Cytotoxic Activity and Composition of Petroleum Ether Extract from Magydaris tomentosa (Desf.) W. D. J. Koch (Apiaceae)
Abstract
:1. Introduction
2. Results and Discussion
| Rt a | Component | Id. b | 1% c | 2% c |
|---|---|---|---|---|
| 63.62 | (Z)-9-Hexadecenoic acid methyl ester; methyl palmitoleate | 1, 2, 3 | 0.1 | |
| 64.54 | Hexadecanoic acid methyl ester; methyl palmitate | 1, 2, 3 | 1.6 | |
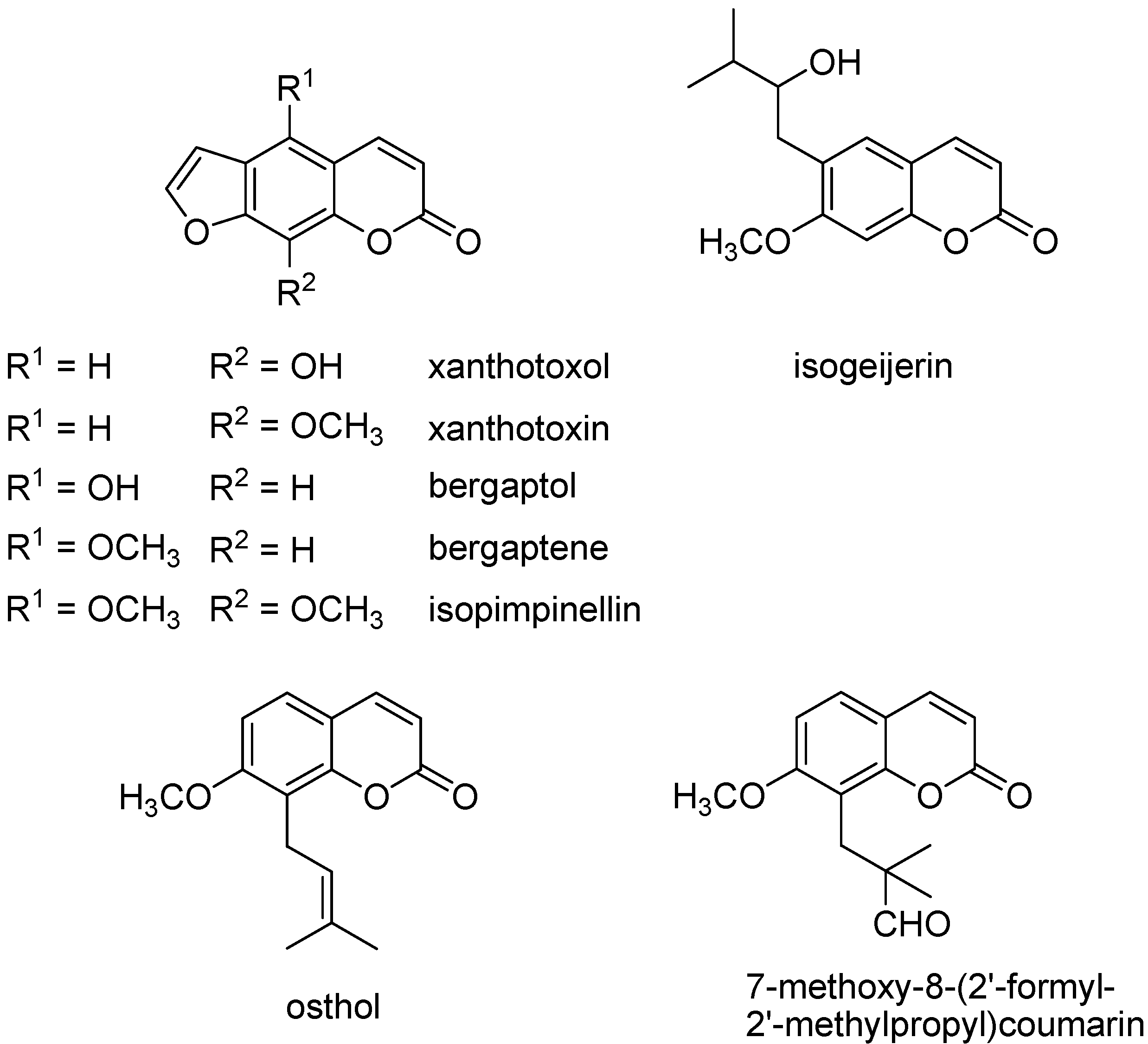
| 68.61 | 5-Demethoxyisoimpinellin; xanthotoxin | 2 | 23.6 | 22.9 |
| 69.57 | Bergaptene | 2 | 3.6 | 3.4 |
| 70.79 | (Z.Z)-9,12-Octadecadienoic acid methyl ester; methyl linoleate | 1, 2, 3 | 0.4 | |
| 71.02 | (Z.Z.Z)-9,12,15-Octadecatrienoic acid methyl ester; methyl linolenate | 1, 2, 3 | 0.3 | |
| 72.05 | Octadecanoic acid methyl ester; methyl stearate | 1, 2, 3 | 0.1 | |
| 72.42 | 7-Methoxy-8-isopentenylcoumarin; osthol | 2 | 6.4 | 5.6 |
| 73.34 | (6E.10E)-7,11,15-Trimethyl-3-methylene-hexadeca-1,6,10,14-tetraene; β-springene | 2 | 0.9 | 1 |
| 75.74 | Isopimpinellin | 2 | 17.8 | 18.8 |
| 76.39 | 7-Methoxy-8-(2'-formyl-2'-methylpropyl)coumarin | 2 | 7.7 | 7.9 |
| 79.6 | Xanthotoxol | 2 | 17.9 | 17.4 |
| 80.74 | Bergaptol | 2 | t | 0.2 |
| 74.91 | 7-Methoxy-6-(2'-oxo-3'-methylbutyl)coumarin; isogeijerin | 2 | 0.9 | 0.5 |
| 84.33 | Pentacosane | 1, 2, 3 | 0.5 | 0.6 |
| 85.34 | Docosanoic acid methyl ester; methyl behenate | 1, 2, 3 | 0.2 | |
| 90.27 | Heptacosane | 1, 2, 3 | 1.2 | 1.4 |
| 91.16 | Tetracosanoic acid methyl ester; methyl lignocerate | 1, 2, 3 | 0.3 | |
| 93.05 | Octacosane | 1, 2, 3 | 0.2 | 0.2 |
| 95.93 | Nonacosane | 1, 2, 3 | 7.2 | 7.6 |
| 96.69 | Hexacosanoic acid methyl ester; methyl cerotate | 1, 2 | 0.2 | |
| 98.41 | Triacontane | 1, 2, 3 | 0.2 | 0.2 |
| 101.03 | Hentriacontane | 1, 2 | 1.8 | 1.9 |
| 101.85 | Octacosanoic acid methyl ester | 1, 2 | 0.1 | |
| 107.73 | Triacontanoic acid methyl ester; methyl melissate | 2 | 0.1 | |
| 112.67 | Hop-22(29)-en-3β-ol | 2 | 0.2 | 0.2 |
| Total | 90.1 | 93.2 |
| IC50 at 72 h | |||
|---|---|---|---|
| J774A.1 | MCF7 | A375 | |
| M. tomentosa flowers petroleum ether extract | 71.52 ± 3.44 | 0.94 ± 0.6 | nd |
| 6-Mercaptopurine | 0.003 | 48.23 | 142.36 |
3. Experimental Section
3.1. Plant Material
3.2. Extraction Procedure
3.3. Reagents
3.4. GC-MS Analysis: Qualitative and Quantitative Analyses of the Extract
3.5. Cell Lines Culture
3.6. MTT Anti-Proliferative Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pignatti, S. Flora d’Italia; Edagricole: Bologna, Italy, 1982; Volume II, p. 211. [Google Scholar]
- Rosselli, S.; Maggio, A.; Bellone, G.; Formisano, C.; Basile, A.; Cicala, C.; Alfieri, A.; Mascolo, N.; Bruno, M. Antibacterial and anticoagulant activities of coumarins isolated from the flowers of Magydaris tomentosa. Planta Med. 2007, 73, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Rosselli, S.; Maggio, A.M.; Bellone, G.; Formisano, C.; Senatore, F.; Bruno, M. A new irregular diterpenoid of biogenetic interest from the flowers of Magydaris tomentosa (Desf.) DC. (Apiaceae). Nat. Prod. Commun. 2007, 2, 5–8. [Google Scholar]
- Curini, M.; Cravotto, G.; Epifano, F.; Giannone, G. Chemistry and biological activity of natural and synthetic prenyloxycoumarins. Curr. Med. Chem. 2006, 13, 199–222. [Google Scholar] [CrossRef] [PubMed]
- Macias, F.A.; Galindo, J.C.G.; Massanet, G.M.; Rodriguez-Luis, F.; Zubia, E. Natural product models as allelochemicals. 2. Allelochemicals from Pilocarpus goudotianus leaves. J. Chem. Ecol. 1993, 19, 1371–1379. [Google Scholar] [CrossRef] [PubMed]
- Conforti, F.; Marrelli, M.; Menichini, F.; Bonesi, M.; Statti, G.; Provenzano, E.; Menichini, F. Natural and synthetic furanocoumarins as treatment for vitiligo and psoriasis. Curr. Drug Ther. 2009, 4, 38–58. [Google Scholar] [CrossRef]
- Messer, A.; Raquet, N.; Lohr, C.; Schrenk, D. Major furocoumarins in grapefruit juice II: Phototoxicity, photogenotoxicity, and inhibitory potency vs. cytochrome P450 3A4 activity. Food. Chem. Toxicol. 2012, 50, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Abdel Hafez, O.M.; Amin, K.M.; Abdel-Latif, N.A.; Mohamed, T.K.; Ahmed, E.Y.; Maher, T. Synthesis and antitumor activity of some new xanthotoxin derivatives. Eur. J. Med. Chem. 2009, 44, 2967–2974. [Google Scholar]
- Fujioka, T.; Furumi, K.; Fujii, H.; Okabe, H.; Mihashi, K.; Nakano, Y.; Matsunaga, H.; Katano, M.; Mori, M. Antiproliferative constituents from Umbelliferae plants. V. A new furanocoumarin and falcarindiol furanocoumarin ethers from the root of Angelica japonica. Chem. Pharm. Bull. 1999, 47, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Maneerat, W.; Prawat, U.; Saewan, N.; Laphookhieo, S. New coumarins from Clausena lansium twigs. J. Braz. Chem. Soc. 2010, 21, 665–668. [Google Scholar] [CrossRef]
- Kleiner, H.E.; Reed, M.J.; DiGiovanni, J. Naturally occurring coumarins inhibit human cytochromes P450 and block benzo[a]pyrene and 7,12-dimethylbenz[a]anthracene DNA adduct formation in MCF-7 cells. Chem. Res. Toxicol. 2003, 16, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.F.; Nordby, H.E. Two new coumarins from grapefruit peel oil. Tetrahedron 1966, 22, 1489–1493. [Google Scholar] [CrossRef]
- Gonzalez, A.G.; Barroso, J.T.; Lopez Dorta, H.; Luis, J.R.; Rodriguez Luis, F. Umbellifera components. XVII. Simple coumarins of Seseli tortuosum L.B.S. Eur. Anal. Quim. 1978, 74, 979–984. [Google Scholar]
- Imai, F.; Kinoshita, T.; Sankawa, U. Constituents of the leaves of Murraya paniculata collected in Taiwan. Chem. Pharm. Bull. 1989, 37, 358–362. [Google Scholar] [CrossRef]
- You, L.; An, R.; Wang, X.; Li, Y. Discovery of novel osthole derivatives as potential anti-breast cancer treatment. Bioorg. Med. Chem. Lett. 2010, 20, 7426–7428. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.-L.; Wang, M.-C.; Chen, L.-G.; Wang, C.-C. Cytotoxic activity of coumarins from the fruits of Cnidium monnieri on leukemia cell lines. Planta Med. 2003, 69, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Diwan, R.; Malpathak, N. Furanocoumarins: Novel topoisomerase I inhibitors from Ruta graveolens L. Bioorg. Med. Chem. 2009, 17, 7052–7055. [Google Scholar] [CrossRef] [PubMed]
- Bianco, G.; Russo, R.; Marzocco, S.; Velotto, S.; Autore, G.; Severino, L. Modulation of macropahge activity by aflatoxins B1 and B2 and their metabolites aflatoxins M1 and M2. Toxicon 2012, 59, 644–650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conforti, F.; Menichini, F.; Formisano, C.; Rigano, D.; Senatore, F.; Bruno, M.; Rosselli, S.; Çelik, S. Anthemis wiedemanniana essential oil prevents LPS-induced production of NO in RAW 264.7 macrophages and exerts antiproliferative and antibacterial activities “in vitro”. Nat. Prod. Res. 2012, 26, 1594–1601. [Google Scholar] [CrossRef] [PubMed]
- Zito, P.; Sajeva, M.; Bruno, M.; Maggio, A.; Rosselli, S.; Formisano, C.; Senatore, F. Essential oil composition of stems and fruits of Caralluma europaea N.E.Br. (Apocynaceae). Molecules 2010, 15, 627–638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Autore, G.; Marzocco, S.; Formisano, C.; Bruno, M.; Rosselli, S.; Jemia, M.B.; Senatore, F. Cytotoxic Activity and Composition of Petroleum Ether Extract from Magydaris tomentosa (Desf.) W. D. J. Koch (Apiaceae). Molecules 2015, 20, 1571-1578. https://doi.org/10.3390/molecules20011571
Autore G, Marzocco S, Formisano C, Bruno M, Rosselli S, Jemia MB, Senatore F. Cytotoxic Activity and Composition of Petroleum Ether Extract from Magydaris tomentosa (Desf.) W. D. J. Koch (Apiaceae). Molecules. 2015; 20(1):1571-1578. https://doi.org/10.3390/molecules20011571
Chicago/Turabian StyleAutore, Giuseppina, Stefania Marzocco, Carmen Formisano, Maurizio Bruno, Sergio Rosselli, Mariem Ben Jemia, and Felice Senatore. 2015. "Cytotoxic Activity and Composition of Petroleum Ether Extract from Magydaris tomentosa (Desf.) W. D. J. Koch (Apiaceae)" Molecules 20, no. 1: 1571-1578. https://doi.org/10.3390/molecules20011571






