Salidroside Regulates Inflammatory Response in Raw 264.7 Macrophages via TLR4/TAK1 and Ameliorates Inflammation in Alcohol Binge Drinking-Induced Liver Injury
Abstract
:1. Introduction
2. Results
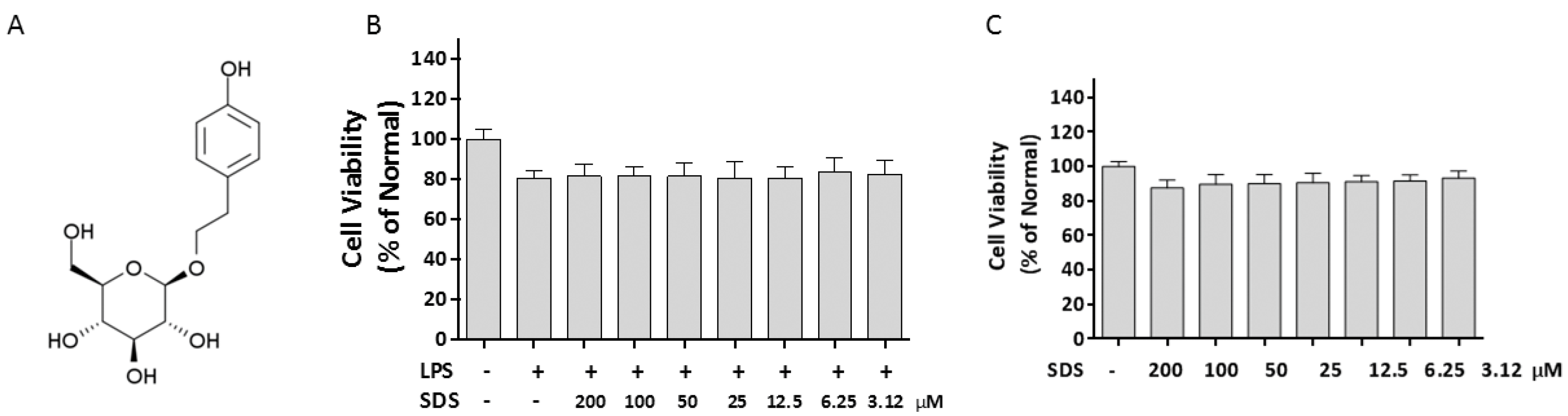
2.1. Effect of SDS on Cell Viability
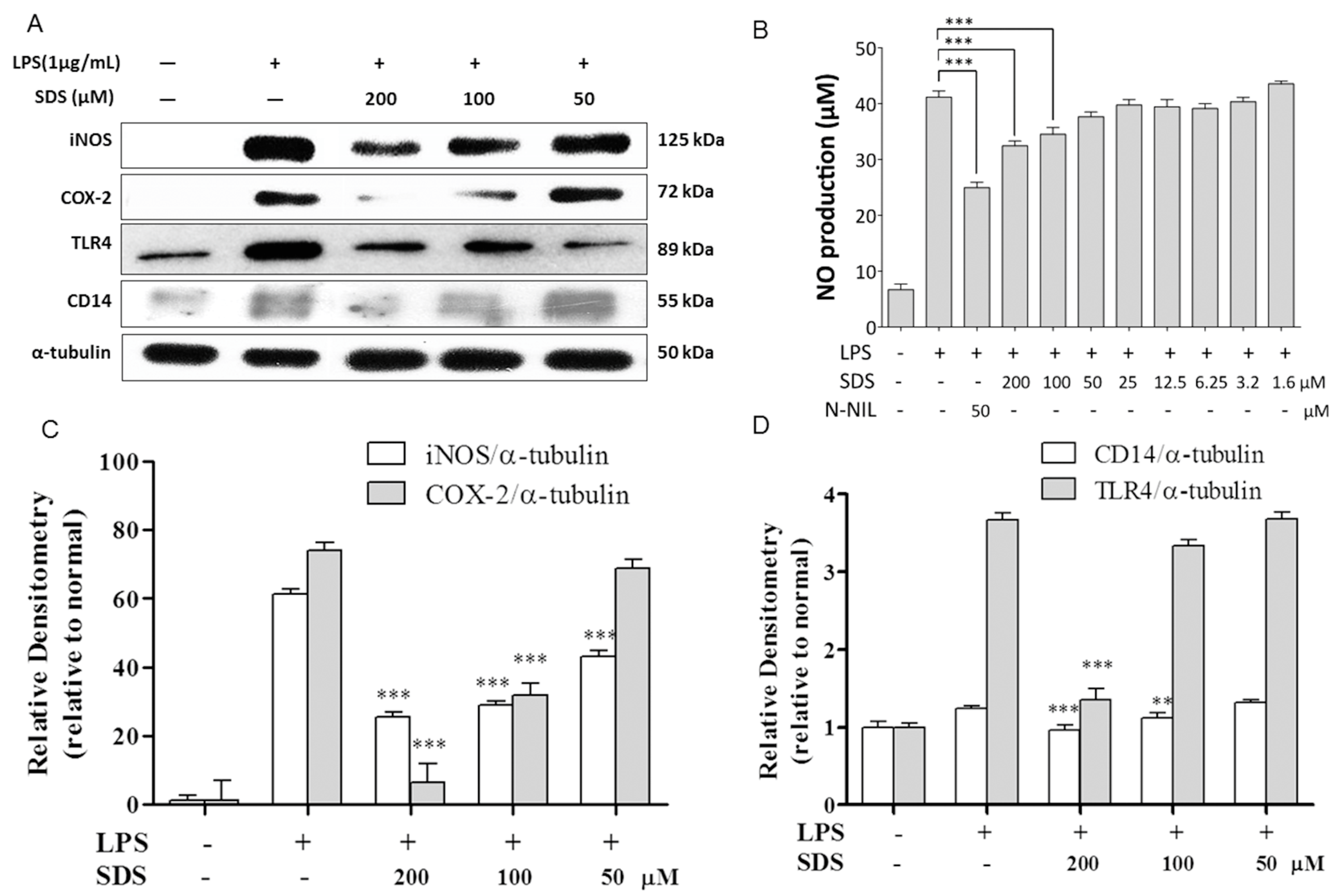
2.2. Effect of SDS on LPS-Induced iNOS and COX-2 Protein Expression
2.3. Effect of SDS on LPS-Induced TLR4 and CD14 Expression
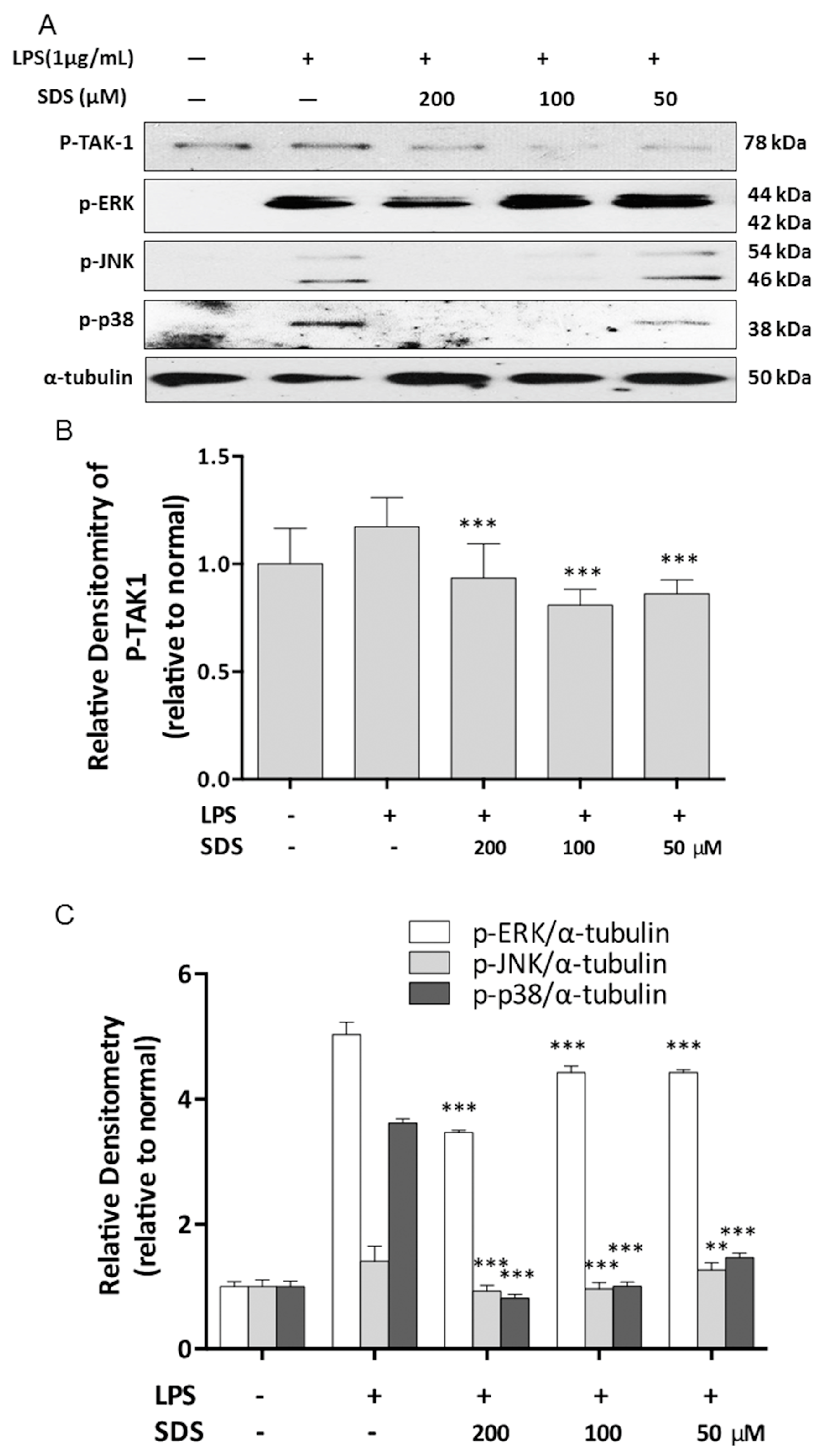
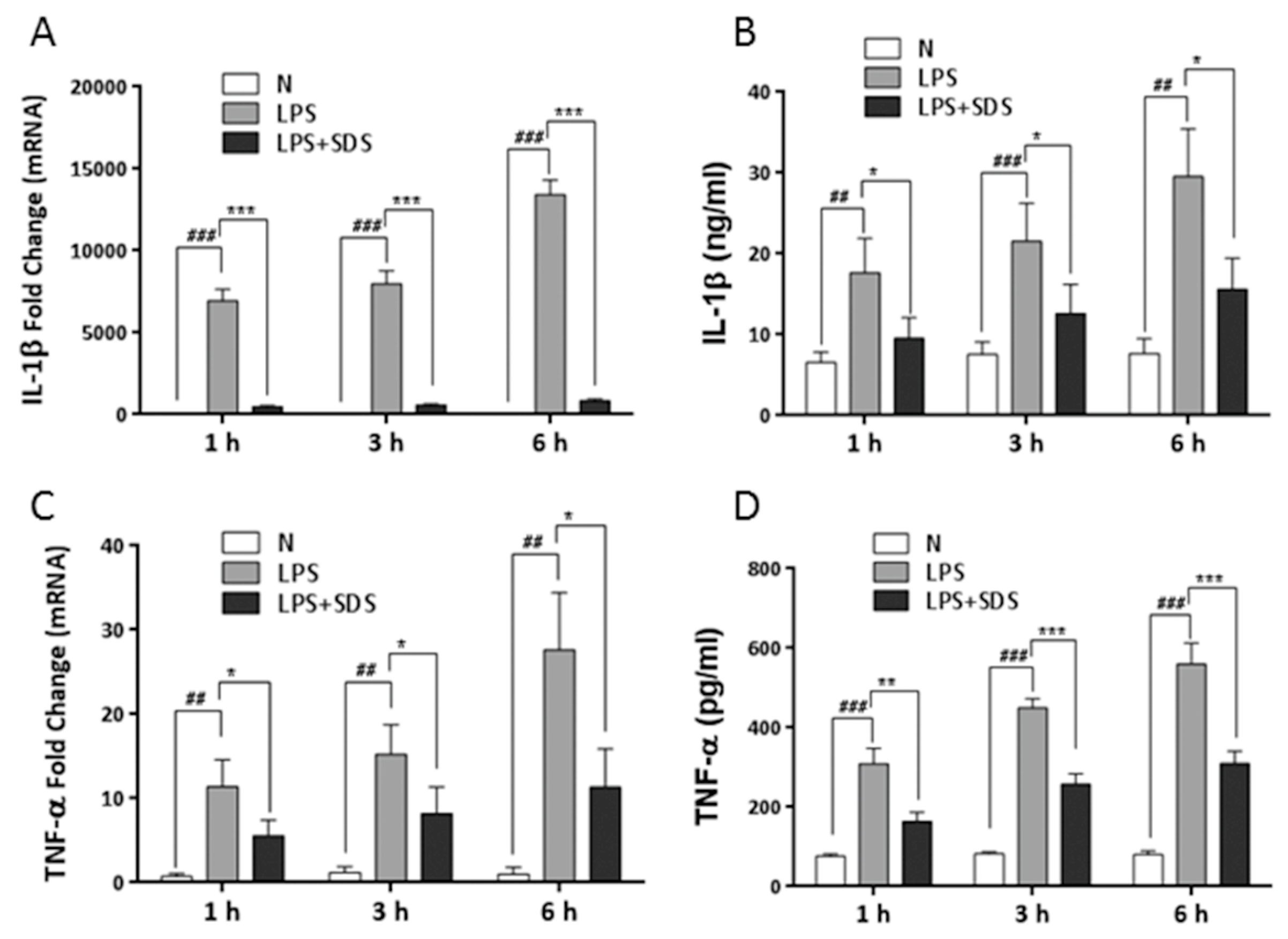
2.4. Effect of SDS on LPS-Induced Phosphorylation of TAK1 and MAPK
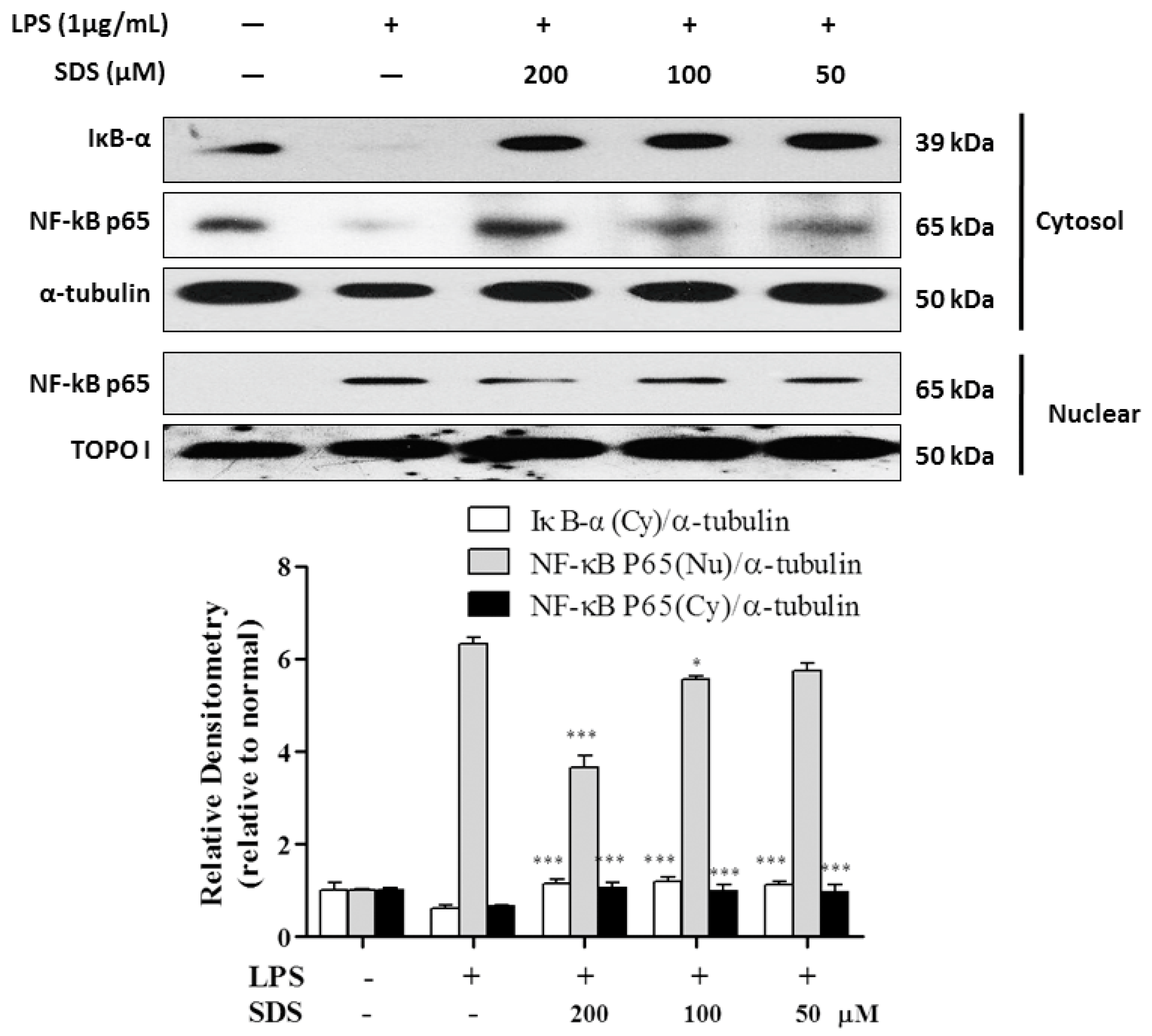
2.5. Effect of SDS on LPS-Induced IκB Degradation and NF-κB Nuclear Translocation
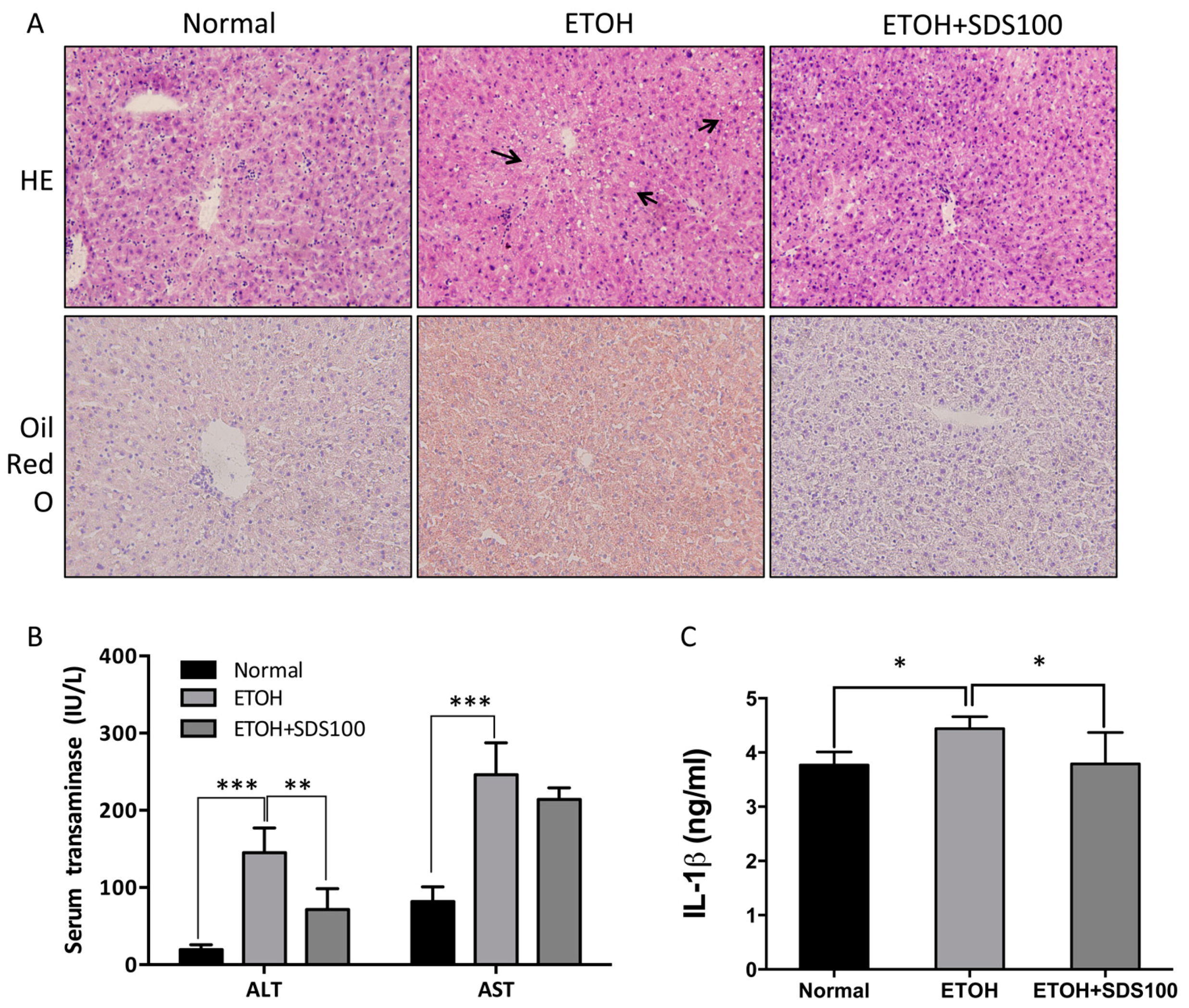
2.6. Effect of SDS on Binge Drinking Induced Liver Injury
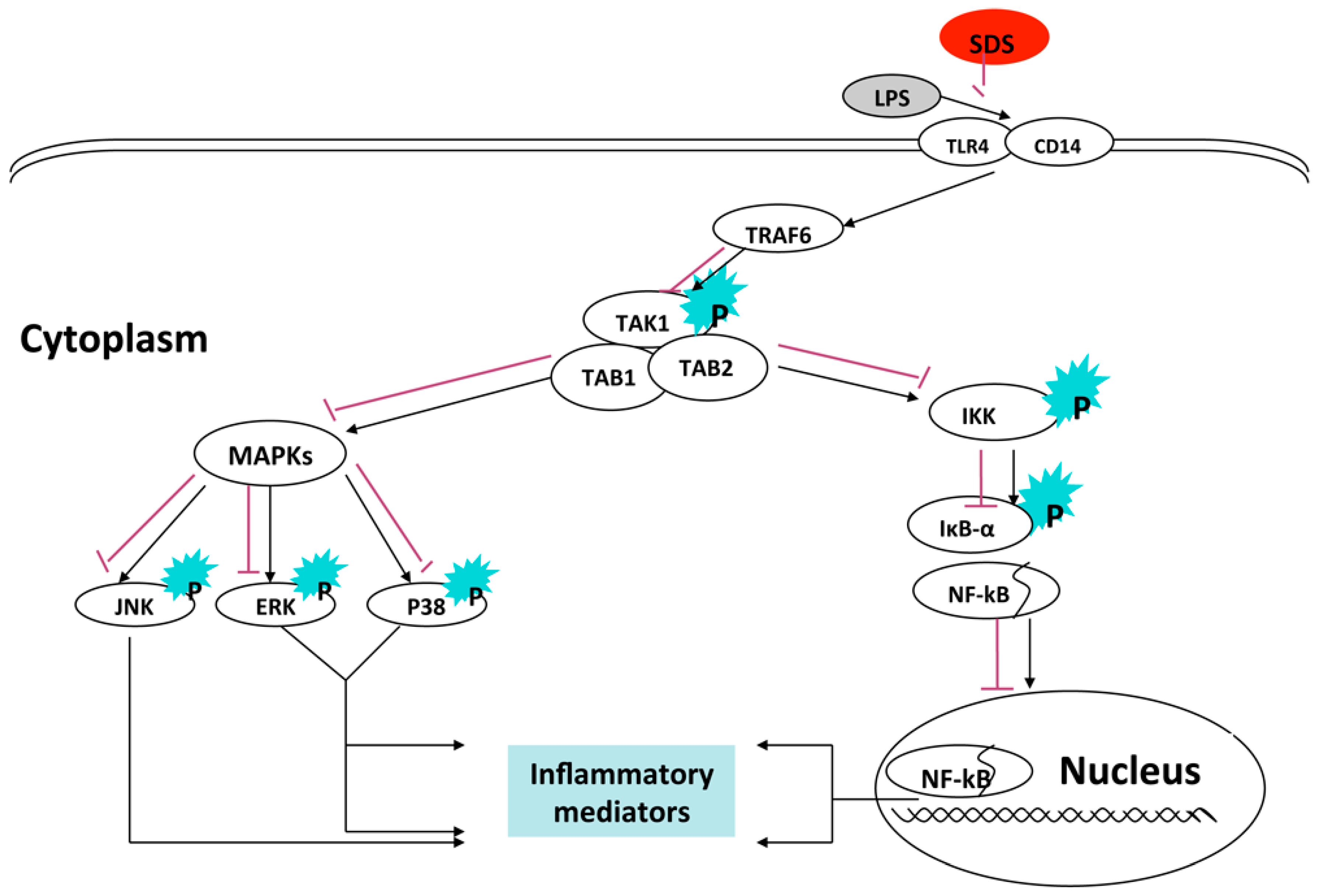
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture
4.3. Measurement of Cell Viability
4.4. Quantification of NO Production
4.5. Binge Drinking Protocol
4.6. Blood Biochemistry and ELISA Assay
4.7. Real-Time RT-PCR
4.8. Histochemistry
4.9. Western Blotting Analysis
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ALT | alanine aminotransferase |
| AST | aspartate aminotransferase |
| ERK | extracellular signal regulated kinase |
| iNOS | inducible nitric oxide synthase |
| COX-2 | cyclooxygenase-2 |
| IκB-α | inhibitory κB-α |
| IKK | IκB kinase |
| IRAK | interleukin-1 receptor-associated kinase |
| JNK | c-Jun terminal kinase |
| LPS | lipopolysaccharide |
| LBP | lipopolysaccharide-binding protein |
| MAPK | mitogen-activated protein kinases |
| MD-2 | myeloid differentiation-2 |
| MyD88 | myeloid differentiation primary response gene 88 |
| NF-κB | nuclear factor-κB |
| TAK1 | TGF-β activated kinase 1 |
| TLR4 | toll-like receptor 4 |
| TRAF6 | TNF receptor associated factor 6 |
| TRIF | toll/IL-1 receptor domain containing adaptor inducing interferon-β |
References
- Li, Q.; Verma, I.M. NF-κB regulation in the immune system. Nat. Rev. Immunol. 2002, 2, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Zandi, E.; Rothwarf, D.M.; Delhase, M.; Hayakawa, M.; Karin, M. The IκB kinase complex (IKK) contains two kinase subunits, IKKα and IKKβ, necessary for IκB phosphorylation and NF-κB activation. Cell 1997, 91, 243–252. [Google Scholar] [CrossRef]
- Hartlage-Rubsamen, M.; Lemke, R.; Schliebs, R. Interleukin-1β, inducible nitric oxide synthase, and nuclear factor-κ are induced in morphologically distinct microglia after rat hippocampal lipopolysaccharide/interferon-gamma injection. J. Neurosci. Res. 1999, 57, 388–398. [Google Scholar] [CrossRef]
- Park, J.S.; Woo, M.S.; Kim, D.H.; Hyun, J.W.; Kim, W.K.; Lee, J.C.; Kim, H.S. Anti-inflammatory mechanisms of isoflavone metabolites in lipopolysaccharide-stimulated microglial cells. J. Pharmacol. Exp. Ther. 2007, 320, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Vanden Berghe, W.; Plaisance, S.; Boone, E.; de Bosscher, K.; Schmitz, M.L.; Fiers, W.; Haegeman, G. P38 and extracellular signal-regulated kinase mitogen-activated protein kinase pathways are required for nuclear factor-κB p65 transactivation mediated by tumor necrosis factor. J. Biol. Chem. 1998, 273, 3285–3290. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Shen, X.Y.; Huang, H.Q.; Xu, S.W.; Yu, Y.; Zhou, C.H.; Chen, S.R.; Le, K.; Wang, Y.H.; Liu, P.Q. Cryptotanshinone suppressed inflammatory cytokines secretion in RAW264.7 macrophages through inhibition of the NF-κB and mapk signaling pathways. Inflammation 2011, 34, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.W.; Yoon, C.H.; Kim, Y.H.; Boo, Y.C.; Park, K.M.; Park, Y.K. Wen-pi-tang-hab-wu-ling-san extract inhibits the release of inflammatory mediators from LPS-stimulated mouse macrophages. J Ethnopharmacol. 2007, 114, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.H.; Kao, Y.C.; Chen, B.C.; Chen, C.H.; Chan, P.; Lee, H.M. Dipyridamole activation of mitogen-activated protein kinase phosphatase-1 mediates inhibition of lipopolysaccharide-induced cyclooxygenase-2 expression in RAW 264.7 cells. Eur. J. Pharmacol. 2006, 541, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.C.; Choi, J.W.; Hong, H.Y.; Lee, S.A.; Hong, S.; Park, E.H.; Kim, S.J.; Lim, C.J. Heme oxygenase-1 mediates the anti-inflammatory effect of mushroom phellinus linteus in LPS-stimulated RAW264.7 macrophages. J. Ethnopharmacol. 2006, 106, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Jeong, H.J.; Kim, D.W.; Sohn, E.J.; Jo, H.S.; Kim, D.S.; Kim, H.A.; Park, E.Y.; Park, J.H.; Son, O.; et al. Pep-1-pon1 protein regulates inflammatory response in RAW264.7 macrophages and ameliorates inflammation in a TPA-induced animal model. PLoS ONE 2014, 9, e86034. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Lee, B.S.; Yi, K.Y.; Paik, S.G. Upstream NF-κB site is required for the maximal expression of mouse inducible nitric oxide synthase gene in interferon-gamma plus lipopolysaccharide-induced RAW264.7 macrophages. Biochem. Biophys. Res. Commun. 1997, 236, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Shepherd, E.G.; Nelin, L.D. Mapk phosphatases-regulating the immune response. Nat. Rev. Immunol. 2007, 7, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Bi, H.; Zhuang, Y.; Liu, C.; Cai, T.; Liu, X.; Zhang, X.; Liu, T.; Ma, Y. Production of salidroside in metabolically engineered Escherichia coli. Sci. Rep. 2014, 4, 6640. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, J.F.; Lou, J.; Yan, C.; Ren, Z.H.; Qiao, H.X.; Hong, D.S. In vitro promoted differentiation of mesenchymal stem cells towards hepatocytes induced by salidroside. J. Pharm. Pharmacol. 2010, 62, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, A.; Hou, R.; Jia, X.; Jiang, W.; Chen, J. Salidroside protects cardiomyocyte against hypoxia-induced death: A hif-1α-activated and VEGF-mediated pathway. Eur. J. Pharmacol. 2009, 607, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Panossian, A.; Hamm, R.; Wikman, G.; Efferth, T. Mechanism of action of rhodiola, salidroside, tyrosol and triandrin in isolated neuroglial cells: An interactive pathway analysis of the downstream effects using rna microarray data. Phytomedicine 2014, 21, 1325–1348. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.L.; Piao, D.M.; Han, X.H.; Nan, J.X. Protective effects of salidroside against acetaminophen-induced toxicity in mice. Biol. Pharm. Bull. 2008, 31, 1523–1529. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Luo, F.; Jiang, W.; Zhu, L.; Gao, J.; He, H.; Wei, T.; Gong, S.; Yan, T. Protective activity of salidroside against ethanol-induced gastric ulcer via the MAPK/NF-κBpathway in vivo and in vitro. Int. Immunopharmacol. 2015, 28, 604–615. [Google Scholar] [CrossRef] [PubMed]
- Guan, S.; Feng, H.; Song, B.; Guo, W.; Xiong, Y.; Huang, G.; Zhong, W.; Huo, M.; Chen, N.; Lu, J.; et al. Salidroside attenuates LPS-induced pro-inflammatory cytokine responses and improves survival in murine endotoxemia. Int. Immunopharmacol. 2011, 11, 2194–2199. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Huang, G.; Xiong, Y.; Liu, J.; Xu, L.; Wang, Z.; Li, G.; Lu, J.; Guan, S. Inhibitory effects of salidroside on nitric oxide and prostaglandin E2 production in lipopolysaccharide-stimulated RAW264.7 macrophages. J. Med. Food 2013, 16, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Ci, X.; Ren, R.; Xu, K.; Li, H.; Yu, Q.; Song, Y.; Wang, D.; Li, R.; Deng, X. Schisantherin a exhibits anti-inflammatory properties by down-regulating NF-κB and mapk signaling pathways in lipopolysaccharide-treated RAW264.7 cells. Inflammation 2010, 33, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Delhase, M. The IκB kinase (IKK) and NF-κB: Key elements of proinflammatory signalling. Semin. Immunol. 2000, 12, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R.; Szabo, G. Interleukin-1 and inflammasomes in alcoholic liver disease/acute alcoholic hepatitis and nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatology 2016, 64, 955–965. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, D.A.; Ko, C.W.; Liu, Y.; Liu, X.; Hise, A.G.; Nunez, G.; Croniger, C.M. Alcohol-induced liver injury is modulated by nlrp3 and nlrc4 inflammasomes in mice. Mediat. Inflamm. 2013, 2013, 751374. [Google Scholar] [CrossRef] [PubMed]
- Beutler, B.; Rietschel, E.T. Innate immune sensing and its roots: The story of endotoxin. Nat. Rev. Immunol. 2003, 3, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lei, T.; Chen, X.; Peng, Y.; Long, H.; Zhou, L.; Huang, J.; Chen, Z.; Long, Q.; Yang, Z. Resistin up-regulates cox-2 expression via TAK1-IKK-NF-κB signaling pathway. Inflammation 2010, 33, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lian, L.H.; Bai, T.; Wu, Y.L.; Wan, Y.; Xie, W.X.; Jin, X.; Nan, J.X. Cryptotanshinone inhibits LPS-induced proinflammatory mediators via tlr4 and TAK1 signaling pathway. Int. Immunopharmacol. 2011, 11, 1871–1876. [Google Scholar] [CrossRef] [PubMed]
- Ishii, K.J.; Koyama, S.; Nakagawa, A.; Coban, C.; Akira, S. Host innate immune receptors and beyond: Making sense of microbial infections. Cell Host Microbe 2008, 3, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R.; Preston-Hurlburt, P.; Janeway, C.A. A human homologue of the drosophila toll protein signals activation of adaptive immunity. Nature 1997, 388, 394–397. [Google Scholar] [PubMed]
- Zeng, K.W.; Fu, H.; Liu, G.X.; Wang, X.M. Icariin attenuates lipopolysaccharide-induced microglial activation and resultant death of neurons by inhibiting TAK1/IKK/NF-κB and jnk/p38 mapk pathways. Int. Immunopharmacol. 2010, 10, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya-Tsuji, J.; Kishimoto, K.; Hiyama, A.; Inoue, J.; Cao, Z.; Matsumoto, K. The kinase TAK1 can activate the NIK-IκB as well as the map kinase cascade in the IL-1 signalling pathway. Nature 1999, 398, 252–256. [Google Scholar] [PubMed]
- Lian, L.H.; Wu, Y.L.; Wan, Y.; Li, X.; Xie, W.X.; Nan, J.X. Anti-apoptotic activity of gentiopicroside in d-galactosamine/lipopolysaccharide-induced murine fulminant hepatic failure. Chem. Biol. Interact. 2010, 188, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.W.; Wu, T.; Qi, J.Y.; Wang, Y.Q.; Luo, X.P.; Ning, Q. Salidroside attenuates LPS-stimulated activation of thp-1 cell-derived macrophages through down-regulation of MAPK/NF-κB signaling pathways. J. Huazhong Univ. Sci. Technol. Med. Sci. 2013, 33, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Bode, C.; Kugler, V.; Bode, J.C. Endotoxemia in patients with alcoholic and non-alcoholic cirrhosis and in subjects with no evidence of chronic liver disease following acute alcohol excess. J. Hepatol. 1987, 4, 8–14. [Google Scholar] [CrossRef]
- Rao, R. Endotoxemia and gut barrier dysfunction in alcoholic liver disease. Hepatology 2009, 50, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Szabo, G.; Bala, S. Alcoholic liver disease and the gut-liver axis. World J. Gastroenterol. 2010, 16, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Marmier, S.; Dentin, R.; Daujat-Chavanieu, M.; Guillou, H.; Bertrand-Michel, J.; Gerbal-Chaloin, S.; Girard, J.; Lotersztajn, S.; Postic, C. Novel role for carbohydrate responsive element binding protein in the control of ethanol metabolism and susceptibility to binge drinking. Hepatology 2015, 62, 1086–1100. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.L.; Lian, L.H.; Jiang, Y.Z.; Nan, J.X. Hepatoprotective effects of salidroside on fulminant hepatic failure induced by d-galactosamine and lipopolysaccharide in mice. J. Pharm. Pharmacol. 2009, 61, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Kanuri, G.; Weber, S.; Volynets, V.; Spruss, A.; Bischoff, S.C.; Bergheim, I. Cinnamon extract protects against acute alcohol-induced liver steatosis in mice. J. Nutr. 2009, 139, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the salidroside are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, P.; Song, S.-Z.; Jiang, S.; Li, X.; Yao, Y.-L.; Wu, Y.-L.; Lian, L.-H.; Nan, J.-X. Salidroside Regulates Inflammatory Response in Raw 264.7 Macrophages via TLR4/TAK1 and Ameliorates Inflammation in Alcohol Binge Drinking-Induced Liver Injury. Molecules 2016, 21, 1490. https://doi.org/10.3390/molecules21111490
Sun P, Song S-Z, Jiang S, Li X, Yao Y-L, Wu Y-L, Lian L-H, Nan J-X. Salidroside Regulates Inflammatory Response in Raw 264.7 Macrophages via TLR4/TAK1 and Ameliorates Inflammation in Alcohol Binge Drinking-Induced Liver Injury. Molecules. 2016; 21(11):1490. https://doi.org/10.3390/molecules21111490
Chicago/Turabian StyleSun, Peng, Shun-Zong Song, Shuang Jiang, Xia Li, You-Li Yao, Yan-Ling Wu, Li-Hua Lian, and Ji-Xing Nan. 2016. "Salidroside Regulates Inflammatory Response in Raw 264.7 Macrophages via TLR4/TAK1 and Ameliorates Inflammation in Alcohol Binge Drinking-Induced Liver Injury" Molecules 21, no. 11: 1490. https://doi.org/10.3390/molecules21111490






