google Scholar] [CrossRef] [PubMed]
- Daub, M.E.; Herrero, S.; Chung, K.-R. Reactive oxygen species in plant pathogenesis: The role of perylenequinone photosensitizers. Antioxid. Redox Signal. 2013, 19, 970–989. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.; Imperial, V.; Haugland, R.; Diwu, Z. Antiviral activities of photoactive perylenequinones. Photochem. Photobiol. 1997, 65, 352–354. [Google Scholar] [CrossRef] [PubMed]
- Nenghui, W.; Zhiyi, Z. Relationship between photosensitizing activities and chemical structure of hypocrellin A and B. J. Photochem. Photobiol. B 1992, 14, 207–217. [Google Scholar] [CrossRef]
- Deininger, M.H.; Weinschenk, T.; Morgalla, M.H.; Meyermann, R.; Schluesener, H.J. Release of regulators of angiogenesis following Hypocrellin-A and-B photodynamic therapy of human brain tumor cells. Biochem. Biophs. Res. Commun. 2002, 298, 520–530. [Google Scholar] [CrossRef]
- Li, Q.; Harvey, L.M.; McNeil, B. Oxidative stress in industrial fungi. Crit. Rev. Biotechnol. 2009, 29, 199–213. [Google Scholar] [CrossRef] [PubMed]
- Imlay, J.A. Pathways of oxidative damage. Annu. Rev. Microbiol. 2003, 57, 395–418. [Google Scholar] [CrossRef] [PubMed]
- Trachootham, D.; Lu, W.; Ogasawara, M.A.; Valle, N.R.-D.; Huang, P. Redox regulation of cell survival. Antioxid. Redox Signal. 2008, 10, 1343–1374. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.J.; Liao, X.R.; Liang, X.H.; Ding, Y.R.; Sun, J.; Zhang, D.B. Induction of hypocrellin production by Triton X-100 under submerged fermentation with Shiraia sp. SUPER-H168. New Biotechnol. 2011, 28, 588–592. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; McNeil, B.; Harvey, L.M. Adaptive response to oxidative stress in the filamentous fungus Aspergillus niger B1-D. Free Radic. Biol. Med. 2008, 44, 394–402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gyöngyösi, N.; Káldi, K. Interconnections of Reactive Oxygen Species Homeostasis and Circadian Rhythm in Neurospora crassa. Antioxid. Redox Signal. 2014, 20, 3007–3023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Castro, C.; del Valle, P.; Rua, J.; Garcia-Armesto, M.R.; Gutierrez-Larrainzar, M.; Busto, F.; de Arriaga, D. Antioxidant defence system during exponential and stationary growth phases of Phycomyces blakesleeanus: Response to oxidative stress by hydrogen peroxide. Fungal Biol. 2013, 117, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Circu, M.L.; Aw, T.Y. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic. Biol. Med. 2010, 48, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Franco, R.; Cidlowski, J.A. Glutathione Efflux and Cell Death. Antioxid. Redox Signal. 2012, 17, 1694–1713. [Google Scholar] [CrossRef] [PubMed]
- Miranda, R.U.; Gomez-Quiroz, L.E.; Mejia, A.; Barrios-Gonzalez, J. Oxidative state in idiophase links reactive oxygen species (ROS) and lovastatin biosynthesis: Differences and similarities in submerged- and solid-state fermentations. Fungal Biol. 2013, 117, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.B.; Luo, J.; Huang, X.Y.; Lu, M.B.; Yu, L.J. Oxidative stress response of Blakeslea trispora induced by H2O2 during beta-carotene biosynthesis. J. Ind. Microbiol. Biotechnol. 2014, 41, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Pang, W.; Wang, J. Effect of oxidative stress on hypocrellin A yield in submerged cultures of endophytic Shiraia sp. A8. Planta Med. 2014, 80, P1N2. [Google Scholar] [CrossRef]
- Ferrigo, D.; Raiola, A.; Bogialli, S.; Bortolini, C.; Tapparo, A.; Causin, R. In Vitro Production of Fumonisins by Fusarium verticillioides under Oxidative Stress Induced by H2O2. J. Agric. Food Chem. 2015, 63, 4879–4885. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Chen, Y. Hypocrellin A, a new drug for photochemotherapy. Kexue Tongbao 1981, 26, 1040–1042. [Google Scholar]
- Kishi, T.; Tahara, S.; Taniguchi, N.; Tsuda, M.; Tanaka, C.; Takahashi, S. New perylenequinones from Shiraia bambusicola. Planta Med. 1991, 57, 376–379. [Google Scholar] [CrossRef] [PubMed]
- Montibus, M.; Pinson-Gadais, L.; Richard-Forget, F.; Barreau, C.; Ponts, N. Coupling of transcriptional response to oxidative stress and secondary metabolism regulation in filamentous fungi. Crit. Rev. Microbiol. 2015, 41, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.-Y.; Roze, L.V.; Linz, J.E. Oxidative Stress-Related Transcription Factors in the Regulation of Secondary Metabolism. Toxins 2013, 5, 683–702. [Google Scholar] [CrossRef] [PubMed]
- Angelova, M.B.; Pashova, S.B.; Spasova, B.K.; Vassilev, S.V.; Slokoska, L.S. Oxidative stress response of filamentous fungi induced by hydrogen peroxide and paraquat. Mycol. Res. 2005, 109, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant. Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Hatem, E.; Berthonaud, V.; Dardalhon, M.; Lagniel, G.; Baudouin-Cornu, P.; Huang, M.E.; Labarre, J.; Chedin, S. Glutathione is essential to preserve nuclear function and cell survival under oxidative stress. Free Radic. Biol. Med. 2014, 67, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Pocsi, I.; Prade, R.A.; Penninckx, M.J. Glutathione, altruistic metabolite in fungi. Adv. Microb. Physiol. 2004, 49, 1–76. [Google Scholar] [PubMed]
- Bai, Z.H.; Harvey, L.M.; McNeil, B. Oxidative stress in submerged cultures of fungi. Crit. Rev. Biotechnol. 2003, 23, 267–302. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.G.; Townsend, C.A. Molecular characterization of the cercosporin biosynthetic pathway in the fungal plant pathogen cercospora nicotianae. J. Am. Chem. Soc. 2016, 138, 4219–4228. [Google Scholar] [CrossRef] [PubMed]
- Chooi, Y.-H.; Tang, Y. Navigating the fungal polyketide chemical space: From genes to molecules. J. Org. Chem. 2012, 77, 9933–9953. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Lee, M.H.; Daub, M.E.; Chung, K.R. Molecular analysis of the cercosporin biosynthetic gene cluster in Cercospora nicotianae. Mol. Mcrobiol. 2007, 64, 755–770. [Google Scholar] [CrossRef] [PubMed]
- Griffith, O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 1980, 106, 207–212. [Google Scholar] [CrossRef]
- Aebi, H. [13] Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [PubMed]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar] [PubMed]
- Carlberg, I.; Mannervik, B. Glutathione reductase. Method Enzymol. 1984, 113, 484–490. [Google Scholar]
- Sample Availability: Not available.

© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, H.; Chen, J.; Gao, R.; Liao, X.; Cai, Y. Adaptive Responses to Oxidative Stress in the Filamentous Fungal Shiraia bambusicola. Molecules 2016, 21, 1118. https://doi.org/10.3390/molecules21091118
Deng H, Chen J, Gao R, Liao X, Cai Y. Adaptive Responses to Oxidative Stress in the Filamentous Fungal Shiraia bambusicola. Molecules. 2016; 21(9):1118. https://doi.org/10.3390/molecules21091118
Chicago/Turabian StyleDeng, Huaxiang, Jiajun Chen, Ruijie Gao, Xiangru Liao, and Yujie Cai. 2016. "Adaptive Responses to Oxidative Stress in the Filamentous Fungal Shiraia bambusicola" Molecules 21, no. 9: 1118. https://doi.org/10.3390/molecules21091118




