HPLC-DPPH Screening Method for Evaluation of Antioxidant Compounds Extracted from Semen Oroxyli
Abstract
:1. Introduction
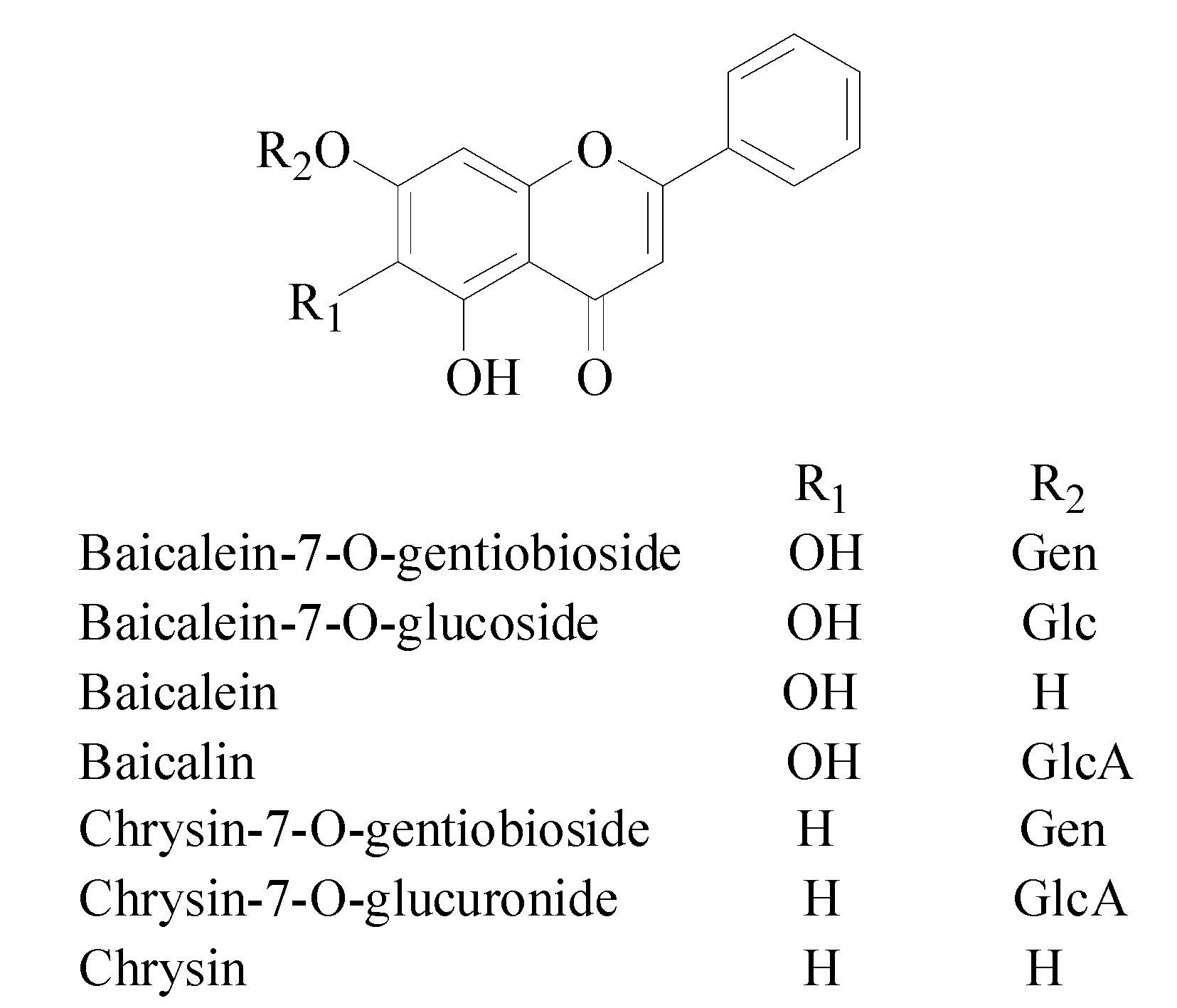
2. Results and Discussion
2.1. Online Post-Column HPLC-DPPH Method
| Concentration of DPPH (mol/L) | S/N | Flow rate of DPPH (mL/min) | S/N |
|---|---|---|---|
| 1 × 10−4 | 19.35 | 0.2 | 19.84 |
| 5 × 10−5 | 18.00 | 0.3 | 42.99 |
| 1 × 10−5 | 23.67 | 0.4 | 53.84 |
| 5 × 10−6 | 9.56 | 0.6 | 76.36 |
| 1 × 10−6 | ND | 0.8 | 133.43 |
| 1 × 10−7 | ND | 1.0 | 255.27 |
| Linearity | LOD (μg) | LOQ (μg) | Repeatability (n = 6) | Precision (n = 6) | Stability (24 h) | |||
|---|---|---|---|---|---|---|---|---|
| Test range (μg) | Regression Equation | r | S/N = 3 | S/N = 10 | RSD (%) | RSD (%) | RSD (%) | |
| Baicalein-7-O-gentiobioside | 1.85–18.48 | Y = 27803.40X + 10715.58 | 0.9991 | 1.16 | 1.85 | 1.58 | 2.27 | 4.94 |
| Baicalein-7-O-glucoside | 1.29–12.90 | Y = 54206.71X − 21149.04 | 0.9990 | 0.97 | 1.29 | 2.29 | 3.73 | 2.58 |
| Baicalin | 1.24–12.42 | Y = 54134.19X − 26188.18 | 0.9992 | 0.87 | 1.24 | 4.24 | 0.90 | 2.17 |
| Baicalein | 2.50–7.50 | Y = 49422.32X − 39602.40 | 0.9992 | 0.06 | 0.57 | 4.50 | 2.17 | 2.43 |

2.2. DPPH Spiking HPLC Analysis Method

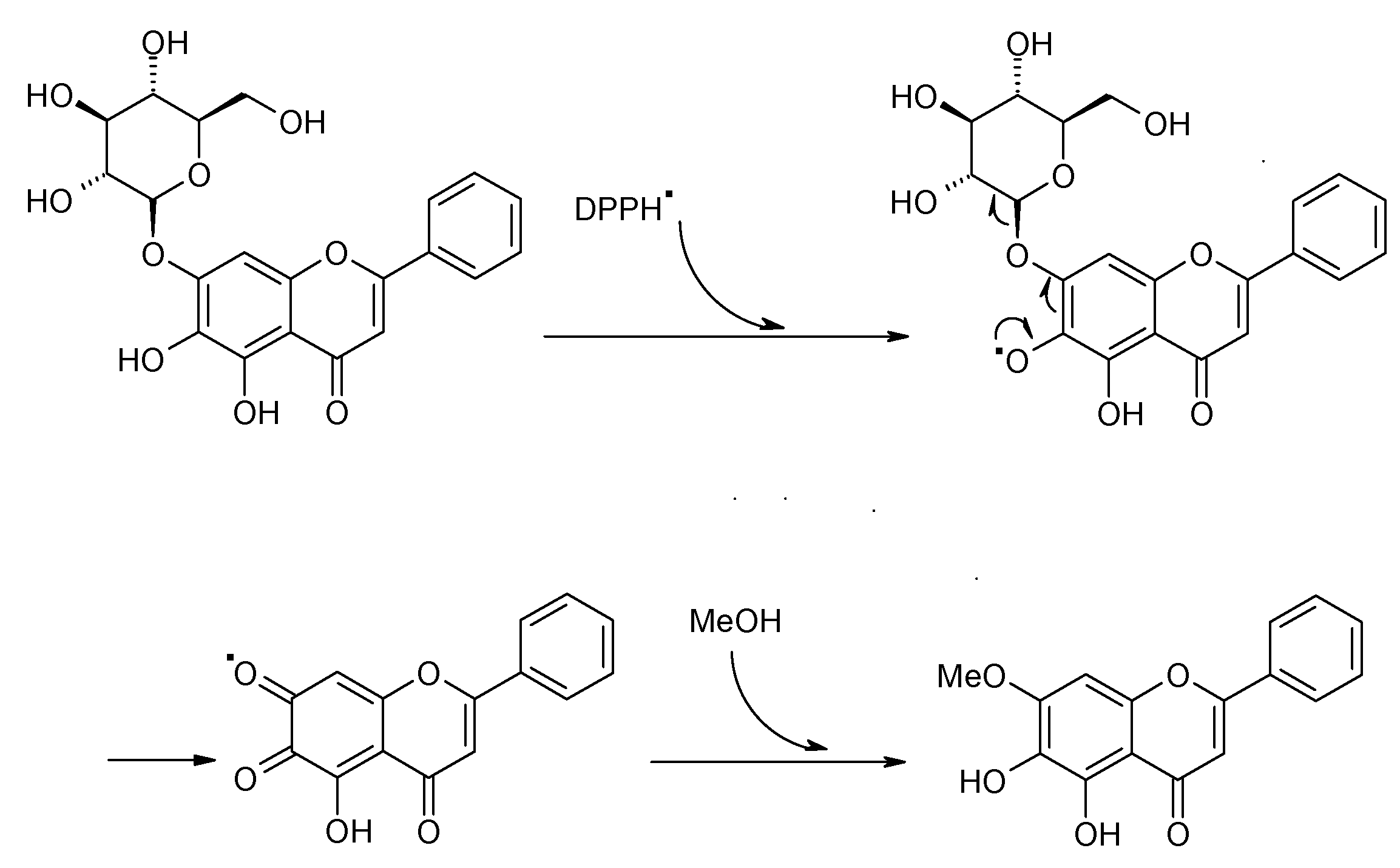
3. Experimental
3.1. General Information
3.2. Plant Materials
3.3. Preparation of Standard and Sample Solutions
3.4. Preparation of DPPH Stock Solution
3.5. HPLC Analysis
3.6. Online Post-Column HPLC-DPPH Assay

3.7. DPPH Spiking HPLC Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- National Commission of Chinese Pharmacopoeia. Pharmacopoeia of People’s Pepublic of China; China medical sciences Press: Beijing, China, 2010; p. 60. [Google Scholar]
- Jiansu New Medical College. The Dictionary of CHINESE Herbal Medicine; Shanghai people publishing house: Shanghai, China, 1977; p. 369. [Google Scholar]
- Babu, T.H.; Manjulatha, K.; Kumar, G.S.; Hymavathi, A.; Tiwari, A.K.; Purohit, M. Gastroprotective flavonoid constituents from Oroxylum indicum Vent. Bioorg. Med. Chem. Lett. 2010, 20, 117–120. [Google Scholar] [CrossRef]
- Chen, Z.L.; Zhao, Z.Y. Chemical studies on Oroxylum indicum (L) vent. (I)-Two new flavone glucosides oroxin A and oroxin B. Acta Pharm. Sin. 1964, 11, 762–767. [Google Scholar]
- Sankara, S.; Naif, A.G.R. Flavonoids of the leaves of Oroxylum indicum and Pajanelia tongifolia. Phytochemistry 1972, 11, 439–440. [Google Scholar] [CrossRef]
- Chen, L.J.; Games, D.E.; Jones, J. Isolation and identification four flavonoid constituents from the seeds of Oroxylum indicum by high-speed counter-current chromatography. J. Chromatogr. A 2003, 988, 95–105. [Google Scholar] [CrossRef]
- Tomimori, T.; Imoto, Y.; Ishida, M.; Kizu, H.; Namba, T. Studies on the Nepalese crude drugs (VIII). On the flavonoid constituents of the seed of Oroxylum indicum Vent. Shoyakugaku Zasshi 1988, 42, 98–101. [Google Scholar]
- Joshi, K.C.; Prakash, L.; Shah, R.K. Chemical examination of roots of Tabebuia rosea and heartwood of Oroxylum indicum. Planta Med. 1977, 31, 257–264. [Google Scholar] [CrossRef]
- Nair, A.G.R.; Joshi, B.S. Oroxindin-A new flavone glucuronide from Oroxylum indicum Vent. Proc. Indian Acad. Sci. 1979, 88A, 323–329. [Google Scholar]
- Rahman, I.; Biswas, S.K.; Kirkham, P.A. Regulation of inflammation and redox signaling by dietary polyphenols. Biochem. Pharmacol. 2006, 72, 1439–1452. [Google Scholar] [CrossRef]
- Kirkham, P.; Rahman, I. Oxidative stress in asthma and COPD: Antioxidants as a therapeutic strategy. Pharmacol. Ther. 2006, 111, 476–494. [Google Scholar] [CrossRef]
- Yan, R.Y.; Cao, Y.Y.; Chen, C.Y.; Dai, H.Q.; Yu, S.X.; Wei, J.L.; Li, H.; Yang, B. Antioxidant flavonoids from the seed of Oroxylum indicum. Fitoterapia 2011, 82, 841–847. [Google Scholar] [CrossRef]
- Cao, Y.Y.; Yan, R.Y.; Yang, L.X.; Guo, J.; Liu, H.L.; Zhang, J.Y.; Yang, B.; Huang, L.Q. Quality evaluation of Semen Oroxyli based on the determination of multiple components with a single reference standard. J. Chromatogr. Sci. 2013, 51, 477–484. [Google Scholar] [CrossRef]
- Li, Y.J.; Chen, J.; Li, Y.; Li, P. Identification and quantification of free radical scavengers in the flower buds of Lonicera species by online HPLC-DPPH assay coupled with electrospray ionization quadrupole time-of-flight tandem mass spectrometry. Biomed. Chromatogr. 2012, 26, 449–457. [Google Scholar] [CrossRef]
- Zhang, Y.P.; Shi, S.Y.; Xiong, X.; Chen, X.Q.; Peng, M.J. Comparative evaluation of three methods based on high-performance liquid chromatography analysis combined with a 2,2'-diphenyl-1-picrylhydrazyl assay for the rapid screening of antioxidants from Pueraria lobata flowers. Anal. Bioanal. Chem. 2012, 402, 2965–2976. [Google Scholar] [CrossRef]
- Ou, Z.Q.; Schmierer, D.M.; Rades, T.; Larsen, L.; McDowell, A. Application of an online post-column derivatization HPLC-DPPH assay to detect compounds responsible for antioxidant activity in Sonchus oleraceus L. leaf extracts. J. Pharm. Pharmacol. 2012, 65, 271–279. [Google Scholar]
- McDermott, G.P.; Noonan, L.K.; Mnatsakanyan, M.; Shalliker, R.A.; Conlan, X.A.; Barnett, N.W.; Francis, P.S. High-performance liquid chromatography with post-column 2,2'-diphenyl-1-picrylhydrazyl radical scavenging assay: Methodological considerations and application to complex samples. Anal. Chim. Acta 2010, 675, 76–82. [Google Scholar] [CrossRef]
- Dapkevicius, A.; van Beek, T.A.; Niederländer, H.A.G. Evaluation and comparison of two improved techniques for the on-line detection of antioxidants in liquid chromatography eluates. J. Chromatogr. A 2001, 912, 73–82. [Google Scholar]
- Koleva, I.I.; Niederländer, H.A.; van Been, T.A. An on-line HPLC method for detection of radical scavenging compounds in complex mixtures. Anal. Chem. 2000, 72, 2323–2328. [Google Scholar] [CrossRef]
- Amić, D.; Davidović-Amić, D.; Beslo, D.; Rastija, V.; Lucić, B.; Trinajstić, N. SAR and QSAR of the antioxidant activity of flavonoids. Curr. Med. Chem. 2007, 14, 827–845. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the all samples here mentioned are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yan, R.; Cao, Y.; Yang, B. HPLC-DPPH Screening Method for Evaluation of Antioxidant Compounds Extracted from Semen Oroxyli. Molecules 2014, 19, 4409-4417. https://doi.org/10.3390/molecules19044409
Yan R, Cao Y, Yang B. HPLC-DPPH Screening Method for Evaluation of Antioxidant Compounds Extracted from Semen Oroxyli. Molecules. 2014; 19(4):4409-4417. https://doi.org/10.3390/molecules19044409
Chicago/Turabian StyleYan, Renyi, Yangyang Cao, and Bin Yang. 2014. "HPLC-DPPH Screening Method for Evaluation of Antioxidant Compounds Extracted from Semen Oroxyli" Molecules 19, no. 4: 4409-4417. https://doi.org/10.3390/molecules19044409




