1. Introduction
Serotonin, or 5-hydroxytryptamine (5-HT), is a monoamine neurotransmitter found in blood platelets, the GI tract and the central nervous system (CNS) of animals and humans [
1]. It is a well-known neurotransmitter in the pathways associated with mood, and its action is known to contribute to the feeling of happiness [
2].
Approximately 90% of the human body’s total 5-HT is located in enterochromaffin cells in the gut, where it is used to regulate intestinal movement [
3,
4]. 5-HT has proven to be an important mediator of gastrointestinal motility [
5,
6]. Based on pharmacological criteria, 5-HT receptors are classified into seven main receptor subtypes. Five of them are present in enteric neurons, enterochromaffin cells, and GI smooth muscle, namely 5-HT
1, 5-HT
2, 5-HT
3, 5-HT
4 and 5-HT
7 [
7,
8]. In the gastrointestinal tract, 5-HT is reported to contribute to the regulation of motility and secretion via several 5-HT receptor types [
9]. Serotonin receptors are activated by the neurotransmitter serotonin, which acts as their natural ligand [
6], and serotonin receptors are the target of a variety of pharmaceutical and illicit drugs, including many antidepressants, antipsychotics, anorectics, antiemetics, gastroprokinetics, antimigraine agents, hallucinogens and entactogens [
10].
G proteins function to transduce the effect of a ligand binding to a cell surface receptor into intracellular signals. Isolated ESMCs were permeabilized with saponin to allow G protein antibodies to enter into the cytoplasm. These antisera, raised against synthetic peptides corresponding to the amino acid sequence of the carboxyl terminal of the G protein α subunit, have been used as effective probes for G protein structure and function [
11]. For example, antibodies raised against the carboxyl terminal region of the α subunit of G
αi2 successfully blocked receptor-mediated adenylyl cyclase inhibition [
12].
Many studies have shown diverse mechanisms by which intestinal smooth muscle contraction occurs [
13,
14]. Contraction of the lower esophageal sphincter by acetylcholine (ACh) is mediated by activation of M
3 muscarinic receptors linked to G
q and to phosphatidylinositol-specific phospholipase C (PI-PLC), as well as by production of inositol 1, 4, 5-trisphosphate (IP
3) and diacylglycerol (DAG). IP3 causes release of Ca
2+ from stores at a concentration sufficient to cause activation of calmodulin (CAM), and Ca
2+ CAM causes activation of myosin light chain kinase (MLC kinase).
Acetylcholine-induced contraction of esophageal muscle is mediated by muscarinic M
2 receptors linked to G
i3-type G proteins, which activate phosphatidylcholine-specific phospholipase C (PC-PLC) and phospholipase D (PLD) to produce DAG. DAG and arachidonic acid (AA), which are generated by cytosolic phospholipase A
2 (cPLA
2), interact to activate protein kinase C (PKC)-ε. The influx of Ca
2+ may independently activate the same phospholipases and produce the same second messengers, potentiating the activation of PKC-ε, which, in turn, is linked to two separate mitogen-activated protein (MAP) kinase pathways. One MAP kinase pathway is dependent on ERK1/ERK2, and the other is dependent on heat-shock protein (HSP) 27-linked p38 MAP kinase [
15].
In many GI studies, guinea pigs, rats, mice, ferrets, dogs and humans have been commonly used as models [
16,
17,
18,
19,
20]. However, the effect of 5-HT on the regulation of intestinal contractions has not been established in felines. Likewise, the involvement of 5-HT receptors in 5-HT-elicited responses has been less extensively investigated in the feline esophagus.
DA-9701 is the standardized extract of the seed of
Pharbitis nil Choisy (Pharbitidis Semen, Convolvulaceae) and the root of
Corydalis yahusuo W.T. Wang (Corydalis Tuber, Papaveraceae) [
21]. We previously found that DA-9701 has strong gastroprokinetic effects and a safety profile superior to conventional prokinetics like cisapride and mosapride [
21]. DA-9701 not only accelerates gastric emptying and the GI transit of meals in normal conditions and conditions that result in an abnormal delay, but also enhances gastric accommodation in conscious dogs [
21].
In the present study, the signaling pathways initiated by 5-HT in feline esophageal muscle cells were identified. Also, the effect of DA-9701 on 5-HT-induced contraction was confirmed. In order to determine the receptor mediating the contraction, selective 5-HT agonists and antagonists were used. Specific G protein antibodies were used to identify the coupling of specific G proteins to effector enzymes, and selective inhibitors were used to characterize the pathways involved in MLC20 (20 kDa regulatory light chain of myosin II) phosphorylation and muscle contraction. Pretreatment with DA-9701 and then treatment with 5-HT and 5-HT agonists elucidated the effect of DA-9701 on 5-HT-induced contraction, and MLC20 phosphorylation was used to determine the effect of DA-9701.
2. Results and Discussion
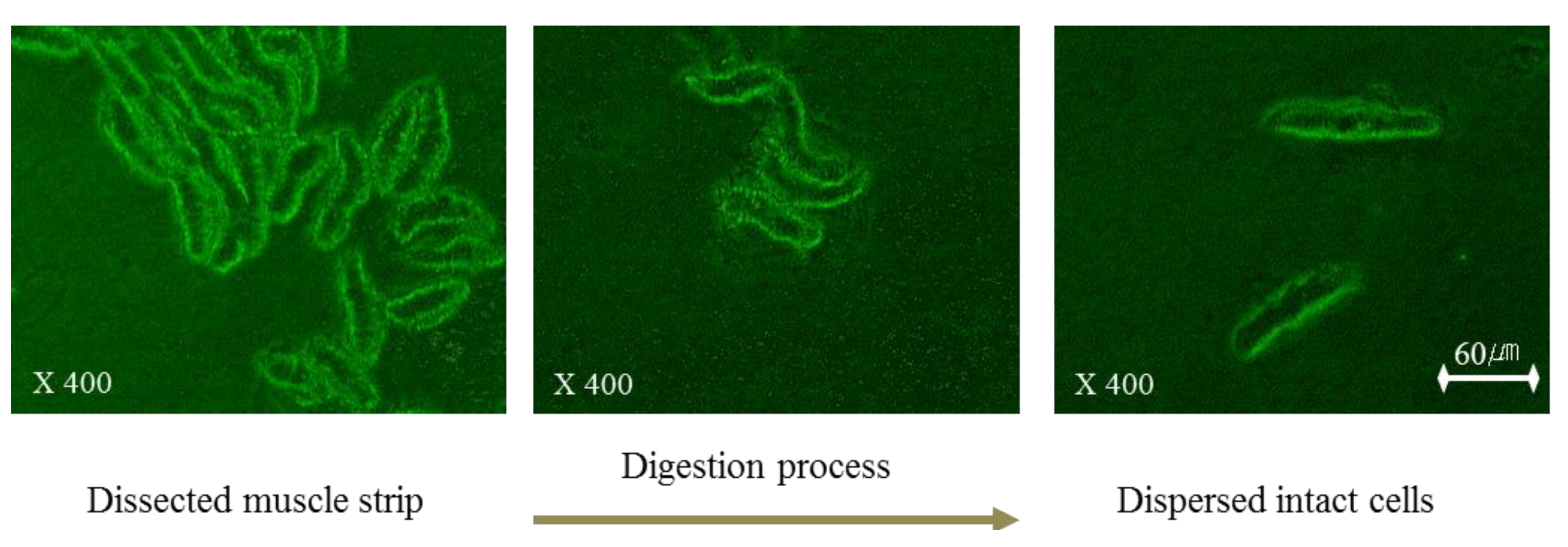
2.1. Identification of Dispersed Esmcs
To assess the dispersing process of esophageal smooth muscle in collagenase buffer, the dissected smooth muscle strip was observed every 5 min by microscopy after an overnight incubation. After this incubation, we observed that ESMCs were separated from the esophageal smooth muscle tissue squares (
Figure 1). After identification of the separate ESMCs, the incubation was ended to prepare the dispersed muscle cell suspension. In
Figure 1, the dispersing process is shown starting on the left and proceeding to the right. Freshly isolated ESMCs were spindle shaped with diverse lengths, ranging from 38–92 µm. The dispersing process was captured using a digital closed-circuit video camera.
Figure 1.
The dispersing process of feline ESMCs.
Figure 1.
The dispersing process of feline ESMCs.
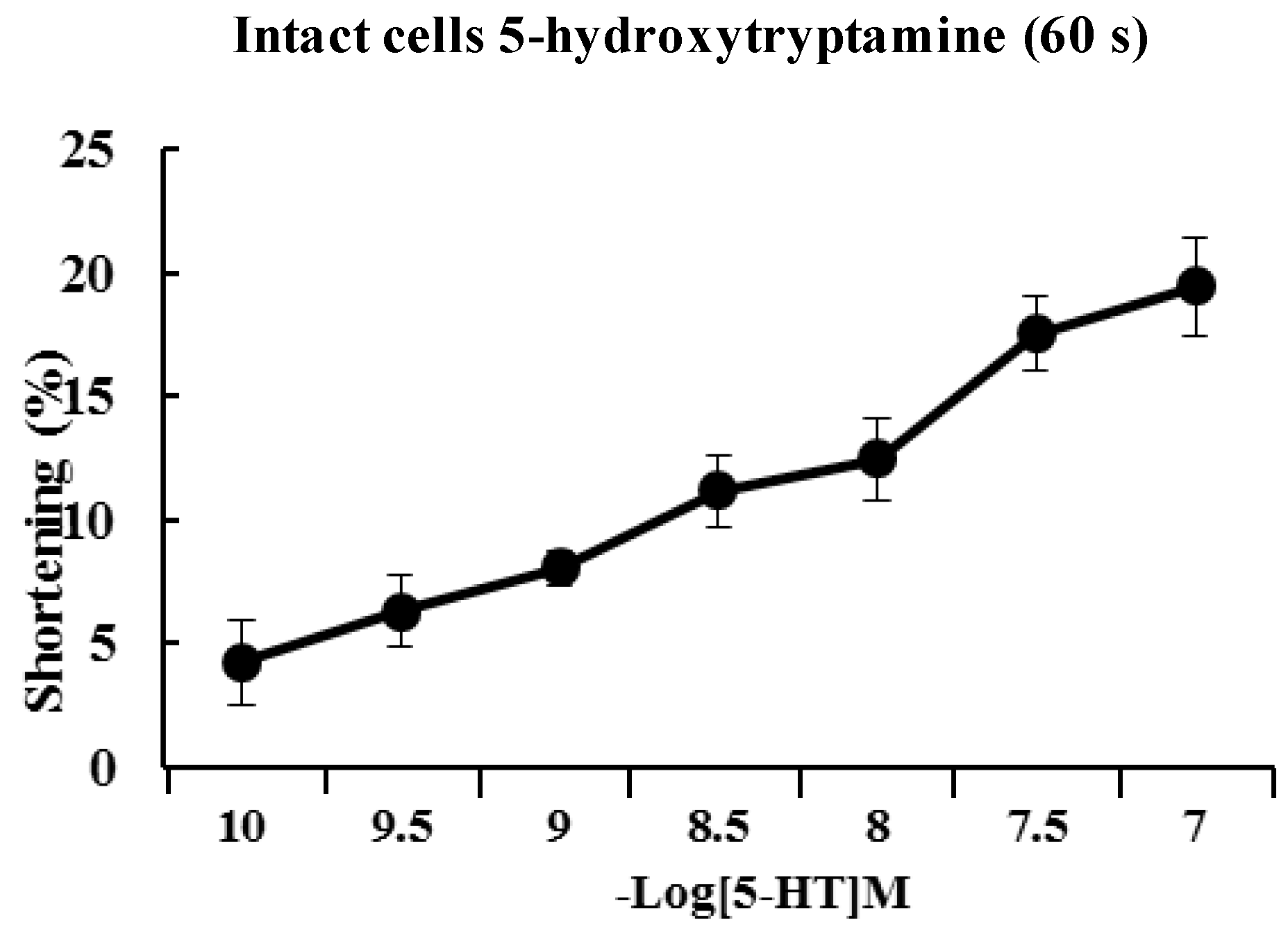
2.2. 5-HT Induces Contraction of Isolated ESMCs
Approximately 90% of the human body’s total serotonin is located within enterochromaffin cells in the gut where it is used to regulate intestinal movements. In the present study, freshly isolated ESMCs were stimulated for 60 s with 10
−10 to 10
−7 M 5-HT. The response to 5-HT was concentration-dependent with a maximal response observed at 10
−7 M (
Figure 2).
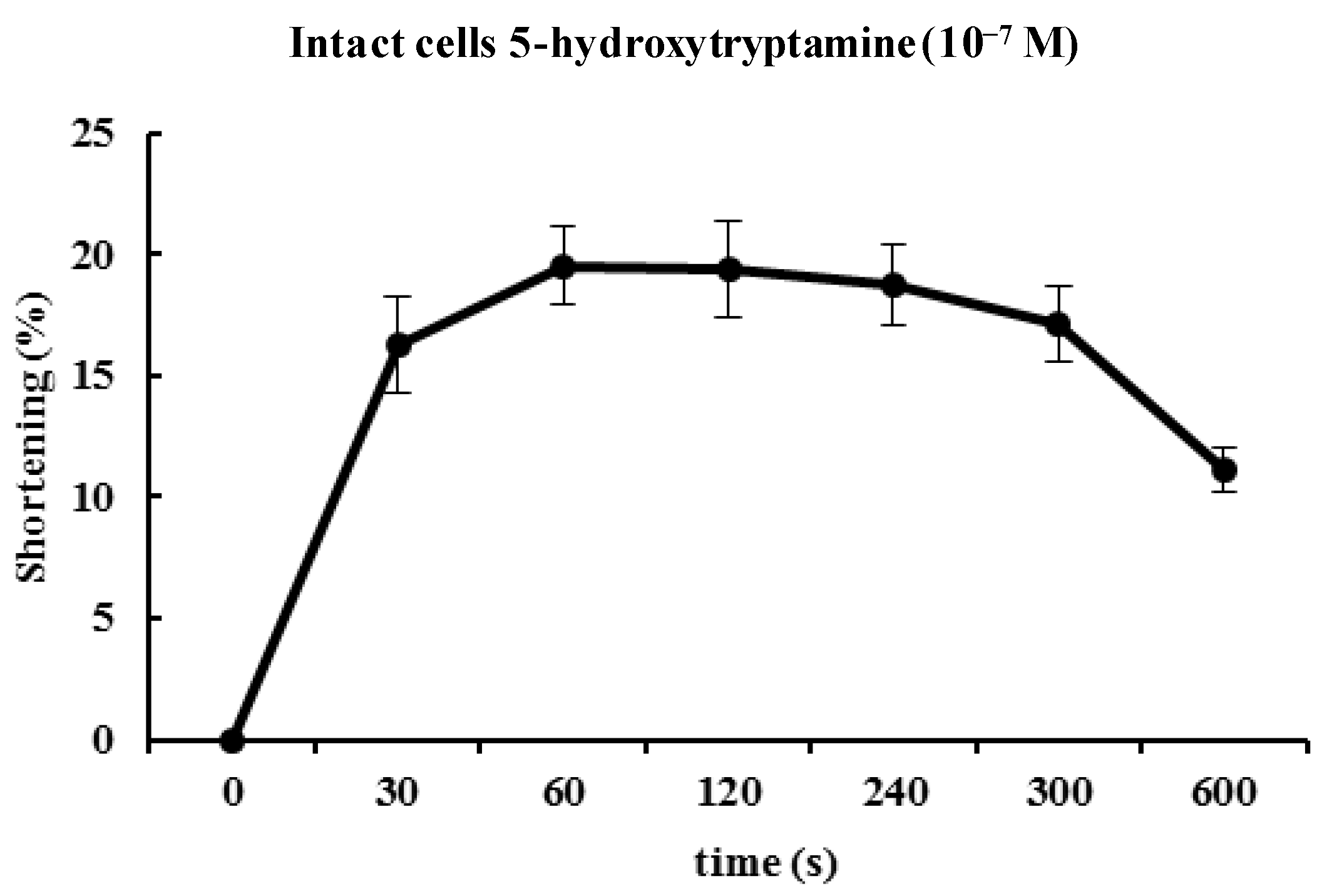
Figure 3 illustrates the time course of 5-HT-induced contractions. The contractions peaked at 60 s and then slowly declined. Based on the concentration-response and time-course data, ESMCs were exposed to 5-HT at a final concentration of 10
−7 M for 60 s in most of the later experiments (
Figure 3).
Figure 2.
Dose-dependent contractile response of cat ESMCs to 5-HT. Data are expressed as the means ± S.E. of four experiments.
Figure 2.
Dose-dependent contractile response of cat ESMCs to 5-HT. Data are expressed as the means ± S.E. of four experiments.
Figure 3.
Time course of the contractile response of cat smooth muscle cells to 5-HT (10−7 M). Data are expressed as the means ± S.E. of four experiments.
Figure 3.
Time course of the contractile response of cat smooth muscle cells to 5-HT (10−7 M). Data are expressed as the means ± S.E. of four experiments.
As mentioned, the contractions showed concentration dependency that reached a maximum at 10−7 M. The cellular response to 5-HT was evoked quickly (in 60 s) and decreased slowly. 5-HT-induced responses were initiated quickly and had prolonged effects in ESMCs. Next, the mechanism of 5-HT-induced contraction in feline ESMCs was explored.
2.3. Effects of a 5-HT Receptor Antagonist on 5-HT-Induced Contraction
To test whether the 5-HT1 or 5-HT2 receptor is involved in 5-HT-induced muscle contraction, freshly isolated ESMCs were pretreated with (A) methysergide (5-HT1A partial agonist and 5-HT2B,C receptor antagonist at 10−8 M, 10−7 M, and 10−6 M) or (B) ketanserin (a selective 5-HT2 receptor antagonist at 10−8 M, 10−7 M, and 10−6 M) for 1 min and then treated with 5-HT (10−7 M). Methysergide did not inhibit 5-HT-induced contraction. Pretreatment with ketanserin, on the other hand, significantly inhibited 5-HT-induced smooth muscle cell contraction.
To identify whether the 5-HT
3 or 5-HT
4 receptor is involved in 5-HT-induced contraction, freshly isolated ESMCs were pretreated with (C) ondansetron (a selective 5-HT
3 receptor antagonist at 10
−9, 10
−8, and 10
−7 M) or (D) GR113808 (a selective 5-HT
4 receptor antagonist at 10
−10, 10
−9 and 10
−8 M) for 1 min and then treated with 5-HT (10
−7 M). Ondansetron and GR113808 significantly inhibited 5-HT-induced smooth muscle cell contraction. These results suggest that 5-HT
2, 5-HT
3 and 5-HT
4 receptors mediate 5-HT-induced contraction (
Figure 4). Based on
Figure 4A, 5-HT
1 receptor seems to be involved in 5-HT-induced contraction. It needs further study to evaluate.
Figure 4.
Effect of a 5-HT antagonist on 5-HT-induced contractions in isolated ESMCs. (A) Effect of a 5-HT1 partial agonist and 5-HT2 antagonist on 5-HT-induced contractions; (B) Effect of a 5-HT2 antagonist on 5-HT-induced contractions; (C) Effect of a 5-HT3antagonist on 5-HT-induced contractions; (D) Effect of a 5-HT4 antagonist on 5-HT-induced contractions. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. control, * p < 0.05 vs. control).
Figure 4.
Effect of a 5-HT antagonist on 5-HT-induced contractions in isolated ESMCs. (A) Effect of a 5-HT1 partial agonist and 5-HT2 antagonist on 5-HT-induced contractions; (B) Effect of a 5-HT2 antagonist on 5-HT-induced contractions; (C) Effect of a 5-HT3antagonist on 5-HT-induced contractions; (D) Effect of a 5-HT4 antagonist on 5-HT-induced contractions. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. control, * p < 0.05 vs. control).
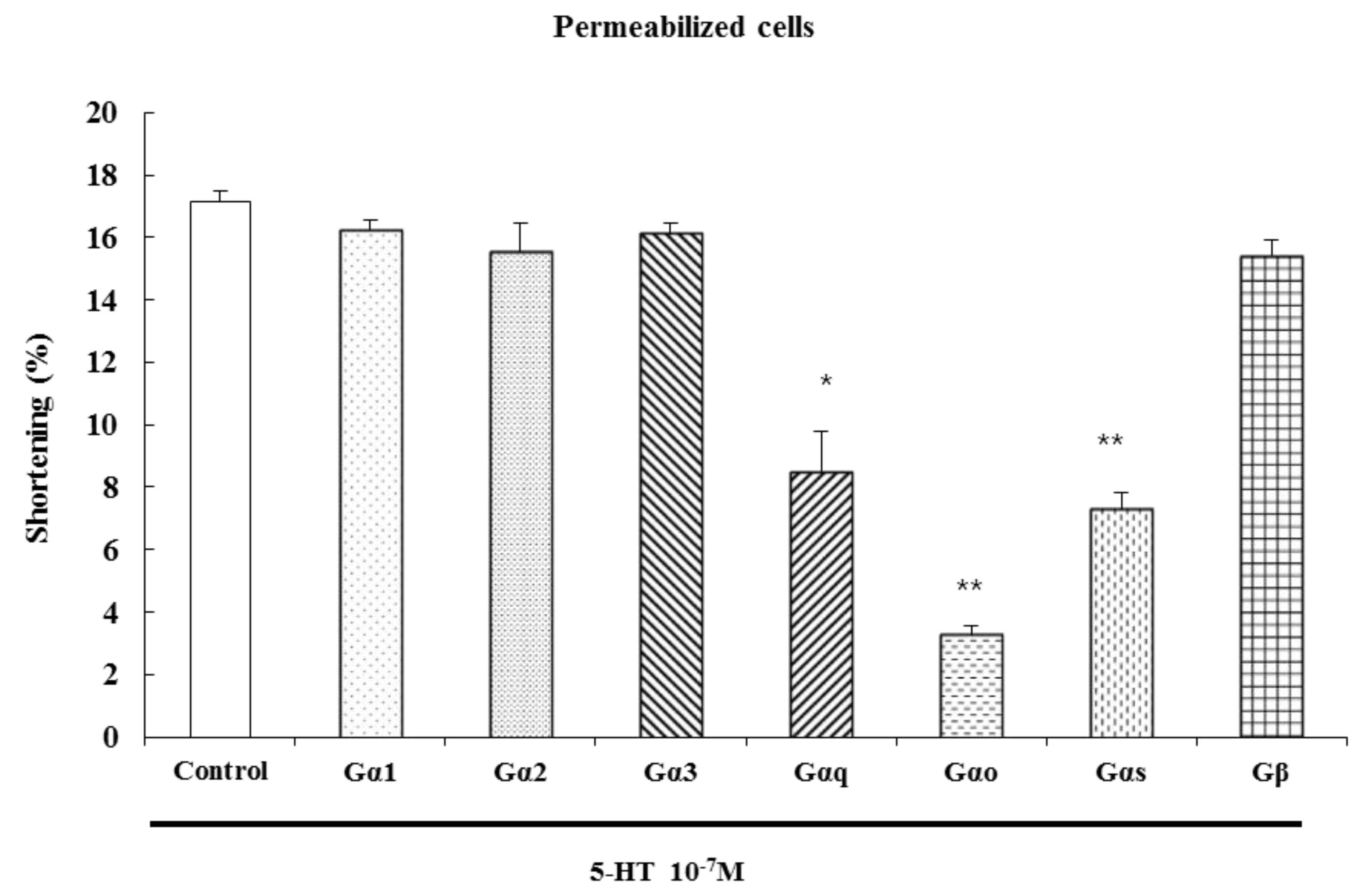
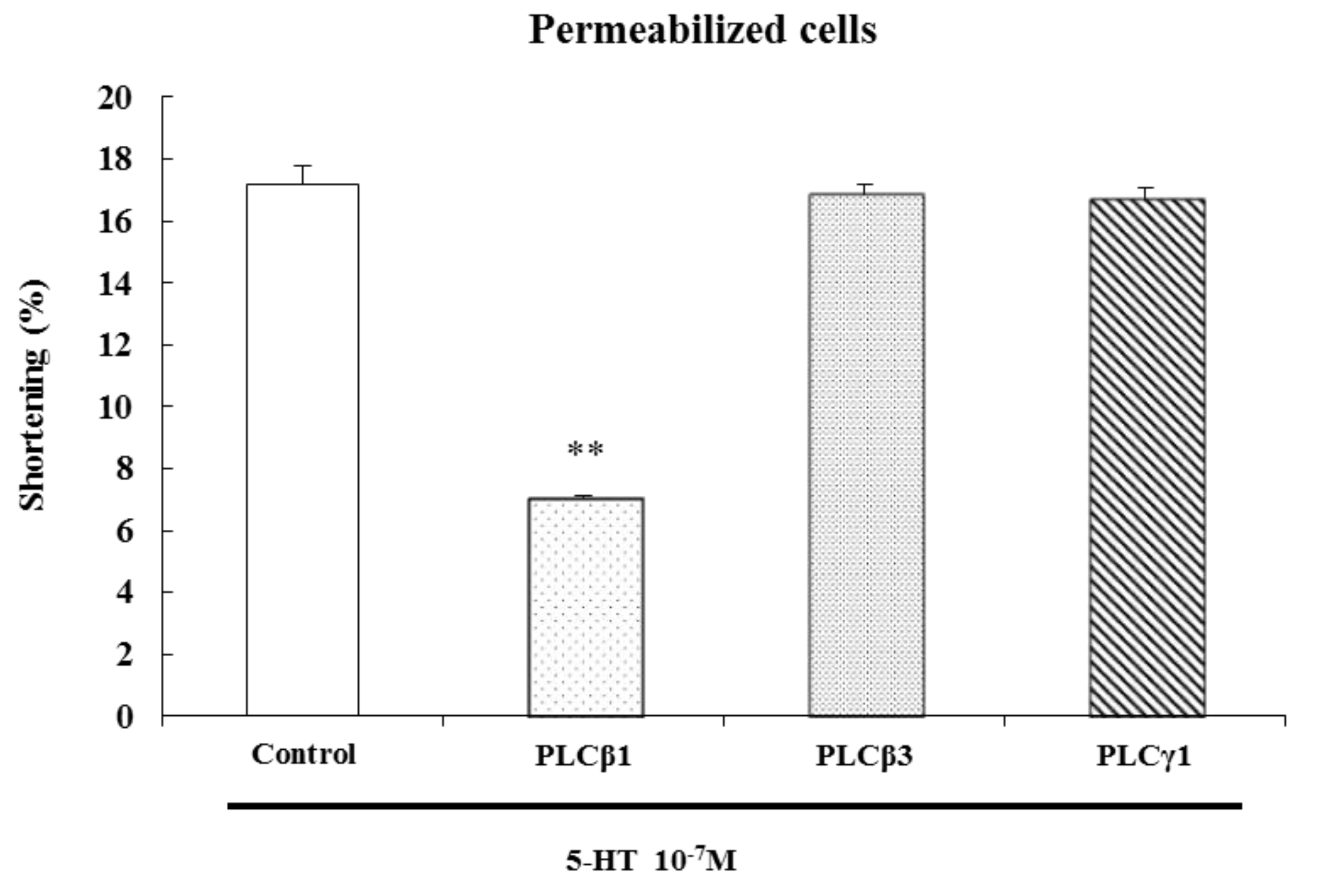
2.4. Characterization of the G Protein Subtypes Involved in 5-HT-Induced Contraction
The serotonin receptors, also known as 5-hydroxytryptamine receptors, or 5-HT receptors, are a group of G protein-coupled receptors and ligand-gated ion channels [
22]. After specific antibodies bind, G protein cannot be activated by signals from receptors, blocking the transduction of the signal to its effectors. We have previously shown that G
αi1, G
αi2, G
αi3, G
β (40 kDa), G
αo (40 kDa), G
αq (42 kDa), and G
αs (46 kDa) proteins exist in cat ESMCs [
23].
To identify the specific G protein involved in 5-HT-induced esophageal contraction, cells were permeabilized with saponin in cytosolic medium containing each G protein antibody (1:200 dilution) to allow diffusion of antibodies into the cell. These antibodies block receptor-induced activation of G proteins by binding to their terminal peptide regions, which normally interact with the receptor. Antibodies to G
αq, G
αo and G
αs inhibited 5-HT-induced contractions, but antibodies to G
αi1, G
αi2, G
αi3, and G
β did not. We firstly confirmed that the antibodies used in these experiments did not affect the contraction of ESMCs by themselves. These data suggest that 5-HT-induced muscle contraction is mediated by G
αq, G
αo, and G
αs proteins (
Figure 5).
Figure 5.
Inhibition of 5-HT-induced contraction in permeabilized ESMCs by antibodies to G protein isoforms. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. control, * p < 0.05 vs. control).
Figure 5.
Inhibition of 5-HT-induced contraction in permeabilized ESMCs by antibodies to G protein isoforms. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. control, * p < 0.05 vs. control).
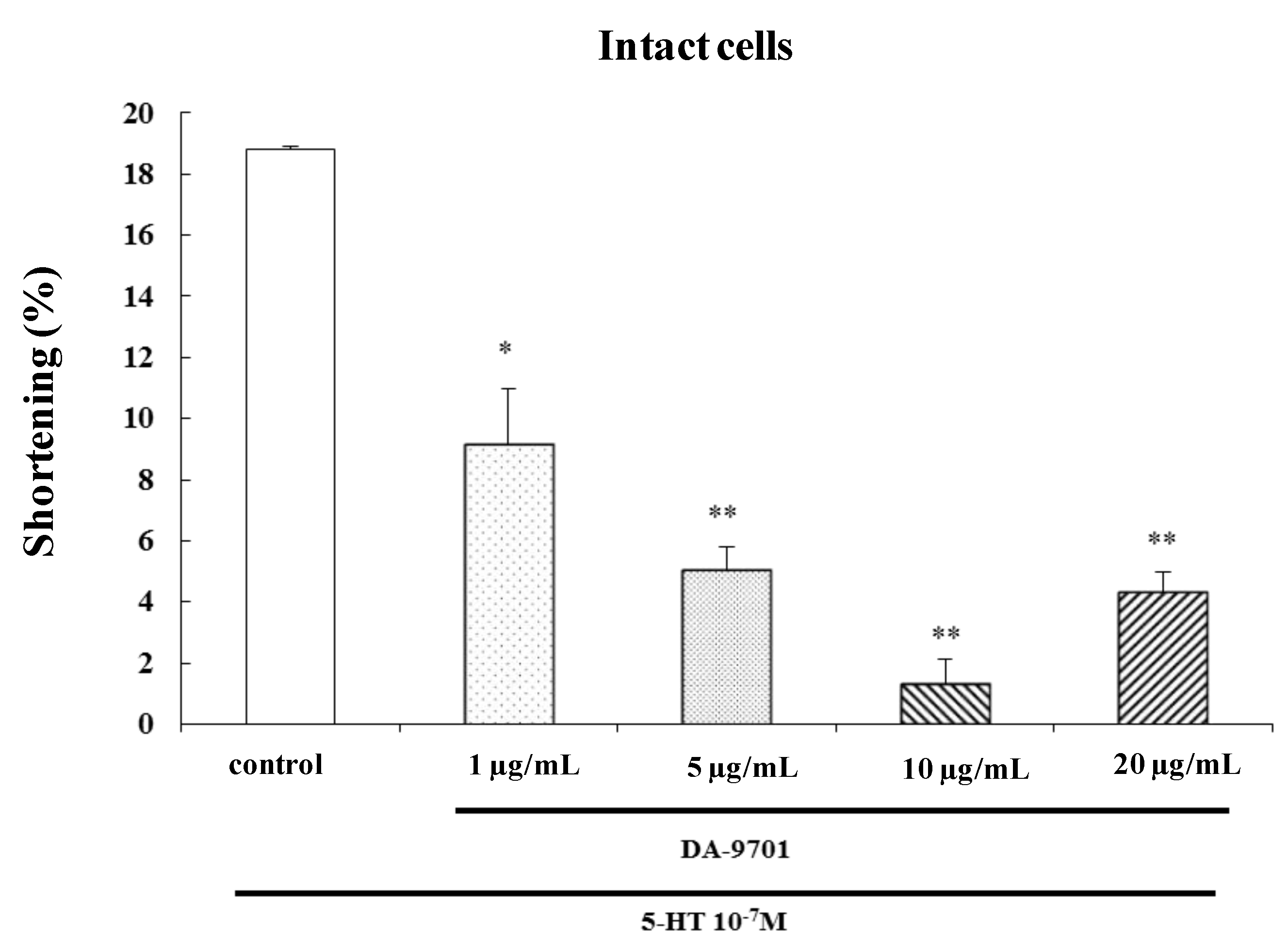
2.7. Effects of DA-9701 on 5-HT-Induced Contraction
To investigate the effect of DA-9701 on 5-HT-induced contraction, freshly dispersed ESMCs were incubated with DA-9701 (1, 5, 10, and 20 μg/mL) for 1 min and then treated with 5-HT (10
−7 M). DA-9701 significantly inhibited 5-HT-induced contraction. The most effective dose of DA-9701 was 10 μg/mL (
Figure 8).
Figure 8.
Effect of DA-9701 on 5-HT-induced contraction in isolated ESMCs. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. control, * p < 0.05 vs. control).
Figure 8.
Effect of DA-9701 on 5-HT-induced contraction in isolated ESMCs. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. control, * p < 0.05 vs. control).
2.8. Effects of DA-9701 on 5-HT Agonist-Induced Contraction
To test the effect of 5-HT receptor agonist-induced contraction, freshly dispersed ESMCs were treated with a 5-HT2 agonist (α-methylserotonin, 10−6 M), a 5-HT3 agonist (2-methyl-5-HT, 10−6 M), and a 5-HT4 agonist (mosapride, 10−7 M). α-Methylserotonin, 2-methyl-5-HT, and mosapride caused feline ESMCs to contract. These results offered additional confirmation that 5-HT2, 5-HT3, and 5-HT4 receptors are involved in 5-HT-induced contraction.
To identify the effect of DA-9701 on 5-HT agonist-induced contraction, freshly dispersed ESMCs were incubated with DA-9701 (10 μg/mL) for 1 min and then treated with a 5-HT
2 agonist (α-methylseroyonin), a 5-HT
3 agonist (2-methyl-5-HT), and a 5-HT
4 agonist (mosapride). Contraction was measured after 1 min. DA-9701 significantly inhibited 5-HT agonist-induced contraction (
Figure 9).
Figure 9.
The effect of DA-9701 on 5-HT agonist-induced contraction of cat ESMCs. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. control).
Figure 9.
The effect of DA-9701 on 5-HT agonist-induced contraction of cat ESMCs. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. control).
2.9. Effects of DA-9701 and 5-HT on MLC20 Phosphorylation
We performed an MLC
20 phosphorylation experiment to determine whether 5-HT-induced phosphorylation of MLC
20 occurred at Ser
19 in dispersed ESMCs, and we observed that 5-HT significantly increased phosphorylation of MLC
20. To identify the effect of DA-9701 on phosphorylation of MLC
20 in 5-HT-induced muscle contraction, freshly dispersed ESMCs were pretreated with DA-9701 (10 μg/mL) for 1 min and then treated with 5-HT (10
−7 M). As shown in
Figure 10, the p-MLC
20/MLC
20 level decreased in DA-9701 treated cells. These results indicate that DA-9701 significantly inhibits phosphorylation of MLC
20 (
Figure 10).
It was previously found that DA-9701 has strong gastroprokinetic effects and a safety profile superior to conventional prokinetics, including cisapride and mosapride [
21]. ESMCs were exposed to various concentrations of DA-9701 and then treated 5-HT. DA-9701 inhibited 5-HT-induced contraction, and the inhibition was maximal at a treatment dose of 10 μg/mL. Increased immunoreactive protein bands of 5-HT-treated cells corresponded to the 20 kDa phosphospecific Ser
19-MLC
20 antibody. These results indicate that activated MLC kinase phosphorylates MLC
20.
Figure 10.
The effect of 5-HT and DA-9701 on phosphorylation of MLC20. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. vehicle, # p < 0.01 vs. control).
Figure 10.
The effect of 5-HT and DA-9701 on phosphorylation of MLC20. Data are expressed as the means ± S.E. of four experiments (Student’s t-test; ** p < 0.01 vs. vehicle, # p < 0.01 vs. control).
3. Experimental
3.1. Materials
5-Hydroxytryptamine hydrochloride, methysergide maleate salt, ketanserin (+)-tartrate salt, ondansetron hydrochloride dihydrate, GR113808 (1-[2-[(methylsulfonyl)amino]ethyl]-4-piperidinyl]methyl-1-methyl-1H-indole-3-carboxylate), α-methylserotonin, mosapride, U73122, ML-9, Y27632, and chelerythrine were from Sigma Chemical Co. (St Louis, MO, USA). 2-methyl-5-hydroxytryptamine HCl (Tocris, Bristol, UK), DA-9701 (Dong-A Pharmaceutical Co., Seoul, Korea ), G protein antibodies (Gi1, Gi2, Gi3, Gq, Gs, Go, and G β), PLC isozyme antibodies (β1, β3, γ1), phosphospecific Ser19-MLC20 antibody, and GAPDH antibody were from Santa Cruz Biotechnology (Santa Cruz, CA, USA) and Ser19-MLC20 antibody was from Cell Signaling Technology (Beverly, MA, USA). Goat anti-rabbit IgG-HRP and goat anti-mouse IgG-HRP were from Zymed Laboratories Inc. (Eccles Avenue, CA, USA). The rainbow molecular weight marker was from Amersham (Arlington Heights, IL, USA). The enhanced chemiluminescence (ECL) agents were from PerkinElmer Life Sciences (Boston, MA, USA). The sodium dodecyl sulfate (SDS) sample buffer was from Owl Scientific Inc. (Woburn, MA, USA). The nitrocellulose membrane, Tris/Glycine/SDS and Tris/Glycine buffers were from BioRad (Richmond, CA, USA). The phosphate-buffered saline (PBS) was from Roche Diagnostics Co. (Indianapolis, IN, USA). RestoreTM Western blot stripping buffer was from Pierce (Rockford, IL, USA). The 4-(2-hydroxyethyl)-1-piperazine-N'-2-ethane sulfonic acid (HEPES), collagenase type F, ammonium persulfate, Ponceau S, bovine serum albumin (BSA), leupeptin, aprotinin, β-mercaptoethanol, N,N,N',N'-tetramethylethylene diamine (TEMED), ethylene glycol-bis-(β-aminoethylether)-N,N,N',N'-tetraacetic acid (EGTA), ethylene diamine tetraacetic acid (EDTA), phenylmethyl-sulfonylfluoride (PMSF) and other reagents were purchased from Sigma Chemical Co. (St Louis, MO, USA).
3.2. Preparation of Dispersed Muscle Cells
Single muscle cells were isolated as previously described [
24]. Muscle strips were incubated overnight in normal potassium HEPES buffer containing 1 mg/mL papain, 1 mM dithiothreitol, 1 mg/mL BSA, and 0.5 mg/mL collagenase (type F, Sigma), and muscle strips were equilibrated with 95% O
2–5% CO
2 to maintain a pH of 7.0 at 31 °C. The composition of normal potassium HEPES buffer was 1 mM CaCl
2, 250 µM EDTA, 10 mM glucose, 10 mM HEPES, 4 mM KCl, 131 mM NaCl, 1 mM MgCl
2, and 10 mM taurine. Twenty-four h later, tissue was warmed to room temperature for 30 min and incubated in a water bath at 31 °C for 30 min. After incubation, the digested tissue was poured over a 360 µm Nitex filter, rinsed in collagenase-free HEPES buffer to remove any trace of collagenase, and then incubated in this solution at 31 °C gassed with 95% O
2–5% CO
2. The cells were allowed to dissociate freely for 10 to 20 min. Suspensions of single muscle cells were harvested by filtration through a 500 µm Nitex mesh [
24]. Before beginning the experiment, the cells were kept at 31 °C for at least 10 min to relax them. Throughout the procedure, care was taken not to agitate the fluid to avoid cell contraction in response to mechanical stress. Animal experiments were approved by the Institutional Animal Care and Use Committee of Chung-Ang University, in accordance with the guide for the Care and Use of Laboratory Animals in Seoul, Korea.
3.3. Preparation of Permeabilized Smooth Muscle Cells
When required, cells were permeabilized to allow the non-diffusible agents, such as G protein antibodies or PLC isozyme antibodies, to diffuse across the intact cell membranes. Notably, contraction in permeabilized cells is not significantly different from that in intact cells [
25,
26,
27,
28,
29]. After completion of the enzymatic phase of the digestion process, the partly digested muscle tissue was washed with an enzyme-free cytosolic buffer with the following composition: 20 mM NaCl, 100 mM KCl, 5.0 mM MgSO
4, 0.96 mM NaH
2PO
4, 1.0 mM EGTA, 0.48 mM CaCl
2, and 2% bovine serum albumin. The cytosolic buffer was equilibrated with 95% O
2–5% CO
2 to maintain a pH of 7.2 at 31 °C. Muscle cells dispersed spontaneously in this medium. The cytosolic buffer contained 0.48 mM CaCl
2 and 1 mM EGTA, yielding 0.18 mM of free Ca
2+ [
30]. After dispersion, the cells were permeabilized by incubation for 5 min in cytosolic buffer that contained saponin (75 µg/mL). After exposure to saponin, the cell suspension was centrifuged at 350 g, and the resulting pellet was washed with saponin-free modified cytosolic buffer that contained antimycin A (10 µM), ATP (1.5 mM), and an ATP-regenerating system that consisted of creatine phosphate (5 mM) and creatine phosphokinase (10 units/mL). After the cells were washed free of saponin, they were re-suspended in modified cytosolic buffer.
3.4. Preparation of Dispersed Muscle Cells
Contraction of isolated muscle cells was measured by scanning micrometry [
31]. An aliquot of cell suspension containing 10
4 muscle cells/mL was added to HEPES medium containing the test agents. The reaction was terminated by addition of acrolein (1% final concentration). A length of 40 to 50 muscle cells treated with a contractile agent was measured at random by a scanning micrometry phase-contrast microscope (model ULWCD 0.30 Olympus, Tokyo, Japan) and by a digital closed-circuit video camera (CCD color camera, Toshiba, Tokyo, Japan) connected to a Macintosh computer (Apple, Cupertino, CA, USA) using the NIH Image 1.57 software program (National Institutes of Health, Bethesda, MD, USA). Measurements of the treated cells were compared to the lengths of untreated cells. Contraction was expressed as the percent reduction in mean cell length from baseline. Using agonists, the time course of contraction consists of a peak contraction followed by a lower, sustained plateau. In the present study, contraction refers to the one that occurred after 60 s of agonist (5-HT) exposure.
3.5. Western Blot Analysis of MLC20 Phosphorylation
Equal amounts of the proteins from each sample were resolved on an SDS-polyacrylamide gel by electrophoresis, and the Bradford reagents determined the supernatant protein concentration, according to the instructions of the manufacturer (Bio-Rad) The absorbance was measured spectrophotometrically at a wavelength of 595 nm.
Phosphorylated MLC
20 was determined by immunoblot analysis using a phospho-specific antibody [
13,
32]. Previously frozen dispersed muscle cell samples were homogenized in a buffer containing 20 mM Tris-HCl (pH 7.4), 0.5 mM EDTA, 0.5 mM EGTA, 1% (
w/v) Triton X-100, 0.01% (
w/v) SDS, 10 μg/mL leupeptin, 10 μg/mL aprotinin, 1 mM PMSF, and 0.7 μg/mL β-mercaptoethanol. Samples of the homogenates were then centrifuged for 10 min at 4 °C, and the supernatants were collected. Aliquots were subjected to electrophoresis on an SDS-polyacrylamide gel. A pre-stained molecular mass marker was also run in an adjacent lane to allow for molecular mass determination of the loading buffer, which consisted of 25 mM Tris (pH 8.3), 192 mM glycine, and 0.1% SDS. The separated proteins were transferred to a 0.45-μm nitrocellulose membrane in transfer buffer (25 mM Tris (pH 8.3), 192 mM glycine, and 20% (
v/v) methanol) using a power supply (Power Pac 1000, Bio-Rad, Melville, NY, USA). To confirm gel-loading uniformity, blots were stained with Ponceau S. After confirmation, membranes were washed with PBS and then incubated in PBS buffer containing 5% non-fat dry milk for 1 h at room temperature to block nonspecific binding. After washing with PBS three times for 5 min, the membrane was incubated overnight with antibodies (1:1000 dilution) to MLC
20 (Ser
19) in a PBS solution containing 0.1% BSA at 4 °C. Then, the membrane was washed with PBS containing 0.05% Tween 20 twice for 5 min, and then the membrane was incubated with horseradish peroxidase-conjugated secondary antibody (1:5000 dilution) for 1 h at room temperature. Enhanced chemiluminescence agents (ECLs; Perkin Elmer) were used to detect immunoreactive bands, which were developed by an X-ray film developer and fixer. Developed films from ECL were scanned and analyzed densitometrically using Scion Image. Phosphorylation of MLC
20 was calculated as the ratio of phosphorylated MLC
20 to total MLC
20.
3.6. Analysis of Data
Data from individual assays represent the mean values from triplicate measurements. The data were expressed as the means ± S.E.M. and analyzed with a Student’s t-test. p < 0.05 was considered statistically significant.
4. Conclusions
In the present study, 5-HT-induced contraction was mediated by 5-HT2, 5-HT3, and 5-HT4 receptors, as determined by the use of 5-HT antagonists. ESMCs were pretreated with DA-9701, followed by treatment with 5-HT2, 5-HT3, and 5-HT4 agonists. 5-HT2, 5-HT3, and 5-HT4 agonists induced cell contraction, and DA-9701 inhibited those actions. Therefore, 5-HT2, 5-HT3, and 5-HT4 receptors were confirmed to mediate 5-HT-induced contractions, and DA-9701 inhibited these 5-HT-induced contractions.
Smooth muscle cells pretreated with DA-9701 and then treated with 5-HT showed decreased immunoreactive protein bands compared to 5-HT-treated cells alone. These bands corresponded to 20-kDa phosphospecific Ser19-MLC20. These results suggest that DA-9701 inhibits phosphorylation of MLC20.
In summary, 5-HT binds to its G protein-coupled receptors to induce smooth muscle contraction in feline ESMCs. 5-HT2, 5-HT3, and 5-HT4 receptors located on esophageal smooth muscle are coupled to Gαq, Gαo, and Gαs. These G proteins activate PLC, which leads to Ca2+/calmodulin-dependent MLCK activation, resulting in MLC20 phosphorylation and cell contraction. DA-9701 inhibits 5-HT-induced contraction by inhibiting phosphorylation of MLC20.