The Anthraquinone Derivatives from the Fungus Alternaria sp. XZSBG-1 from the Saline Lake in Bange, Tibet, China
Abstract
:1. Introduction
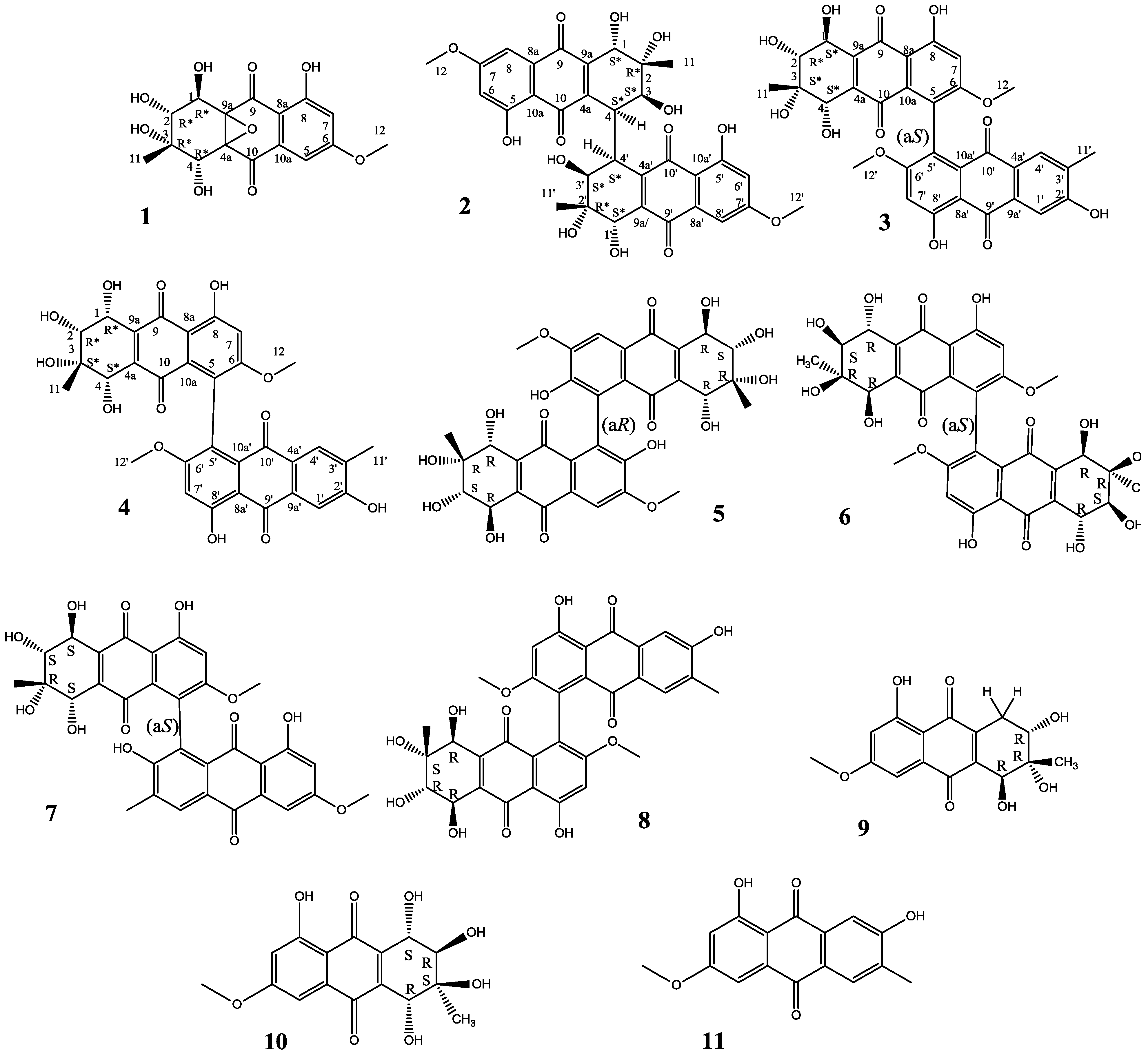
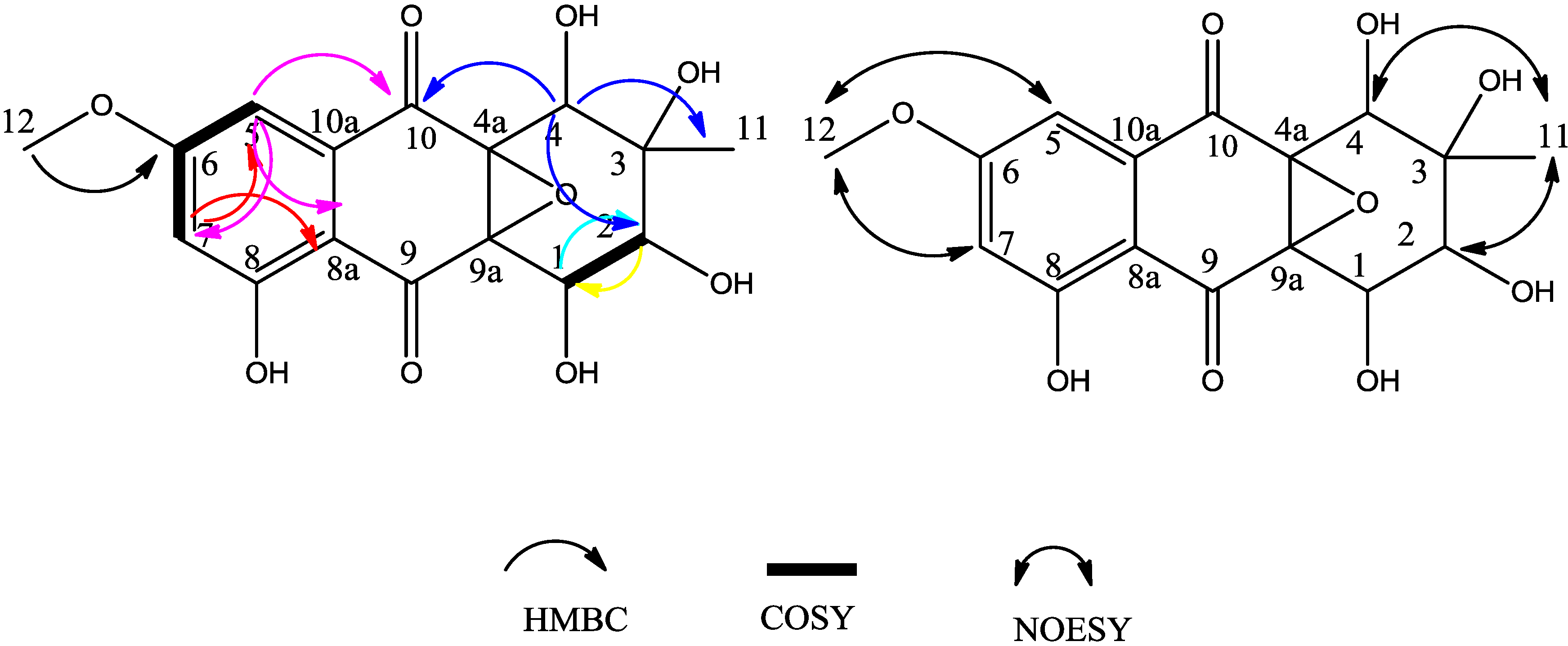
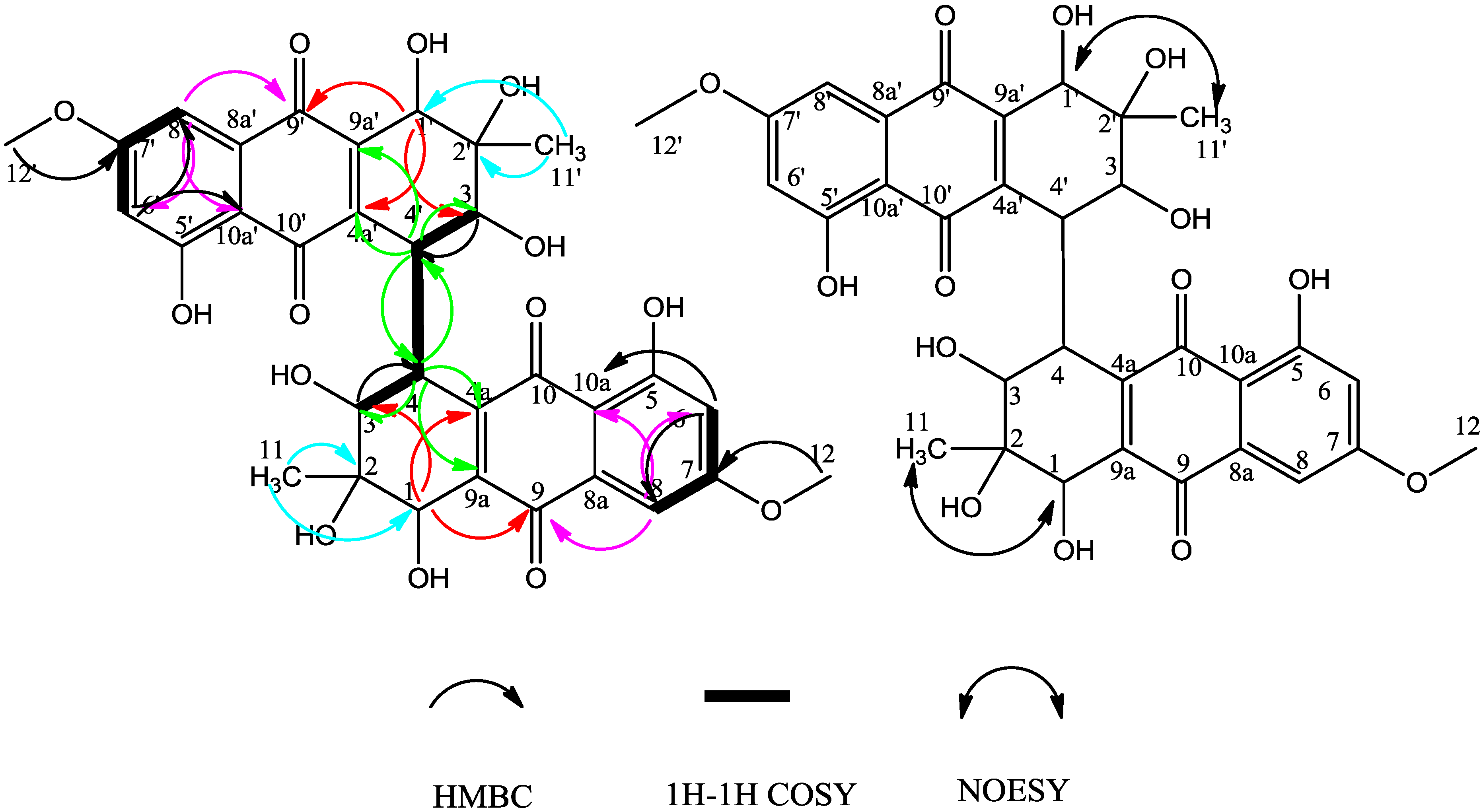
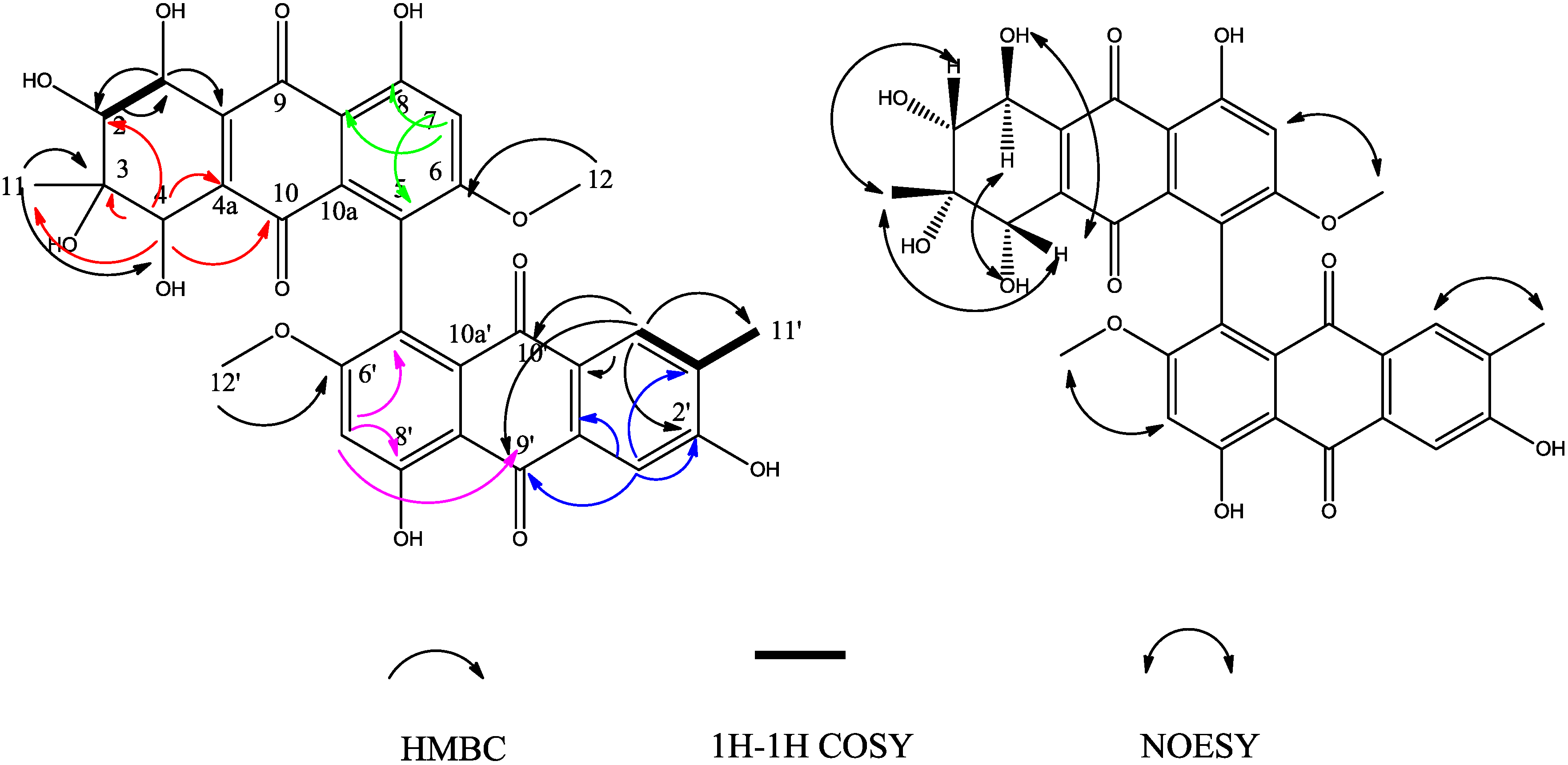
2. Results and Discussion
| Position | Compound 1 | Position | Compound 2 | ||||
|---|---|---|---|---|---|---|---|
| δC, (ppm) | δH (ppm) (mult., J in Hz) | HMBC | δC, (ppm) | δH (ppm) (mult., J in Hz) | HMBC | ||
| 1 | 67.28 | 4.42 d (8.1) | C-2 | 1,1' | 67.78 | 4.38 d (4.32) | C-2, 3, 4a, 9, 9a, 11 |
| 2 | 71.84 | 3.27 d (8.1) | C-1 | 2,2' | 71.69 | ||
| 3 | 74.14 | 3,3' | 69.59 | 3.77 br. d (7.8,4.8 overlap) | C-4 | ||
| 4 | 67.64 | 4.43 s | C-2, 3, 10, 11 | 4,4' | 42.7 | 3.82 dd (1.3, 4.8) | C-3, 4', 4a, 9a |
| 4a | 68.37 | 4a,4a' | 149.43 | ||||
| 5 | 106.19 | 6.91 d (2.48) | C-6, 7, 8a, 10 | 5,5' | 163.04 | ||
| 6 | 165.49 | 6,6' | 105.49 | 6.80 d (2.5) | C-5, 7, 8, 10a | ||
| 7 | 106.98 | 6.81 d (2.48) | C-5, 6, 8, 8a | 7,7' | 164.89 | ||
| 8 | 162.11 | 8,8' | 105.63 | 7.05 d (2.5) | C-6, 7, 9, 10a | ||
| 8a | 110.0 | 8a,8a' | 133.69 | ||||
| 9 | 193.61 | 9,9' | 183.08 | ||||
| 9a | 67.16 | 9a,9a' | 140.62 | ||||
| 10 | 191.12 | 10,10' | 188.59 | ||||
| 10a | 134.46 | 10a,10a' | 109.91 | ||||
| 11 | 21.78 | 1.12 s | C-2, 3, 4 | 11,11' | 22.22 | 1.13 s | C-1, 2, 3 |
| 12 | 56.43 | 3.86 s | C-6 | 12,12' | 56.08 | 3.91 s | C-7 |
| 1-OH | 1,1'-OH | 5.12 d (4.32) | C-1, 2, 9a | ||||
| 2-OH | 2,2'-OH | 4.14 s | C-1 | ||||
| 3-OH | 3,3'-OH | 4.09 d (7.8) | C-3, 4 | ||||
| 4-OH | 5,5'-OH | 12.26 s | C-5, 6, 10a | ||||
| 8-OH | 11.16 s | ||||||
| Position | Compound 3 | Compound 4 | ||||||
|---|---|---|---|---|---|---|---|---|
| δC, (ppm) | δH (ppm) (mult., J in Hz) | HMBC | NOE | δC, (ppm) | δH (ppm) (mult., J in Hz) | HMBC | NOE | |
| 1 | 68.39 | 4.47 dd (5.78, 7.06) | C-2, 9a | H-4-OH | 68.45 | 4.48 dd (5.60, 6.87) | C-2, 4a, 9a, 9 | H-4 |
| 2 | 73.78 | 3.57 dd (7.06, 7.06) | C-1 | H-11 | 73.74 | 3.55 dd (6.87, 6.87) | C-1 | H-11 |
| 3 | 72.83 | 72.87 | ||||||
| 4 | 68.22 | 4.03 d (6.93) | C-2, 3, 4a, 10, 11 | H-11,1-OH | 68.27 | 4.05 d (6.77) | C-2, 9a, 10, 4a, 11 | H-1, 11 |
| 4a | 143.31 | 143.42 | ||||||
| 5 | 122.86 | 122.59 | ||||||
| 6 | 164.28 | 164.33 | ||||||
| 7 | 103.77 | 6.93 s | C-5, 8, 8a | 103.77 | 6.92 s | C-6, 8, 8a, 9 | H-12' | |
| 8 | 163.63 | 163.73 | ||||||
| 8a | 109.27 | 109.3 | ||||||
| 9 | 188.77 | 188.79 | ||||||
| 9a | 142.78 | 142.61 | ||||||
| 10 | 184.09 | 183.88 | ||||||
| 10a | 128.86 | 129.02 | ||||||
| 11 | 22.18 | 1.13 s | C-3, 4 | H-2,4,1-OH | 22.22 | 1.13 s | C-3, 4 | H-2, 4 |
| 12 | 56.74 | 3.70 s | C-6 | H-7 | 56.71 | 3.69 s | C-6 | H-7 |
| 1-OH | 5.64 d (7.06) | C-1, 3, 9a | H-4 | 4.98 d (5.60) | C-1, 2, 9a | H-4-OH | ||
| 2-OH | 4.36 s | C-2, 4 | 4.81 d (6.87) | C-1, 2, 3 | H-4-OH | |||
| 3-OH | 4.85 d (6.93) | C-1 | 4.38 s | C-2, 4, 11 | H-1-OH | |||
| 4-OH | 5.04 d (5.78) | C-2, 4, 4a | H-1 | 5.44 d (6.77) | C-3, 4, 4a | H- 2-OH | ||
| 8-OH | 13.04 s | C-7, 8, 8a | 13.04 s | C-7, 8, 8a | ||||
| 1' | 110.44 | 7.56 s | C- 2', 3', 9', 9a' | 110.42 | 7.55 s | C-2', 3', 9', 9a', 10' | H-2'-OH | |
| 2' | 161.19 | 161.24 | ||||||
| 3' | 125.25 | 125.27 | ||||||
| 4' | 130.18 | 7.70 d (0.56) | C-3', 4a', 10', 11' | H-11' | 130.33 | 7.70 d (0.69) | C-2', 4a', 9', 10', 11' | H-11' |
| 4a' | 132.39 | 132.38 | ||||||
| 5' | 121.81 | 123.12 | ||||||
| 6' | 164.18 | 165.25 | ||||||
| 7' | 104.01 | 6.94 s | C- 5', 8', 8a', 9' | 103.54 | 6.94 s | C- 6, 8', 8a', 9' | H-8'-OH | |
| 8' | 164.85 | 165.23 | ||||||
| 8a' | 109.98 | 109.92 | ||||||
| 9' | 186.7 | 186.61 | ||||||
| 9a' | 132.2 | 132.2 | ||||||
| 10' | 181.1 | 180.97 | ||||||
| 10a' | 131.48 | 130.43 | ||||||
| 11' | 16.03 | 2.20 s | C-2',4' | H-4' | 16.09 | 2.19 s | C-2', 3', 4' | H-4' |
| 12' | 56.65 | 3.68 s | C-6' | H-7' | 56.83 | 3.72 s | C-6’ | H-7' |
| 2'-OH | 11.07 br. s | 11.03 br. s | H-1' | |||||
| 8'-OH | 13.60 s | C-7', 8', 8a' | 13.66 s | C-7', 8', 8a' | H-7' | |||
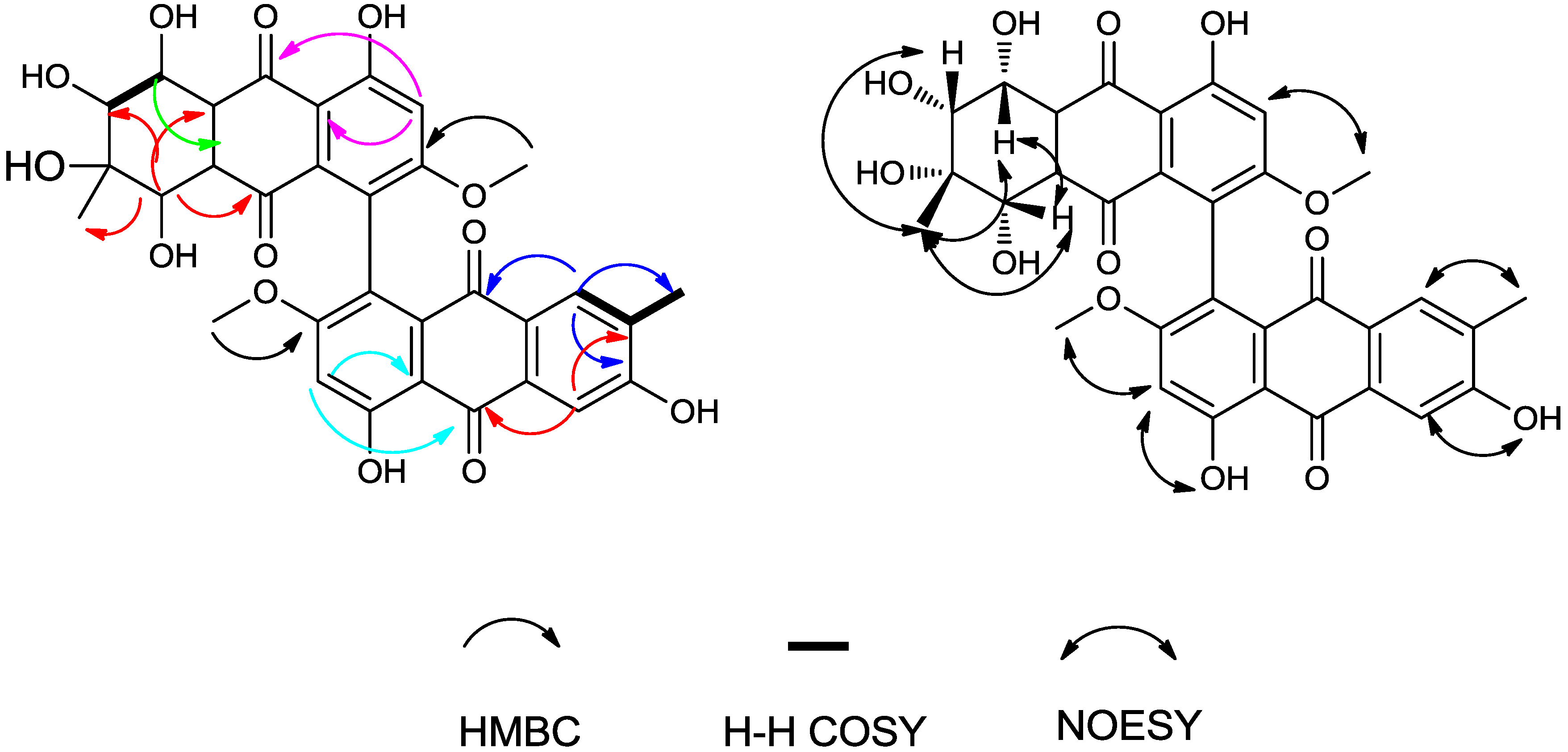
| Compound | IC50 (μM) | ||
|---|---|---|---|
| MCF-7/ADR | HCT-116 | HeLa | |
| 1 | 18.48 | 23.24 ± 2.85 | 43.74 ± 4.02 |
| 2 | >100 | >50 | >50 |
| 3 | ND | 32.38 ± 1.57 | >50 |
| 4 | >50 | >50 | >100 |
| 5 | >100 | >100 | >100 |
| 6 | >100 | >100 | >100 |
| 7 | >100 | >100 | >100 |
| 8 | 44.99 | >50 | >50 |
| 9 | >100 | 3.03 ± 0.05 | 8.09 ± 0.89 |
| 10 | >100 | >100 | >100 |
| 11 | >100 | >100 | >100 |
| EPI | 2.36 | 0.96 ± 0.02 | 0.48 ± 0.03 |
| Comp. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | Genistein * |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IC50 (μM) | >100 | 72.0 | 7.2 | >100 | >400 | >100 | >100 | >100 | >100 | >100 | 83.5 | 13.6 |
3. Experimental Section
3.1. General Procedures
3.2. Fungal Material
3.3. Identification of Fungal Cultures
3.4. Fermentation, Extraction, and Isolation of Alternaria sp. XZSBG-1
3.5. Biological Assays
3.5.1. Antitumor Activity in Vitro
Cell Culture
Assessment of Antitumor Activity by MTT Assay
3.5.2. Enzyme Assays
4. Conclusions
Supplementary Materials
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lin, Y.C.; Wu, X.Y.; Deng, Z.J.; Wang, J.; Zhou, S.N.; Vrijmoed, L.L.P.; Jones, E.B.G. The metabolites of the mangrove fungus Verruculina enalia No. 2606 from a salt lake in the Bahamas. Phytochemistry 2002, 59, 469–471. [Google Scholar]
- Chen, W.Y.; Wyk, V.B.-E.; Vermaak, I.; Viljoen, M.A. Cape aloes—A review of the phytochemistry, pharmacology and commercialsation of Aloe ferox. Phytochem. Lett. 2012, 5, 1–12. [Google Scholar] [CrossRef]
- Shukla, V.; Joshi, G.P.; Rawat, M.S.M. Lichens as a potential natural source of bioactive compounds: A review. Pytochem. Rev. 2010, 9, 303–314. [Google Scholar] [CrossRef]
- Mishra, B.B.; Singh, D.D.; Kishore, N.; Tiwari, V.K.; Tripathi, V. Antifungal constituents isolated from the seeds of Aegle marmelos. Phytochemistry 2010, 71, 230–234. [Google Scholar] [CrossRef]
- Marcello, L. Anthraquinones: Analytical techniques as a novel tool to investigate on the triggering of biological targets. Curr. Drug Targets 2011, 12, 366–380. [Google Scholar] [CrossRef]
- Suemitsu, R.; Horiuchi, K.; Kubota, M.; Okamatsu, T. Production of alterporriols, altersolanols and macrosporin by Alternaria porri and A. Solani. Phytochemistry 1990, 29, 1509–1511. [Google Scholar] [CrossRef]
- Ohnishi, K.; Suemitsu, R.; Kubota, M.; Matano, H.; Yamada, Y. Biosyntheses of alterporriol D and E by Alternaria porri. Phytochemistry 1991, 30, 2593–2595. [Google Scholar] [CrossRef]
- Phuwapraisirisa, P.; Rangsan, J.; Siripong, P.; Tip-pyang, S. New antitumour fungal metabolites from Alternaria porri. Nat. Prod. Res. 2009, 23, 1063–1071. [Google Scholar] [CrossRef]
- Debbab, A.; Aly, A.H.; Edrada-Ebel, R.A.; Wray, V.; Müller, W.E.G.; Totzke, F.; Zirrgiebel, U.; Schächtele, C.; Kubbutat, M.H.G.; Lin, W.H.; et al. Bioactive metabolites from the endophytic fungus Stemphylium globuliferum isolated from Mentha pulegium. J. Nat. Prod. 2009, 72, 626–631. [Google Scholar] [CrossRef]
- Debbab, A.; Aly, A.H.; Edrada-Ebel, R.A.; Wray, V.; Pretsch, A.; Pescitelli, G.; Kurtan, T.; Proksch, P. New anthracene derivatives- structure elucidation and antimicrobial activity. Eur. J. Org. Chem. 2012, 2012, 1351–1359. [Google Scholar] [CrossRef]
- Huang, C.H.; Pan, J.H.; Chen, B.; Yu, M.; Huang, H.B.; Zhu, X.; Lu, Y.J.; She, Z.G.; Lin, Y.C. Three bianthraquinone derivatives from the mangrove endophytic fungus Alternaria sp. ZJ9–6B from the South China Sea. Mar. Drugs 2011, 9, 832–843. [Google Scholar] [CrossRef]
- Zheng, C.J.; Shao, C.L.; Guo, Z.Y.; Chen, J.F.; Deng, D.S.; Yang, K.L.; Chen, Y.Y.; Fu, X.M.; She, Z.G.; Lin, Y.C.; et al. Bioactive hydroanthraquinones and anthraquinones dimmers from a soft coral-derived Alternaria sp. fungus. J. Nat. Prod. 2012, 75, 189–197. [Google Scholar] [CrossRef]
- Okamura, N.; Haraguchi, H.; Hashimoto, K.; Yagi, A. Altersolanol-related antimicrobial compounds from a strain of Alternaria solani. Phytochemistry 1993, 34, 1005–1009. [Google Scholar] [CrossRef]
- Okamura, N.; Yagi, A.; Haraguchi, H.; Hashimoto, K. Simultaneous high-performance liquid chromatographic determination of altersolanol A, B, C, D, E and F. J. Chromatogr. A 1993, 630, 418–422. [Google Scholar] [CrossRef]
- Höller, U.; Gloer, J.B.; Wicklow, D.T. Biologically active polyketide metabolites from an undetermined fungicolous Hyphomycete resembling Clasdosporium. J. Nat. Prod. 2002, 65, 876–882. [Google Scholar] [CrossRef]
- Kanamaru, S.; Honma, M.; Murakami, T.; Tsushima, T.; Kudo, S.; Tanaka, K.; Nihei, K.; Nehira, T.; Hashimoto, M. Absolute stereochemistry of altersolanol A and alterporriols. Chirality 2012, 24, 137–146. [Google Scholar] [CrossRef]
- Huang, C.H.; Jin, H.; Song, B.; Zhu, X.; Zhao, H.X.; Cai, J.J.; Lu, Y.J.; Chen, B.; Lin, Y.C. The cytotoxicity and anticancer mechanisms of alterporriol L, a marine bianthraquinone, agaist MCF-7 human breast cancer cells. Appl. Microbiol. Biotechnol. 2012, 93, 777–785. [Google Scholar] [CrossRef]
- Chen, H.; Zhong, L.L.; Long, Y.H.; Li, J.; Wu, J.H.; Liu, L.; Chen, S.P.; Lin, Y.C.; Li, M.F.; Zhu, X.; et al. Studies on the synthesis of derivatives of marine-derived bostrycin and their structure-activity relationship against tumor cells. Mar. Drugs 2012, 10, 932–952. [Google Scholar] [CrossRef]
- Du, Z.Y.; Liu, R.R.; Shao, W.Y.; Mao, X.P.; Ma, L.; Gu, L.Q.; Huang, Z.S.; Chan, A.S.C. Alpha-glucosidase inhibition of natural curcuminoids and curcumin analogs. Eur. J. Med. Chem. 2006, 41, 213–218. [Google Scholar] [CrossRef]
- Stewart, C.N., Jr.; Via, L.E. A rapid CTAB DNA isolation technique useful for RAPD fingerprinting and other PCR applications. Biotechniques 1993, 14, 748–749. [Google Scholar]
- Sample Availability: Sample of the compound 4 is available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, B.; Shen, Q.; Zhu, X.; Lin, Y. The Anthraquinone Derivatives from the Fungus Alternaria sp. XZSBG-1 from the Saline Lake in Bange, Tibet, China. Molecules 2014, 19, 16529-16542. https://doi.org/10.3390/molecules191016529
Chen B, Shen Q, Zhu X, Lin Y. The Anthraquinone Derivatives from the Fungus Alternaria sp. XZSBG-1 from the Saline Lake in Bange, Tibet, China. Molecules. 2014; 19(10):16529-16542. https://doi.org/10.3390/molecules191016529
Chicago/Turabian StyleChen, Bin, Qiong Shen, Xun Zhu, and Yongcheng Lin. 2014. "The Anthraquinone Derivatives from the Fungus Alternaria sp. XZSBG-1 from the Saline Lake in Bange, Tibet, China" Molecules 19, no. 10: 16529-16542. https://doi.org/10.3390/molecules191016529




