Vasorelaxation Induced by a New Naphthoquinone-Oxime is Mediated by NO-sGC-cGMP Pathway
Abstract
:1. Introduction
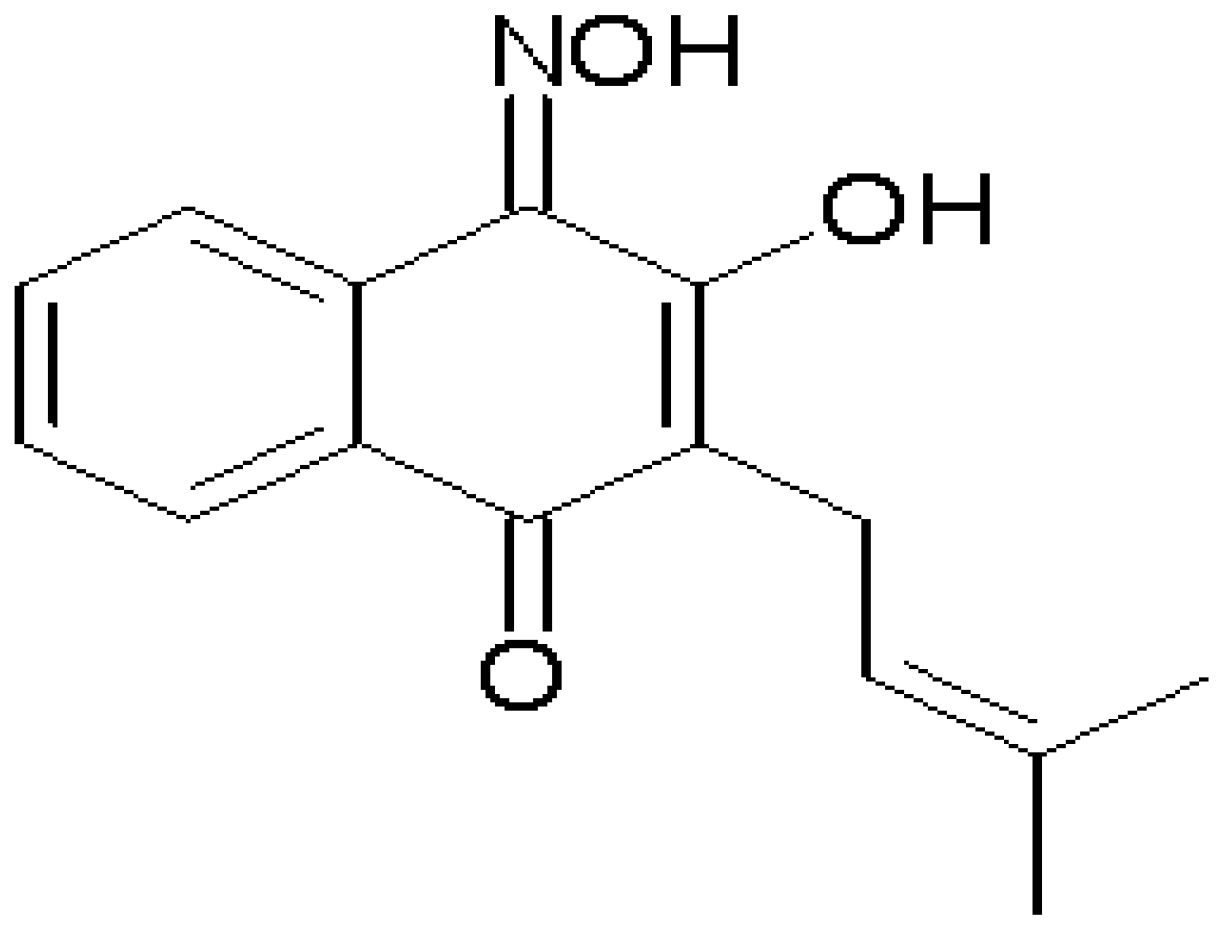
2. Results and Discussion
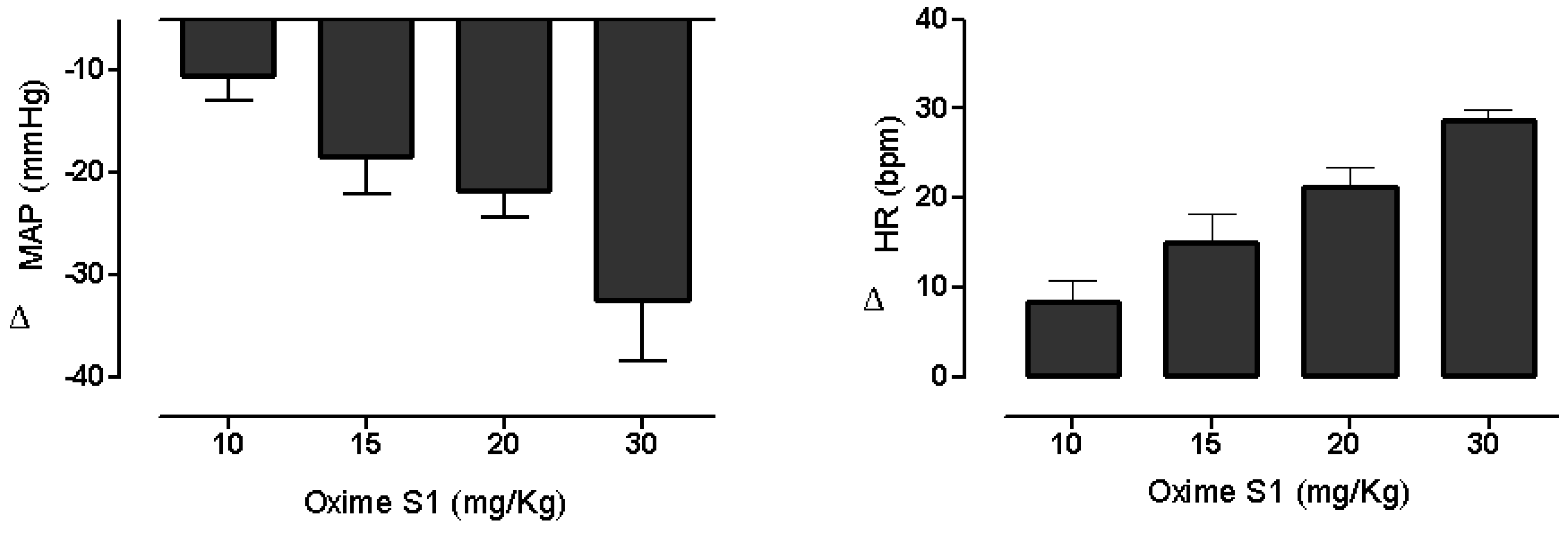
2.1. Oxime S1 Reduces Blood Pressure in Conscious Rats
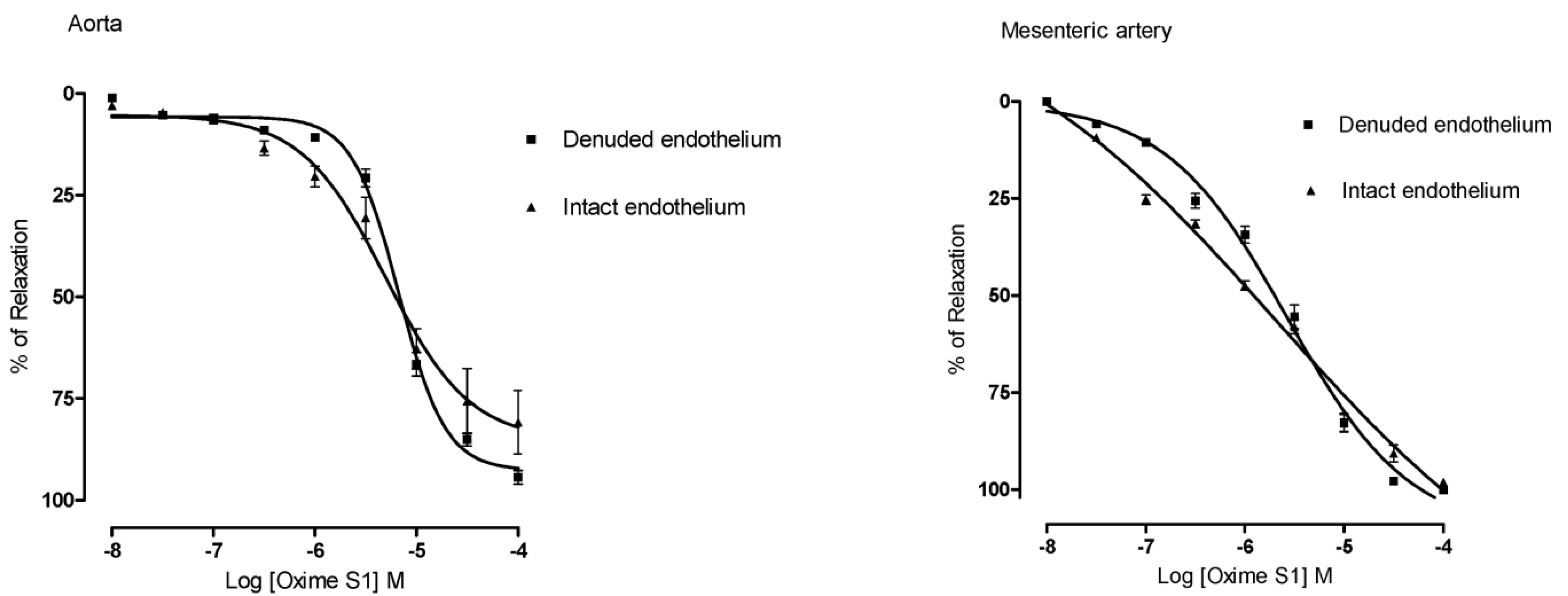
2.2. Oxime S1 Produces Endothelium-Independent Relaxations in Both Aorta and Superior Mesenteric Artery Rings
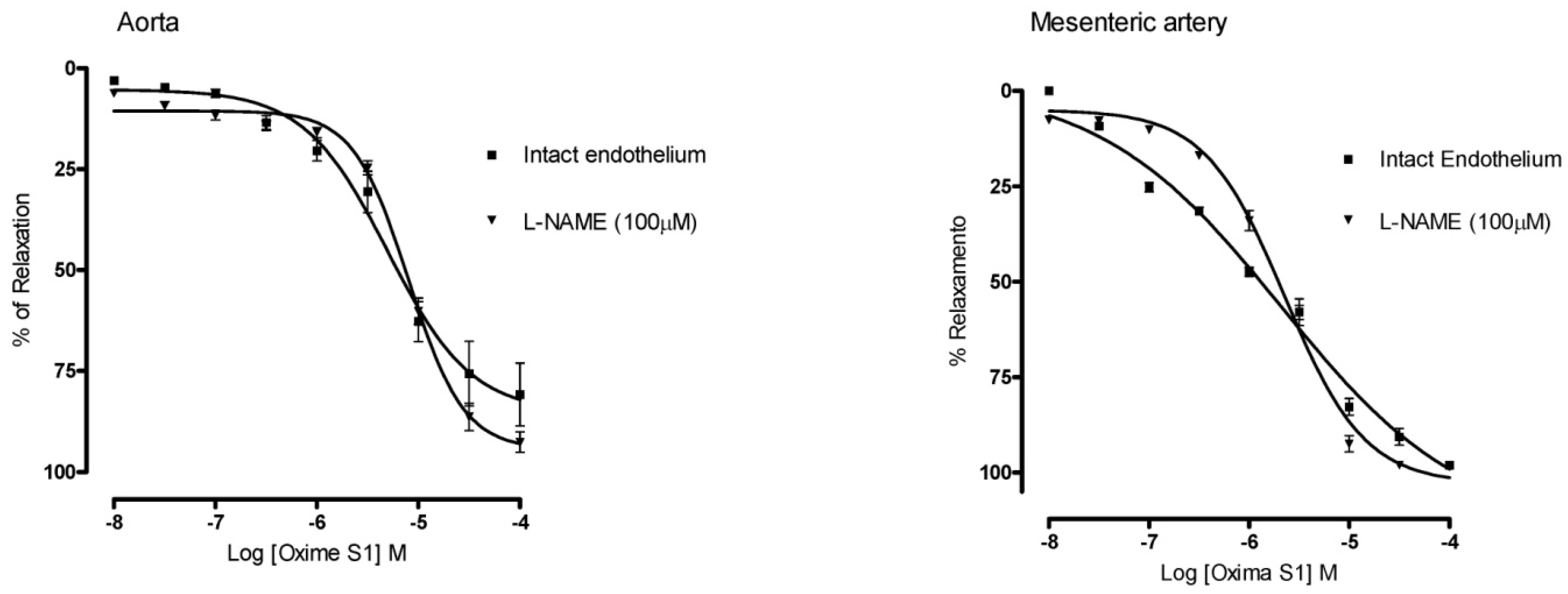
2.3. Oxime S1 Produces Vasorelaxation via Activation of the NO-sGC-cGMP Pathway
 ) in aorta; PTIO (▲) or ODQ (●) in mesenteric artery. Each data point represents the mean and the s.e.m. from 7 different experiments.
) in aorta; PTIO (▲) or ODQ (●) in mesenteric artery. Each data point represents the mean and the s.e.m. from 7 different experiments.
 ) in aorta; PTIO (▲) or ODQ (●) in mesenteric artery. Each data point represents the mean and the s.e.m. from 7 different experiments.
) in aorta; PTIO (▲) or ODQ (●) in mesenteric artery. Each data point represents the mean and the s.e.m. from 7 different experiments.
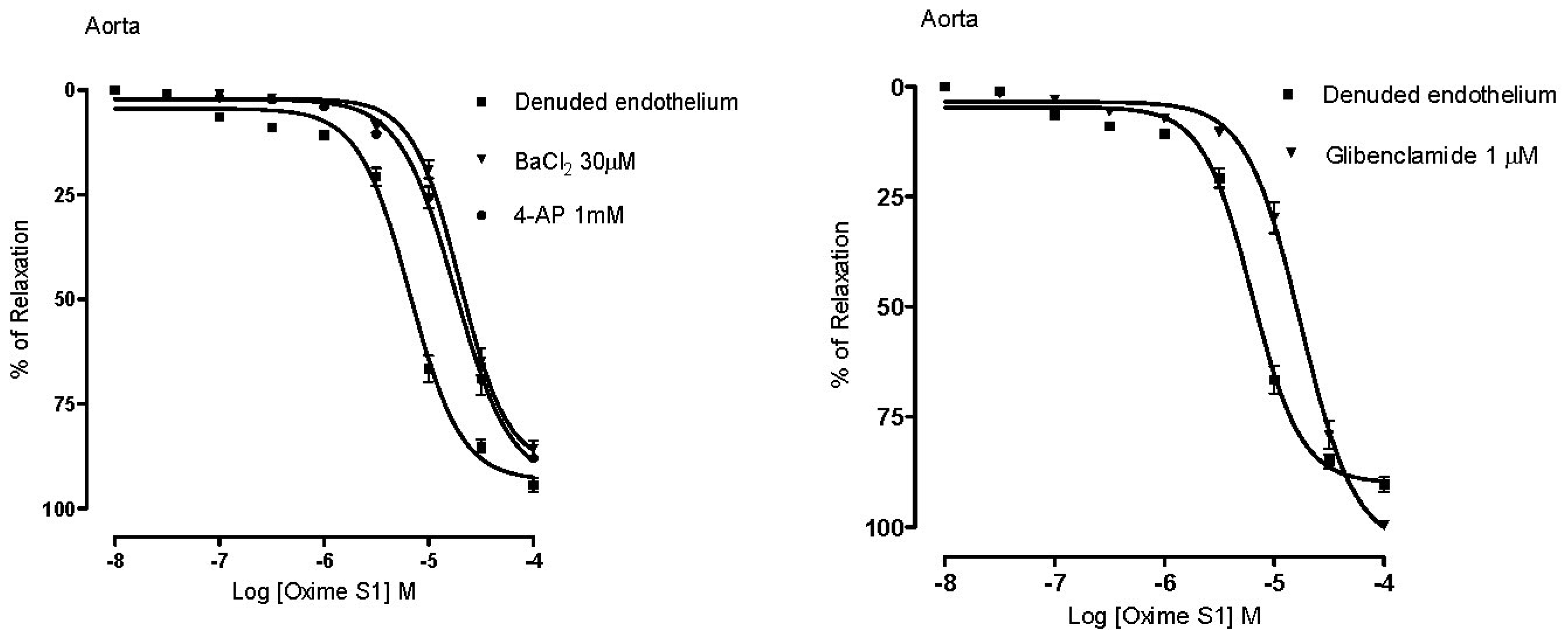
2.4. Vasorelaxation Induced by Oxime S1 is also Mediated by Activation of K+ Channels


2.5. Oxime S1 Releases Nitric Oxide When in Solution Acting as a Nitric Oxide Donor

3. Experimental
3.1. Animals
3.2. Drugs and Solutions
3.3. Cardiovascular Parameters in Vivo
3.4. Studies on Aortic and Mesenteric Artery Rings Isolated from Rats
3.5. Determination of Nitric Oxide Release Using NO Microsensors
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Palmer, R.M.J.; Ferrige, A.G.; Moncada, S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 1987, 327, 524–526. [Google Scholar] [CrossRef]
- Moncada, S.; Higgs, E.A. The Discovery of nitric oxide and its role in vascular biology. Br. J. Pharmacol. 2006, 147, S193–S201. [Google Scholar] [CrossRef]
- Moncada, S.; Higgs, E.A. The l-arginine-nitric oxide pathway. N. Engl. J. Med. 1993, 29, 2002–2012. [Google Scholar]
- Hishikawa, K.; Nakaki, T.; Suzuki, H.; Kato, R.; Saruta, T. l-Arginine as an antihypertensive agent. Cardiovasc. Pharmacol. 1992, 20, S196–S197. [Google Scholar] [CrossRef]
- Moncada, S.; Higgs, E.A. Molecular mechanisms and therapeutic strategies related to nitric oxide. FASEB J. 1995, 9, 1319–1330. [Google Scholar]
- Rajapakse, N.; Mattson, D.L. Role of l-arginine in nitric oxide production in health and hypertension. Clin. Exp. Pharmacol. Physiol. 2009, 36, 249–255. [Google Scholar] [CrossRef]
- Boucher, J.L.; Genet, A.; Vadon, S.; Delaforge, M.; Mansuy, D. Formation of nitrogen oxides and citrulline upon oxidation of N-omegahydroxy-l-arginine by hemeproteins. Biochem. Biophys. Res. Commun. 1992, 184, 1158–1164. [Google Scholar]
- Jousserandot, A.; Boucher, J.L.; Henry, Y.; Niklaus, B.; Clement, B.; Mansuy, D. Microsomal cytochrome P450 dependent oxidation of N-hydroxyguanidines, amidoximes, and ketoximes: Mechanism of the oxidative cleavage of their C=N(OH) bond with formation of nitrogen oxides. Biochemistry 1998, 37, 17179–17191. [Google Scholar]
- Caro, A.A.; Cederbaum, A.I.; Stoyanovsky, D.A. Oxidation of the ketoximeacetoxime to nitric oxide by oxygen radical-generating systems. Nitric Oxide 2001, 5, 413–424. [Google Scholar]
- Vetrovsky, P.; Boucher, J.L.; Schott, C.; Beranova, P.; Chalupsky, K.; Callizot, N.; Muller, B.; Entlicher, G.; Mansuy, D.; Stoclet, J.C. Involvement of NO in the endothelium-independent relaxing effects of N(omega)-hydroxy-l-arginine and other compounds bearing a C=NOH function in the rat aorta. J. Pharmacol. Exp. Ther. 2002, 303, 823–830. [Google Scholar] [CrossRef]
- Moali, C.; Brolo, M.; Custot, J.; Sari, M.A.; Boucher, J.L.; Stuehr, D.J.; Mansuy, D. Recognition of a-amino acids bearing various C=NOH functions by nitric oxide synthase and arginase involves very different structural determinants. Biochemistry 2000, 39, 8208–8218. [Google Scholar]
- Beranova, P.; Chalupsky, K.; Kleschyov, A.L.; Schott, C.; Boucher, J.L.; Mansuy, D.; Munzel, T.; Muller, B.; Stoclet, J.C. NN-hydroxy-l-arginine homologues and hydroxylamine as nitric oxide-dependent vasorelaxant agents. Eur. J. Pharmacol. 2005, 516, 260–267. [Google Scholar] [CrossRef]
- Braga, V.A.; Medeiros, I.A.; Ribeiro, T.P.; França-Silva, M.S.; Botelho-Ono, M.S.; Guimarães, D.D. Angiotensin-II-induced reactive oxygen species along the SFO-PVN-RVLM pathway: Implications in neurogenic hypertension. Braz. J. Med. Biol. Res. 2011, 44, 871–876. [Google Scholar] [CrossRef]
- Veras, R.C.; Rodrigues, K.G.; Alustau, M.C.; Araújo, I.G.; de Barros, A.L.; Alves, R.J.; Nakao, L.S.; Braga, V.A.; Silva, D.F.; de Medeiros, I.A. Participation of nitric oxide pathway in the relaxation response induced by E-cinnamaldehyde oxime in superior mesenteric artery isolated from rats. J. Cardiovasc. Pharmacol. 2013, 62, 58–66. [Google Scholar] [CrossRef]
- Schott, C.A.; Bogen, C.M.; Vetrovsky, P.; Berton, C.C.; Stoclet, J.C. Exogenous NG-hydroxy-l-arginine causes nitrite production in vascular smooth muscle cells in the absence of nitric oxide synthase activity. FEBS Lett. 341, 203–207.
- Chalupsky, K.; Gadea, I.; Entlicher, G.; Stoclet, J.C.; Muller, B. Relaxant effect of oxime derivatives in isolated rat aorta: Role of nitric oxide (NO) formation in smooth muscle. Biochem. Pharmacol. 2004, 67, 1203–1214. [Google Scholar] [CrossRef]
- Friebe, A.; Koesling, D. Regulation of nitric oxide-sensitive guanylyl cyclase. Circ. Res. 2003, 93, 96–105. [Google Scholar]
- Moncada, S.; Radomski, M.W.; Palmer, R.M. Endothelium-derivated relaxing factor identification as nitric oxide and role in the control of vascular tone and platelet function. Biochem. Pharmacol. 1988, 37, 2495–2500. [Google Scholar] [CrossRef]
- Mistry, D.I.; Garland, C.J. Nitric oxide (NO)-induced activation of large conductance Ca2+-dependent K+ channels (BKCa) in smooth muscle cells isolated from the rat mesenteric artery. Br. J. Pharmacol. 1998, 124, 1131–1140. [Google Scholar] [CrossRef]
- Birschmann, I.; Walter, B. Physiology and pathophysiology of vascular signaling controlled by guanosine 3',5'-cyclic monophosphate-dependent protein kinase. Acta Biochim. Pol. 2004, 51, 397–404. [Google Scholar]
- Boerth, N.J.; Dey, N.B.; Cornwell, T.L.; Lincoln, T.M. Cyclic GMP-dependent protein kinase regulates vascular smooth muscle cell phenotype. J. Vasc. Res. 1997, 34, 245–259. [Google Scholar]
- Sand, A.; Andersson, E.; Fried, G. Nitric oxide donors mediate vasodilation in human placental arteries partly through a direct effect on potassium channels. Placenta 2006, 27, 181–190. [Google Scholar] [CrossRef]
- Moncada, S.; Higgs, E.A. Nitric oxide and the vascular endothelium. Handb. Exp. Pharmacol. 2006, 176, 213–254. [Google Scholar]
- França-Silva, M.S.; Luciano, M.N.; Ribeiro, T.P.; Silva, J.S.; Santos, A.F.; França, K.C.; Nakao, L.S.; Athayde-Filho, P.F.; Braga, V.A.; Medeiros, I.A. The 2-nitrate-1,3-dibuthoxypropan, a new nitric oxide donor, induces vasorelaxation in mesenteric arteries of the rat. Eur. J. Pharmacol. 2012, 690, 170–175. [Google Scholar] [CrossRef]
- França-Silva, M.S.; Monteiro, M.M.; Queiroz, T.M.; Santos, A.F.; Athayde-Filho, P.F.; Braga, V.A. The new nitric oxide donor 2-nitrate-1,3-dibuthoxypropan alters autonomic function in spontaneously hypertensive rats. Auton. Neurosci. 2012, 171, 28–35. [Google Scholar] [CrossRef]
- Silva, T.M.S.; Camara, C.A.; Barbosa, T.P.; Soares, A.Z.; da Cunha, L.C.; Pinto, A.C.; Vargas, M.D. Molluscicidal activity of synthetic lapachol amino and hydrogenated derivatives. Bioorg. Med. Chem. 2005, 13, 193–196. [Google Scholar]
- Braga, V.A. Dietary salt enhances angiotensin-II-induced superoxide formation in the rostral ventrolateral medulla. Auton. Neurosci. 2010, 155, 14–18. [Google Scholar] [CrossRef]
- Ribeiro, T.P.; Porto, D.L.; Menezes, C.P.; Antunes, A.A.; Silva, D.F.; de Sousa, D.P.; Nakao, L.S.; Braga, V.A.; Medeiros, I.A. Unraveling the cardiovascular effects induced by alpha-terpineol: A role for the NO-cGMP pathway. Clin. Exp. Pharmacol. Physiol. 2010, 37, 811–816. [Google Scholar]
- Furchgott, F.F.; Zawadzki, J.V. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature 1980, 288, 373–376. [Google Scholar] [CrossRef]
- Botelho-Ono, M.S.; Pina, H.V.; Sousa, K.H.; Nunes, F.C.; Medeiros, I.A.; Braga, V.A. Acute superoxide scavenging restores depressed baroreflex sensitivity in renovascular hypertensive rats. Auton. Neurosci. 2011, 159, 38–44. [Google Scholar] [CrossRef]
- Burmeister, M.A.; Young, C.N.; Braga, V.A.; Butler, S.D.; Sharma, R.V.; Davisson, R.L. In vivo bioluminescence imaging reveals redox-regulated activator protein-1 activation in paraventricular nucleus of mice with renovascular hypertension. Hypertension 2011, 57, 289–297. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dantas, B.P.V.; Ribeiro, T.P.; Assis, V.L.; Furtado, F.F.; Assis, K.S.; Alves, J.S.; Silva, T.M.S.; Camara, C.A.; França-Silva, M.S.; Veras, R.C.; et al. Vasorelaxation Induced by a New Naphthoquinone-Oxime is Mediated by NO-sGC-cGMP Pathway. Molecules 2014, 19, 9773-9785. https://doi.org/10.3390/molecules19079773
Dantas BPV, Ribeiro TP, Assis VL, Furtado FF, Assis KS, Alves JS, Silva TMS, Camara CA, França-Silva MS, Veras RC, et al. Vasorelaxation Induced by a New Naphthoquinone-Oxime is Mediated by NO-sGC-cGMP Pathway. Molecules. 2014; 19(7):9773-9785. https://doi.org/10.3390/molecules19079773
Chicago/Turabian StyleDantas, Bruna P. V., Thaís P. Ribeiro, Valéria L. Assis, Fabíola F. Furtado, Kívia S. Assis, Jeziane S. Alves, Tania M.S. Silva, Celso A. Camara, Maria S. França-Silva, Robson C. Veras, and et al. 2014. "Vasorelaxation Induced by a New Naphthoquinone-Oxime is Mediated by NO-sGC-cGMP Pathway" Molecules 19, no. 7: 9773-9785. https://doi.org/10.3390/molecules19079773



