Immobilization of Trichoderma harzianum α-Amylase on Treated Wool: Optimization and Characterization
Abstract
:1. Introduction
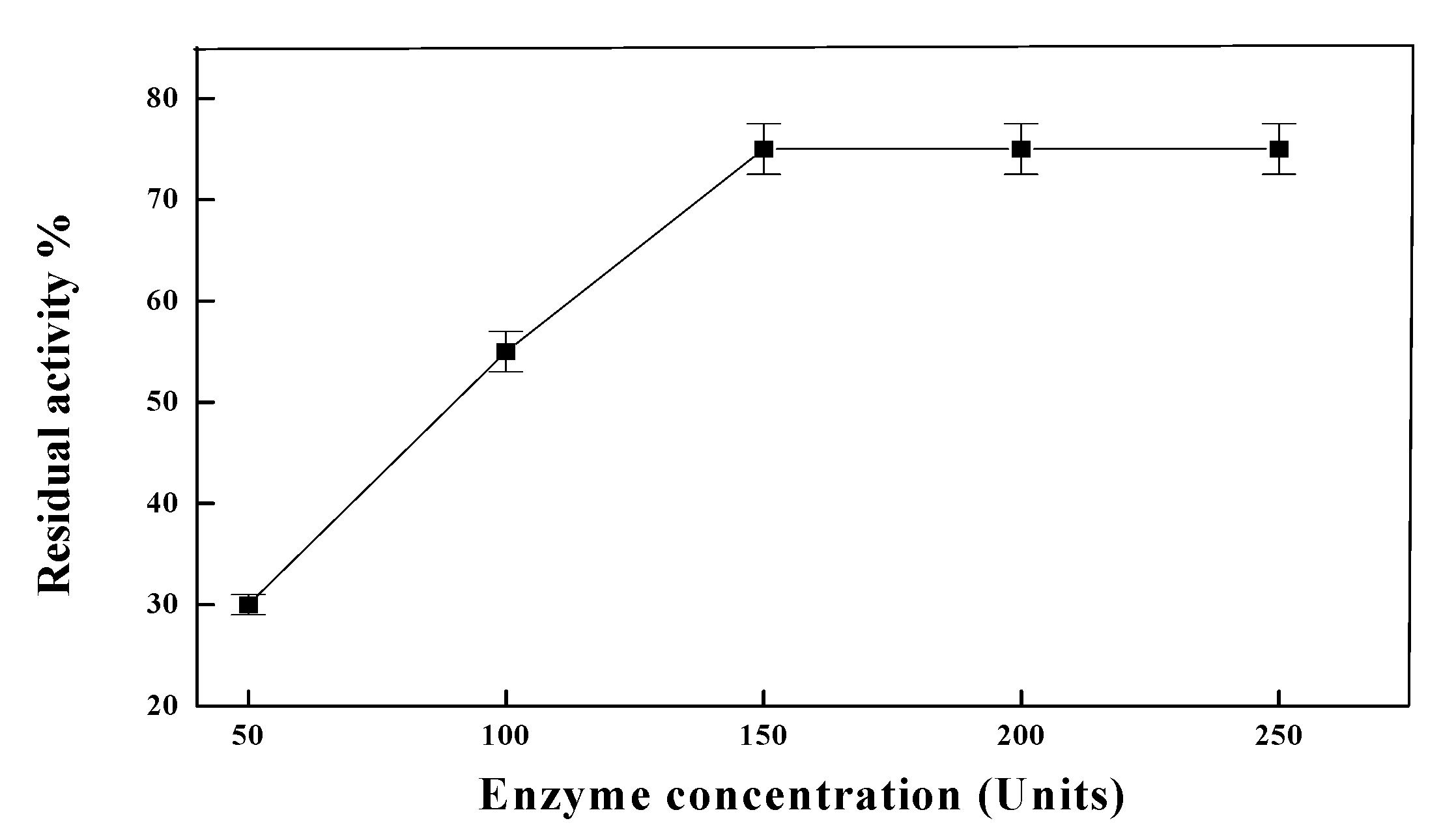
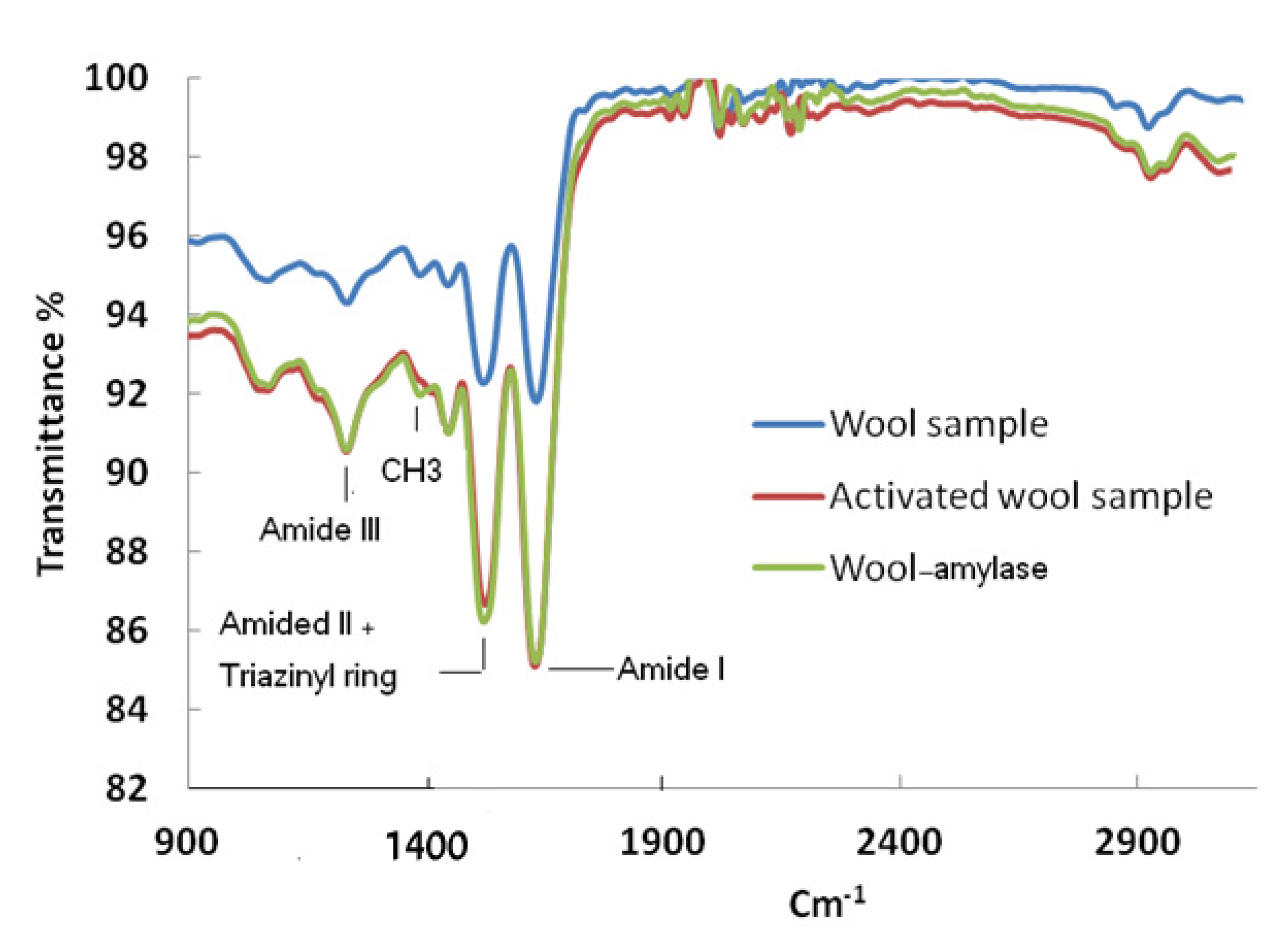
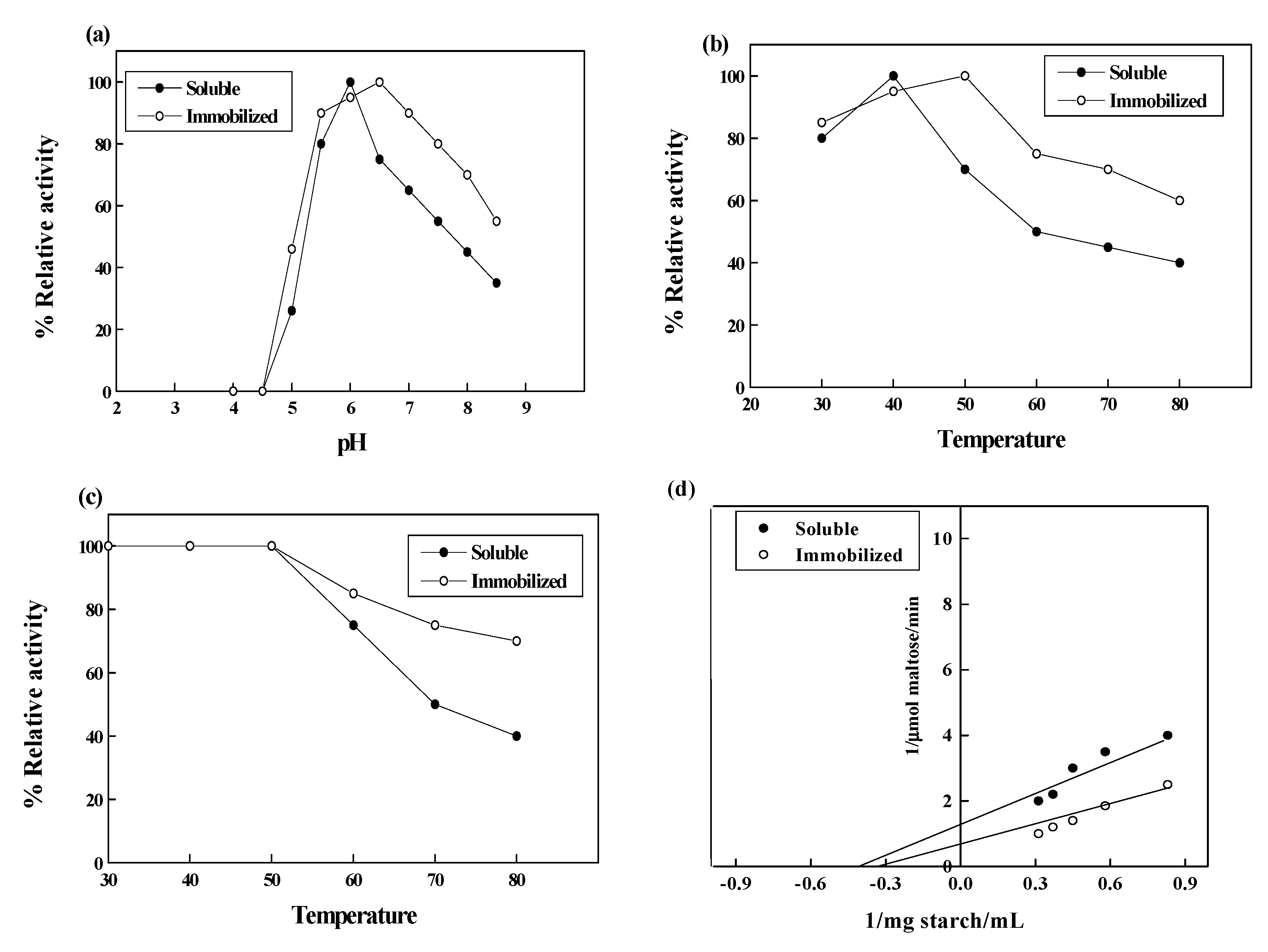
2. Results and Discussion
| Cyanuric Chloride % | Immobilization Efficiency % | |
|---|---|---|
| pH 5.5 | pH 7.2 | |
| 2 | 15 ± 0.7 | 25 ± 1.3 |
| 4 | 50 ± 2.8 | 70 ± 3.8 |
| 6 | 30 ± 1.7 | 45 ± 2.1 |
| 8 | 12 ± 0.7 | 22 ± 1.1 |


| Substrate | Relative Activity % | |
|---|---|---|
| Soluble α-Amylase | Immobilized α-Amylase | |
| Starch | 95 ± 2.5 | 100 ± 3.1 |
| Glycogen | 80 ± 1.8 | 90 ± 2.6 |
| Amylopectin | 68 ± 2.1 | 80 ± 2.0 |
| Amylose | 70 ± 2.2 | 75 ± 1.5 |
| α-Cyclodextrin | 41 ± 1.6 | 53 ± 1.2 |
| β-Cyclodextrin | 42 ± 1.2 | 49 ± 1.6 |
| Metal Ion | Relative Activity % | |
|---|---|---|
| Soluble α-Amylase | Immobilized α-Amylase | |
| Control | 100 ± 2.5 | 100 ± 3.1 |
| Cu2+ | 70 ± 2.3 | 94 ± 2.8 |
| Ni2+ | 78 ± 2.4 | 113 ± 2.5 |
| Ca2+ | 109 ± 1.8 | 120 ± 2.3 |
| Zn2+ | 91 ± 2.3 | 102 ± 3.2 |
| Co2+ | 115 ± 1.7 | 125 ± 2.8 |
| Pb2+ | 70 ± 2.2 | 90 ± 3.0 |
| Hg2+ | 15 ± 0.6 | 60 ± 2.0 |
3. Experimental
3.1. T. harzianum α-Amylase
3.2. Preparation of Support
3.3. Immobilization Procedure

3.4. Characterization of Wool and Immobilized Wool-α-Amylase
3.5. α-Amylase Assay
3.6. Reusability of Immobilized Enzyme
3.7. Effect of pH and Temperature
3.8. Determination of Kinetic Constant
3.9. Substrate Specificity
3.10. Effect of Metal Ions
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pandya, P.H.; Jarsa, R.V.; Newalkar, B.; Bhalt, P.N. Studies on the activity and stability of immobilized α-amylase in ordered mesoporous silicas. Microporous. Mesoporous. Mater. 2005, 77, 67–77. [Google Scholar] [CrossRef]
- Gupta, R.; Gigars, P.; Mohapatra, H.; Goswami, V.K.; Chauhan, B. Microbial α-amylase: A biotechnological perspective. Process Biochem. 2003, 38, 1599–1616. [Google Scholar] [CrossRef]
- Walsh, G. Industrial enzymes: An introduction. In Biochemistry and Biotechnology; Walsh, G., Ed.; Wiley: NewYork, NY, USA, 2002; p. 454. [Google Scholar]
- Sidhu, G.S.; Sharma, P.; Chakrabart, T.; Gupta, J.K. Strain improvement for the production thermostable α-amylase. Enzym. Microb. Technol. 1997, 21, 525–530. [Google Scholar] [CrossRef]
- Sassolas, A.; Blum, L.J.; Leca-Bouvier, B.D. Immobilization strategies to develop enzymatic biosensors. Biotechnol. Adv. 2012, 30, 489–511. [Google Scholar] [CrossRef]
- Asgher, M.; Shahid, M.; Kamal, S.; Iqbal, H.M.N. Recent trends and valorization of immobilization strategies and ligninolytic enzymes by industrial biotechnology. J. Mol. Cataly. B-Enzym. 2014, 101, 56–66. [Google Scholar]
- Cao, L. Carrier-bound immobilized enzymes. In Principles, Application and Design; Wiley–VCH: Weinheim, Germany, 2006. [Google Scholar]
- Mateo, C.; Palomo, J.M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzym. Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Brena, B.; González-Pombo, P.; Batista-Viera, F. Immobilization of enzymes: A literature survey. Methods Mol. Biol. 2009, 1051, 15–31. [Google Scholar]
- Fernandez-Lafuente, R. Stabilization of multimeric enzymes: Strategies to prevent subunit dissociation. Enzym. Microb. Technol. 2009, 45, 405–418. [Google Scholar] [CrossRef]
- Hwang, E.T.; Gu, M.B. Enzyme stabilization by nano/microsized hybrid materials. Eng. Life Sci. 2013, 13, 49–61. [Google Scholar] [CrossRef]
- Cao, L. Immobilized enzymes: Science or art? Curr. Opin. Chem. Biol. 2005, 9, 217–226. [Google Scholar] [CrossRef]
- Arica, M.Y.; Hasirci, V.; Alaeddinoglu, N.G. Covalent immobilization of α-amylase onto pHEMA microspheres: Preparation and application to fixed bed reactor. Biomaterials 1995, 15, 761–768. [Google Scholar]
- Sun, Y.-M.; Chen, J.-P.; Chu, D.-H. Preparation and characterization of α-amylase-immobilized thermal-responsive composite hydroget membranes. J. Biomed. Mater. Res. 1999, 45, 125–132. [Google Scholar] [CrossRef]
- Tumturk, H.; Aksoy, S.; Hasirci, N. Covalent immobilization of α-amylase onto poty(2-hydroxyethyl methacrylate) and poly(styrene-2-hydroxyethyl methacrylate) microspheres and the effect of Ca2+ ions on the enzyme activity. Food Chem. 2000, 68, 259–266. [Google Scholar] [CrossRef]
- Bayramoglu, G.; Yilmaz, M.; Arica, M.Y. Immobilization of a thermostable α-amylase onto reactive membranes: Kinetics characterization and application to continuous starch hydrolysis. Food Chem. 2004, 84, 591–599. [Google Scholar] [CrossRef]
- Kahraman, M.V.; Bayramoglu, G.; Kayaman-Apohan, N.; Gungor, A. α-Amylase immobilization on functionalized glass beads by covalent attachment. Food Chem. 2007, 104, 1385–1392. [Google Scholar] [CrossRef]
- Turunc, O.; Kahraman, M.V.; Akdemir, Z.S.; Kayaman-Apohan, N.; Gungor, A. Immobilization of α-amylase onto cyclic carbonate bearing hybrid material. Food Chem. 2009, 112, 992–997. [Google Scholar]
- Noda, T.; Suda, S.F. Sweet potato α-amylase immobilized on chitosan beads and its application in semi-continuous production of maltose. Carbohydr. Polym. 2001, 44, 189–195. [Google Scholar] [CrossRef]
- Tanyolac, D.; Yuruksoy, B.I.; Ozdural, A.R. Immobilization of a thermostable α-amylase, Thermamyl, onto nitrocellulose membrane by Cibacron Blue F3GA dye binding. Biochem. Eng. J. 1998, 2, 179–186. [Google Scholar] [CrossRef]
- Kurimoto, A.; Tanabe, T.; Tachibana, A.; Yamauchi, K.J. Keratin sponge: Immobilization of lysozyme. J. Biosci. Bioeng. 2003, 96, 307–309. [Google Scholar]
- Monier, M.; El-Sokkary, A.M.A.; Sarhan, A.A. Immobilization of Candida rugosa lipase on modified natural wool fibers. React. Funct. Polym. 2010, 70, 122–128. [Google Scholar] [CrossRef]
- Quirke, M.E. 1,3,5-Triazines. In Comprehensive Heterocyclic Chemistry; Katritzky, A.R., Rees, C.W., Eds.; Pergamon Press: Oxford, UK, 1984; Volume 3, pp. 457–530. [Google Scholar]
- Smolin, E.M.; Rapoport, L. Cyanuric acid and derivatives. In The Chemistry of Heterocyclic Compounds; Weissberger, A., Ed.; Interscience Publishers INC.: New York, NY, USA, 1959; Volume 13, pp. 17–48. [Google Scholar]
- Mohamed, S.A.; Darwish, A.A.; El-Shishtawy, R.M. Immobilization of horseradish peroxidase on activated wool. Process Biochem. 2013, 48, 649–655. [Google Scholar] [CrossRef]
- Mohamed, S.A.; Azhar, E.I.; Ba-Akdah, M.M.; Tashkandy, N.R.; Kumosani, T.A. Production, purification and characterization of α-amylase from Trichoderma harzianum grown on mandarin peel. Afr. J. Microbiol. Res. 2011, 5, 930–940. [Google Scholar]
- Singh, V.; Singh, D. An efficient carrier matrix for amylase immobilization. Process Biochem. 2013, 48, 96–102. [Google Scholar] [CrossRef]
- Tuzmen, N.; Kalburcu, T.; Denizli, A. α-Amylase immobilization onto dye attached magnetic beads: Optimization and characterization. J. Mol. Cataly. B-Enzym. 2012, 78, 16–23. [Google Scholar] [CrossRef]
- Wang, F.; Gu, Z.; Cui, Z.; Liu, L. Comparison of covalent immobilization of amylase on polystyrene pellets with pentaethylenehexamine and pentaethylene glycol spacers. Bioresour. Technol. 2011, 102, 9374–9379. [Google Scholar]
- Wu, Z.; Qi, W.; Wang, M.; Wang, Y.; Su, Z.; He, R. Chelate immobilization of amylase on metal ceramic powder: Preparation, characterization and application. Biochem. Eng. J. 2013, 77, 190–197. [Google Scholar]
- Akkaya, B.; Yenidunya, A.F.; Acalla, R. Production and immobilization of a novel thermoalkalophilic extracellular amylase from bacilli isolate. Int. J. Biol. Macromol. 2012, 50, 991–995. [Google Scholar]
- Bayramoglu, G.; Denizli, A.; Arica, M.Y. Membrane with incorporated hydrophobic ligand for hydrophobic interaction with proteins: Application to lipase adsorption. Polym. Int. 2002, 51, 966–972. [Google Scholar]
- Martinek, K.; Kilbanov, A.M.; Goldmacher, V.S.; Berezin, I.V. The principles of enzyme stabilization. Biochim. Biophys. Acta 1977, 485, 1–12. [Google Scholar]
- Pascoal, A.M.; Mitidieri, S.; Fernández, K.F. Immobilization of α-amylase from Aspergillus niger onto polyaniline. Bioresour. Technol. 2011, 89, 300–306. [Google Scholar]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, A.; Torres, R.; Fernández-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef]
- Alexandratos, S.; Hong, M.J. Enhanced metal ion affinities by supported ligand synergistic interaction in bifunctional polymer-supported aminomethylphosphonates. Sep. Sci. Technol. 2002, 37, 2587–2605. [Google Scholar] [CrossRef]
- Fahmy, A.S.; Bagos, V.B.; Mohammed, T.M. Immobilization of Citrullus vulgaris urease on cyanuric chloride dear-cellulose ether: Preparation and properties. Biores. Technol. 1998, 64, 121–129. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for the determination of reducing sugar. Anal. Chem. 1959, 31, 426–429. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the soluble and immobilized α-amylases are available from the authors.
© 2014 by the authors. licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mohamed, S.A.; Khan, J.A.; Al-Bar, O.A.M.; El-Shishtawy, R.M. Immobilization of Trichoderma harzianum α-Amylase on Treated Wool: Optimization and Characterization. Molecules 2014, 19, 8027-8038. https://doi.org/10.3390/molecules19068027
Mohamed SA, Khan JA, Al-Bar OAM, El-Shishtawy RM. Immobilization of Trichoderma harzianum α-Amylase on Treated Wool: Optimization and Characterization. Molecules. 2014; 19(6):8027-8038. https://doi.org/10.3390/molecules19068027
Chicago/Turabian StyleMohamed, Saleh A., Jalaluddin A. Khan, Omar A. M. Al-Bar, and Reda M. El-Shishtawy. 2014. "Immobilization of Trichoderma harzianum α-Amylase on Treated Wool: Optimization and Characterization" Molecules 19, no. 6: 8027-8038. https://doi.org/10.3390/molecules19068027






