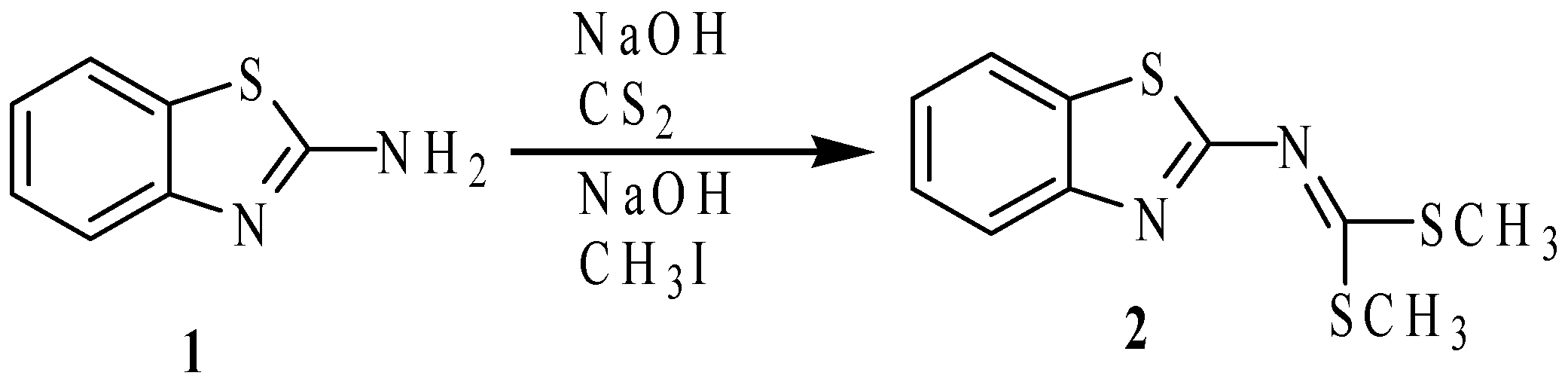
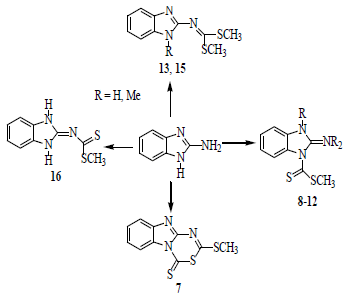
2.1. Synthesis
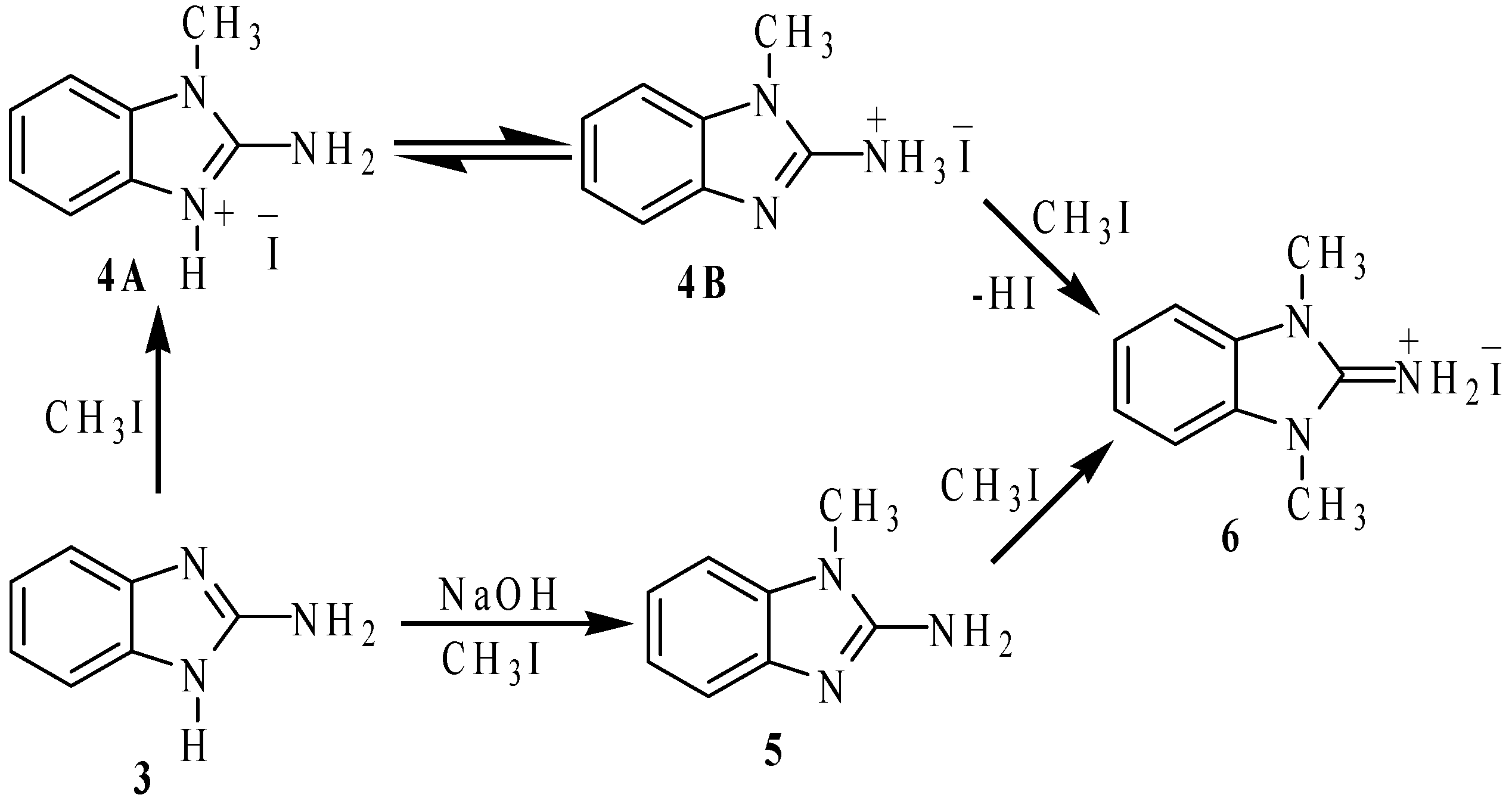
To investigate the nitrogen nucleophilicity, we reacted 2-aminobenzimidazole with methyl iodide (MeI) in ethanol as solvent (
Scheme 2). If one molar equivalent of MeI was added, a mixture of the iodide salts: 1-methyl-2-ammoniumbenzimidazole (
4, 30%), 1,3-dimethyl-2-ammoniumbenzimidazole (
6, 30%) and 2-ammoniobenzimidazole (30%) were observed in the
1H-NMR spectrum. To explain this result, we propose that 2-aminobenzimidazole is methylated to produce the iodide salt of the
N-methylated compound
4A as intermediate, which is immediately transformed into the more stable tautomer
4B. When two molar equivalents of methyl iodide were used, the basic imidazolic nitrogen atom of the iodide salt
4B is methylated to give the dimethylated iodide salt
6 (60%). The remaining 2-aminobenzimidazole traps the generated HI to give the iodide salt of 2-ammoniumbenzimidazole (30%), as side product.
Scheme 2.
Methylation reactions of 2-aminobenzimidazole (3).
Scheme 2.
Methylation reactions of 2-aminobenzimidazole (3).
In order to isolate the
N-methylbenzimidazole
5, we reacted 2-aminobenzimidazole (
3) with one molar equivalent of sodium hydroxide using as DMF solvent, followed by one molar equivalent of MeI, as depicted in
Scheme 2. Under these conditions, the reaction afforded a 1:1:1 mixture of
5, the dimethylated iodide salt
6 and unreacted
3. The same reaction with two molar equivalents of MeI affords
6 as the only product in 90% yield.
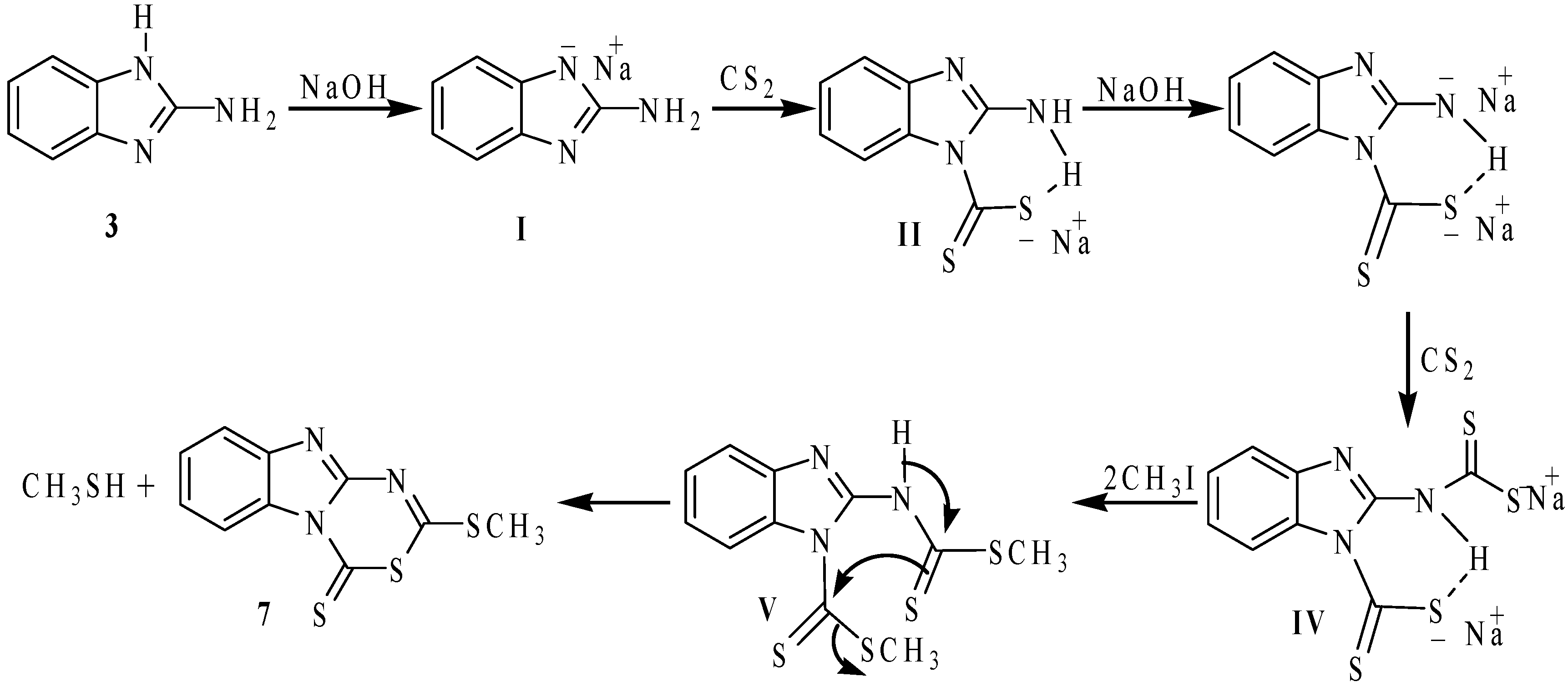
On these bases, a detailed study and the characterization of the compounds derived from the reaction of 2-aminobenzimidazole (
3) with carbon disulfide in basic media followed by methylation with methyl iodide, using DMF as solvent were performed. When
3 was reacted with CS
2 and CH
3I in basic (NaOH) medium, following the reported procedure for the synthesis of compound
2 [
8], the reaction failed to give the analogous compound, and instead a 1:3 mixture of compounds
7 and
8 were observed in the
1H- and
13C-NMR spectra (
Scheme 3). With separation purposes, this mixture was dissolved in ethanol. 2-Methylthio-4
H-[1,3,5]thiadiazino[3,4-
a]benzimidazole-4-thione (
7) was isolated from the mixture as an insoluble yellow solid and after recrystallization from chloroform, it was obtained as yellow crystals in 15% yield. 2-Aminobenzimidazole-1-carbodithioic acid methyl ester (
8) precipitated from the ethanol solution, as yellow crystals in 40% yield as the previously described polymorph [
15]. In this reaction, the small quantity of compound
7 is formed from
8 because CH
3I, NaOH and CS
2 were added in 20% molar excess. Compounds
7 and
8 were obtained in 40% and 66% yield, respectively, when the reactions were carried out in stoichiometric quantities. These results are in agreement with the stronger acidic character of the imidazolic hydrogen atom than that of the exocyclic 2-amino group.
Scheme 3.
Reaction of 2-aminobenzimidazole (3) with CS2 and methyl iodide in basic media.
Scheme 3.
Reaction of 2-aminobenzimidazole (3) with CS2 and methyl iodide in basic media.
The formation of compounds
7 and
8 can be explained by assuming that the benzimidazolate
I, obtained from the reaction of
3 with NaOH, which reacts with one molar equivalent of CS
2 to give the thiocarbamate
II that, after methylation, affords compound
8. The intermediate
III, formed by a second molar equivalent of NaOH, reacts with a second molar equivalent of CS
2 to produce the dithiocarbamate dianion
IV. The subsequent methylation of
IV with two molar equivalents of MeI, affords the proposed intermediate compound
V, which is finally cyclized to give the heterocyclic compound
7. This mechanistic proposal is depicted in
Scheme 4.
Scheme 4.
Mechanistic pathway to obtain compound 7.
Scheme 4.
Mechanistic pathway to obtain compound 7.
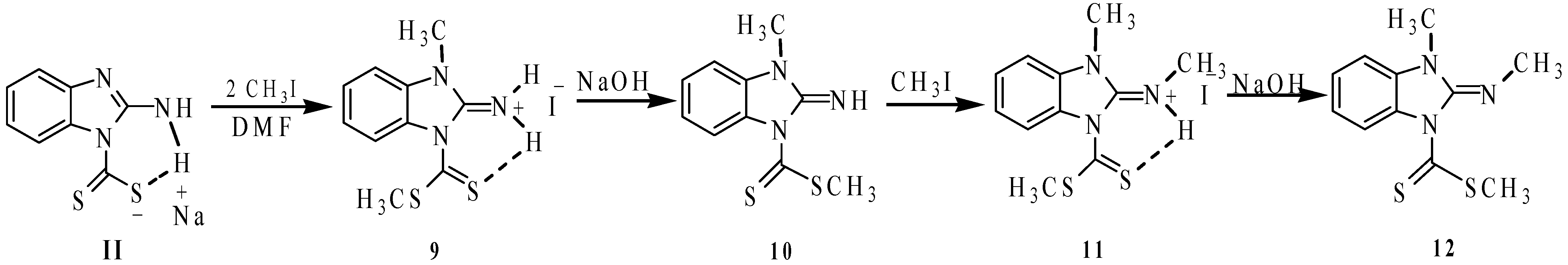
In order to obtain the compound
10, we reacted 2-aminobenzimidazole (
3) with exactly one molar equivalent of NaOH and one molar equivalent of CS
2 followed by the addition of two molar equivalents of MeI. Under these conditions, the iminium iodide salt
9 was formed. Compound
9 was neutralized with NaOH to give 2-imino-3-methyl-2,3-dihydro-benzimidazol-1-carbodithioic acid methyl ester (
10). Subsequent methylation at the imine nitrogen of compound
10 was performed to give
N-(1-methyl-3-(methylthiocarbonothioyl)-1
H-benzo[
d]imidazol-2(3
H)-ylidene)methanaminium iodide (
11). 3-Methyl-2-methylimino-2,3-dihydro-benzoimidazole-1-carbodithioic acid methyl ester (
12) was obtained by deprotonating
11 with NaOH. The sequence of reactions is depicted in
Scheme 5.
Scheme 5.
Synthetic method to get compounds 9–12.
Scheme 5.
Synthetic method to get compounds 9–12.
The reaction of 2-aminobenzimidazole with NaOH and CS
2 in an equimolar ratio, in refluxing DMF by 8 h was carried out, then one molar equivalent of NaOH and two molar equivalents of CH
3I were subsequently added. Under these conditions, a white solid precipitates from the aqueous-DMF solution. The solid compound was purified by recrystallization in ethanol and white crystals were obtained in 25% yield. This compound was characterized by NMR and X-ray diffraction analysis and the structure corresponded to dimethyl 1
H-benzo[
d]imidazol-2-ylcarbonodithioimidate (
13,
Scheme 6). The reaction proceeded through the intermediacy of the exocyclic sodium amide
VII whose formation is favored by heating.
Scheme 6.
Synthetic pathway to access to compounds 13–17.
Scheme 6.
Synthetic pathway to access to compounds 13–17.
When three molar equivalents of methyl iodide were used in the methylation reaction to obtain
13, the hydroiodide salt of dimethyl 1-methyl-1
H-benzo[
d]imidazol-2-ylcarbonodithioimidate (
14) precipitated from the aqueous-DMF solution and was crystallized from ethanol. The structure of this compound was analyzed by X-ray diffraction (
vide infra). After neutralization of
14 with one equivalent of NaOH, dimethyl 1-methyl-1
H-benzo[
d]imidazol-2-ylcarbonodithioimidate (
15) was obtained quantitatively as a white solid. This compound has already been obtained from 1-methyl-2-aminobenzimidazole whose NMR data and X-ray diffraction structure has been reported elsewhere [
16].
In order to obtain (1,3-dihydrobenzoimidazol-2-ylidene)-dithiocarbamic acid methyl ester (16), the intermediate VIII was prepared in situ and then methylated with one molar equivalent of CH3I, to afford compound 16 in mixture with the iodide salt of 2-(bis(methylthio)methyleneamino)-1H-benzo[d]imidazol-3-ium (17) which was separated as a precipitate from ethanol solution.
2.2. Molecular Structure in Solution by NMR
A complete list of
1H and
13C-NMR data of compounds
7 and
9–
14 and
16–
17 is given in
Table 1 and
Table 2, respectively, to support the proposed structures.
Table 1.
1H-NMR chemical shifts of compounds
7,
9–
14,
16 and
17.
![Molecules 19 13878 i001]()
Table 1.
1H-NMR chemical shifts of compounds 7, 9–14, 16 and 17. ![Molecules 19 13878 i001]()
| Comp. | H4 | H5 | H6 | H7 | NH | NCH3 | SCH3 |
|---|
| 7 b | 7.82 | 7.43 | 7.54 | 9.00 | – | – | 2.80 |
| 9 a | 7.58 | 7.46 | 7.31 | 7.24 | 9.3 | 3.67 | 2.91 |
| 10 b | 6.85 | 7.19 | 6.98 | 8.10 | 7.3 | 3.41 | 2.78 |
| 11 a | 7.59 | 7.41 | 7.31 | 7.61 | 9.1 | 3.71, 3.10 | 2.96 |
| 12 b | 6.77 | 6.93 | 7.11 | 8.12 | – | 3.40, 3.25 | 2.71 |
| 13 b | 7.50 | 7.10 | 7.11 | 7.32 | 12.2 | – | 2.57 |
| 14 a | 7.78 | 7.47 | 7.45 | 7.60 | 8.6 | 3.77 | 2.73 |
| 16 | 7.24 | 7.49 | 7.49 | 7.24 | 13.0 | – | 2.44 |
| 17 | 7.10 | 7.40 | 7.40 | 7.10 | – | – | 2.55 |
Table 2.
13C-NMR chemical shifts of compounds 7, 9–14, 16 and 17.
Table 2.
13C-NMR chemical shifts of compounds 7, 9–14, 16 and 17.
| Comp. | C2 | C4 | C5 | C6 | C7 | C8 | C9 | C10 | C11 | NCH3 | SCH3 |
|---|
| 7 a | 172.3 | 127.9 | 120.4 | 125.8 | 118.0 | 131.8 | 142.4 | 182.8 | 172.4 | – | 14.3 |
| 9 b | 148.6 | 125.6 | 111.9 | 124.8 | 111.6 | 129.4 | 131.0 | 200.3 | – | 30.6 | 22.8 |
| 10 a | 151.5 | 125.9 | 122.3 | 121.4 | 118.9 | 129.1 | 132.7 | 201.6 | – | 28.5 | 20.4 |
| 11 a | 149.0 | 125.7 | 124.8 | 111.3 | 111.3 | 129.7 | 131.1 | 201.4 | – | 31.6, 31.3 | 23.0 |
| 12 b | 145.5 | 124.4 | 120.1 | 112.5 | 106.3 | 131.0 | 134.5 | 203.3 | – | 36.0, 29.8 | 21.5 |
| 13 a | 153.2 | 118.9 | 121.8 | 118.9 | 111.1 | 133.0 | 143.0 | 173.5 | – | – | 16.1 |
| 14 a | 149.2 | 114.7 | 125.3 | 125.8 | 114.7 | 130.8 | 132.1 | 185.8 | – | 30.7 | 16.7 |
| 16 b | 151.2 | 112.8 | 124.2 | 124.2 | 112.8 | 129.1 | 129.1 | 205.6 | – | – | 18.6 |
| 17 a | 152.9 | 114.8 | 121.8 | 121.8 | 114.8 | 138.0 | 138.0 | 173.4 | – | – | 15.9 |
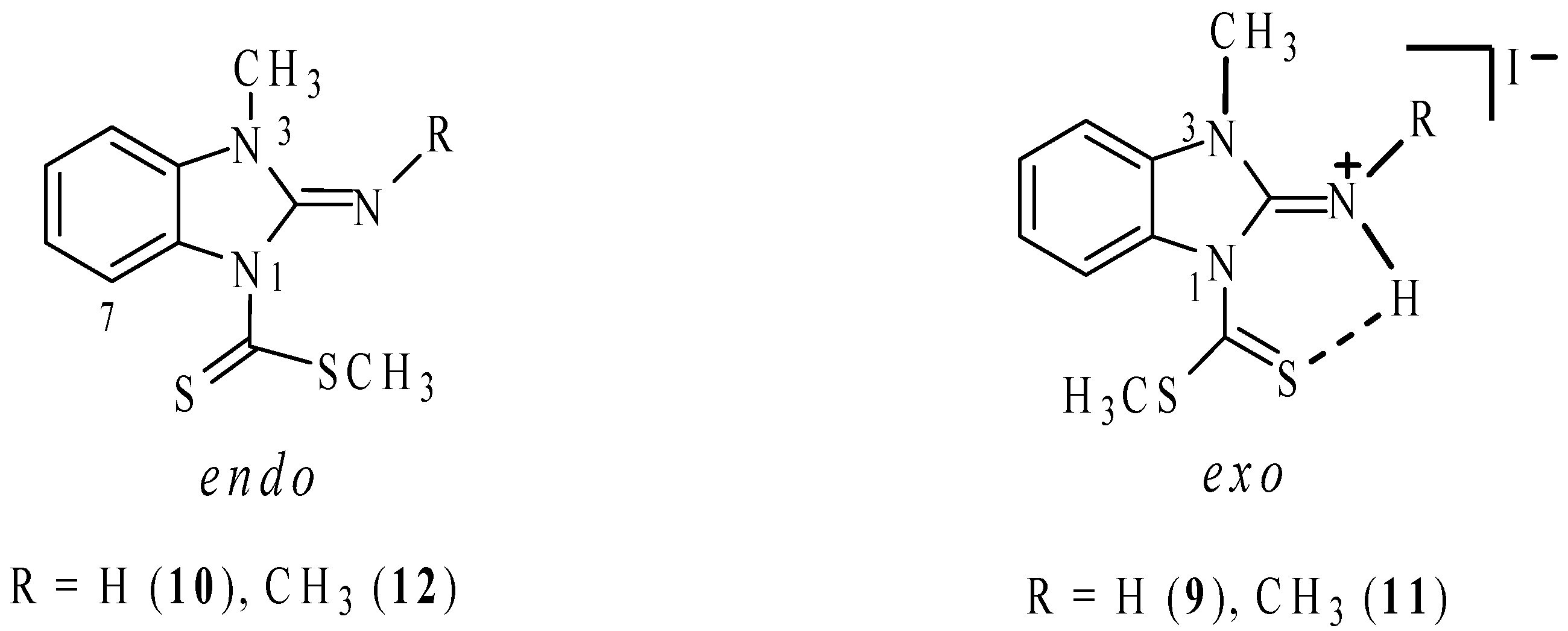
The chemical shift of H7 in compounds
7 and
9–
12, is sensitive to the conformation of the C=S group. It appears as a doublet at δ 9.0 in compound
7 because of the deshielding effect of the neighbouring thiocarbonyl group. In this context, it is worth noting that the chemical shift of H7 for the neutral compounds
10 and
12, is approximately at 8.1; this shift to higher frequencies suggests that the preferred conformation of the thiocarbonyl group on the NMR time scale is
endo (
Figure 1). In contrast, the hydrogen atom on the exocyclic nitrogen atom in imminium compounds
9 and
11, forms a hydrogen bond with the sulfur atom of the thiocarbonyl group. This interaction has the effect to fix the conformation of the thiocarbonyl group in
exo disposition, as has been reported for compound
8, [
15] shifting H7 to lower frequencies at 7.24 and 7.61 ppm, respectively (
Scheme 7).
Figure 1.
Endo conformers of compounds 10 and 12 and exo ones of compounds 9 and 11.
Figure 1.
Endo conformers of compounds 10 and 12 and exo ones of compounds 9 and 11.
Scheme 7.
Isomerization of 11-exo in DMSO-d6 solution. nOes are represented by double headed curved arrows.
Scheme 7.
Isomerization of 11-exo in DMSO-d6 solution. nOes are represented by double headed curved arrows.
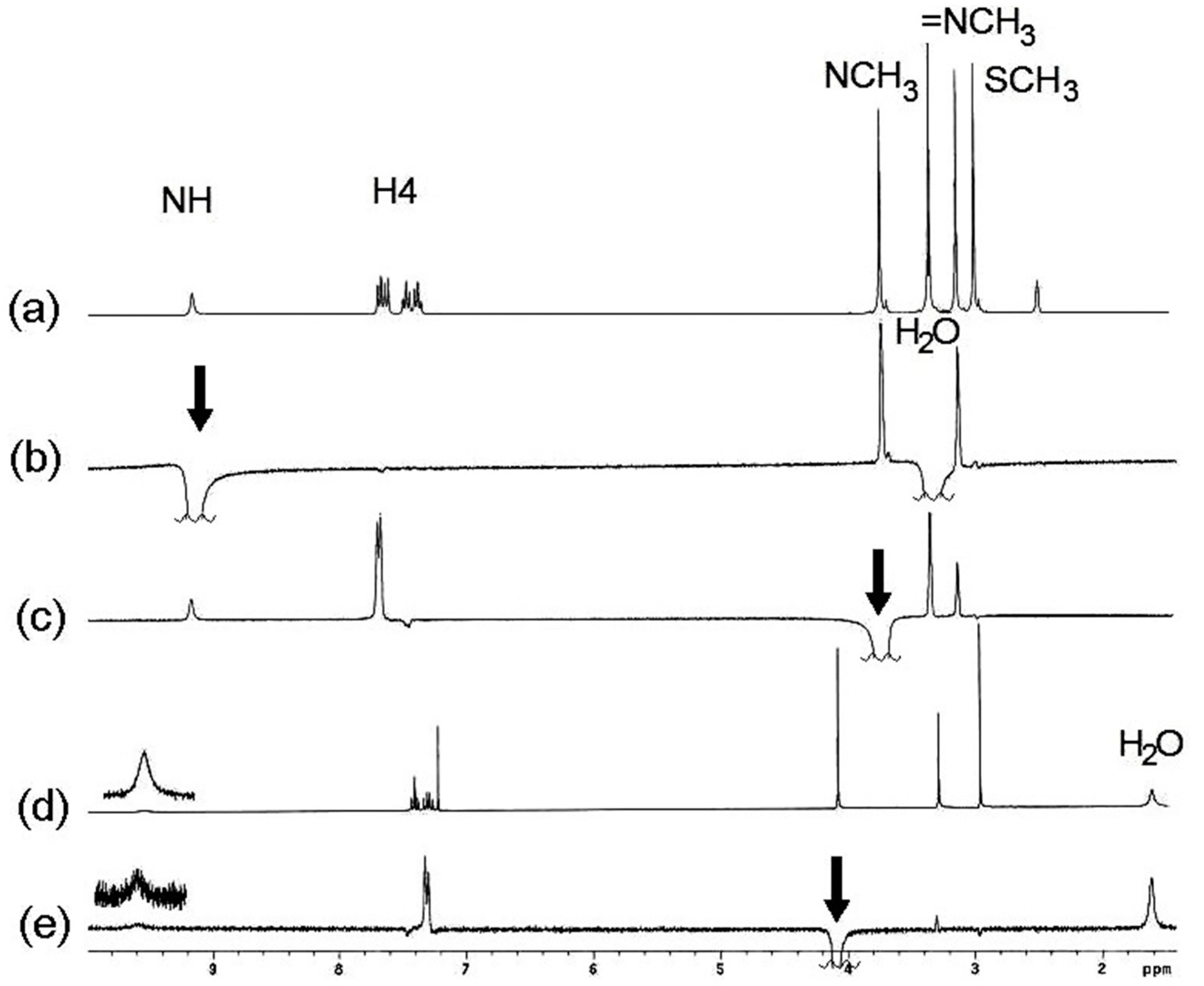
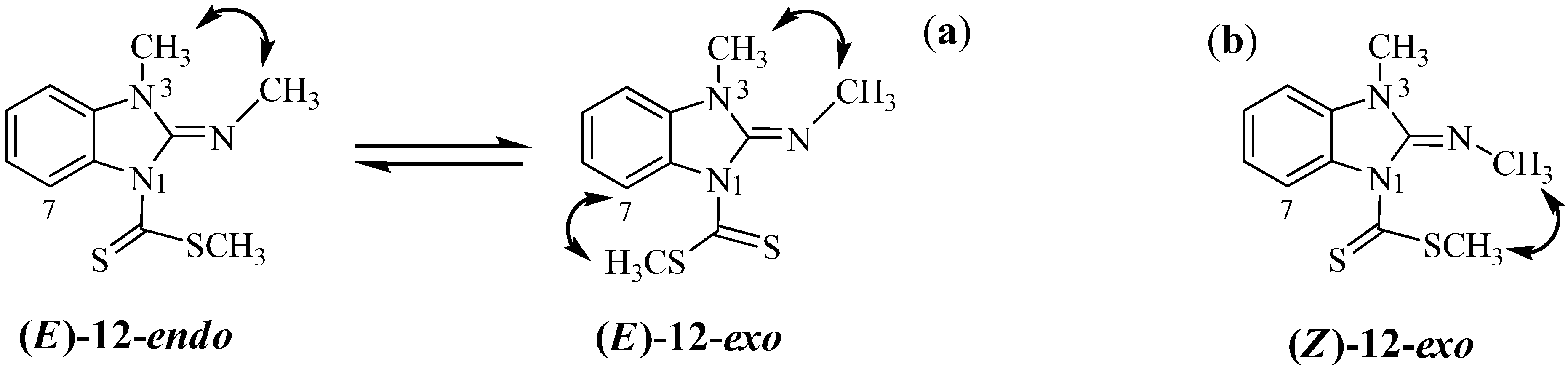
In order to confirm the stereochemistry of compound
11, nOe experiments were carried out. Irradiation of the NH gave a nOe on both N-CH
3 groups (
Figure 2b) and the irradiation of the N3-CH
3 protons gave a nOe on both the CH
3 and NH protons of the exocylic C=N group (
Figure 2c), in DMSO-
d6 solution. After the irradiation of the N3-CH
3 signal, nOe was observed only on the NH signal in CDCl
3 solution (
Figure 2e). In every case, the nOe was not observed on the SCH
3 signal. These findings can be explained due to an isomerization process mediated by the participation of the lone pair of electrons on N3 (
Scheme 7). In DMSO-
d6 solution both
E and
Z isomers are in a fast equilibrium in the
1H-NMR time scale, whereas in CDCl
3 solution the last isomer is the preferred.
Figure 2.
(a) 1H-NMR spectrum of compound 11 in DMSO-d6; (b) After irradiation of the NH signal, nOes on both CH3 groups are observed; (c) nOe on the NH, exocyclic NCH3 and H4 after irradiation of the N3-CH3 signal; (d) 1H-NMR spectrum of compound 11 in CDCl3; (e) Irradiation of N3-CH3 gave a nOe on NCH3 in CDCl3.
Figure 2.
(a) 1H-NMR spectrum of compound 11 in DMSO-d6; (b) After irradiation of the NH signal, nOes on both CH3 groups are observed; (c) nOe on the NH, exocyclic NCH3 and H4 after irradiation of the N3-CH3 signal; (d) 1H-NMR spectrum of compound 11 in CDCl3; (e) Irradiation of N3-CH3 gave a nOe on NCH3 in CDCl3.
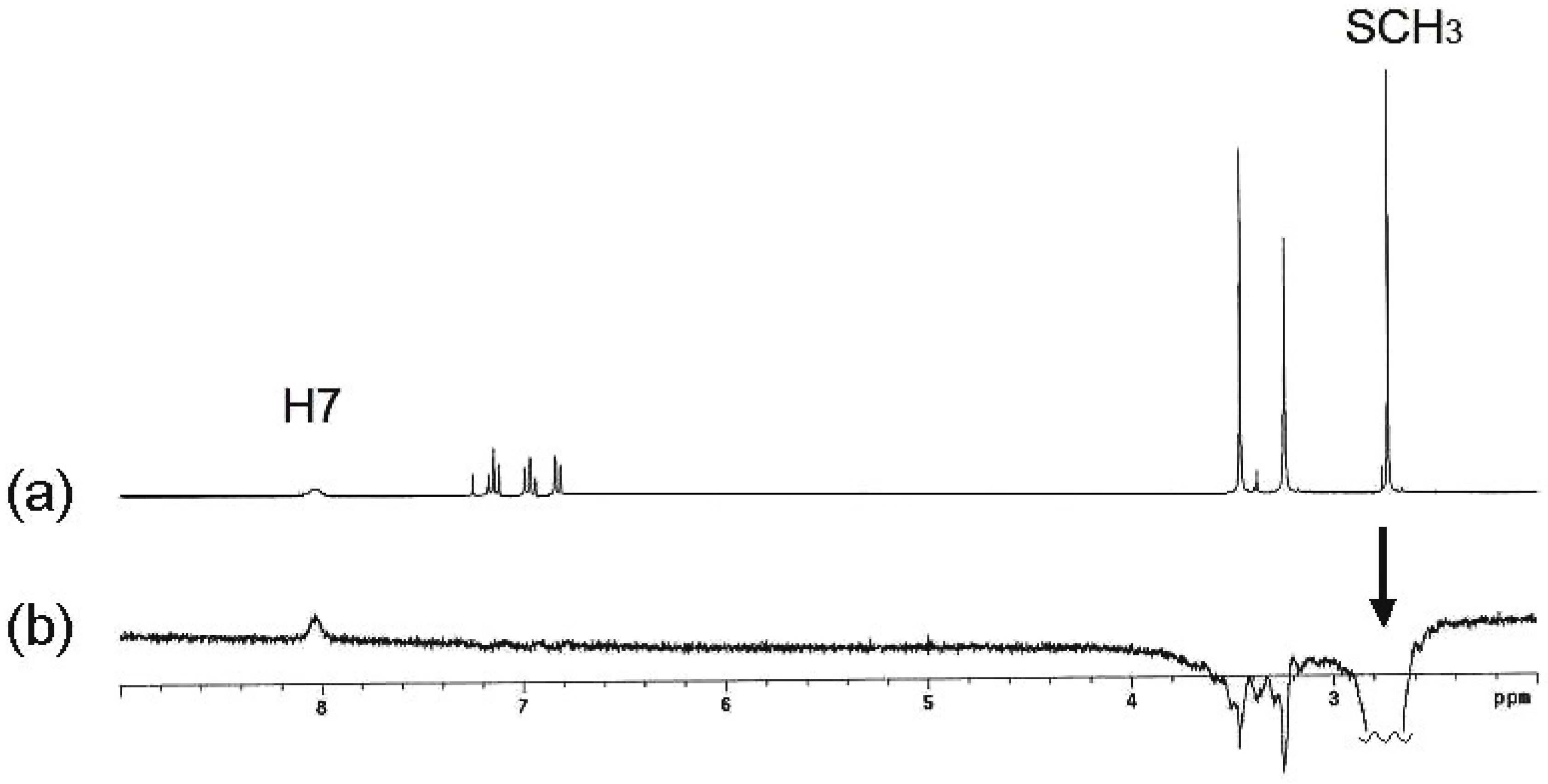
The shift of this equilibrium to the (
E)-
11-
exo isomer in polar solvents, explains the isolation of the neutral compound
12 with both NCH
3 groups in
syn disposition and preference for the
endo rotamer, on the
1H-NMR time scale. The stereochemistry of compound (
E)
-12 was confirmed by nOe experiments (
Figure 3). After irradiation of the SCH
3 signal, both in DMSO-
d6 and CDCl
3 solutions, a very small nOe was observed on the H7 proton but not on the exocyclic NCH
3, as expected for the (
Z)
-12-exo isomer (
Scheme 8). nOe on N3-CH
3 was useless because the close proximity with the chemical shift of the exocyclic N-CH
3.
Figure 3.
(a) 1H-NMR spectrum of compound 12 in CDCl3; (b) After irradiation of the SCH3 signal, nOe on H7 was observed.
Figure 3.
(a) 1H-NMR spectrum of compound 12 in CDCl3; (b) After irradiation of the SCH3 signal, nOe on H7 was observed.
Scheme 8.
Endo-exo equilibrium in compound (E)-12. Expected nOes are represented by double headed curved arrows in E (a) and Z (b) isomers of compound 12.
Scheme 8.
Endo-exo equilibrium in compound (E)-12. Expected nOes are represented by double headed curved arrows in E (a) and Z (b) isomers of compound 12.
2.3. Molecular Structure of Compounds 7, 13 and 14 by X-Ray Diffraction
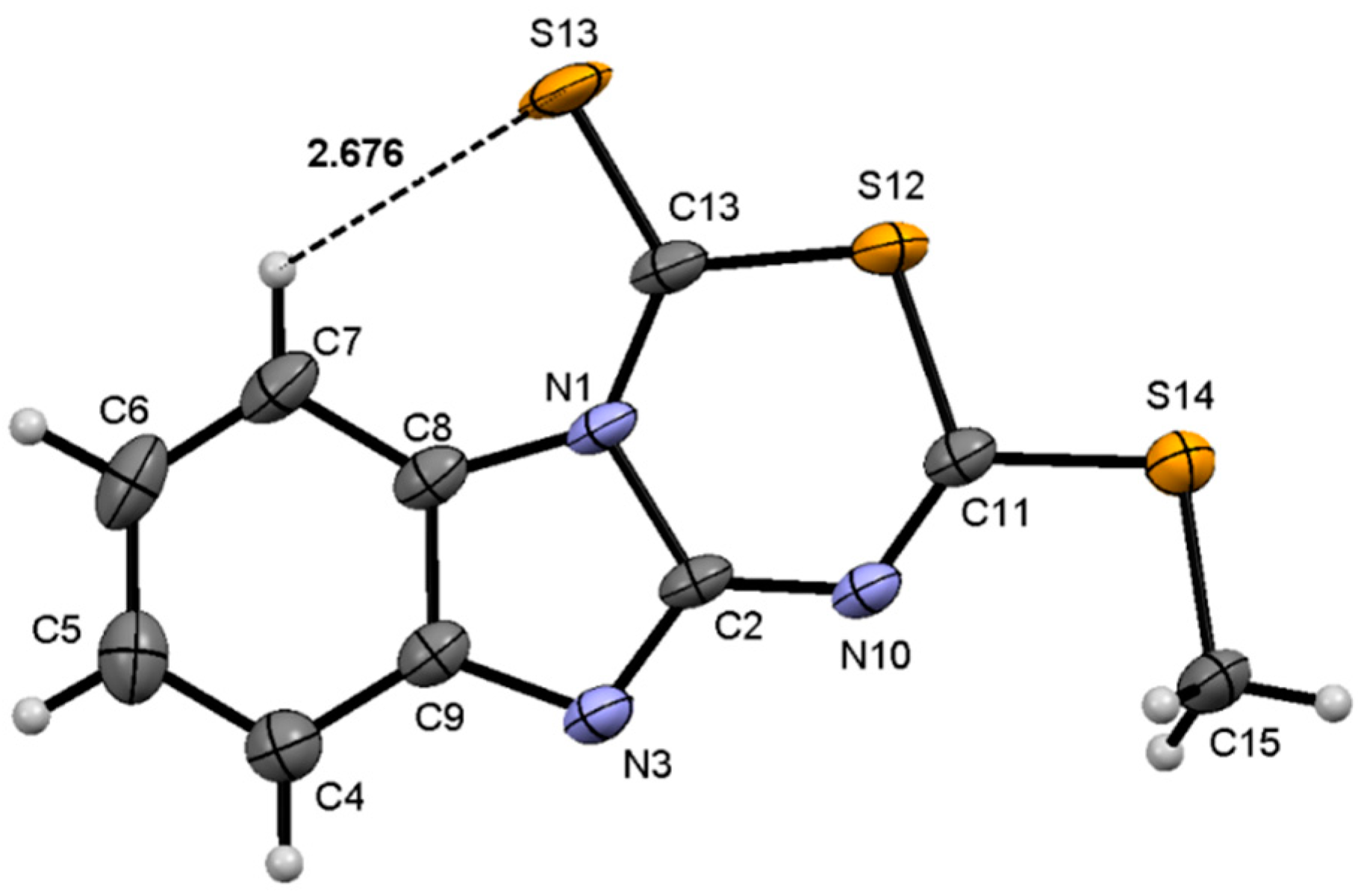
Analysis of the X-ray diffraction structure of compound
7 (
Figure 4) shows the new fusioned heterocycle in the same plane of the benzimidazole ring. The values of the torsion angles N(10)–C(11)–S(12)–C(13) of 2.0(2)° and N(10)–C(2)–N(1)–C(13) of 2.4(4)°, are representative of this condition. These molecular arrangement, explains the interaction of the phenyl hydrogen with the sulfur atom whose geometric parameters are: H(7)∙∙∙S(13) of 2.68 Å and C(7)–H(7)∙∙∙S(13) of 121°; this interaction is responsible for the high frequency shift of the phenyl H7 observed in the
1H NMR spectrum. The bond distances N(3)―C(2) of 1.301(3) Å and N(10)–C(2) of 1.355(3) Å are of intermediate value for single and double bond character, whereas the N(1)–C(2) and N(10)–C(11) distances of 1.433(3) Å and 1.286(4) Å, are characteristics for a single and a double bonds, respectively. The bond distances and angles are in agreement with the proposed structure.
Figure 4.
Molecular structure of compound 7. Selected bond lengths (Å) and angles (°): S(12)–C(11) 1.760(2), S(12)–C(13) 1.744(2), S(13)–C(13) 1.629(2), S(14)–C(11) 1.739(2), S(14)–C(15) 1.796(3), N(3)–C(2) 1.301(3), N(3)–C(9) 1.394(3), N(1)–C(2) 1.433(3), N(1)–C(13) 1.372(3), N(10)–C(2) 1.355(3), N(10)–C(11) 1.286(4), C(11)–S(12)–C(13) 103.77(12), C(11)–S(14)–C(15) 101.18(13), C(2)–N(1)–C(13) 126.11(19), C(2)–N(10)–C(11) 121.6(2), N(1)–C(2)–N(3) 112.31(18), S(12)–C(11)–N(10) 126.27(16), N(10)–C(2)–N(1)–C(13) 2.4(4), N(1)–C(2)–N(10)–C(11) −4.1(3), C(7)–C(8)–N(1)–C(13) −3.0(4), N(10)–C(11)–S(12)–C(13) 2.0(2), N(10)–C(11)–S(14)–C(15)–6.1(2).
Figure 4.
Molecular structure of compound 7. Selected bond lengths (Å) and angles (°): S(12)–C(11) 1.760(2), S(12)–C(13) 1.744(2), S(13)–C(13) 1.629(2), S(14)–C(11) 1.739(2), S(14)–C(15) 1.796(3), N(3)–C(2) 1.301(3), N(3)–C(9) 1.394(3), N(1)–C(2) 1.433(3), N(1)–C(13) 1.372(3), N(10)–C(2) 1.355(3), N(10)–C(11) 1.286(4), C(11)–S(12)–C(13) 103.77(12), C(11)–S(14)–C(15) 101.18(13), C(2)–N(1)–C(13) 126.11(19), C(2)–N(10)–C(11) 121.6(2), N(1)–C(2)–N(3) 112.31(18), S(12)–C(11)–N(10) 126.27(16), N(10)–C(2)–N(1)–C(13) 2.4(4), N(1)–C(2)–N(10)–C(11) −4.1(3), C(7)–C(8)–N(1)–C(13) −3.0(4), N(10)–C(11)–S(12)–C(13) 2.0(2), N(10)–C(11)–S(14)–C(15)–6.1(2).
Figure 5.
Molecular structure of compound 13. Selected bond lengths (Å) and angles (°): S(12)–C(11) 1.745(2), S(12)–C(13) 1.786(3), S(14)–C(11) 1.748(2), S(14)–C(15) 1.795(3), N(1)–C(2) 1.338(3), N(1)–C(8) 1.383(3), N(3)–C(2) 1.335(3), N(3)–C(9) 1.388(3), N(10)–C(2) 1.383(3), N(10)–C(11) 1.273(3), C(11)–S(12)–C(13) 101.10(14), C(11)–S(14)–C(15) 104.73(16), C(2)–N(10)–C(11) 121.95(18), N(1)–C(2)–N(3) 113.69(17), N(1)–C(2)–N(10) 125.16(19), N(3)–C(2)–N(10) 120.78(19), S(12)–C(11)–S(14) 117.10(15), S(12)–C(11)–N(10) 120.23(17), S(14)–C(11)–N(10) 122.67(17), N(1)–C(2)–N(10)–C(11) −71.3(3), N(3)–C(2)–N(10)–C(11) 116.2(2), N(10)–C(11)–S(12)–C(13) −1.0(2), S(12)–C(11)–S(14)–C(15) 7.4(2), N(10)–C(11)–S(14)–C(15) −173.0(2), S(12)–C(11)–N(10)–C(2) 179.93(18), S(14)–C(11)–N(10)–C(2) 0.4(3).
Figure 5.
Molecular structure of compound 13. Selected bond lengths (Å) and angles (°): S(12)–C(11) 1.745(2), S(12)–C(13) 1.786(3), S(14)–C(11) 1.748(2), S(14)–C(15) 1.795(3), N(1)–C(2) 1.338(3), N(1)–C(8) 1.383(3), N(3)–C(2) 1.335(3), N(3)–C(9) 1.388(3), N(10)–C(2) 1.383(3), N(10)–C(11) 1.273(3), C(11)–S(12)–C(13) 101.10(14), C(11)–S(14)–C(15) 104.73(16), C(2)–N(10)–C(11) 121.95(18), N(1)–C(2)–N(3) 113.69(17), N(1)–C(2)–N(10) 125.16(19), N(3)–C(2)–N(10) 120.78(19), S(12)–C(11)–S(14) 117.10(15), S(12)–C(11)–N(10) 120.23(17), S(14)–C(11)–N(10) 122.67(17), N(1)–C(2)–N(10)–C(11) −71.3(3), N(3)–C(2)–N(10)–C(11) 116.2(2), N(10)–C(11)–S(12)–C(13) −1.0(2), S(12)–C(11)–S(14)–C(15) 7.4(2), N(10)–C(11)–S(14)–C(15) −173.0(2), S(12)–C(11)–N(10)–C(2) 179.93(18), S(14)–C(11)–N(10)–C(2) 0.4(3).
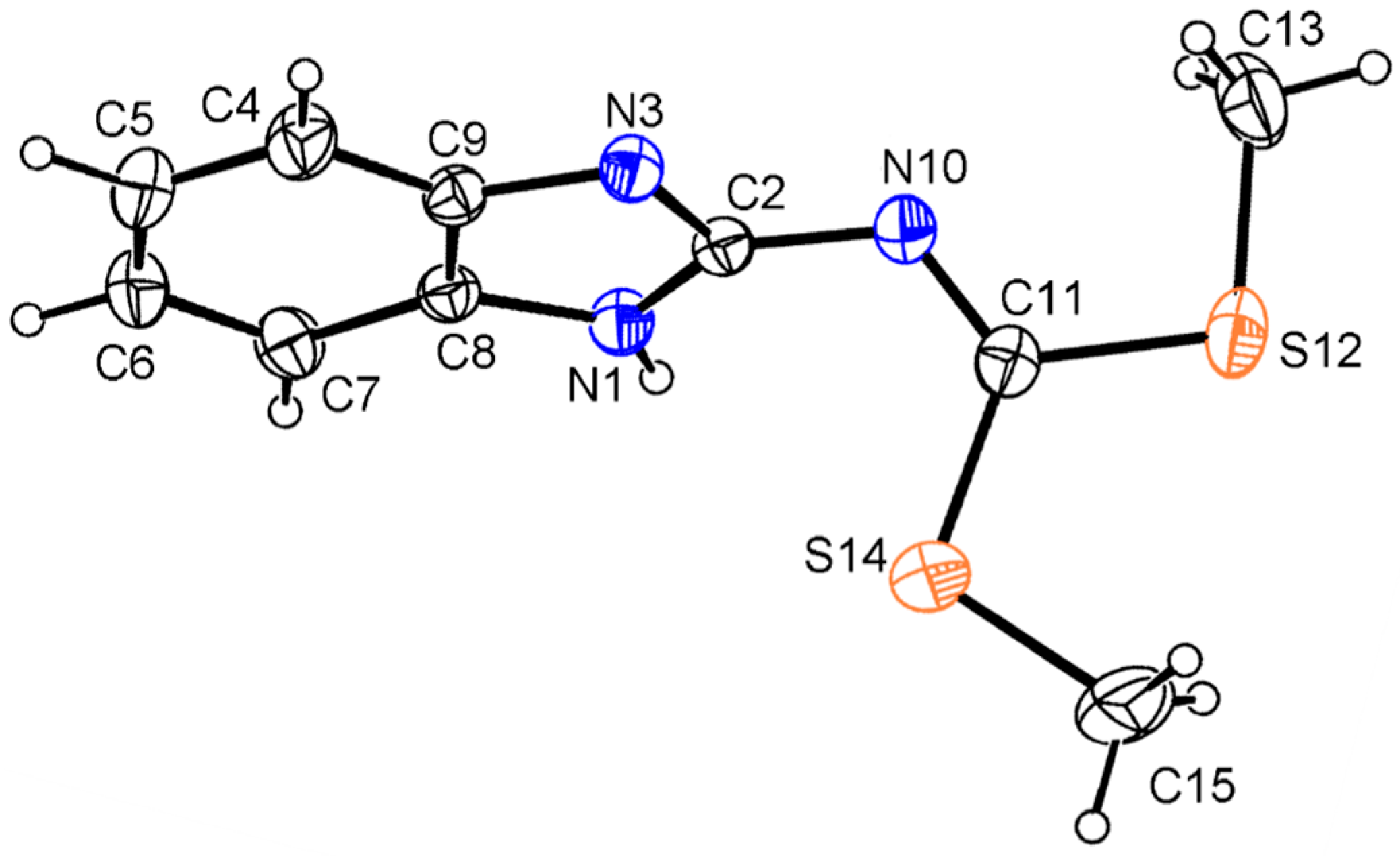
The molecular structure of compound
13 is depicted in
Figure 5. Benzimidazole NH prefers to form intermolecular N–H∙∙∙N, instead of intramolecular hydrogen bonding interactions to give a polymeric supramolecular structure. Thus the N=C(SMe)
2 moiety is free for rotation, being located out of the mean benzimidazole ring plane as shown by the values of the torsion angles of −71.3(3)° for N(1)–C(2)–N(10)–C(11) and 116.2(2)° for N(3)–C(2)–N(10)–C(11). This geometric feature contrasts with the planar structure observed for the analogous derivatives of 2-aminobenzothiazole and 2-amino-1-methyl benzimidazole [
16]. The N(1)–(2), N(3)–(2) and N(10)–(2) distance values of 1.338(3), 1.335(3) and 1.383(3) Å, are intermediate between single and a double bond character, compared with N(10)–C(11) of 1.273(3) Å, which has a double bond character.
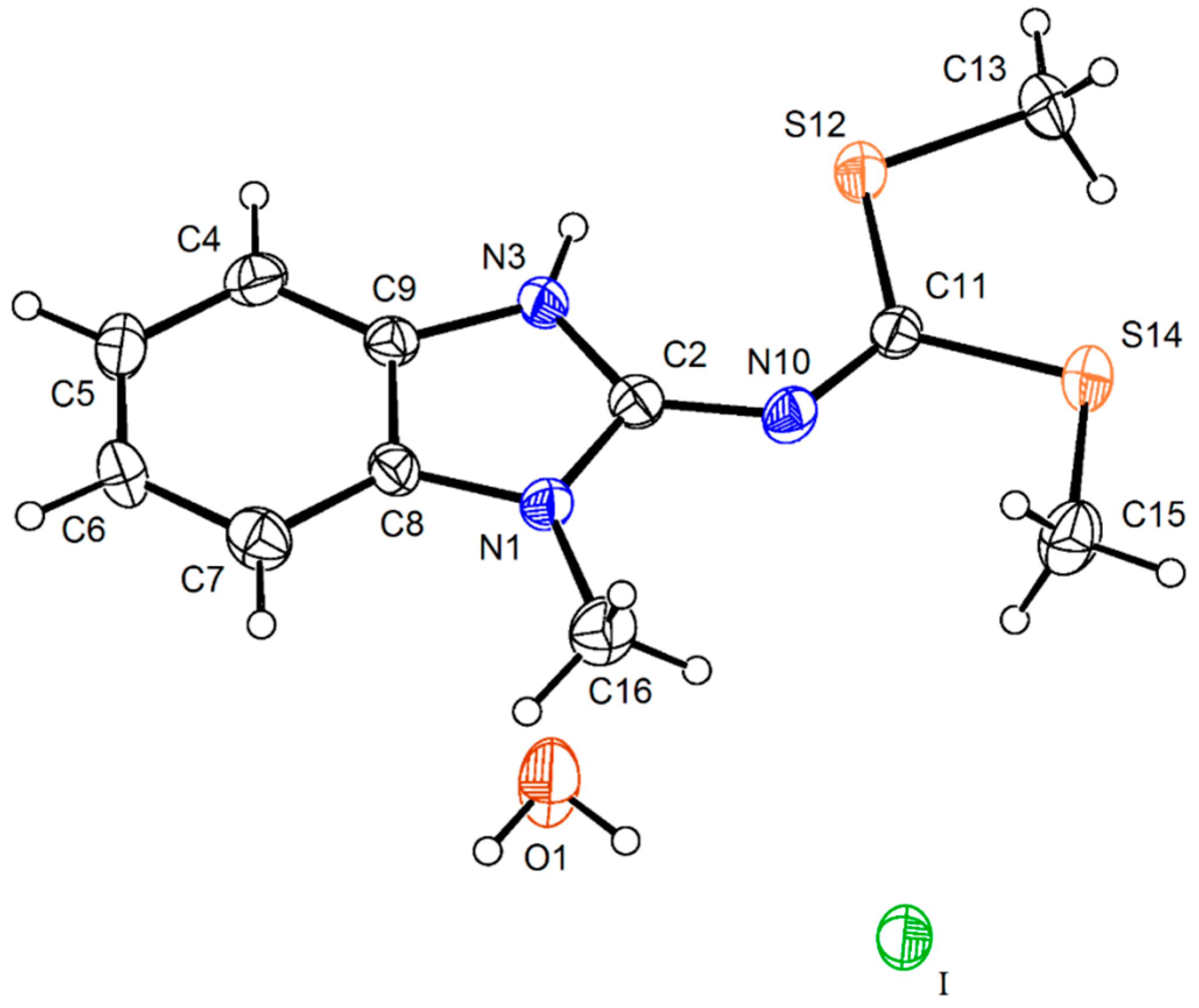
Compound
14 crystalized with one molecule of water (
Figure 6). The intramolecular N(3)–H(3)∙∙∙S(12) hydrogen bonding interaction gives shape to a six membered ring with a N(3)∙∙∙S(12) distance of 3.081(6) Å and N(3)–H(3)∙∙∙S(12) angle of 120° which forces the planarity of the molecule. The angles N(1)–C(2)–N(10)–C(11) of 179.8(7)° and N(3)–C(2)–N(10)–C(11) of −0.8(14)° confirm the planarity of the N=C(SMe)
2 an the value of N(10)–C(2) bond length of 1.342(10) Å, the strengthening of this bond. There is an intermolecular O1∙∙∙S12 interaction of 3.302(9) Å, instead of the S∙∙∙S interaction observed in the crystal structure of the neutral compound
15 [
15].
Figure 6.
Molecular structure of the iodide salt 14 at 30% of probability. Selected bond lengths (Å) and angles (°): S(12)–C(11) 1.743(9), S(12)–C(13) 1.791(8), S(14)–C(11) 1.731(8), S(14)–C(15) 1.794(10), N(1)–C(2) 1.356(10), N(1)–C(8) 1.378(9), N(1)–C(16) 1.458(12), N(3)–C(2) 1.345(10), N(3)–C(9) 1.382(9), N(10)–C(2) 1.342(10), N(10)–C(11) 1.289(10), C(4)–C(5) 1.391(11), C(2)–N(1)–C(16) 125.3(6), C(2)–N(10)–C(11) 129.2(7), N(1)–C(2)–N(3) 107.9(6), N(1)–C(2)–N(10) 118.4(7), N(3)–C(2)–N(10) 133.8(7), S(12)–C(11)–S(14) 116.9(4), S(12)–C(11)–N(10) 123.7(6), S(14)–C(11)–N(10) 119.5(6), N(1)–C(2)–N(10)–C(11) 179.8(7), N(3)–C(2)–N(10)–C(11) −0.8(14), N(10)–C(11)–S(12)–C(13) −177.8(7), S(12)–C(11)–S(14)–C(15) 176.5(5), N(10)–C(11)–S(14)–C(15) −3.5(7), S(12)–C(11)–N(10)–C(2) 1.5(11), S(14)–C(11)–N(10)–(2) −178.5(6).
Figure 6.
Molecular structure of the iodide salt 14 at 30% of probability. Selected bond lengths (Å) and angles (°): S(12)–C(11) 1.743(9), S(12)–C(13) 1.791(8), S(14)–C(11) 1.731(8), S(14)–C(15) 1.794(10), N(1)–C(2) 1.356(10), N(1)–C(8) 1.378(9), N(1)–C(16) 1.458(12), N(3)–C(2) 1.345(10), N(3)–C(9) 1.382(9), N(10)–C(2) 1.342(10), N(10)–C(11) 1.289(10), C(4)–C(5) 1.391(11), C(2)–N(1)–C(16) 125.3(6), C(2)–N(10)–C(11) 129.2(7), N(1)–C(2)–N(3) 107.9(6), N(1)–C(2)–N(10) 118.4(7), N(3)–C(2)–N(10) 133.8(7), S(12)–C(11)–S(14) 116.9(4), S(12)–C(11)–N(10) 123.7(6), S(14)–C(11)–N(10) 119.5(6), N(1)–C(2)–N(10)–C(11) 179.8(7), N(3)–C(2)–N(10)–C(11) −0.8(14), N(10)–C(11)–S(12)–C(13) −177.8(7), S(12)–C(11)–S(14)–C(15) 176.5(5), N(10)–C(11)–S(14)–C(15) −3.5(7), S(12)–C(11)–N(10)–C(2) 1.5(11), S(14)–C(11)–N(10)–(2) −178.5(6).