Antileishmanial and Cytotoxic Compounds from Valeriana wallichii and Identification of a Novel Nepetolactone Derivative †
Abstract
:1. Introduction
2. Results and Discussion
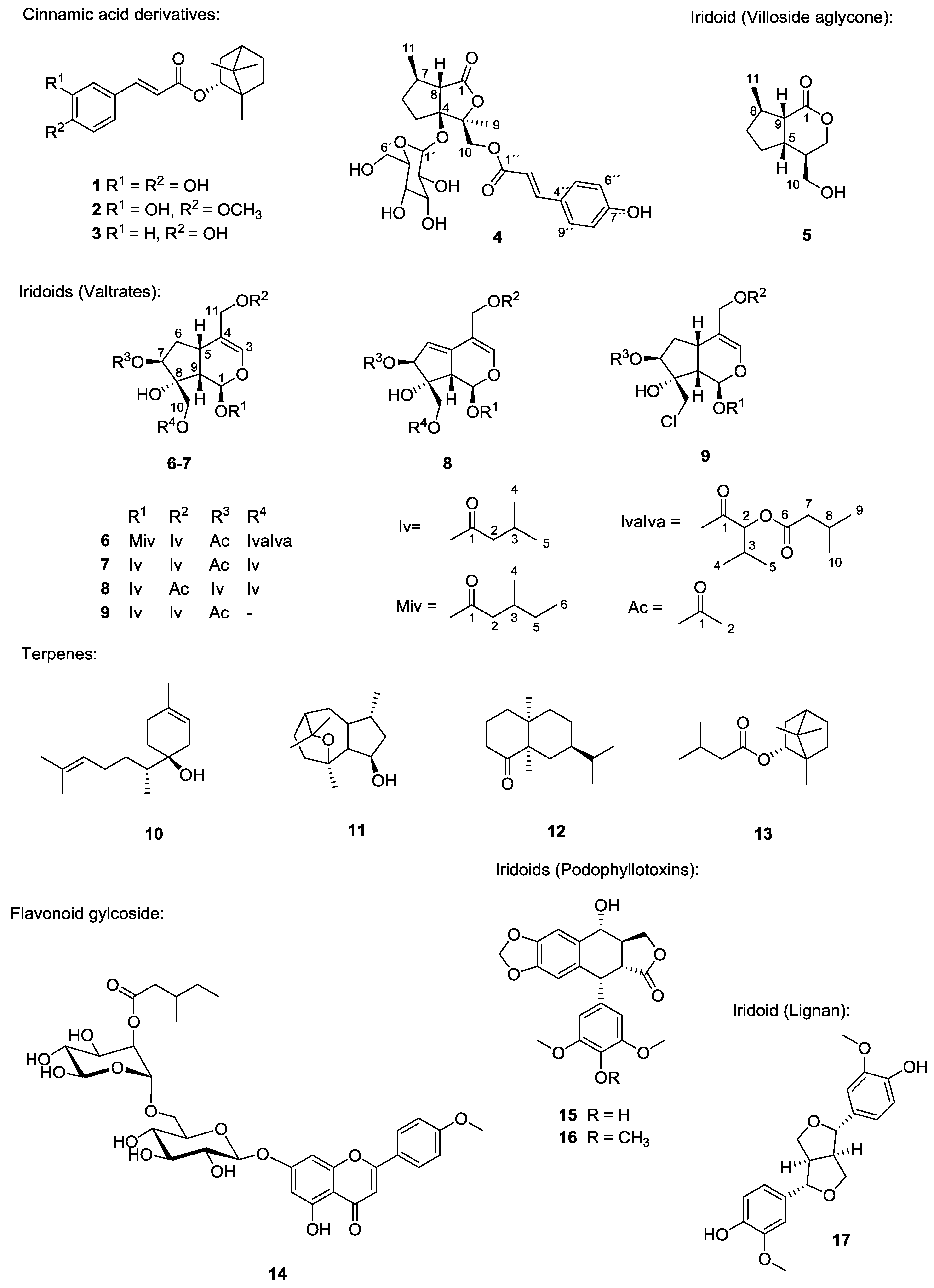
2.1. Bioassay-Guided Fractionation of the Extract
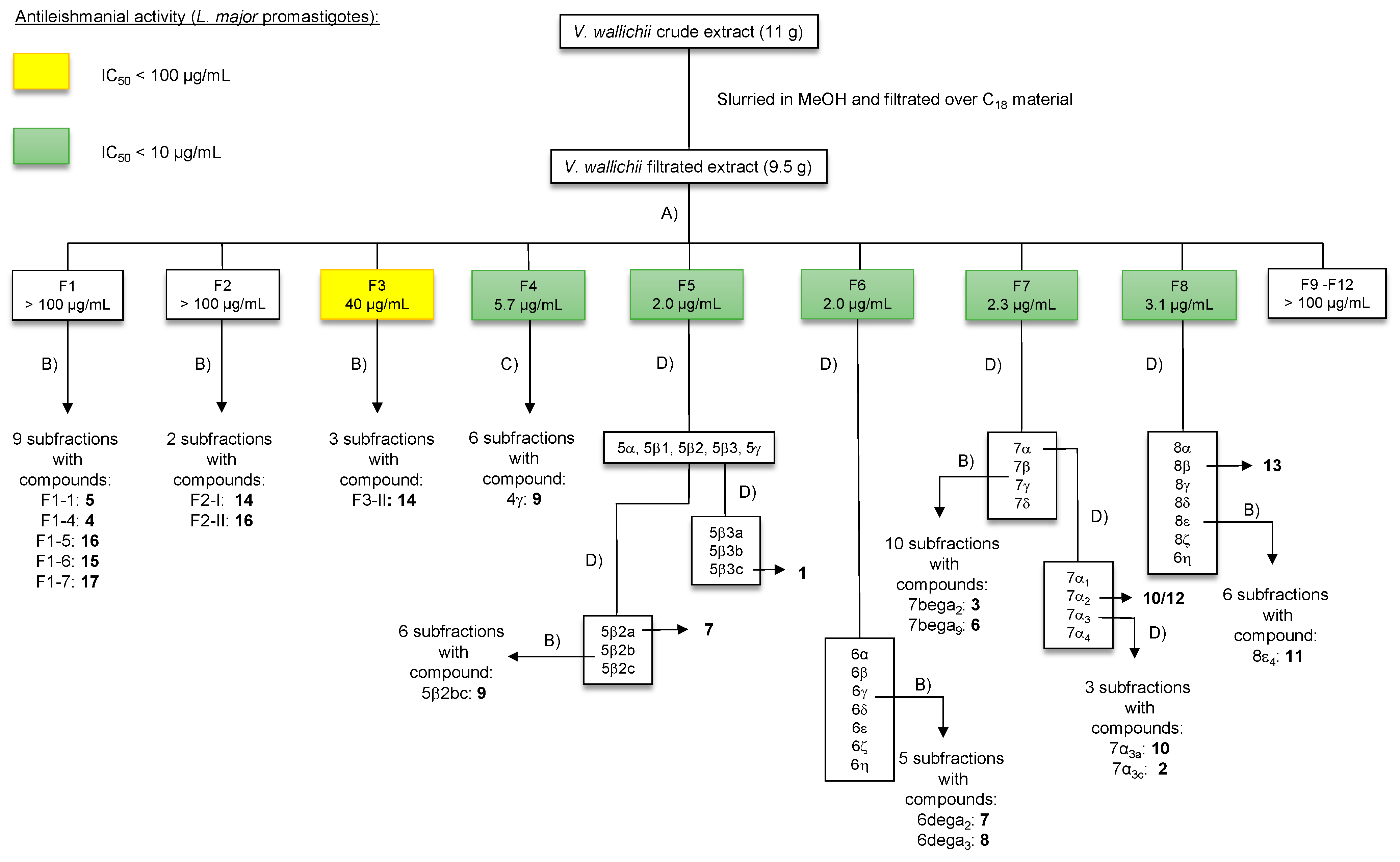
2.2. Structure Elucidation of Novel Nepetolactone Derivative 4

2.3. Structure Elucidation of Villoside Aglycone 5
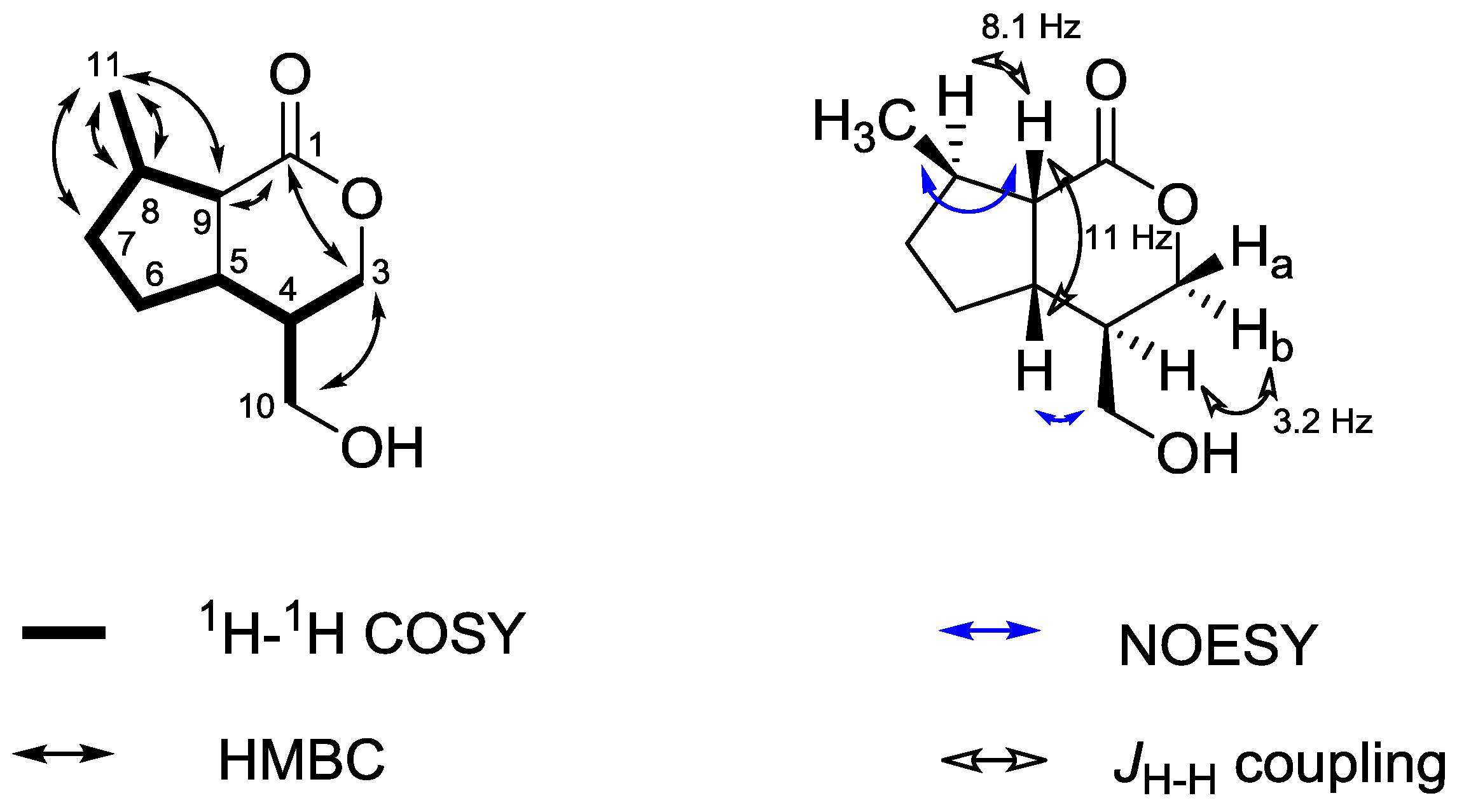
2.4. Valtrates with Antileishmanial Activity
| Position | 13C of 6 | 1H of 6 | 13C of 7 | 1H of 7 | 13C of 8 [20] | 1H of 8 | 13C of 9 [17] | 1H of 9 [17] | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 89.2 | 6.22 (d; 4.7; 1H) | 89.2 | 6.27 (d; 4.2; 1H) | 92.5 | 6.25 (d; 10.0; 1H) | 89.4 | 6.19 (d; 5.1; 1H) | |
| 3 | 141.0 | 6.44 (br s; 1H) | 140.9 | 6.44 (br s; 1H) | 148.0 | 6.68 (br s; 1H) | 141.0 | 6.45 (br s; 1H) | |
| 4 | 112.8 | 112.9 | 108.7 | 113.0 | |||||
| 5 | 31.8 | 2.90 (dd; 9.2; 11.0; 1H) | 31.4 | 2.90 (dd; 7.8; 8.0; 1H) | 139.1 | 32.6 | 2.91 (m; 1H) | ||
| 6 | 34.9 | 2.1 (m; 2H) | 34.7 | 2.12 (m; 2H) | 117.6 | 5.77 (t; 2.8; 1H) | 35.0 | 2.04–2.13 (m; 2H) | |
| 7 | 80.1 | 4.99 (t; 4.5; 1H) | 80.4 | 5.03 (t; 4.7; 1H) | 83.1 | 5.47 (d; 2.8; 1H) | 80.0 | 5.01 (t; 3.9; 1H) | |
| 8 | 80.9 | 80.9 | 80.2 | 81.8 | |||||
| 9 | 44.5 | 2.42 (dd; 6.1; 15.0; 1H) | 44.7 | 2.44 (dd; 4.1; 9.6; 1H) | 48.4 | 2.94 (dd; 2.5; 10.0; 1H) | 45.4 | 2.45 (dd; 5.9; 8.1; 1H) | |
| 10 | 66.8 | 4.29 (d; 11.5; 1H) 4.35 (d; 11.5; 1H) | 66.6 | 4.22 (s; 2H) | 65.5 | 4.32 (d; 11.6; 1H) 4.39 (d; 11.6; 1H) | 49.1 | 3.71 (d; 11.4; 1H) 3.82 (d; 11.4; 1H) | |
| 11 | 63.4 | 4.42 (d; 12.3; 1H) 4.62 (d; 12.3; 1H) | 63.3 | 4.42 (d; 12.3; 1H) 4.62 (d; 12.3; 1H) | 60.9 | 4.64 (d; 12.5; 1H) 4.71 (d; 12.5; 1H) | 63.3 | 4.42 (d; 12.3; 1H) 4.60 (d; 12.3; 1H) | |
| R1 | 1 | 171.3 | 171.1 | 170.8 d | 171.1 | ||||
| 2 | 41.3 | 2.40 (m; 2H) | 43.3 b | 2.23 (m; 4H) | 43.2 | 2.32 (d; 7.0; 2H) | 43.4 g | 2.09–2.13 (m; 4H) | |
| 3 | 31.8 | 1.90 (m; 1H) | 25.6 c | 2.07–2.15 (m; 3H) | 25.7 | 2.05–2.09 (m; 2H) | 25.7 h | 2.00–2.06 (m; 2H) | |
| 4 | 19.5 | 0.97 (m; 3H) | 22.3 a | 0.96 (m; 18H) | 22.3 f | 0.93 (d; 6.6; 6H) g | 22.4 i | 0.97 (d; 6.7; 6H) g | |
| 5 | 29.3 | 1.40 (m; 2H) | 22.3 a | 0.96 (m; 18H) | 22.3 f | 0.93 (d; 6.6; 6H) g | 22.4 i | 0.97 (d; 6.7; 6H) g | |
| 6 | 11.2 | 0.90 (m; 3H) | |||||||
| R2 | 1 | 169.7 | 173.0 | 170.8 d | 172.9 | ||||
| 2 | 43.0 | 2.30–2.35 (m; 4H) | 43.5 | 2.19 (m; 2H) | 20.9 | 2.04 (s; 3H) | 43.3 g | 2.09–2.13 (m; 4H) | |
| 3 | 25.7 | 2.11 (m; 2H) | 25.7 c | 2.07–2.15 (m; 3H) | 25.6 h | 2.00–2.06 (m; 2H) | |||
| 4 and 5 | 22.4 | 0.98 (d; 6.9; 12H) | 22.4 a | 0.96 (m; 18H) | 22.4 i | 0.95 (d; 6.6; 6H) g | |||
| R3 | 1 | 170.2 | 170.3 | 171.8 | 169.8 | ||||
| 2 | 21.0 | 2.07 (s; 3H) | 21.0 | 2.05 (s; 3H) | 43.4 | 2.14 (m; 2H) | 21.0 | 2.08 (s; 3H) | |
| 3 | 25.6 e | 2.05–2.09 (m; 2H) | |||||||
| 4 and 5 | 22.3 f | 0.95 (d; 6.6; 6H) g | |||||||
| R4 | 1 | 173.2 | 172.9 | 173.8 | |||||
| 2 | 76.9 | 4.74 (d; 4.8; 1H) | 43.1 b | 2.23 (m; 4H) | 43.0 | 2.20 (m; 2H) | |||
| 3 | 30.0 | 2.23 (m; 1H) | 25.7 c | 2.07–2.15 (m; 3H) | 25.6 e | 2.17–2.25 (m; 1H) | |||
| 4 | 18.8 | 1.02 (m; 3H) | 22.3 a | 0.96 (m; 18H) | 22.3 f | 0.99 (d; 6.6; 6H) g | |||
| 5 | 17.3 | 1.00 (m; 3H) | 22.3 a | 0.96 (m; 18H) | 22.3 f | 0.99 (d; 6.6; 6H) g | |||
| 6 | 172.9 | ||||||||
| 7 | 43.0 | 2.30–2.35 (m; 4H) | |||||||
| 8 | 25.7 | 2.11 (m; 2H) | |||||||
| 9 and 10 | 22.4 | 0.98 (d; 6.9; 12H) |
2.5. Antileishmanial Activity and Cytotoxicity
| Compound | IC50 (L. major) [µg/mL] | IC50 (J774.1) [µg/mL] |
|---|---|---|
| Bornyl caffeate (1) | 48.8 * | 8.3 * |
| 2 | 16.7 | 34.7 |
| 3 | 12.2 | 8.6 |
| 4 | >100 | >100 |
| 5 | >100 | 17.5 |
| 6 | 1.9 a | <0.8 a |
| 7 | 0.8 | <0.8 |
| 8 | 1.7 * | 1.0 * |
| 9 | 2.3 | 1.9 |
| β-Bisabolol (10) | 52.2 | 37.1 |
| α-Kessyl alcohol (11) | 5.8 b | 1.7 b |
| Valeranone (12) | 60.8 | 42.6 |
| Bornyl isovalerate (13) | >100 * | >100 * |
| Linarin-O-2-methylbutyrate (14) | >100 | 10.8 |
| 15 | >100 | <0.8 |
| 16 | >100 | <0.8 |
| Pinoresinol (17) | >100 | <0.8 |
| Miltefosine | 36.2 * | 56.5 * |
3. Experimental Section
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Characterization of Novel Nepetolactone Derivative {[(3S,4S,7R,8S)-1-Oxo-3,7-dimethyl-4-(O-β-d-glucopyranosyl)oxy]hexahydrocyclopenta[c]furan-3-yl}-methyl-trans-4-hydroxycinnamate (4)
3.5. Characterization of Villoside Aglycone (4R,5S,8R,9S)-4-(Hydroxymethyl)-8-methylhexahydro-cyclopenta[c]pyranone (5)
3.6. Antileishmanial and Cytotoxicity Assays
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sah, S.P.; Mathela, C.; Chopra, K. Valeriana wallichii: A phyto-pharmacological review. J. Pharm. Res. 2010, 3, 2337–2339. [Google Scholar]
- Khuda, F.; Iqbal, Z.; Khan, A.; Nasir, F. Antimicrobial and anti-inflammatory activities of leaf extract of Valeriana wallichii DC. Pak. J. Pharm. Sci. 2012, 25, 715–719. [Google Scholar] [PubMed]
- Agnihotri, S.; Wakode, S.; Ali, M. Chemical composition, antimicrobial and topical anti-inflammatory activity of Valeriana jatamansi Jones. essential oil. J. Essent. Oil Bear. Pl. 2011, 14, 417–422. [Google Scholar] [CrossRef]
- Dua, V.; Alam, M.; Pandey, A.; Rai, S.; Chopra, A.; Kaul, V.; Dash, A. Insecticidal activity of Valeriana jatamansi (Valerianaceae) against mosquitoes. J. Am. Mosq. Control Assoc. 2008, 24, 315–318. [Google Scholar] [CrossRef] [PubMed]
- Ming, D.S.; Yu, D.Q.; Yang, Y.Y.; He, C.H. The structures of three novel sesquiterpenoids from Valeriana jatamansi Jones. Tetrahedron Lett. 1997, 38, 5205–5208. [Google Scholar] [CrossRef]
- Sudhanshu, N.R.; Sandhya, M.; Ekta, M. Evaluation of antioxidant properties of Valeriana wallichi to scavenge free radicals. Asian J. Pharm. Clin. Res. 2012, 5, 238–240. [Google Scholar]
- Thusoo, S.; Gupta, S.; Sudan, R.; Kour, J.; Bhagat, S.; Hussain, R.; Bhagat, M. Antioxidant activity of essential oil and extracts of Valeriana jatamansi roots. BioMed. Res. Int. 2014. [Google Scholar] [CrossRef]
- Ghosh, S.; Debnath, S.; Hazra, S.; Hartung, A.; Thomale, K.; Schultheis, M.; Kapkova, P.; Schurigt, U.; Moll, H.; Holzgrabe, U. Valeriana wallichii root extracts and fractions with activity against Leishmania spp. Parasitol. Res. 2011, 108, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Glaser, J.; Schultheis, M.; Hazra, S.; Hazra, B.; Moll, H.; Schurigt, U.; Holzgrabe, U. Antileishmanial lead structures from nature: Analysis of structure-activity relationships of a compound library derived from caffeic acid bornyl ester. Molecules 2014, 19, 1394–1410. [Google Scholar] [CrossRef] [PubMed]
- Sobolev, A.P.; Brosio, E.; Gianferri, R.; Segre, A.L. Metabolic profile of lettuce leaves by high-field NMR spectra. Magn. Reson. Chem. 2005, 43, 625–638. [Google Scholar] [CrossRef] [PubMed]
- Eisenbraun, E.J.; Browne, C.E.; Irvin-Willis, R.L.; McGurk, D.J.; Eliel, E.L.; Harris, D.L. Structure and stereochemistry of 4aβ,7α,7aβ-nepetalactone from Nepeta mussini and its relationship to the 4aα,7α,7aα -and 4aα,7α,7aβ-nepetalactones from N. cataria. J. Org. Chem. 1980, 45, 3811–3814. [Google Scholar] [CrossRef]
- Lin, S.; Chen, T.; Liu, X.-H.; Shen, Y.-H.; Li, H.-L.; Shan, L.; Liu, R.-H.; Xu, X.-K.; Zhang, W.-D.; Wang, H. Iridoids and lignans from Valeriana jatamansi. J. Nat. Prod. 2010, 73, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, H.; Yokokawa, Y.; Endo, T. Studies on the constituents of Patrinia villosa Juss. Yakugaku Zasshi 1973, 93, 607–611. [Google Scholar] [PubMed]
- Fuzzati, N.; Wolfender, J.; Hostettmann, K.; Msonthi, J.; Mavi, S.; Molleyres, L. Isolation of antifungal valepotriates from Valeriana capense and the search for valepotriates in crude Valerianaceae extracts. Phytochem. Anal 1996, 7, 76–85. [Google Scholar] [CrossRef]
- Bos, R.; Hendriks, H.; Scheffer, J.J.C.; Woerdenbag, H.J. Cytotoxic potential of valerian constituents and valerian tinctures. Phytomedicine 1998, 5, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Shen, Y.-H.; Li, H.-L.; Yang, X.-W.; Chen, T.; Lu, L.-H.; Huang, Z.-S.; Liu, R.-H.; Xu, X.-K.; Zhang, W.-D.; et al. Acylated iridoids with cytotoxicity from Valeriana jatamansi. J. Nat. Prod. 2009, 72, 650–655. [Google Scholar]
- Lin, S.; Zhang, Z.-X.; Chen, T.; Ye, J.; Dai, W.-X.; Shan, L.; Su, J.; Shen, Y.-H.; Li, H.-L.; Liu, R.-H.; et al. Characterization of chlorinated valepotriates from Valeriana jatamansi. Phytochemistry 2013, 85, 185–193. [Google Scholar]
- Bounthanh, C.; Bergmann, C.; Beck, J.P.; Haag-Berrurier, M.; Anton, R. Valepotriates, a new class of cytotoxic and antitumor agents. Planta Med. 1981, 41, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Finner, E.; David, S.; Thies, P. Über die Wirkstoffe des Baldrians. Planta Med. 1984, 50, 4–6. [Google Scholar] [CrossRef] [PubMed]
- Koch, U.; Hölzl, J. The compounds of Valeriana alliariifolia. Planta Med. 1985, 51, 172–173. [Google Scholar] [CrossRef]
- Xu, J.; Li, Y.; Guo, Y.; Guo, P.; Yamakuni, T.; Ohizumi, Y. Isolation, structural elucidation, and neuroprotective effects of iridoids from Valeriana jatamansi. Biosci. Biotechnol. Biochem. 2012, 76, 1401–1403. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.P.; Li, E.W.; Zhang, Q.; Yuan, C.S.; Jia, Z.J. Five new iridoids from Patrinia rupestris. Chem. Biodivers. 2006, 3, 762–770. [Google Scholar] [CrossRef] [PubMed]
- Minyard, J.P.; Thompson, A.C.; Hedin, P.A. Constituents of the cotton bud. VIII. β-Bisabolol, a new sesquiterpene alcohol. J. Org. Chem. 1968, 33, 909–911. [Google Scholar]
- Itô, S.; Kodama, M.; Nozoe, T.; Hikino, H.; Hikino, Y.; Takeshita, Y.; Takemoto, T. Structure and absolute configuration of α-kessyl alcohol and kessyl glycol. Tetrahedron 1967, 23, 553–563. [Google Scholar] [CrossRef]
- Nishiya, K.; Kimura, T.; Takeya, K.; Itokawa, H. Sesquiterpenoids and iridoid glycosides from Valeriana fauriei. Phytochemistry 1992, 31, 3511–3514. [Google Scholar] [CrossRef] [PubMed]
- Vite, G.D.; Spencer, T.A. Synthesis of (+)-7-epivaleranone and (+)-valeranone. J. Org. Chem. 1988, 53, 2560–2565. [Google Scholar] [CrossRef]
- Gränicher, F.; Christen, P.; Kapetanidis, I. Essential oils from normal and hairy roots of Valeriana officinalis var. sambucifolia. Phytochemistry 1995, 40, 1421–1424. [Google Scholar] [CrossRef]
- Mathela, C.S.; Chanotiya, C.S.; Sammal, S.S.; Pant, A.K.; Pandey, S. Compositional diversity of terpenoids in the himalayan Valeriana genera. Chem. Biodivers. 2005, 2, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, J.; Liu, H.; Zhou, L.; Liu, Z.; Wang, J.; Han, J.; Yu, Z.; Yang, F. Chemical analysis and biological activity of the essential oils of two valerianaceous species from China: Nardostachys chinensis and Valeriana officinalis. Molecules 2010, 15, 6411–6422. [Google Scholar] [CrossRef] [PubMed]
- Houghton, P.J. The biological activity of valerian and related plants. J. Ethnopharmacol. 1988, 22, 121–142. [Google Scholar] [CrossRef] [PubMed]
- Bos, R.; Woerdenbag, H.J.; Hendriks, H.; Smit, H.F.; Wikström, H.V.; Scheffer, J.J. Composition of the essential oil from roots and rhizomes of Valeriana wallichii DC. Flavour Fragr. J. 1997, 12, 123–131. [Google Scholar] [CrossRef]
- Sati, S.; Mathela, C. Essential oil composition of Valeriana hardwickii var. arnottiana from the Himalayas. Flavour Fragr. J. 2005, 20, 299–301. [Google Scholar] [CrossRef]
- Chari, V.M.; Jordan, M.; Wagner, H.; Thies, P.W. A 13C-NMR study of the structure of an acyl-linarin from Valeriana wallichii. Phytochemistry 1977, 16, 1110–1112. [Google Scholar] [CrossRef]
- Bringmann, G.; Thomale, K.; Bischof, S.; Schneider, C.; Schultheis, M.; Schwarz, T.; Moll, H.; Schurigt, U. A novel Leishmania major amastigote assay in 96-well format for rapid drug screening and its use for discovery and evaluation of a new class of leishmanicidal quinolinium salts. Antimicrob. Agents Chemother. 2013, 57, 3003–3011. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of novel compounds 4 and 5 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glaser, J.; Schultheis, M.; Moll, H.; Hazra, B.; Holzgrabe, U. Antileishmanial and Cytotoxic Compounds from Valeriana wallichii and Identification of a Novel Nepetolactone Derivative. Molecules 2015, 20, 5740-5753. https://doi.org/10.3390/molecules20045740
Glaser J, Schultheis M, Moll H, Hazra B, Holzgrabe U. Antileishmanial and Cytotoxic Compounds from Valeriana wallichii and Identification of a Novel Nepetolactone Derivative. Molecules. 2015; 20(4):5740-5753. https://doi.org/10.3390/molecules20045740
Chicago/Turabian StyleGlaser, Jan, Martina Schultheis, Heidrun Moll, Banasri Hazra, and Ulrike Holzgrabe. 2015. "Antileishmanial and Cytotoxic Compounds from Valeriana wallichii and Identification of a Novel Nepetolactone Derivative" Molecules 20, no. 4: 5740-5753. https://doi.org/10.3390/molecules20045740





