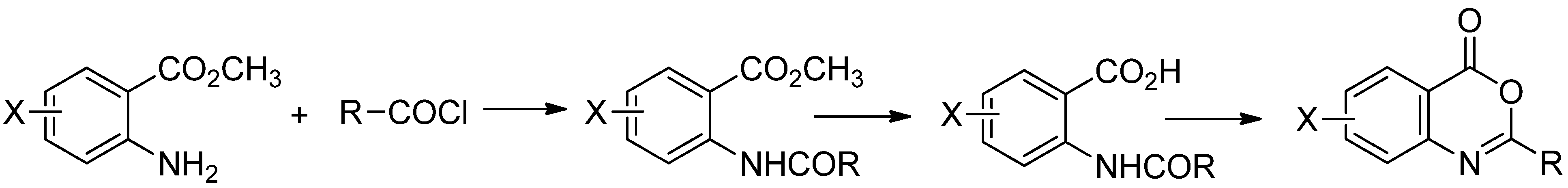
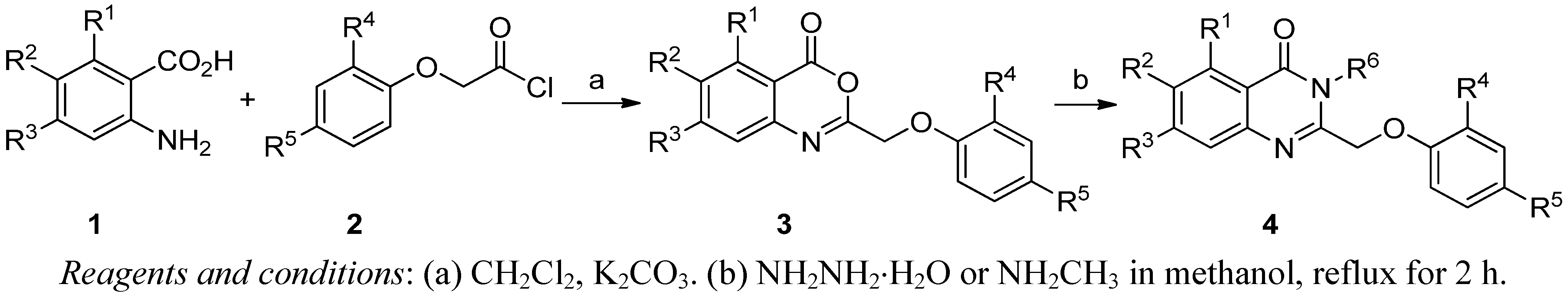
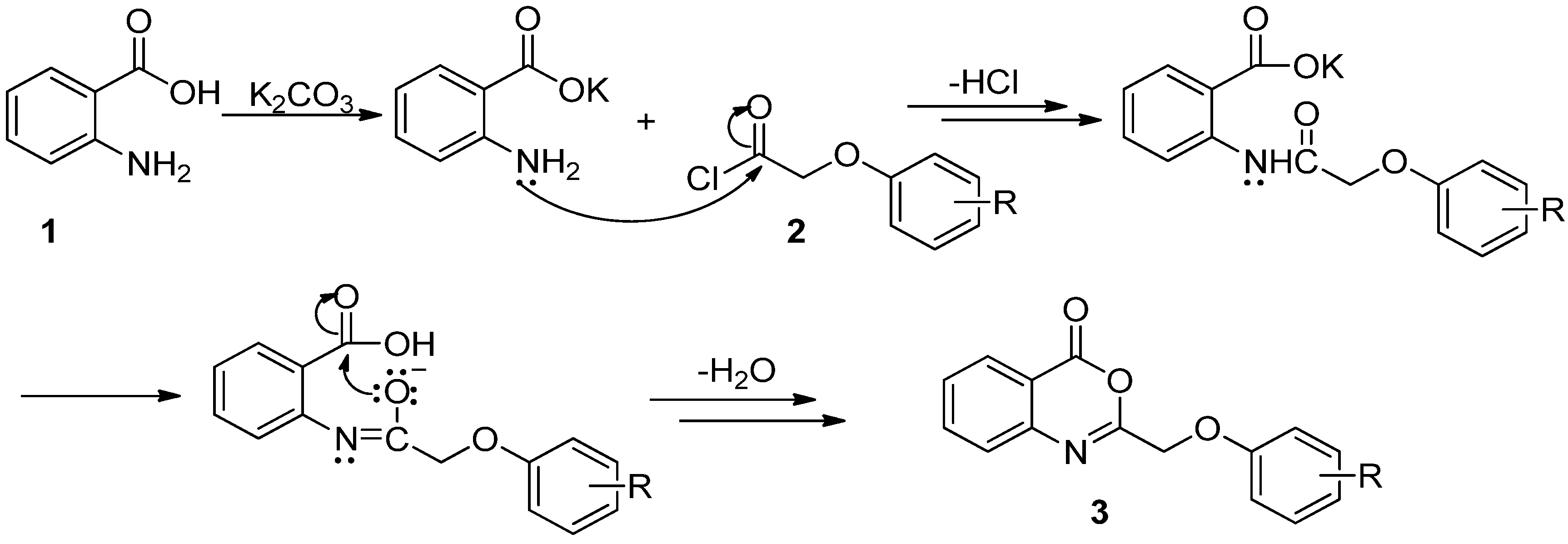
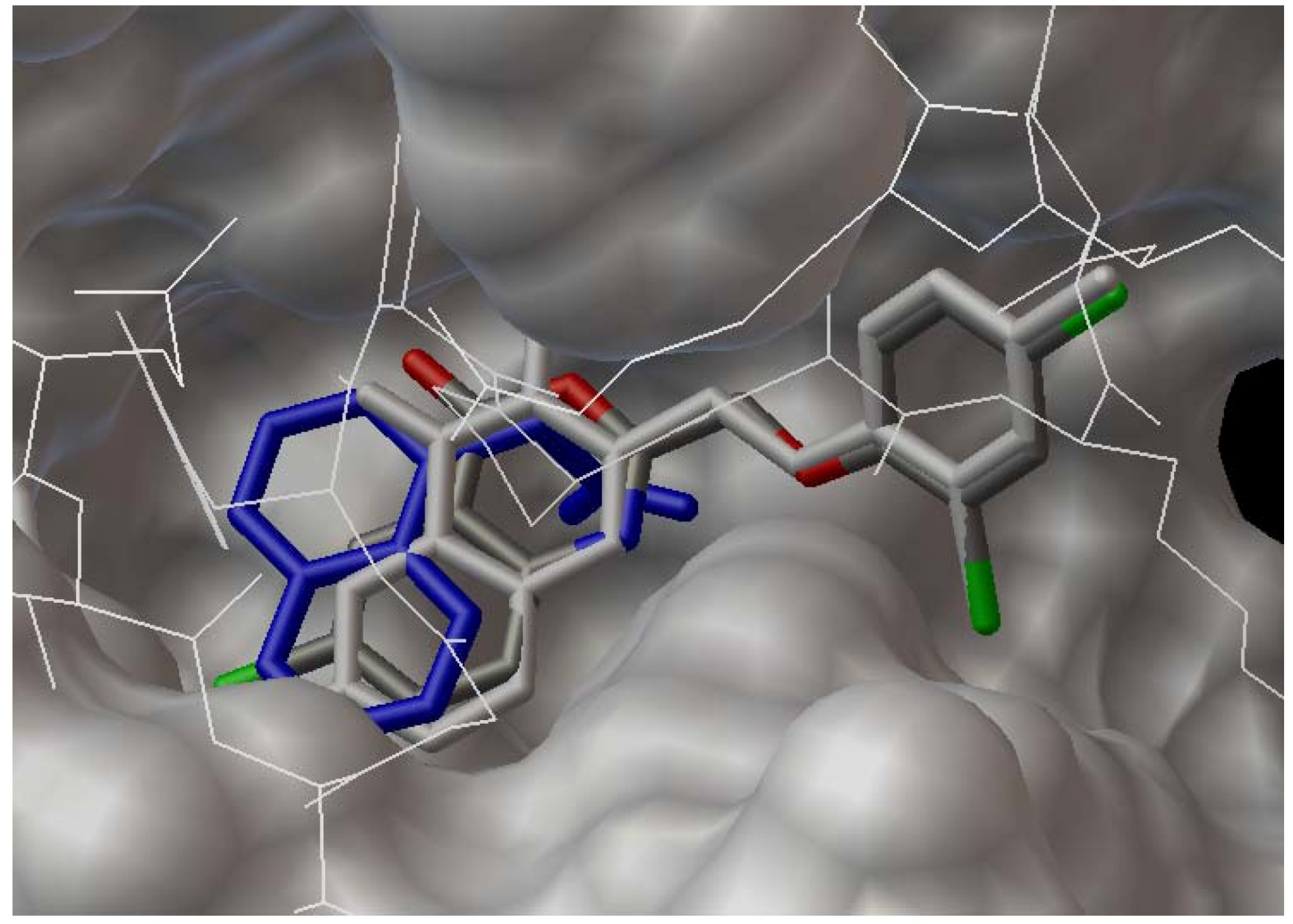
3.2. Synthesis
Step 1. Synthesis of phenoxyacetic chlorides. Phenoxyacetic chlorides were synthesized according to the reported method [
18]. Briefly, the mixture of phenoxyacetic acid (10 mmol) and thionyl chloride (6–10 mL) was reacted for 3 h under reflux until no further gaseous HCl was released. Excess thionyl chloride was distilled off under reduced pressure, giving the corresponding phenoxyacetic chlorides (~98% yield) as brown oils. The phenoxyacetic chlorides were used in the next step without further purification.
Step 2. General synthesis of 2-phenoxymethyl-4H-3,1-benzoxazin-4-ones 3a–
w. In a 250-mL flask, anthranilic acid (10 mmol) and anhydrous potassium carbonate (20 mmol) were mixed in dry CH
2Cl
2 (100 mL) under stirring in an ice bath, then phenoxyacetic chloride (15 mmol) in CH
2Cl
2 (10 mL) was added dropwise. After the addition, the solution was stirred for 2 h at room temperature. The mixture was filtered and washed with CH
2Cl
2. The filtrate was washed with saturated brine and the organic layer was dried with sodium sulfate. After evaporating the solvent the residue was chromatographed on silica gel using the mixed solvents of ether/petroleum ether (volumetric ratio: 1:9). Molecular structures, melting points and yields are summarized in
Table 4.
Table 4.
Molecular structures, yields, and melting points of synthesized 2-phenoxy-4H-3,1-benzoxazin-4-ones 3.
Table 4.
Molecular structures, yields, and melting points of synthesized 2-phenoxy-4H-3,1-benzoxazin-4-ones 3.
| No. | R1 | R2 | R3 | R4 | R5 | Appearance | Mp/°C | Yield/% |
|---|
| 3a | H | H | H | H | H | pale yellow solid | 87.9~92.1 | 85.6 |
| 3b | Cl | H | H | H | H | white solid | 118.3~120.9 | 87.1 |
| 3c | H | Cl | H | H | H | pale yellow solid | 113.1~115.2 | 79.8 |
| 3d | H | H | Cl | H | H | pale yellow solid | 117.3~119.9 | 87.1 |
| 3e | H | CH3 | H | H | H | pink solid | 133.3~135.1 | 85.2 |
| 3f | H | OCH3 | OCH3 | H | H | pale yellow solid | 132.1~134.3 | 76.1 |
| 3g | H | H | H | H | Cl | white solid | 147.5~149.2 | 89.1 |
| 3h | Cl | H | H | H | Cl | white solid | 144.3~145.7 | 80.1 |
| 3i | H | Cl | H | H | Cl | pale yellow solid | 153.2~154.6 | 79.5 |
| 3j | H | H | Cl | H | Cl | pale yellow solid | 189.9~191.8 | 83.3 |
| 3k | H | CH3 | H | H | Cl | pale yellow solid | 143.7~145.6 | 92.3 |
| 3l | H | OCH3 | OCH3 | H | Cl | white solid | 118.5~119.3 | 90.8 |
| 3m | H | H | H | Cl | Cl | white solid | 118.5~119.3 | 73.5 |
| 3n | Cl | H | H | Cl | Cl | white solid | 161.9~163.2 | 81.1 |
| 3o | H | Cl | H | Cl | Cl | white solid | 157.9~160.1 | 78.6 |
| 3p | H | H | Cl | Cl | Cl | white solid | 139.6~142.5 | 88.1 |
| 3q | H | OCH3 | OCH3 | Cl | Cl | white solid | 161.9~163.2 | 90.2 |
| 3r | H | H | H | H | F | white solid | 112.5~114.6 | 92.2 |
| 3s | Cl | H | H | H | F | white solid | 155.9~156.7 | 77.3 |
| 3t | H | Cl | H | H | F | white solid | 159.8~161.7 | 87.1 |
| 3u | H | H | Cl | H | F | white solid | 123.6~124.9 | 89.9 |
| 3v | H | CH3 | H | H | F | white solid | 130.8~131.9 | 91.4 |
| 3w | H | OCH3 | OCH3 | H | F | white solid | 144.7~146 | 96.2 |
2-(Phenoxymethyl)-4H-3,1-benzoxazin-4-one (3a). 1H-NMR (400 MHz, CDCl3): δ 5.018 (s, 2H, -CH2O-), 7.028 (d, J = 17.2 Hz, 3H, Ar-H), 7.303~7.341 (m, 2H, Ar-H), 7.550~7.587 (m, 1H, Ar-H), 7.668 (d, J = 8.4 Hz, 1H, Ar-H), 7.819~7.857 (m, 1H, Ar-H), 8.227 (d, J = 8 Hz, 1H, Ar-H). IR (KBr) ν: 3020,1763, 1695, 1606, 1496, 1243, 760 cm−1. EI-MS m/z (%): 253.04 (M+, 44.47), 77.03 (53.73), 132.05 (61.88), 253.04 (44.74), 145.97 (100.00).
5-Chloro-2-(phenoxymethyl)-4H-3,1-benzoxazin-4-one (3b). 1H-NMR (400 MHz, CDCl3): δ 4.99 (s, 2H, -CH2O-), 7.22 (d, J = 6.8 Hz, 3H, Ar-H), 7.303~7.321 (m, 2H, Ar-H), 7.571 (d, J = 4 Hz, 2H, Ar-H), 7.696 (d, J = 4 Hz, 1H, Ar-H). IR (KBr) ν: 3023, 1751, 1659, 1587, 1492, 1230, 787 cm−1. EI-MS m/z (%): 286.92 (M+, 23.63), 77.09 (27.30), 181.97 (32.70), 179.91 (100.00).
6-Chloro-2-(phenoxymethyl)-4H-3,1-benzoxazin-4-one (3c). 1H-NMR (400 MHz, CDCl3): δ 5.011 (s, 2H, -CH2O-), 7.027 (d, J = 8 Hz, 3H, Ar-H), 7.306~7.344 (m, 2H, Ar-H), 7.411 (d, J = 4.1 Hz, 1H, Ar-H), 7.632 (d, J = 4 Hz, 1H, Ar-H), 8.188 (d, J = 3.6 Hz, 1H, Ar-H). IR (KBr) ν: 3009, 1780, 1671, 1516, 1232, 748 cm−1. EI-MS m/z (%): 28.99 (M+, 39.33), 77.08 (37.04), 179.97 (39.62), 166.01 (100.00).
7-Chloro-2-(phenoxymethyl)-4H-3,1-benzoxazin-4-one (3d). 1H-NMR (400 MHz, CDCl3): δ 5.010 (s, 2H, -CH2O-), 7.021 (d, J = 6.4 Hz, 3H, Ar-H), 7.303~7.342 (m, 2H, Ar-H), 7.516 (d, J = 8.8 Hz, 1H, Ar-H), 7.660 (s, 1H, Ar-H), 8.146 (d, J = 8.4 Hz, 1H, Ar-H). IR (KBr) ν: 2924, 1673, 1602, 1515, 1241, 850 cm−1. EI-MS m/z (%): 286.94 (M+, 32.76), 77.05 (32.95), 123.96 (28.54), 179.93 (100.00).
6-Methyl-2-(phenoxymethyl)-4H-3,1-benzoxazin-4-one (3e). 1H-NMR (400 MHz, CDCl3): δ 2.391 (s, 3H, -CH3), 5.000 (s, 2H, -CH2O-), 7.035 (d, J = 8.4 Hz, 3H, Ar-H), 7.296~7.335 (m, 2H, Ar-H), 7.560 (d, J = 8 Hz, 1H, Ar-H), 7.629 (s, 1H, Ar-H), 8.016 (s, 1H, Ar-H). IR (KBr) ν: 3021, 2947, 1751, 1657, 1585, 1492, 1206, 787 cm−1. EI-MS m/z (%): 267.01 (M+, 34.95), 77.04 (41.69), 160.03 (41.17), 146.00 (100.00).
6,7-Dimethoxy-2-(phenoxymethyl)-4H-3,1-benzoxazin-4-one (3f). 1H-NMR (400 MHz, CDCl3): δ 3.995 (d, J = 3.2 Hz, 6H, -OCH3), 5.000 (s, 2H, -CH2O-), 7.036 (d, J = 8 Hz, 3H, Ar-H), 7.089 (s, 1H, Ar-H), 7.342~7.271 (m, 2H, Ar-H), 7.531 (s, 1H, Ar-H). IR (KBr) ν: 2947, 2873, 1751, 1659, 1587, 1492, 1222, 755 cm−1. EI-MS m/z (%): 313.11 (M+, 25.99), 220.07 (41.27), 192.12 (100.00).
2-((4-Chlorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3g). 1H-NMR (400 MHz, CDCl3): δ 4.986 (s, 2H, -OCH2-), 6.973 (d, J = 9.6 Hz, 2H, Ar-H), 7.264 (d, J = 16 Hz, 2H, Ar-H), 7.577 (d, J = 16.8 Hz, 1H, Ar-H), 7.645~7.664 (m, 1H, Ar-H), 7.823~8.65 (m, 1H, Ar-H), 8.228 (d, J = 9.2 Hz, 1H, Ar-H). IR (KBr) ν: 2932, 1671, 1582, 1514, 1494, 1293, 753 cm−1. EI-MS m/z (%): 286.97 (M+, 32.55), 77.05 (34.92), 90.10( 38.11), 132.05 (95.12), 146.02 (100.00).
5-Chloro-2-((4-chlorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3h). 1H-NMR (400 MHz, CDCl3): δ 4.591 (s, 2H, -CH2O-), 6.953 (d, 8.8 Hz, 2H, Ar-H), 7.216 (d, J = 8.4 Hz, 1H, Ar-H), 7.296~7.389 (m, 3H, Ar-H), 8.330 (s, 1H, Ar-H). 13C-NMR (CDCl3) δ: 68.49, 116.59, 119.04, 125.55, 126.99, 129.11, 129.44, 131.64, 136.38, 142.11, 156.22, 166.19, 170.38. IR (KBr) ν: 2952, 1673, 1580, 1515, 1495, 1241, 749 cm−1. EI-MS m/z (%): 321.82 (M+, 5.60), 166.05 (55.61), 240.07 (25.21), 180.03 (100.00).
6-Chloro-2-((4-chlorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3i). 1H-NMR (400 MHz, CDCl3): δ 4.976 (s, 2H, -OCH2-), 6.96 (d, J = 8.8 Hz, 2H, Ar-H), 7.273 (d, J = 8.4 Hz, 2H, Ar-H), 7.603 (d, J = 8.4 Hz, 1H, Ar-H), 7.777 (d, J = 10.8 Hz, 1H, Ar-H), 8.182~8.188 (s, 1H, Ar-H). IR (KBr) ν: 2987, 1697, 1582, 1520, 1493, 1242, 755 cm−1. EI-MS m/z (%): 320.90 (M+, 25.82), 110.97 (27.73), 179.94 (22.04), 165.95 (100.00).
7-Chloro-2-((4-chlorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3j). 1H-NMR (400 MHz, CDCl3): δ 4.980 (s, 2H, -CH2O-), 6.953 (d, 8.8 Hz, 2H, Ar-H), 6.957 (d, J = 8.8 Hz, 2H, Ar-H), 7.261 (d, J = 17.2 Hz, 2H, Ar-H), 7.422 (d, J = 5.6 Hz, 2H, Ar-H), 7.651 (s, 1H, Ar-H), 8.112 (d, J = 4.4 Hz, 2H, Ar-H). 13C-NMR (CDCl3) δ: 67.40, 104.22, 114.94, 116.54, 116.69, 123.80, 125.47, 129.26, 129.39, 132.96, 138.63, 141.08, 155.83, 159.06, 167.29. IR (KBr) ν: 2933, 1760, 1665, 1518, 1445, 1280, 817 cm−1. EI-MS m/z (%): 320.98 (M+, 32.11), 123.99 (28.18), 166.00 (58.01), 179.89 (100.00).
2-((4-Chlorophenoxy)methyl)-6-methyl-4H-3,1-benzoxazin-4-one (3k). 1H-NMR (400 MHz, CDCl3): δ 2.380 (s, 3H, -CH3), 4.966 (s, 2H,-OCH2-), 6.966 (d, J = 8.4 Hz, 2H, Ar-H), 7.262 (d, J = 8.8 Hz, 2H, Ar-H), 7.543 (d, J = 6.8 Hz, 1H, Ar-H), 7.633 (d, J = 2.4 Hz, 1H, Ar-H), 8.017 (s, 1H, Ar-H). 13C-NMR (CDCl3) δ: 21.26, 66.74, 104.11, 116.29, 116.88, 126.91, 128.24, 129.48, 137.88, 139.66, 143.38, 148.23, 156.39, 158.77. IR (KBr) ν: 2973, 2945, 1749, 1662, 1618, 1492, 1247, 820 cm−1. EI-MS m/z (%): 301.04 (M+, 28.66), 128.14 (32.95), 171.08 (25.40), 146.06 (100.00).
2-((4-Chlorophenoxy)methyl)-6,7-dimethoxy-4H-3,1-benzoxazin-4-one (3l). 1H-NMR (400 MHz, CDCl3): δ 3.994 (d, J = 4 Hz, 6H, -OCH3), 4.967 (s, 2H, -OCH2-), 6.967 (d, J = 9.2 Hz, 2H, Ar-H), 7.072 (s, 1H, Ar-H), 7.279 (d, J = 4 Hz, 2H, Ar-H), 7.528 (s, 1H, Ar-H). 13C-NMR (CDCl3) δ: 56.43, 56.52, 66.67, 107.55, 108.15, 109.72, 116.29, 126.94, 129.51, 141.91, 150.13, 156.53, 158.56. IR (KBr) ν: 2972, 2847, 1749, 1661, 1615, 1492, 1242, 833 cm−1. EI-MS m/z (%): 346.95 (M+, 10.22), 220.01 (46.59), 192.01 (100.00).
2-((2,4-Dichlorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3m). 1H-NMR (400 MHz, CDCl3): δ 5.055 (s, 2H, -OCH2-), 7.012 (d, J = 8.8 Hz, 1H, Ar-H), 7.187 (d, J = 10 Hz, 1H, Ar-H), 7.419 (s, 1H, Ar-H), 7.590 (d, J = 7.2 Hz, 1H, Ar-H), 7.639~7.657 (m, 1H, Ar-H), 7.825~7.846 (m, 1H, Ar-H), 8.231~8.240 (s, J = 7.2 Hz, 1H, Ar-H). 13C-NMR (CDCl3) δ: 67.95, 104.67, 15.72, 124.71, 127.25, 127.69, 128.72, 129.23, 130.42, 136.77, 142.12, 139.81, 152.49, 156.71. IR (KBr) ν: 3039, 1773, 1654, 1591, 1477, 1237, 773 cm−1. EI-MS m/z (%): 321 (M+, 100.00), 119.08 (38.56), 132.04 (42.89), 160.98 (67.85).
5-Chloro-2-((2,4-dichlorophenoxy)methyl)-4H-benzo[3,1]oxazin-4-one (3n). 1H-NMR (400 MHz, CDCl3): δ 5.055 (s, 2H, -OCH2-), 7.012 (d, J = 8.8 Hz, 1H, Ar-H), 7.187 (d, J = 10 Hz, 1H, Ar-H), 7.419 (s, 1H, Ar-H), 7.590 (d, J = 7.2 Hz, 1H, Ar-H), 7.639~7.657 (m, 1H, Ar-H), 7.825~7.846 (m, 1H, Ar-H), 8.231~8.240 (s, J = 7.2 Hz, 1H, Ar-H). IR (KBr) ν: 2924, 1752, 1659, 1579, 1477, 1243, 819 cm−1. EI-MS m/z (%): 355.86 (M+, 100.00), 152.98 (47.48), 166.02 (67.72), 179.96 (48.40).
6-Chloro-2-((2,4-dichlorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3o). 1H-NMR (400 MHz, CDCl3): δ 5.043 (s, 2H, -OCH2-), 6.984 (d, J = 8.4 Hz, 1H, Ar-H), 7.175 (d, J = 2.4 Hz,1H, Ar-H), 7.422 (s, 1H, Ar-H), 7.597 (d, J = 8.4 Hz, 1H, Ar-H), 7.764~7.791 (m, 1H, Ar-H), 8.182~8.188 (s, 1H, Ar-H). 13C-NMR (CDCl3) δ: 67.67, 118.19, 118.33, 122.47, 124.59, 127.67, 128.04, 130.41, 134.97, 137.04, 143.91, 152.29, 156.87, 157.38. IR (KBr) ν: 3068 cm−1, 1781 cm−1, 1689 cm−1, 1474 cm−1, 1283 cm−1, 794 cm−1. EI-MS m/z(%): 354.96 (M+, 9.17), 168.1 (37.70), 319.91 (46.05), 166.08 (100.00).
7-Chloro-2-((2,4-dichlorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3p). 1H-NMR (400 MHz, CDCl3): δ 5.047 (s, 2H, -OCH2-), 6.982 (d, J = 6 Hz, 1H, Ar-H), 7.175~7.258 (m, 2H, Ar-H), 7.424 (s, 1H, Ar-H), 7.539 (d, J = 8.4 Hz, 1H, Ar-H), 7.645 (s, 1H, Ar-H), 8.153 (d, J = 8.4 Hz, 1H, Ar-H). IR (KBr) ν: 2925, 1736, 1660, 1579, 1492, 1245, 802 cm−1. EI-MS m/z (%): 355.96 (M+, 14.59), 179.98 (78.12), 319.87 (93.89), 166.05 (100.00).
2-((2,4-Dichlorophenoxy)methyl)-6,7-dimethoxy-4H-3,1-benzoxazin-4-one (3q). 1H-NMR (400 MHz, CDCl3): δ 3.999 (d, J = 5.2 Hz, 6H, -OCH3), 5.031 (s, 2H, -OCH2-), 7.003 (s, 1H, Ar-H), 7.055 (s, 1H, Ar-H), 7.415 (s, 1H, Ar-H), 7.421 (s, 1H, Ar-H), 7.528 (s, 1H, Ar-H). 13C-NMR (CDCl3) δ: 56.39, 56.50, 67.81, 107.49, 108.13, 109.69, 115.59, 124.33, 127.38, 127.63, 130.29, 141.79, 150.21, 152.46, 155.89, 156.41, 158.42. IR (KBr) ν: 2927, 2803, 1736, 1651, 1580, 1492, 1247, 803 cm−1. EI-MS m/z (%): 381.98 (M+, 89.40), 191.99 (91.64), 206.03 (78.72), 383.89 (51.06), 221.02 (100.00).
2-((4-Fluorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3r). 1H-NMR (400 MHz, CDCl3): δ 4.9796 (s, 2H, -OCH2-), 6.971 (d, J = 8.6 Hz, 2H, Ar-H), 7.156 (d, J = 8 Hz, 2H, Ar-H), 7.560~7.596 (m, 1H, Ar-H), 7.664 (d, J = 8.4 Hz, 1H, Ar-H), 7.825~7.845 (m, 1H, Ar-H), 8.229 (d, J = 8.8 Hz, 1H, Ar-H). IR (KBr) ν: 2923, 1757, 1693, 1505, 1453, 1273, 787 cm−1. EI-MS m/z (%): 271.02 (M+, 53.36), 90.11 (31.46), 145.98 (83.03), 132.06 (100.00).
5-Chloro-2-((4-fluorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3s). 1H-NMR (600 MHz, CDCl3): δ 4.945 (s, 2H, -CH2O-), 6.983 (d, J = 2.4 Hz, 2H, Ar-H), 6.995 (d, J = 8.4 Hz, 2H, Ar-H), 7.548~7.577 (m, 2H, Ar-H), 7.686 (s, 1H, Ar-H). IR (KBr) ν: 2923, 1756, 1694, 1583, 1505, 1272, 892 cm−1. EI-MS m/z (%): 305.02 (M+, 25.81), 124.04 (17.28), 166.00 (25.57), 181.99 (30.12), 179.95 (100.00).
6-Chloro-2-((4-fluorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3t). 1H-NMR (600 MHz, CDCl3): δ 4.963 (s, 2H, -OCH2-), 6.5 (d, J = 4.2 Hz, 2H, Ar-H), 7.010 (d, J = 9.6 Hz, 2H, Ar-H), 7.609 (d, J = 9 Hz, 1H, Ar-H), 7.872 (d, J = 9 Hz, 1H, Ar-H), 8.183 (s, 1H, Ar-H). IR (KBr) ν: 2923, 1747, 1660, 1606, 1504, 1211, 830 cm−1. EI-MS m/z (%): 305.00 (M+, 28.86), 124.05 (21.22), 168.05 (31.99), 166.01 (100.00).
7-Chloro-2-((4-fluorophenoxy)methyl)-4H-3,1-benzoxazin-4-one (3u). 1H-NMR (600 MHz, CDCl3): δ 4.964 (s, 2H, -CH2O-), 6.985 (d, J = 4.8 Hz, 2H, Ar-H), 6.997 (d, J = 7.2 Hz, 2H, Ar-H), 7.533 (d, J = 4.8 Hz, 1H, Ar-H), 7.656 (s, 1H, Ar-H), 8.147 (d, J = 8.4 Hz, 1H, Ar-H). IR (KBr) ν: 2923, 1671, 1661, 1509, 1437, 1223, 772 cm−1. EI-MS m/z (%): 305.00 (M+, 38.01), 124.03 (27.72), 166.03 (51.89), 305.00 (38.01), 180.00 (100.00).
2-((4-Fluorophenoxy)methyl)-6-methyl-4H-3,1-benzoxazin-4-one (3v). 1H-NMR (400 MHz, CDCl3): δ 2.492 (s, 3H, -CH3), 4.960 (s, 2H, -OCH2-), 6.986 (d, J = 1.2 Hz, 2H, Ar-H), 7.001 (d, J = 1.2 Hz, 2H, Ar-H), 7.556 (d, J = 7.6 Hz, 1H, Ar-H), 7.647 (d, J = 8.4 Hz, 1H, Ar-H), 8.017 (s, 1H, Ar-H). 13C-NMR (CDCl3) δ: 66.95, 115.51, 115.93, 116.12, 116.16, 116.20, 126.99, 129.61, 129.90, 143.17, 146.56, 153.75, 156.70, 157.78, 158.82, 159.09. IR (KBr) ν: 2961, 2923, 1671, 1509, 1437, 1223, 772 cm−1. EI-MS m/z (%): 285.02 (M+, 26.17), 104.08 (14.48), 160.06 (23.87), 146.01 (100.00).
2-((4-Fluorophenoxy)methyl)-6,7-dimethoxy-4H-3,1-benzoxazin-4-one (3w). 1H-NMR (400 MHz, CDCl3): δ 3.991 (d, J = 12 Hz, 6H, -OCH3), 4.963 (s, 2H, -OCH2-), 6.968 (d, J = 1.2 Hz, 2H, Ar-H), 7.005 (d, J = 4 Hz, 2H, Ar-H), 7.087 (s, 1H, Ar-H), 7.532 (s, 1H, Ar-H). IR (KBr) ν: 2920, 2806, 1670, 1611, 1507, 1433, 1222, 771 cm−1. EI-MS m/z (%): 331.03 (M+, 13.11), 220.04 (45.51), 191.99 (100.00).
Step 3. Synthesis of 2-phenoxymethyl–3H
-quinazolin-4-ones 4a–
s. 2-phenoxymethyl-3
H-quinazolin-4-ones
4a–s were synthesized according to the reported method [
19]. Briefly, 2-phenoxyacetic-4
H-3.1-benzoxazin-4-one (5 mmol) was heated with hydrazine hydrate (80% aqueous solution, 10 mL) or methylamine (33% methanol solution, 10 mL) in ethanol (5 mL) under reflux for 3 h. The clear and hot solution was filtered and then cooled down to room temperature. Crystals appeared and were collected to obtain the desired product with high purity. Molecular structures, melting points and yields are summarized in
Table 5.
Table 5.
Molecular structures, yields, and melting points of synthesized 2-phenoxy-3H-quinazolin-4-ones 4.
Table 5.
Molecular structures, yields, and melting points of synthesized 2-phenoxy-3H-quinazolin-4-ones 4.
| No. | R1 | R2 | R3 | R4 | R5 | R6 | Appearance | Mp/°C | Yield/% |
|---|
| 4a | Cl | H | H | H | Cl | NH2 | white solid | 191.8~193.3 | 70.1 |
| 4b | H | Cl | H | H | Cl | NH2 | white solid | 178.1~179.4 | 60.7 |
| 4c | H | H | Cl | H | Cl | NH2 | white solid | 129.5~131.6 | 59.1 |
| 4d | H | CH3 | H | H | Cl | NH2 | white solid | 177.3~178.6 | 69.2 |
| 4e | H | OCH3 | OCH3 | H | Cl | NH2 | white solid | 202.8~204.1 | 81.3 |
| 4f | H | H | H | Cl | Cl | NH2 | white solid | 214.6~215.7 | 80.5 |
| 4g | Cl | H | H | Cl | Cl | NH2 | white solid | 201.5~202 | 76.4 |
| 4h | H | Cl | H | Cl | Cl | NH2 | white solid | 223.6~224.9 | 90.5 |
| 4i | H | H | Cl | Cl | Cl | NH2 | white solid | 129.1~131.3 | 85.6 |
| 4j | H | OCH3 | OCH3 | Cl | Cl | NH2 | white solid | 272.3~273.1 | 89.3 |
| 4k | Cl | H | H | H | Cl | CH3 | white solid | 184.9~185.7 | 65.1 |
| 4l | H | Cl | H | H | Cl | CH3 | white solid | 179.9~181.2 | 71.1 |
| 4m | H | H | Cl | H | Cl | CH3 | white solid | 202.4~203.9 | 67.3 |
| 4n | H | CH3 | H | H | Cl | CH3 | white solid | 196.4~197.6 | 68.3 |
| 4o | H | OCH3 | OCH3 | H | Cl | CH3 | white solid | 201.8~202.7 | 70.9 |
| 4p | H | H | H | Cl | Cl | CH3 | white solid | 172.1~173.4 | 81.7 |
| 4q | H | Cl | H | Cl | Cl | CH3 | white solid | 240.1~241.8 | 84.6 |
| 4r | H | H | Cl | Cl | Cl | CH3 | white solid | 210.4~211.4 | 78.9 |
| 4s | H | OCH3 | OCH3 | Cl | Cl | CH3 | white solid | 243.9~245.2 | 91.3 |
3-Amino-5-chloro-2-((4-chlorophenoxy)methyl)-3H-quinazolin-4-one (4a). 1H-NMR (400 MHz, CDCl3): δ 4.411 (s, 2H, -CH2O-), 5.585 (s, 2H, N-NH2), 6.611(d, J = 7.6 Hz, 2H, Ar-H), 7.060 (d, J = 2.4 Hz, 2H, Ar-H), 7.496 (d, J = 8.0 Hz, 1H, Ar-H), 7.546 (d, J = 12.4 Hz, 2H, Ar-H). IR (KBr) ν: 3421, 2923, 1673, 1608, 1490, 1432, 1240, 771 cm−1. EI-MS m/z (%): 335.18 (M+, 67.58), 75.06 (82.84), 100.26 (66.29), 179.23 (100.00).
3-Amino-6-chloro-2-((4-chlorophenoxy)methyl)-3H-quinazolin-4-one (4b). 1H-NMR (600 MHz, CDCl3): δ 5.189 (s, 2H, -CH2O-), 5.293 (s, 2H, N-NH2), 6.995 (d, J = 8.4 Hz, 2H, Ar-H), 7.273 (d, J = 10.8 Hz, 2H, Ar-H), 7.699 (d, J = 9.6 Hz, 1H, Ar-H), 7.716 (d, J = 10.8Hz, 1H, Ar-H), 8.255 (s, 1H, Ar-H). IR (KBr) ν: 3435, 2987, 1747, 1681, 1537, 1494, 1242, 721 cm−1. EI-MS m/z (%): 335.06 (M+, 15.93), 208.11 (33.32), 335.06 (15.93), 179.06 (100.00).
3-Amino-7-chloro-2-((4-chlorophenoxy)methyl)-3H-quinazolin-4-one (4c). 1H-NMR (400 MHz, CDCl3): δ 5.168 (s, 2H, -CH2O-), 5.292 (s, 2H, N-NH2), 6.995 (d, J = 8.4 Hz, 2H, Ar-H), 7.272 (d, J = 8.4 Hz, 2H, Ar-H), 7.476 (d, J = 8.4 Hz, 1H, Ar-H), 7.749 (s, 1H, Ar-H), 8.109 (d, J = 4.4 Hz, 1H, Ar-H). IR (KBr) ν: 3480, 2983, 1679, 1550, 1493, 1245, 773 cm−1. EI-MS m/z, (%): 335.11 (M+, 19.55), 178.04 (33.81), 180.97 (31.33), 179.06 (100.00).
3-Amino-2-((4-chlorophenoxy)methyl)-6-methyl-3H-quinazolin-4-one (4d). 1H-NMR (400 MHz, CDCl3): δ 2.495 (s, 3H, Ar-CH3), 5.188 (s, 2H, -CH2O-), 5.278 (s, 2H, N-NH2),6.993 (d, J = 9.2 Hz, 2H, Ar-H), 7.254 (d, J = 8.4 Hz, 2H, Ar-H), 7.587 (d, J = 4.4 Hz, 1H, Ar-H), 7.629 (d, J = 4.8 Hz, 1H, Ar-H), 8.052 (s, 1H, Ar-H). IR (KBr) ν: 3466, 2964, 2924, 1747, 1676, 1550, 1494, 1244, 788 cm−1. EI-MS m/z (%): 315.07 (M+, 10.46), 188.11 (31.79), 159.02 (100.00).
3-Amino-2-((4-chlorophenoxy)methyl)-6,7-dimethoxy-3H-quinazolin-4-one (4e). 1H-NMR (400 MHz, CDCl3): δ 3.725 (s, 3H, N-CH3), 4.002 (s, 6H, -OCH3), 5.130 (s, 2H, -CH2O-), 5.285 (s, 2H, N-NH2), 7.013 (d, J = 8.8 Hz, 1H, Ar-H), 7.154 (s, 1H, Ar-H), 7.271 (d, J = 8.0 Hz, 1H, Ar-H), 7.573 (s, 1H, Ar-H). IR (KBr) ν: 3401, 2923, 2788, 1676, 1602, 1491, 1432, 1223, 773 cm−1. EI-MS m/z (%): 361.27 (M+, 7.07), 205.08 (69.78), 234.14 (100.00).
3-Amino-2-((2,4-dichlorophenoxy)methyl)-3H-quinazolin-4-one (4f). 1H-NMR (400 MHz, CDCl3): δ 5.373 (s, 2H, -CH2O-), 5.376 (s, 2H, -NH2), 7.132 (d, J = 4.8 Hz, 1H, Ar-H), 7.220 (d, J = 8.4 Hz, 1H, Ar-H), 7.386 (s, 1H, Ar-H), 7.556 (d, J = 6.8 Hz, 1H, Ar-H), 7.768~7.791 (m, 2H, Ar-H), 8.302 (d, J = 8.0, 1H, Ar-H). IR (KBr) ν: 3421, 2923, 1673, 1604, 1598, 1447, 1232, 771 cm−1. EI-MS m/z (%): 337.12 (M+, 1.62), 144.11 (30.64), 300.12 (72.15), 302.17 (16.80), 145.15 (100.00).
3-Amino-5-chloro-2-((2,4-dichlorophenoxy)methyl)-3H-quinazolin-4-one (4g). 1H-NMR (400 MHz, CDCl3): δ 5.295 (s, 2H, -CH2O-), 5.326 (s, 2H, -NH2), 7.191 (d, J = 8.2 Hz, 1H, Ar-H), 7.206 (d, J = 4.8 Hz, 1H, Ar-H), 7.248 (d, J = 11.2 Hz, 1H, Ar-H), 7.535 (s, 1H, Ar-H), 7.636 (d, J = 5.2 Hz, 2H, Ar-H), 8.302 (d, J = 8.0, 1H, Ar-H). IR (KBr) ν: 3490, 2992, 1676, 1608, 1479, 1432, 1233, 773 cm−1. EI-MS m/z (%): 369.06 (M+, 3.50), 178.09 (27.76), 336.13 (30.28), 179.05 (100.00).
3-Amino-6-chloro-2-((2,4-dichlorophenoxy)methyl)-3H-quinazolin-4-one (4h). 1H-NMR (400 MHz, CDCl3): δ 5.342 (s, 2H, -CH2O-), 5.351 (s, 2H, -NH2), 7.115 (d, J = 2.0 Hz, 1H, Ar-H), 7.396 (d, J = 2.4 Hz, 1H, Ar-H), 7.697 (s, 1H, Ar-H), 7.708 (d, J = 2.0 Hz, 1H, Ar-H), 7.713 (d, J = 4.8 Hz, 1H, Ar-H), 8.268 (s, 1H, Ar-H). IR (KBr) ν: 3325, 3099, 1681, 1576, 1489,1253, 832 cm−1. EI-MS m/z (%): 368.90 (M+, 4.37), 208.10 (41.88), 334.0 (72.00), 366.00 (51.84), 179.01 (100.00).
3-Amino-7-chloro-2-((2,4-dichlorophenoxy)methyl)-3H-quinazolin-4-one (4i). 1H-NMR (600 MHz, CDCl3): δ 4.553 (s, 2H, -CH2O-), 4.599 (s, 2H, -NH2), 6.815 (d, J = 4.8 Hz, 1H, Ar-H), 6.837 (d, J = 2.4 Hz, 1H, Ar-H), 7.167 (s, 1H, Ar-H), 7.207 (d, J = 8.8 Hz, 1H, Ar-H), 7.228 (d, J = 5.2 Hz, 1H, Ar-H), 7.405 (s, 1H, Ar-H). 13C-NMR (CDCl3) δ: 69.21, 117.26, 118.65, 122.89, 124.09, 124.49, 127.62, 128.07, 130.31, 131.39, 139.07, 147.78, 151.98, 160.11,164.05. IR (KBr) ν: 3395, 2929, 1673, 1610, 1501, 1483, 1251,771 cm−1. EI-MS m/z(%):368.99 (M+, 2.58), 179.07 (86.13), 181.13 (31.11), 334.14 (50.05), 336.15 (21.99), 162.02 (100.00).
3-Amino-2-((2,4-dichlorophenoxy)methyl)-6,7-dimethoxy-3H-quinazolin-4-one (4j). 1H-NMR (400 MHz, CDCl3): δ 3.871 (s, 3H, -OCH3), 3.886 (s, 3H, -OCH3), 5.434 (s, 2H, -CH2O-), 5.638 (s, 2H, -NH2), 7.006 (s, 1H, Ar-H), 7.192 (d, J = 8.8 Hz, 1H, Ar-H), 7.320 (d, J = 2.4 Hz, 1H, Ar-H), 7.445 (s, 1H, Ar-H), 7.618 (s, 1H, Ar-H). IR (KBr) ν: 3390, 2927, 1673, 1610, 1599, 1501, 1253, 806 cm−1. EI-MS m/z (%): 395.04 (M+, 6.84), 205.05 (65.95), 234.06 (100.00).
5-Chloro-2-((4-chlorophenoxy)methyl)-3-methyl-3H-quinazolin-4-one (4k). 1H-NMR (400 MHz, CDCl3): δ 3.684 (s, 3H, N-CH3), 5.124 (s, 2H, -CH2O-), 6.986 (d, J = 8.8 Hz, 2H, Ar-H), 7.270 (d, J = 8.0 Hz, 2H, Ar-H), 7.507 (d, J = 9.2 Hz, 1H, Ar-H), 7.603 (d, J = 3.2 Hz, 1H, Ar-H). IR (KBr) ν: 2990, 2923, 1675, 1600, 1479, 1425, 1245, 785 cm−1. EI-MS m/z (%): 334.00 (M+, 6.00), 168.06 (20.78), 209.01 (18.86), 166.29 (100.00).
6-Chloro-2-((4-chlorophenoxy)methyl)-3-methyl-3H-quinazolin-4-one (4l). 1H-NMR (600 MHz, CDCl3): δ 3.733 (s, 3H, N-CH3), 5.155 (s, 2H, -CH2O-), 6.995 (d, J = 9.0 Hz, 2H, Ar-H), 7.272 (d, J = 9.0 Hz, 2H, Ar-H), 7.667 (d, J = 9.0 Hz, 1H, Ar-H), 7.702 (d, J = 9.0 Hz, 1H, Ar-H), 8.260 (s, 1H, Ar-H). IR (KBr) ν: 3096, 2924, 1677, 1602, 1481, 1245, 785 cm−1. EI-MS m/z (%): 334.12 (M+, 13.55), 166.03 (67.05), 207.04 (100.00).
7-Chloro-2-((4-chlorophenoxy)methyl)-3-methyl-3H-quinazolin-4-one (4m). 1H-NMR (400 MHz, CDCl3): δ 3.720 (s, 3H, N-CH3), 5.147 (s, 2H, -CH2O-), 6.993 (d, J = 9.0 Hz, 2H, Ar-H), 7.267 (d, J = 8.8 Hz, 2H, Ar-H), 7.459 (d, J = 6.8 Hz, 1H, Ar-H), 7.698 (s, 1H, Ar-H), 8.213 (d, J = 4.8 Hz, 1H, Ar-H). IR (KBr) ν: 3038, 2923, 1674, 1596, 1482, 1421, 1240, 781 cm−1. EI-MS m/z (%): 334.09 (M+, 11.78), 167.96 (24.42), 209.17 (19.90), 166.00(100.00).
2-((4-Chlorophenoxy)methyl)-3,6-dimethyl-3H-quinazolin-4-one (4n). 1H-NMR (400 MHz, CDCl3): δ 2.498 (s, 3H, Ar, -CH3), 3.726 (s, 3H, N-CH3), 5.152 (s, 2H, -CH2O-), 6.997 (d, J = 5.2 Hz, 2H, Ar-H), 7.259 (d, J = 9.2 Hz, 2H, Ar-H), 7.585 (d, J = 2.0 Hz, 1H, Ar-H), 7.599 (d, J = 4.0 Hz, 1H, Ar-H), 8.082 (s, 1H, Ar-H). IR (KBr) ν: 3080, 2924, 1674, 1599, 1490, 1423, 1241, 782 cm−1. EI-MS m/z (%): 314.23 (M+, 16.06), 146.15 (79.66), 158.07 (77.83), 187.13 (100.00).
2-((4-Chlorophenoxy)methyl)-6,7-dimethoxy-3-methyl-3H-quinazolin-4-one (4o). 1H-NMR (400 MHz, CDCl3): δ 4.007 (s, 3H, -OCH3), 4.035 (s, 3H, -OCH3), 5.176 (s,2H, -CH2O-), 6.992 (d, J = 4.8 Hz, 1H, Ar-H), 7.091 (s, 1H, Ar-H), 7.265 (d, J = 8.8 Hz, 1H, Ar-H), 7.602 (s, 1H, Ar-H). IR (KBr) ν: 3042, 2900, 1675, 1598, 1487, 1424, 1240, 816 cm−1. EI-MS m/z (%): 360.18 (M+, 6.41), 192.11 (19.88), 204.11 (18.44), 233.16 (100.00).
2-((2,4-Dichlorophenoxy)methyl)-3-methy-3H-quinazolin-4-one (4p). 1H-NMR (400 MHz, CDCl3): δ 3.806 (s, 3H, N-CH3), 5.264 (s, 2H, -CH2O-), 7.176 (d, J = 4.8 Hz, 1H, Ar-H), 7.190 (d, J = 2.0 Hz, 1H, Ar-H), 7.389 (s, 1H, Ar-H), 7.329~7.533 (m, 1H, Ar-H), 7.707 (d, J = 8.0 Hz, 1H, Ar-H), 7.755~7.775 (m, 1H, Ar-H), 8.305 (d, J = 8.0 Hz, 1H, Ar-H). IR (Br) ν: 2984, 1676, 1570, 1476, 1251, 773 cm−1. EI-MS m/z (%): 334.12 (M+, 5.76), 144.08 (35.49), 173.09 (51.15), 299.09 (76.34), 132.05 (100.00).
6-Chloro-2-((2,4-dichlorophenoxy)methyl)-3-methyl-3H-quinazolin-4-one (4q). 1H-NMR (400 MHz, CDCl3): δ 3.798 (s, 3H, N-CH3), 5.235 (s, 2H, -CH2O-), 7.016 (d, J = 4.8 Hz, 1H, Ar-H), 7.112 (d, J = 8.4 Hz, 1H, Ar-H), 7.233 (d, J = 4.8 Hz, 1H, Ar-H), 7.341 (s, 1H, Ar-H), 7.418 (d, J = 8.8 Hz, 1H, Ar-H), 7.421 (s, 1H, Ar-H). IR (KBr) ν: 2970, 1675, 1602, 1480, 1426, 1290, 788 cm−1. EI-MS m/z (%): 370.10 (M+, 5.19), 178.09 (55.72), 333.03 (51.79), 207.09 (100.00).
7-Chloro-2-((2,4-dichlorophenoxy)methyl)-3-methyl-3H-quinazolin-4-one (4r). 1H-NMR (400 MHz, CDCl3): δ 3.789 (s, 3H, N-CH3), 5.237 (s, 2H, -CH2O-), 7.145 (d, J = 9.0 Hz, 1H, Ar-H), 7.206 (d, J = 9.0 Hz, 1H, Ar-H), 7.394 (s, 1H, Ar-H), 7.472 (d, J = 8.4 Hz, 1H, Ar-H), 7.694 (s, 1H, Ar-H), 8.225 (d, J = 8.4 Hz, 1H, Ar-H). IR (KBr) ν: 2924, 2854, 1675, 1602, 1480, 1426, 1290, 788 cm−1. EI-MS m/z (%): 367.79 (M+, 2.31), 207.08 (25.72), 333.05 (37.41), 166.13 (100.00).
2-((2,4-Dichlorophenoxy)methyl)-6,7-dimethoxy-3-methyl-3H-quinazolin-4-one (4s) 1H-NMR (400 MHz, CDCl3): δ 3.799 (s, 3H, N-CH3), 4.007(s, 6H, -OCH3), 5.229 (s, 2H, -CH2O-), 7.006 (s, 1H, Ar-H), 7.182 (d, J = 4.8 Hz, 1H, Ar-H), 7.221 (d, J = 2.0 Hz, 1H, Ar-H), 7.341 (s, 1H, Ar-H), 7.418 (s, 1H, Ar-H), 7.421 (s, 1H, Ar-H). IR (KBr) ν: 306, 2835, 1673, 1637, 1497, 1245, 761 cm−1. EI-MS m/z (%): 394.07 (M+, 23.01), 192.08 (17.63), 204.11 (15.13), 233.11 (100.00).